Abstract
High-intensity focused ultrasound (HIFU)–mediated drug delivery is a relatively novel technique used to deliver drugs to a targeted location in the body. High-intensity focused ultrasound–mediated drug delivery has a broad range of applications, such as tumor therapy, treating central nervous diseases, transsclera drug delivery, and cardiovascular treatments. Targeted treatments prove to be advantageous to systemic treatments due to the reduction in the associated side effects. Thus, this literature review focuses on the various applications of HIFU-mediated drug delivery as well as the mechanism involved. This article is intended to supply the reader with a detailed description of how this technique can be used as well as describe its potential to surpass other treatment methods. Further discussion on the efficiency, limitations, and future of HIFU-mediated drug delivery is addressed. Furthermore, the gaps in the published literature, relative to this topic, are discussed. Ultimately, HIFU-mediated drug delivery is a developing technique that could provide patients with exciting treatment options.
Cancer is a prevalent condition affecting millions of individuals worldwide. According to the United Kingdom’s Cancer Research Center, 1 there are more than 200 different forms of cancer. Many patients living with cancer are not eligible for certain forms of treatment due to tumor size, location, and multi-focality or multi-centricity. 2 A high-intensity focused ultrasound (HIFU)–mediated drug delivery system, or sonophoresis, has the capability to provide patients with a potential alternative. 3 This novel approach provides patients with a targeted noninvasive treatment option for a variety of different cancers as well as other disorders. 3 Sonophoresis is a process that can accelerate the absorption of a localized treatment compared to other types of cancer treatments that are more invasive or damaging to the patient. 3 This technique is considered a form of HIFU.
Cancer is a devastating disease that can oftentimes ravish the body. Management of cancer often involves harsh treatment options over the course of several months or years. A clear issue with cancer treatment is the use of aggressive chemicals and radiation. One of the shortcomings is the lack of targeting, which results in several undesirable side effects for many patients. Those who receive chemotherapy can sometimes find it difficult to attend work and live normally.
According to the World Health Organization 4 (WHO), cancer rates are on a steep incline, and it is predicted that approximately 15 million new cases of cancer will be reported by the year 2020. The average of all cancer survival rates is approximately 50%; therefore, finding different treatment options is crucial. 5 The most popular forms of treatment for cancer are chemotherapy, radiation, and surgery. These treatment options prove to be painful and uncomfortable for the patient for long durations. Some forms of treatment are limited because of tumor specifics. 2 Efforts are being made to reduce dose-limiting toxicity and improve localized drug delivery. High-intensity focused ultrasound–mediated drug delivery could provide a noninvasive and safe approach to treating a variety of cancers within the body.
Purpose of the Article
The purpose of this article is to explore and highlight the effect of HIFU-mediated drug delivery as a potential treatment option for various forms of cancer as well as educate others about this up-and-coming-treatment option. High-intensity focused ultrasound has the potential to offer patients more choices for their treatment. High-intensity focused ultrasound–mediated drug delivery is not as well known as other cancer treatment options and has the potential to combat numerous types of cancers without adversely affecting the body, much like other cancer treatment options. Other cancer treatment options affect patients’ quality of life, whereas HIFU-mediated drug delivery could increase patients’ quality of life by targeting only the tumor. The goal is to lower the prevalence of cancer worldwide with noninvasive treatments, and this treatment technique has that potential.
Methodology and Procedures
The articles selected for review had to be analyzed for their methodology and results of their clinical experiments. Each journal article contained research pertaining to HIFU with regard to using and exploring the effects of drug delivery techniques.
The author conducted a thorough literature search in preparing this article. The primary electronic search tools used were Google Scholar and PubMed Central, and the following terms were used in the search: (a) ultrasound, (b) HIFU, and (c) drug delivery. No Boolean strings were used in any of the above searches. The criteria used in the article search included (a) peer review, (b) full text, (c) English language, and (d) articles published within the past 5 years. Articles were excluded from the literature search if they were (a) not full text, (b) not written in English, (c) wikis, or (d) not peer reviewed. Web sites were excluded unless they were related to (a) HIFU, (b) drug delivery, or (c) ultrasound. Using these research methods, a total of 27 peer-reviewed sources was used to supply the information on HIFU-mediated targeted drug delivery.
