Abstract
Osteoporosis, a high-incidence chronic disease, is known to cause physical pain, even leading to disability of patients, and creates a vast social burden. To improve the shortcomings of the current clinical osteoporosis treatment drugs, several studies have designed a multitude of nanomedicines to optimize traditional drugs based on the TOP–DOWN design. Fascinating nanomedicine designs are breathing vitality into traditional osteoporosis drugs. However, considering the unique pathological characteristics of osteoporosis and clinical treatment needs, we should also pay attention to the problems outside the spotlight. Here, we reviewed the current clinical osteoporosis treatment drugs, summarized their limitations, and generalized the nanomedicines designed based on the TOP–DOWN strategy for the mentioned deficiencies. In addition, we explored the issues outside the spotlight of the nanomedicine design for the treatment of osteoporosis because of the gap between laboratory design strategy and clinical demands and then determined the rational design of nanomedicines for osteoporosis.
Introduction
Osteoporosis, a systemic disease that seriously harms human health, can lead to long-term bedrest or even disability. 1 According to the International Osteoporosis Society (IOF), osteoporosis affects one in three women and one in five men over the age of 50 years worldwide. 2 The impact of osteoporosis on human health is increasing, considering the growing global aging population and underdiagnosis of osteoporosis in less developed regions.3,4
Osteoporosis can be categorized into primary types (postmenopausal and age-related) and secondary types (resulting from medications or diseases). The disease progresses through an imbalance in bone remodeling—a process governed mainly by osteoclasts (OCs) and osteoblasts (OBs), which resorb bone and form new bone, respectively. Excessive OC activity driven by receptor activator of nuclear factor kappa-B ligand/receptor activator of nuclear factor kappa-B/osteoprotegerin (RANKL/RANK/OPG) pathway signaling, combined with reduced OB function, leads to net bone loss. This imbalance is accentuated in postmenopausal women due to the deficiency of estrogen, which upregulates OC formation and activity.5–7
Therefore, the common pharmacological interventions include antiresorptive agents (e.g. bisphosphonates and denosumab) that inhibit OC activity and anabolic drugs (e.g. teriparatide) that stimulate OB activity.8–10 However, the potential side effects, low bioavailability, and lack of targeting hinder the clinical efficacy of these traditional drugs in the treatment of osteoporosis.11–13
Recently, advances in nanotechnology have offered promising alternatives for osteoporosis treatment. Nanodrug delivery systems improve bioavailability, enable targeted delivery to bone tissue, and reduce off-target effects, representing a novel strategy for restoring bone homeostasis.14–16
Here, we review the design of nanomedicine for osteoporosis based on the TOP–DOWN strategy in recent years. Furthermore, we point out the issues that have not attracted adequate attention in treating osteoporosis with nanomedicine and provide an outlook on the rational design of nanomedicines for osteoporosis (Figure 1).

Traditional drugs and multifunctional nanomedicines for the treatment of osteoporosis, and issues that might be neglected in nanomedicine design.
Disadvantages in the clinical application of traditional medicine to treat osteoporosis
To better understand the effect of nanomedicines on improving the treatment of osteoporosis, the shortcomings of several common traditional clinical drugs used in osteoporosis treatment are described below.
Deficiency of targeting
Hormone therapy (HT) is one of the most effective and common treatments for osteoporosis. 17 As a representative drug of HT, estrogen has been proven to be the main regulatory factor for bone metabolism.18,19 Estrogen deficiency in women after menopause causes bone loss and osteoporosis, and the regulation of bone metabolism in men is also closely related to estrogen. 20 As an estrogen, 17β-estradiol (E2) has been approved by the Food and Drug Administration owing to its high safety and efficacy.21,22 However, E2 lacks bone-targeting ability, and nonselective accumulation in nonskeletal organs such as the breasts, heart, and uterus can cause serious side effects, including but not limited to cardiovascular disease, breast cancer, ovarian cancer, and endometrial cancer. 17 Given that the majority of patients with osteoporosis are postmenopausal older women, these effects greatly limit the clinical use of HT. As E2 is administered systemically, its lack of targeting capability is unfavorable for increasing the local bone density and reducing side effects.