Drug Delivery Mechanism
Sonography is a diverse medical application that is used for both diagnostic evaluations and treatment interventions. Recent advances in technology have made the use of sonography more well known and diverse. High-intensity focused ultrasound–mediated drug delivery is being studied as a treatment option for various forms of cancer and is demonstrating promising results. A HIFU drug delivery system provides a simple and isolated approach to precisely control the release of therapeutics while suppressing detrimental effects to the surrounding tissue. 6
Ultrasonic energy is focused and directed toward a small area of interest, which is crucial for drug targeting. When ultrasonic waves are applied to a biological system, cavitation, local tissue heating, and radiation forces can occur. 7 These localized biological changes can increase permeation of membranes, which can be used to initiate localized drug delivery. 7 As sound travels through the body, the acoustic energy is absorbed, diverged, and deflected. 7 A portion of the energy is absorbed into the surrounding tissues, in the form of heat. Body temperature will rise significantly if the rate of heat generation outweighs the rate of heat dissipation. 7 This concept proves to be useful when using temperature-sensitive nanoparticles to deliver drugs to targeted regions of the body. A HIFU treatment procedure is either magnetic resonance imaging (MRI) or ultrasound guided, with MRI guidance being most common. 8 Magnetic resonance imaging (a) can monitor tissue temperatures in real time, (b) can assess for tissue damage, and (c) allows for precise targeted treatment. 8
Using ultrasound to deliver drugs to a targeted location is still a novel concept. The exact mechanism and how the tissues are affected still remain unclear. 9 When ultrasonic waves propagate through the body, the energy can be expressed in different forms, such as acoustic radiation force and heat generation cavitation. 3 These forms of energy can create different bio-effects, which can generate a positive or negative effect on the tissues. 3
Different mechanisms can be used to release drug-filled nanoparticles into targeted locations, such as nonthermal delivery and thermal delivery. Other mechanisms such as the use of endocytosis and active membrane transport are also being considered for effective ways to deliver drugs to areas of interest. 9 During nonthermal delivery, cavitation is induced by acoustic waves. Acoustic cavitation under the influence of ultrasound can be described as the formation, growth, and collapse of microbubbles in an aqueous solution. 10 When acoustic waves propagate through the tissues, oscillating cycles of compression and rarefaction occur. 7 Pressure changes during cycles of compression and rarefaction influence the bubble’s growth and eventually its collapse. 10 Different ultrasound intensities can influence the behavior of the bubbles. Low-intensity acoustic waves create a more stable cavitation state and can lead to sheer stress and friction to surrounding tissues. 9 However, high-intensity acoustic waves can result in microjets and shockwaves, which can create temporary pores in surrounding cell membranes and vessel walls, promoting cell/tissue permeability and increased drug uptake. 9
Thermal effects are another HIFU-mediated drug delivery mechanism. Drug delivery using the thermal effect mechanism uses a focused increase in temperature to release drugs to a targeted location. 9 Hyperthermia can result, which is known to increase cellular uptake of anticancer medications. 9 High-intensity focused ultrasound–induced hyperthermia has shown to significantly enhance the delivery of anticancer treatments to targeted tumor sites in vivo. 9
Drug delivery systems using temperature-sensitive liposomes heat the tissues to a threshold temperature (approximately 40°C), which causes the nanocarrier to release its contents at a targeted site. 11 Mild hyperthermia is induced at approximately 40°C to 45°C and can increase liposome tumor accumulation, also known as the enhanced permeability and retention effect. 11 However, this retention and permeation effect does not guarantee uniform delivery due to the tumor’s interstitial matrix and abnormal vasculature. 7
Pulsed focused ultrasound induces mild hyperthermia by using short bursts of acoustic waves. The tissue is allowed to cool, therefore reducing the harmful effects of extreme tissue heating. 7 Hyperthermia enhances response of radiotherapy and chemotherapy treatments by improving tumor oxygenation. 7 By using pulsing ultrasound exposure, drug delivery is enhanced and can lead to improved drug deposition as well as a better therapeutic effect. 7 Research shows that temperatures above 44°C can result in a rapid loss of perfusion, thereby decreasing the effectiveness of drug delivery. 11 However, 40°C is necessary to trigger the release from temperature-sensitive nanocarriers. 11 This stresses the importance of real-time temperature monitoring and control for effective treatment.
Nanocarriers
Drug-loaded microbubbles
Gas-filled microbubbles are widely used as a contrast agent for sonographic applications, but filling these bubbles with treatment regimens offers a new form of targeted treatment. 10 These microbubbles are injected into the blood vessels and pass harmlessly through the body until they come into contact with the ultrasound beam. Once exposed to increased ultrasonic energy, the bubbles burst, releasing the contained drug regimen and creating temporary pores in surrounding cell membranes and vessel walls (sonophoresis) (Figures 1 and 2). This process of opening cell membranes aids in promoting cell and tissue permeability and increases drug uptake.3,9 Microbubbles, due to their cavitation effects, are most useful for their sonophoresis capabilities, especially for drug delivery across the blood–brain barrier (BBB).
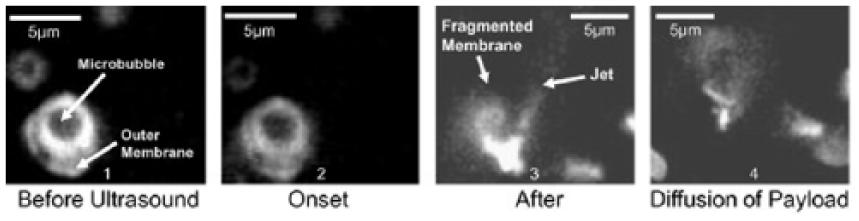
Fluorescent image demonstrating the activation of drug-filled microbubbles upon interaction with high-intensity focused ultrasound showing subsequent rupture of the outer membrane. Reprinted with permission. 12
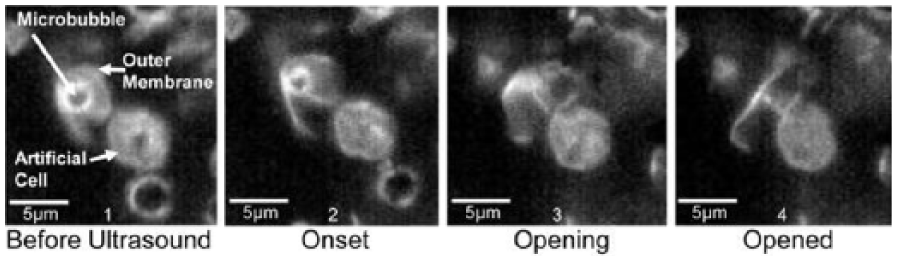
Fluorescent image sequence demonstrating the interaction between a drug-filled microbubble and an artificial cell membrane after acoustic activation. Reprinted with permission. 12
Microbubbles, although short lived, prove to be great contrast agents. Specifically, a microbubble was created to detect liver tumors by using a phosphatidylserine-stabilized perfluorobutane, called Sonazoid. 3 Sonazoid microbubbles are taken up only by Kupffer cells in the liver, and this property makes this delivery system so special. 3 Approximately 10 minutes post-injection, the healthy liver parenchyma will be enhanced by the contrast agent, whereas the tumor-lacking Kupffer cells will be negatively enhanced. 3 Once the location of the tumor has been specified, another nanocarrier containing a chemotherapy regimen could be used.