Low bioavailability
Another problem in the clinical application of traditional osteoporosis drugs is their low bioavailability. Raloxifene (RLX) is a selective estrogen receptor modulator with estrogen-like activity in the treatment of osteoporosis.23,24 Due to its low solubility and high permeability, it is classified as a class II drug according to the Biopharmaceutics Classification System. 25 Although the absorption rate of RLX after oral administration can reach 60%, its final bioavailability is only 2% due to first-pass glucuronidation. 26 Statins were first known as lipid-lowering agents and were later discovered to exert anabolic effects during osteoporosis treatment; however, they exhibit low bioavailability in clinical treatment.27,28 As small-molecule drugs, statins can promote bone formation and inhibit bone resorption; however, first-pass metabolism in the liver reduces their systemic utilization to approximately 2%–4%. 29
Potential side effects
In addition to the lack of targeting and low bioavailability, potential side effects affect the clinical use of the current drugs. 30 The effect of bisphosphonates (BPs) on osteoporosis was first described in 1968, and they are commonly used to prevent the loss of bone density in osteoporosis and similar bone disorders. 31 However, various side effects (such as gastroesophageal irritation, jaw osteonecrosis, severe bone turnover suppression, prostate cancer, and multiple myeloma) caused by long-term administration affect the clinical application of BPs in osteoporosis treatment.32,33 Teriparatide, a recombinant human parathyroid hormone 1-34 (rhPTH 1-34) consisting of the first 34 N-terminal amino acids of the hormone, 34 has a dual effect on bone formation and resorption by adjusting the RANKL/OPG ratio. 35 When administered intermittently at low doses, teriparatide promotes bone formation. However, frequent low-dose administration may cause allergic reactions, itching, and infections at the injection site. Continuous administration at high doses can cause bone resorption and hypercalcemia.35–37
Improvement of traditional osteoporosis treatment by nanomedicine
To overcome the shortcomings of such traditional drugs and achieve better clinical efficacy, the TOP–DOWN strategy has been introduced to design and modify these drugs using nanomedicine architectures. The TOP–DOWN design considers clinical needs as the starting point, subdivides the existing problems in the clinical treatment, and develops solutions one by one.38–40 Its advantages include the following: 1. More diverse drug dosage forms enable more flexible routes of administration, increasing bioavailability and patient treatment compliance. 2. It provides more diverse modification methods. 3. A higher number of precise release sites improve the targeting ability of the drugs, increase drug retention time in the target organ, and achieve better therapeutic effects with smaller drug doses to reduce side effects. Nanomedicines designed via the TOP–DOWN approach can achieve excellent therapeutic effects at lower dosages and improve treatment safety.
More flexible dosage form
The dosage form of traditional drugs is limited by their physicochemical properties, while nanomedicines enrich their dosage forms. Oral administration is commonly used in clinical practice due to its high safety and patient compliance; however, it suffers from reduced bioavailability due to the first-pass effect.41,42 Hosny et al. used nanostructured lipid carriers (NLCs) to increase the bioavailability of RLX by improving its solubility. 23 The broken-down lipids in NLCs interact with bile salts in the presence of intestinal enzymes, facilitating the formation of mixed micelles that absorb RLX. The micelles undergo selective lymphatic absorption or are taken up by M cells in Peyer’s patches in the intestine, bypassing premature first-pass metabolism, thereby enhancing RLX bioavailability. 43 Compared with commercially available tablets, NLC formulations improve the absorption rate of RLX. The bioavailability of RLX in NLC was approximately 3.85 times that of commercially available tablets based on AUC0–inf (area under the plasma concentration–time curve from time 0 extrapolated to infinite time) calculations.
Due to the high polarity and low lipophilicity of BPs, the local transdermal administration of nanocrystallized BPs has been widely studied to increase the local concentration by penetrating the stratum corneum, which limits transdermal drug absorption.44,45 Li et al. prepared a transdermal patch of chitosan–alendronate sodium nanoparticles (CS-AL-NP) using solvent casting–evaporation technology supplemented by external ultrasound to treat osteoporosis. 46 The transdermal CS-AL-NP showed a higher plasma calcium suppression ability and bioavailability than molecular AL, preventing osteoporosis by locally inhibiting OC absorption function. Kaur et al. developed a hydrogel-based nanoemulsion with a small size to deliver fluvastatin. 29 The nanoemulsion increases drug permeability, avoids the first-pass effect of liver metabolism, reduces side effects, and provides a facile transdermal administration procedure.
New possibilities brought by modification
Through various modifications, nanomedicines can improve traditional drugs, including but not limited to extending blood circulation time and enhancing bone-targeting ability.47,48
Surface modification with PEG (polyethylene glycol), albumin, and other molecules can extend the circulation time of nanomedicines in the body.49,50 Altaani et al. developed a novel delivery system: self-assembling chitosan structures formed in the presence of peptides or proteins. 34 They also used low molecular weight chitosan to prepare teriparatide polyelectrolyte complexes (PECs) by adding hydroxypropyl beta-cyclodextrin. This structure enhances the bioavailability of oral teriparatide via chitosan, opening tight junctions of enterocytes and enhancing permeation into systemic circulation. Low molecular weight chitosan can also interact with the intestinal mucosa through electrostatic forces, avoiding a sudden increase in blood drug concentration after intravenous injection and achieving long-term drug release. 51
Modifications of nanomedicines can improve their bone-targeting ability. Chen et al. used a 17β-estradiol (E2)-laden mesoporous silica-coated up conversion nanoparticle with a surface modification of ethylenediaminetetraacetic acid (EDTA) to treat osteoporosis. 17 Through the surface adhesion of EDTA, the bone affinity of the E2-loaded up conversion nanoparticles was improved. Tetracycline (TC) not only has broad-spectrum antibacterial properties but can also bind to hydroxyapatite (HAP), acting as a bone-targeting agent.52,53 Thus, it is used as a bone-targeting molecule for the treatment of bone diseases. 54 Wang et al. constructed phospholipid-amorphous calcium carbonate (ACC) hybrid nanoparticles modified with TC to load simvastatin for osteoporosis treatment. 55 In vitro, HAP absorption assays using fluorescein isothiocyanate (FITC)-labeled ODA (octadecyl amine) showed that TC modification increased the uptake of TC/ACC particles by MC3T3-E1 cells by approximately 70%. In vivo experiments showed that the fluorescence intensity of indocyanine green (ICG)–labeled TC/ACC particles in the femur and tibia was 25.72% higher than that of the control group after TC modification. Both in vitro and in vivo results demonstrated that TC/ACC could accumulate in bone tissue, indicating its potential for bone-targeted delivery (Figure 2). Bone homing peptides and exosomes from specific sources are also used to enhance the bone-targeting efficacy of nanomedicines. Vhora et al. synthesized ionizable lipids with long octadecyl chains and head groups of histidine, a cytoprotective amino acid. 56 The pDNA of BMP-9 and a bone homing peptide were used to modify lipid-nucleic acid nanoparticles (LNPs), which effectively promoted bone regeneration in OVX rats. These LNPs exhibited low cytotoxicity, nonsignificant reactive oxygen species (ROS) induction, and high transfection efficiency.

TC-modified ACC nanoparticles loaded with SIM for the treatment of osteoporosis. 55 Copyright ©2021 Ivyspring International Publisher. ACC: amorphous calcium carbonate; BMPs: bone morphogenetic proteins; SIM: simvastatin; TC: tetracycline; PL: phospholipid; SA: stearic acid.
More precise release sites
Osteoporosis is a systemic metabolic disease; however, its actual damage is reflected in osteoporotic fractures in older patients at specific sites, where minor injuries in daily activities can cause serious consequences.57,58 Therefore, increasing bone density at the prone sites of osteoporotic fractures is key to treatment.