Nanodroplets
Microbubbles can prove to be disadvantageous as drug delivery carriers. Microbubbles have a short lifespan and are too large to traverse the endothelial gap to penetrate most tumors. 13 Under the influence of an acoustic field, liquid droplets have the ability to change phase and become a gas, releasing the contained drug into the surrounding tissues. 13 This phenomenon is known as acoustic droplet vaporization. 13 These nanoparticles can remain stable in an aqueous solution much longer compared with air-filled microbubbles of the same size. 13 This improves the nanodroplet’s half-life in tissues and can range from 4 days to 65 days. 13 Research shows that nanodroplets filled with chemotherapeutic drugs, such as paclitaxel, remain stabilized within the droplet and are successfully released when activated by ultrasound, resulting in effective tumor regression. 13 Fluorocarbons, such as perfluorocarbons (PFCs), can be used as a contrast agent to visualize and validate the correct location for drug delivery. 13 Currently, acoustic droplet vaporization still remains in the preclinical stages and has proven to be successful in animal models; studies demonstrate tumor regression and metastasis suppression in various forms of cancer, such as pancreatic and breast tumors. 13 Nanodroplets are more advantageous to microbubbles due to nanodroplets having a more stable cavitation, which makes acoustic droplet vaporization a better candidate for targeted drug delivery by providing minimal damage to surrounding tissues. 13
Liposomes
Liposomes are another form of thermosensitive drug carriers and are one of the few drug delivery units being used in clinical trials. 11 Liposomes are a lipid bilayer vesicle and can carry any drug of choice 9 (Figure 3). The lipid membrane can be altered to achieve a number of functions including antimicrobial properties. 9 Liposomes are often used as the shell of nanobubbles due to their low immunogenicity, degradability, and good biocompatibility. 9 Microbubble-liposome complexes have a unique advantage to other thermosensitive drug carriers. 14 These particles are excellent contrast agents and yield a strong acoustic signal. 14 The liposome can be filled with various therapeutic agents, such as chemotherapies, genetic material, and molecular drugs, making the liposome quite diverse. 14 The microbubble-liposome complex has been successful in significantly reducing tumor size. 14
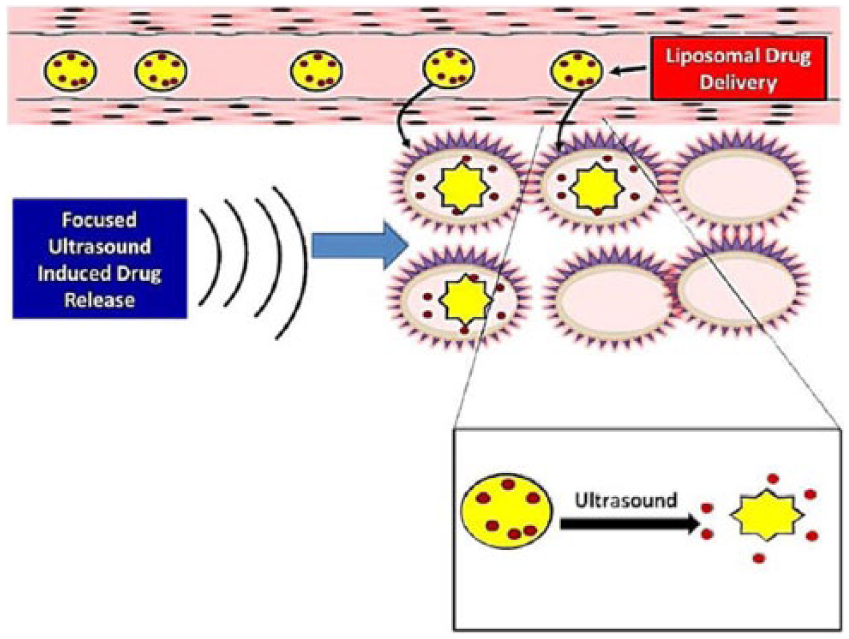
Ultrasound-activated liposome mechanism. Liposomes burst upon exposure to ultrasonic waves, leading to an effective targeted drug delivery. Reprinted with permission. 15
Some advantages to using liposomes are that they have an enhanced permeability and retention effect, which has shown to be effective when treating tumors. 7 However, the permeability and retention effect does not always guarantee a uniform delivery. 7 These thermosensitive carriers can be activated when they come in contact with ultrasound waves. Using liposomes combined with mild hyperthermia is significantly more effective in treating tumor growth compared with free drugs. 7
Applications
Targeted tumor therapy