Physical stimulation, such as ultrasound, photothermal treatment, magnetic fields, and extracorporeal shock wave (ESW), can mediate the real-time release of drugs from nanocarriers at specific sites according to clinical demands. 59 Utilizing liposomes that are easily disrupted by ultrasound, Crasto et al. designed an ultrasound-triggered liposome to encapsulate recombinant human BMP-2 (rhBMP-2). 60 The release of rhBMP-2 from liposomes was positively correlated with ultrasonic pressure and exposure time. An ultrasound-triggered rhBMP-2 delivery system implanted into the hind leg muscles of mice induced local bone formation only in the ultrasound-exposed area. Therefore, a spatiotemporally controllable responsive design may provide a good prospect for the clinical application of nanomedicine in osteoporosis treatment.
ESW has a wide range of applications in bone and muscle diseases.61–63 ESW not only promotes osteogenesis but also mediates drug release through prominent cavitation effect.64,65 Therefore, ESW is a promising method for controlled release in bone diseases. Previously, Liang et al. established an ESW-mediated controlled release system for nanomedicine and developed hollow HAP nanocrystals loaded with zoledronate (ZOL) as smart nanoplatforms (ZOL-hNPs). 66 With ESW mediation, ZOL on the surface of nanoplatforms was rapidly desorbed and released at the specified osteoporosis site within 100 min. Subsequently, proton neutralization and Ca2+ release were accelerated through ESW-induced decomposition of hNPs, thereby achieving sequential treatment of osteoporosis (Figure 3).
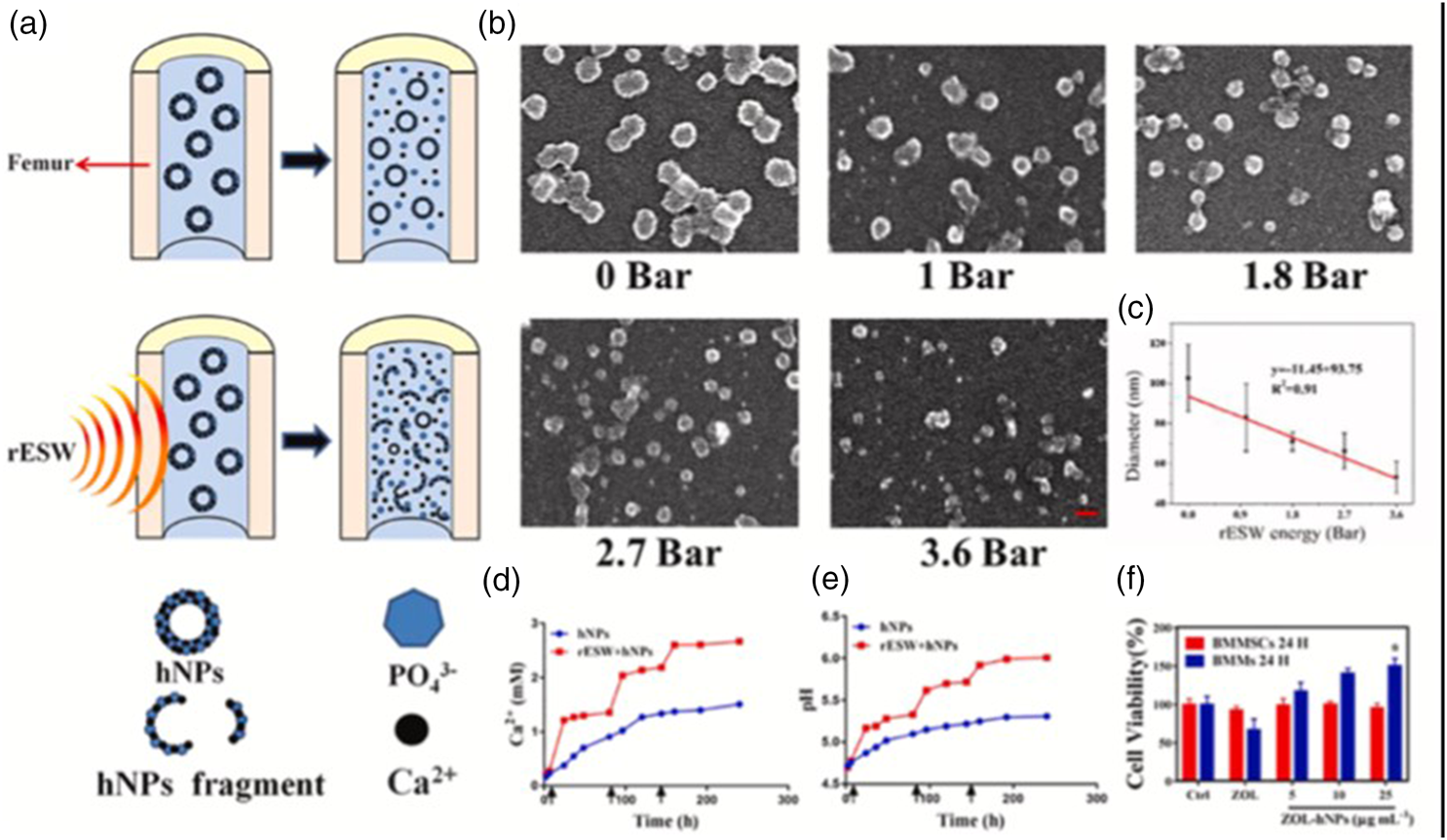
ESW-mediated hNP fragmentation and Ca2+ release. (a) ESW can mediate the fragmentation of hNPs in the femoral bone marrow cavity of rats, thereby releasing Ca2+. (b) SEM images of hNPs processed via ESW with different energy levels. (c) Correlation between ESW energy and particle size of hNPs. (d) ESW promotes Ca2+ release. (e) ESW affects local pH and (f) the effect of hNPs on the viability of bone marrow macrophages and bone marrow mesenchymal stem cells. 66 ESW: extracorporeal shock wave; hNPs: hydroxyapatite nanoparticles; Ca: calcium; SEM: scanning electron microscopy.
New exploration of nanomedicines in the treatment of osteoporosis
With the support of nanotechnology, several nanomedicines have been designed to modify traditional drugs, and numerous new nanomaterials have been reported to be effective in treating osteoporosis. Structured nano-HAP particles can promote osteogenesis and also serve as a matrix to supplement bone. 67 Nano-Au nanoparticles (AuNPs) can regulate the acidic microenvironment of OCs to inhibit excessive bone resorption.68,69 Fullerenol nanoparticles70,71 and iron oxide nanoparticles (IONPs)72,73 can block the fusion of OC precursors by inhibiting the functional actin ring structure of OCs, thus further inhibiting OC maturation and bone resorption.
Giulia et al. developed strontium (Sr)-containing HAP nanoparticles (SrHAn) and used them to treat OBs and OCs. 74 Sr2+ has dual functions, simultaneously regulating OB anabolic processes and OC catabolic processes via calcium-sensitive receptors (CaSR). Treatment in mouse femurs showed that compared with the control group, SrHAn significantly upregulated the expression of OB-related genes such as BGLAP (osteocalcin), IBSP (bone sialoprotein), and SP7 (Osteria) and significantly downregulated the expression of RANKL (tumor necrosis factor ligand superfamily member 11), which is associated with OC activity. This result verifies the dual effects of SrHAn in the treatment of osteoporosis.
AuNPs are widely used in drug delivery, disease diagnosis, and tissue engineering due to their excellent biocompatibility and relatively simple synthesis methods. Bai et al. focused on nanomedicine for osteoporosis and found that AuNPs can inhibit excessive OC bone resorption by regulating the acidic microenvironment. 69 The extracellular acidic microenvironment required by OCs for bone resorption is regulated by V-ATPases, a functional structure assembled from V0 and V1 domains in the membrane. With a high number of carboxyl groups, internalized AuNPs accumulate on the intracellular vesicle membrane. By interacting with the V0 domain of V-ATPase, AuNPs affect the recruitment of the V1 domain and ultimately interfere with acid secretion by OCs.