Many cancers are inoperable due to their location and nature. Luckily, HIFU-mediated drug delivery offers an alternative treatment method for these individuals. For example, pancreatic cancer affects approximately 47 000 individuals with a mortality rate of about 98%. 8 Of these individuals affected, only about 15% to 20% are eligible for surgery. 8 Pancreatic cancer, such as pancreatic ductal adenocarcinoma, is known for having poor tumor vascularization, resulting in an inadequate response to conventional chemotherapy. 16 High-intensity focused ultrasound–mediated drug delivery holds promise for treating individuals with cancers such as pancreatic ductal adenocarcinomas due to its ability to enhance drug extravasation, drug concentration, and diffusion. 16
Treating patients suffering from prostate cancer proves to be a difficult task due to the heterogeneous and specific nature of these cancers. 17 Several treatment strategies have been used over the years, but approximately 35% of patients within 10 years have a relapse. 17 Tumor tissues are generally characterized by having irregular blood flow, poorly organized vasculature, and decreased lymphatic drainage. 17 Drug delivery systems exploit these characteristics by inducing the enhanced permeability and retention effect. Endothelial junctions of blood vessels in normal tissues are extremely small and measure approximately 7 nm, whereas tumor endothelial pores for most tumor types range from 100 nm to 780 nm, allowing nanocarriers to enter only diseased tissues, which increases the bioavailability of the drug in the tumor. 17
Zhou 18 evaluated the efficiency of chemotherapeutic agents combined with nanoparticles injected in tumor cells and activated with ultrasound. This study demonstrated efficient tumor growth reduction and complete elimination after 2 months. 18 Treatment with chemotherapeutic agents alone exhibited necrotic regions within the tumor, whereas chemotherapeutics coupled with ultrasound energy demonstrated dramatic tissue necrosis. 18 Ranjan et al 19 studied the effects of free chemotherapeutic agents (Doxorubicin) compared with drug-encapsulated low temperature sensitive liposomes (LTSLs) on rabbits with thigh muscle tumors. The LTSLs revealed a 7.6-fold greater tumor drug delivery compared with the free Doxorubicin 19 (Figure 4). With regard to specificity, drug concentrations in the tumor using LTSLs were 43.5-fold greater compared with free drugs. 19
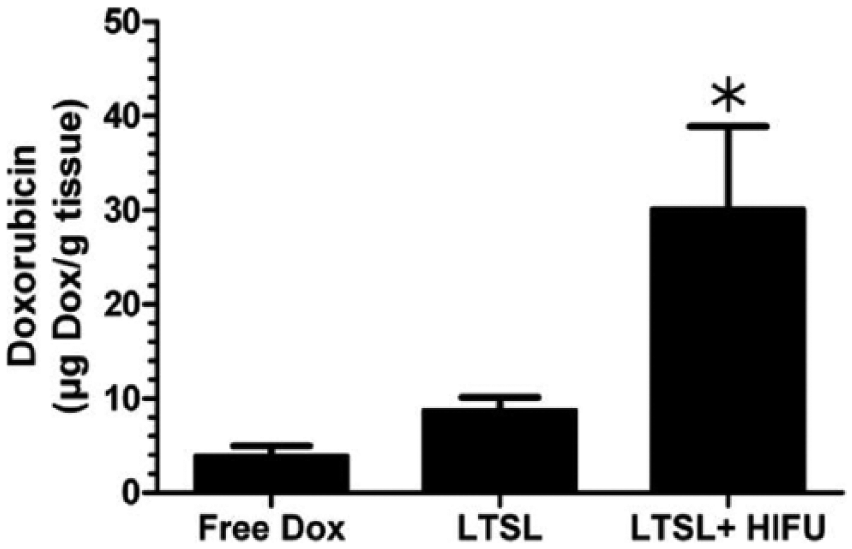
Doxorubicin identified in rabbit tumor following treatment of free Doxorubicin, LTSL, or LTSL+HIFU. Data show mean Doxorubicin concentration within the tumor with a standard error of mean (n = 5). Dox, Doxorubicin; HIFU, high-intensity focused ultrasound; LTSL, low temperature sensitive liposome. Reprinted with permission. 19
Magnetically charged nanoparticles are another strategy employed to treat tumors via drug delivery. 20 Superparamagnetic iron oxide is used as a contrast agent and also as a means for a more targeted drug delivery. 20 These nanocarriers can be magnetically directed to the tumor and activated by HIFU. 20 These contrast agents are used to provide real-time locations of drugs during MRI-guided HIFU treatments. 21 Paramagnetic agents allow the drugs to be tracked and released at a specific location in the body. However, there are some disadvantages to using these contrast agents. These paramagnetic contrast agents alter the specificity of the temperature, creating a temperature reading error range of −4°C to +3°C, depending on the concentration used.21,22 The temperature range alters the effectiveness of treatment and puts the patient in danger of negative bioeffects, such as heat-generated cavitation or tissue ablation. Huang et al 20 performed a study on mice infected with tumors; those treated with HIFU-mediated drug delivery showed half the growth rate compared to those injected with saline. High-intensity focused ultrasound–mediated drug delivery systems hold promise for slowing the progression of tumor growth and metastasis, as well as treating various cancers.