Fullerenol nanoparticles, as polyhydroxylated derivatives of fullerenes, can inhibit OC differentiation, block the fusion of OC precursor cells, and eventually prevent OC bone resorption (Figure 4).70,71 Based on experimental results, Väänänen et al. suggested that fullerenol nanoparticles interact with actin. SAXS and HR-AFM results showed the adhesion of fullerenol nanoparticles on F-actin and interference with Ca2+-induced actin bundling. The sealing zones (SZs) and ruffled borders (RBs) are the characteristic functional structures of OCs, both rich in actin. 75 The obstruction of actin bundling by fullerenol nanoparticles inhibits the formation of SZs and disrupts the acidic microenvironment required for OCs. The reduction in the RB structure restricts OCs from performing bone resorption.

Fullerenol nanoparticles inhibit osteoclast function by affecting actin bundles. (a) Interactions between F-actin, fullerenol nanoparticles, and bundlers and (b) an overall description of the different bundle changes induced by fullerenol nanoparticles. 71
This strategy of targeting actin for osteoporosis treatment is also used in the design and development of IONPs. IONPs have also been found to possess bone-targeting properties. Liu et al. investigated the effect of IONPs on bone marrow macrophage (BMM) differentiation into OCs. 72 They found that IONPs disturb the formation of actin ring structures in BMMs, and the formation of ring structures was significantly inhibited in both size and number in a dose-dependent manner (Figure 5). The actin structures were not formed when the cells were treated with ferucarbotran at concentrations higher than 0.01 mg/mL or ferumoxytol at concentrations higher than 0.1 mg/mL. Liu et al. suggested that IONPs trigger the upregulation of p62, which recruits CYLD and enhances the deubiquitination of TRAF6. TRAF6 plays an essential role in regulating RANKL signaling and ultimately affects actin ring formation. 76 Downstream activation of NF-κB and MAPK signaling was decreased, resulting in the reduced expression of genes related to OC formation.

IONPs inhibited RANKL-induced osteoclast formation. (a) IONPs inhibited osteoclast formation during differentiation and maturation but had no effect on proliferation and fusion and (b) molecular mechanism of IONP inhibition of RANKL-induced osteoclast formation. 72 IONP: iron oxide nanoparticles; RANKL: receptor activator of nuclear factor kappa-B ligand.
Characterization of nanomedicines in terms of bone-targeting ability
In vitro experiments are employed to preliminarily evaluate the bone-targeting potential of nanomedicines. The key methods include HAP-binding assays, in which nanoparticles are incubated with synthetic HAP or bone samples, and their binding affinity is quantified by measuring the reduction in supernatant nanoparticle concentration under physiologically relevant conditions. 77 Additionally, cellular uptake and retention studies using OB-like cells (e.g. MC3T3-E1) or OCs (e.g. from macrophage cell lines) allow the quantification and visualization of nanoparticle internalization via flow cytometry and confocal microscopy. A comparison of targeted versus nontargeted formulations underscores the benefit of specific ligands. Furthermore, the flow conditions or co-culture systems better mimic the dynamic bone marrow microenvironment, revealing cell-specific uptake mechanisms.