Central nervous diseases
Diseases of the central nervous system are difficult to treat with medications due to their inability to penetrate the BBB. Approximately 100% of large molecule drugs and 98% of small molecule drugs are prevented from crossing the BBB from systemic circulation. 23 Intranasal (IN) drug delivery is one noninvasive technique being studied. The nasal cavity possesses a rich vasculature and has demonstrated drug passage through the BBB in small animals to treat conditions such as Parkinson’s disease, Alzheimer’s disease, Huntington’s disease, autism spectrum disorder, depression, and stroke. 23 Clinical trials on humans reveal that IN delivery of insulin is used to slow the progression of Alzheimer’s disease. 23 Pathways, such as the olfactory and trigeminal nerves, make it possible for therapeutic agents to pass through to the brain. 23 However, this method delivers drugs to the entire brain. By using HIFU-mediated drug delivery, targeted areas of the brain can be treated by therapeutic agents.
Microbubbles activated by HIFU can disrupt the BBB by creating temporary pores, resulting in minimal to no tissue damage. 23 The opening of these pores is reversible and will close within several hours to several days, depending on the acoustic constraints used. 23 The pore size can also be adjusted to allow for smaller or larger molecules to pass and can be manipulated by using various microbubbles and acoustic parameters. 23 Intranasal administration offers a simple, painless, and noninvasive approach to deliver drugs directly to the brain, eliminating the use of systemic delivery and thus reducing associated side effects. 23 Future applications of this technique are the utilization of stem cell delivery to the brain to treat traumatic brain injury, stroke, and neurodegenerative diseases. 7 Chen et al 23 evaluated the efficiency of IN drug delivery techniques in 10 mice compared with mice receiving drugs systemically. Significant drug accumulation was noted in the targeted area of the brain in mice receiving ultrasound-mediated IN treatment; therefore, the bioavailability and biodistribution of the drug had increased compared with mice receiving treatment without ultrasound activation. 23
Transsclera drug delivery
There are many disorders affecting the eye, but the eye’s anatomic and physiologic barriers limit the effectiveness of drug treatment regimens. 24 Diabetic retinopathy, age-related macular degeneration, retinitis pigmentosa, and glaucoma are the most common cause of irreversible blindness worldwide. 25 Age-related macular degeneration affects 1.7 million Americans older than 65 years, and approximately 200 000 new cases arise each year. 25 Ultrasound biomicroscopy is used to provide high-quality diagnostic imaging of the anterior portion of the eye, and HIFU has proved to be an effective treatment for reducing intraocular pressure. 24 The blood–retinal barrier proves to be a challenge for drug delivery using conventional treatment options. 24 Ocular ointments are the easiest method of drug delivery but do not stay on the eye long enough. 25 Therefore, drug absorption is limited to a few minutes and the bioavailability is low. 25 High-intensity focused ultrasound–mediated drug delivery could be a potential treatment option for ocular disorders.