In vivo studies are essential for validation of targeting within complex biological environments. Fluorescence imaging (e.g. in vivo imaging system (IVIS)) permits noninvasive, real-time tracking of biodistribution, with fluorescence intensity quantified in the regions of interest over time. Positron emission tomography (PET), using radiolabeled nanocarriers, provides high sensitivity and spatial resolution for quantifying accumulation in deep bones such as the vertebrae and pelvis. 78 Biodistribution analysis remains the gold standard. After euthanizing animals at set intervals, the bones and organs are excised and processed. Bone samples are cleaned and digested to release the drug or nanomaterial, which is quantified via liquid chromatography–tandem mass spectrometry (LC–MS/MS) (for drugs) or inductively coupled plasma mass spectrometry (ICP-MS) (for metal nanoparticles). The results are expressed as % injected dose per gram of tissue (%ID/g), facilitating a comparison between the target and nontarget organs (e.g. liver and spleen) and the calculation of bone selectivity indices. 79
Targeting the nanomedicine inside the bone is also a challenge in osteoporosis. The strategies of delivery include ligand–receptor interactions. Common ligands include BPs and tetracyclines, which exhibit high affinity toward bone mineral HAP and enhance nanocarrier accumulation. However, their excessively strong, nonspecific binding often limits penetration into the marrow beyond the bone surface. Alternatively, osteotropic peptides (e.g. Asp8) offer more specific targeting by binding to both mineral and cellular receptors. 80 Targeting the bone marrow microenvironment requires alternative strategies. Modulating the nanoparticle size (<200 nm) and surface charge (neutral/negative) facilitates extravasation through sinusoid pores and prolongs circulation. The ligands targeting specific bone cells enable cellular internalization. A more advanced approach involves the use of bone marrow-homing cells as natural carriers for nanoparticles, exploiting their innate tropism for precise delivery into the marrow (Table 1). 81
Advantages and disadvantages of various nanomedicines for osteoporosis treatment.

Limitations in the design of nanomedicine for osteoporosis treatment
Despite the emergence of numerous nanomedicines and nanotechnologies, numerous issues inhibit their use in clinical treatment. Some of these issues originate from the unique pathological characteristics of osteoporosis, while others are caused by special clinical prevention and treatment goals.
Spatiotemporal coupling of the pathological process of osteoporosis
The cause of osteoporosis is the disruption of the delicate dynamic balance between OCs and OBs.82,83 Therefore, treatment strategies focus on inhibiting bone resorption or promoting anabolism. However, it is easy to overlook that excessive inhibition of OCs or promotion of OBs may be unfavorable for establishing a new balance. In addition to destroying the normal bone structure, OCs precisely clear away old and morbid bone structures to promote OB generation in new bones. 84 The sequence of events always proceeds from bone resorption to bone formation, and these two processes are tightly coupled in space and time. 85 Due to their structural characteristics, nanomedicines have the advantage of acting as multifunctional delivery systems that can promote OBs and inhibit OCs simultaneously. Therefore, the study of the effect of nanomedicines on the coupling of OCs and OBs and the evaluation of the curative effect on osteoporosis are attracting increasing interest.
Insufficient preclinical research pharmacodynamic evaluation model
It is necessary to consider patient heterogeneity in the clinical treatment of all diseases. In women, osteoporosis is mainly associated with a drop in serum estrogen levels after menopause, while in men, it is mainly caused by aging. 18 A significant translational bottleneck lies in the limited predictive power of the existing preclinical animal models. However, most preclinical studies of nanomedicine still mainly use ovariectomized female rats as osteoporosis model animals, and differential treatment of osteoporosis in different sexes has not been explored. 15
The OVX rodent model, a standard model for studying postmenopausal osteoporosis, effectively models rapid estrogen-deficient bone loss but fails to replicate the slower age-related bone loss observed in older individuals of both sexes. Consequently, nanomedicine efficacy in OVX animals may not translate to age-related osteoporosis. This gap is exacerbated by the near absence of studies in aged or male models, overlooking key pathophysiological differences. 86 Furthermore, these models neglect clinical comorbidities such as diabetes or kidney disease, which alter the bone microenvironment and drug metabolism, potentially changing the efficacy and safety of a therapy. 87 The additional translational challenges include the following: (a) scalability and manufacturing hurdles in achieving reproducible, good manufacturing practice (GMP)–compliant production; (b) immunogenicity risks from repeated dosing, which are rarely assessed in long-term studies; and (c) complex regulatory pathways for defining standards and demonstrating bioequivalence of multicomponent nanomedicines.