In New Zealand, ultrasound has been used in conjunction with microbubbles to enhance drug delivery to the posterior portion of the eye. 24 Nabili et al 24 evaluated the effectiveness of transsclera drug delivery in rabbits. Frequencies of 400 and 600 kHz were used to cross-penetrate the blood–retinal barrier. 24 Sonophoresis was used to create pores to disrupt the blood–retinal barrier, therefore increasing drug uptake. 24 Drug penetration was most effective at 400 kHz. The permeability of the cornea had increased 2.8 times at 400 kHz compared with 2.4 times at 600 kHz. 23 No ocular tissue damage was observed and the drug delivery efficiency was noted to be 1.5 to 2 times more effective. 24 Other possibilities for ocular delivery involving ultrasound application include delivering antiviral and antifungal drugs to treat other eye diseases, such as scleritis, keratitis, and herpetic eye disease. 24
Cardiovascular drug delivery
The third leading cause of death in the United States and the single greatest long-term expenditure is stroke. 26 There are three types of strokes: ischemic, hemorrhagic, and embolic. Ischemic strokes are the most common and account for approximately 80% of all strokes. 26 These strokes are caused by a blockage in the arteries, mainly due to atherosclerosis. Ultrasound-activated microbubbles are successful in treating stroke by increasing the rate of clot dissolution. 26 Unger et al 26 demonstrated that microbubbles were effective in treating pigs with myocardial infarctions. The effects of HIFU-mediated sonothrombolysis were studied on 26 pigs with acute occlusions. 26 Pigs were injected with microbubbles infused at a rate of 2.5 to 3.0 mL/min and exposed to pulses of 5 Hz for 5 seconds. 25 Intimal thickness was measured at 30-, 60-, and 90-minute intervals. 26 Pigs treated with targeted ultrasound showed a significant reduction in infarct size and increased blood flow compared with pretreatment values. 26
Medications are on the market to prevent neointimal formation, such as rapamycin, but can be harmful in higher doses, leading to an increased risk of developing cancer and decreased immune function. 27 By targeting rapamycin, associated risks are reduced. Studies have been performed on small animal models and have shown to be an effective way to treat cardiovascular injuries. 27 Currently, clinical trials have already begun in Europe to treat individuals with stroke. 27
Discussion
This literature review explored and reviewed various journal articles to answer each of the research questions, such as (a) why HIFU is more superior to other treatment options, (b) how HIFU-mediated drug delivery works, (c) how effective HIFU is for drug delivery, and (d) what are the limitations of HIFU-mediated drug delivery. To determine the potential of HIFU-mediated drug delivery compared with other treatment options, both prospective and retrospective articles were used. As far as explaining how HIFU-mediated drug delivery works, multiple sources were used to explain the mechanism involved. However, not enough information has been discovered to fully understand the mechanism involved. Furthermore, the effectiveness of HIFU-mediated drug delivery was explored as well as its future applications. This technique has numerous applications, such as tumor therapy, treating central nervous diseases, transsclera drug delivery, and cardiovascular treatments. Last, multiple creditable sources were used to answer what limitations are present in HIFU-mediated drug delivery.
The research related to HIFU-mediated drug delivery has advanced immensely over the years and is expected to continue. For this technology to grow, several aspects hold exciting potential. The mechanism related to interactions between ultrasound energy and drug delivery vehicles and the subsequent bioeffects need to be further investigated. Nanocarriers need further development to enhance penetration into endothelial junctions as well as improvement of circulation lifespan to create a more effective drug delivery. Furthermore, the interactions between different drugs and ultrasound need to be further investigated to know what drugs are most useful for the different organs and tissues being treated, taking into consideration the tradeoff between drug delivery and cell viability. Last, experiments need to be conducted on larger animal models to transition to human clinical trials.
Conclusion
High-intensity focused ultrasound–mediated drug delivery has a broad range of applications, such as tumor therapy, treating central nervous diseases, transsclera drug delivery, and cardiovascular treatments. When compared to other physical treatment methods, focused ultrasound is more advantageous. High-intensity focused ultrasound has the ability to penetrate deep into tissues and target a precise location in the body with a diameter of approximately 1 mm. It also can be meticulously controlled, and most important, ultrasound is noninvasive. 16 Currently, HIFU-mediated drug delivery shows positive results in treating tumors in animal models as well as delivering drugs to targeted regions. 16 Although HIFU-mediated drug delivery holds great promise as an effective treatment modality, there are some drawbacks and challenges that need to be explored.
Limitations
The bioeffects related to the interaction between nanocarriers and ultrasound energy are not clearly understood. 9 To achieve the anticipated outcome, specific nanocarriers must be selected based on their specific properties. Nanocarriers, also known as nanoparticles, must be small enough to travel through the bloodstream but large enough to prevent renal excretion. 9 When ultrasonic energy waves propagate through the body, the energy can be expressed in different forms such as acoustic radiation force and heat generation cavitation. 3 These forms of energy can create different bioeffects, which can generate a positive or negative effect on the tissues. 3 The mechanism related to the delivery of drugs using a focused ultrasound beam must be fully understood for clinical trials to be safely conducted on humans.
Future
The complexity of the microscopic interactions between ultrasound energy and cells makes it difficult to reveal the mechanism that supports HIFU-mediated drug delivery; therefore, further study is required. 28 Approval for these nanocarriers by the US Food and Drug Administration (FDA) may be a slow process and may affect the interest of pharmaceutical companies to undertake development due to the given risk and expenses related to clinical trials. 13 Clinical trials are already being conducted in other countries for cardiovascular applications, such as treatment for stroke. 26 The FDA gives guidelines to study bioeffects on animal models prior to providing authorization to conduct human clinical trials. 26 Although there is a copious amount of literature concerning HIFU-mediated drug delivery, there remains a need to generate higher levels of research evidence on its efficacy. It is imperative that nanocarriers are thoroughly examined to be sure that they are biocompatible, are biodistributed, and achieve clearance before they can be deemed safe for clinical use. 29
Gaps in the Literature
Advances in medicine are being made on a daily basis. If one treatment modality can prove to be more effective than another, it is essential to educate health care providers and the public about these advances. High-intensity focused ultrasound–mediated drug delivery systems have the potential to decrease cancer mortality and morbidity rates as well as many more applications. Currently, HIFU-mediated drug delivery is a relatively novel technique and still requires more research. 3 The mechanism involving drugs and ultrasound, effect of ultrasound application, and consequence of tissue heating are not completely understood. 16
High-intensity focused ultrasound–mediated targeted drug delivery poses many different challenges. Sound waves have the ability to move molecules and can be expressed through heat generation or acoustic radiation force. 3 These mechanisms cause different biological changes that can increase permeation of membranes, which can be used to enhance drug concentration and uptake at the target site. 3 It is crucial to understand the underlying mechanism with regard to HIFU-mediated drug delivery systems because treatments could potentially do more harm than good.
High-intensity focused ultrasound has the potential to have positive and negative effects on tissues, whereas positive effects include targeted drug release, increased drug carrier, and drug internalization by tumor cells. 16 However, increased drug carrying microbubbles can evoke a potentially negative cellular response, such as edema, inflammation, and/or hemorrhage. 16 More research needs to be conducted on various drug delivery carriers, such as microbubbles and liposomes, to better understand their biological effects under nonionizing radiation. 3
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