Special requirements for controlled release in bone
Physical stimulation–responsive nanomedicines have attracted significant attention in the field of controlled drug release. 11 Bone is a highly hierarchical tissue with a dense cortical structure in the outer layer and loose cancellous bone in the inner layer. It is necessary to consider the shielding effect of the hard bone cortex upon physical stimulation when designing nanomedicine. For example, ultrasonic waves scatter as they pass through the bone cortex, causing energy attenuation. 88 The intrinsic tendency to attenuate sound waves at a given frequency can be expressed by its attenuation coefficient (expressed as α, in dB/mHz × cm). Cortical bone has a very high attenuation coefficient (α = 20), making it almost impermeable to ultrasound. 89 However, if the power of ultrasonic waves is arbitrarily increased, the heat generated by ultrasonic attenuation will also increase, resulting in damage to normal tissues. 90
The application of photothermal-mediated nanomedicine release is also influenced by the bone structure. A representative example is near-infrared (NIR) light. NIR light (wavelength between 750 and 1000 nm) exhibits good tissue penetration, high biocompatibility, and low scattering performance, which can mediate the release of temperature-sensitive materials by increasing local tissue temperature. 91 Although the tissue penetration depth of NIR light is much deeper than that of visible and ultraviolet light, it is inefficient when penetrating bone tissue. 92 Studies have shown that the efficiency of NIR light in penetrating bone is inversely correlated with bone thickness, with an 810-nm laser penetrating only 0.5% of a human skull and scalp tissue with a thickness of 1.9 cm. 93 Magnetic control is also a common means of mediating targeted drug release. 94 Although magnetically controlled methods have less effect on the penetration depth and can selectively accumulate magnetic nanomedicine locally, this controlled release method cannot distinguish between muscle and bone within the same magnetic field region.
Because of these structural properties of bone, it is necessary to identify a stimulation method that is more suitable for controlled drug release in bone, such as ESW. ESW is a bone-adaptive stimulation method that has been clinically applied in the treatment of orthopedic diseases such as nonunion and femoral head necrosis. 95 ESW demonstrates deep penetration, and focused shock waves can deliver energy to the depth of the femoral head without generating excess heat. 96 The above characteristics make ESW an excellent method for the local controlled release of nanomedicines. However, only a few studies have explored the application of ESW for controlling local release, and the more precise dose–effect relationship of ESW energy and frequency warrants further investigation.
Limitations of nanomedicines in osteoporosis
A primary concern is the long-term biocompatibility and potential off-target toxicity of nanomaterials. For example, despite the excellent safety profile of several constituents such as poly(lactic-co-glycolic acid) (PLGA) and chitosan in short-term studies, their chronic accumulation upon repeated administration, which is a requisite for osteoporosis management, remains poorly understood. Nondegradable or slowly degradable nanoparticles can accumulate in the mononuclear phagocyte system, particularly in the liver and spleen, potentially leading to chronic inflammation, granuloma formation, or organ dysfunction. 97 Furthermore, the bone marrow itself is a highly sensitive organ; a high, nonspecific payload of nanocarriers in the marrow cavity could inadvertently disrupt the delicate balance of hematopoiesis or the marrow microenvironment, potentially suppressing vital cellular functions. The potential for engineered nanomaterials to induce unintended immune responses or oxidative stress in bone cells also warrants careful, long-term investigation.
From a technical standpoint, the quest for effective bone-targeting strategies remains challenging. Although ligands such as tetracycline and BPs improve bone homing, they primarily target the mineralized bone matrix (HAP). This presents a delivery dilemma. Osteoporosis is a disease of the bone marrow microenvironment, where the key cellular players (OCs, OBs, and their precursors) reside. To reach these stromal and hematopoietic cells, nanoparticles are required to extravasate from the marrow sinusoids and navigate the marrow space, which, in osteoporosis, is often infiltrated with adipose tissue, potentially hindering diffusion. 98 Therefore, high mineral affinity does not automatically equate to high bioavailability at the precise cellular site of action within the marrow.
Moreover, the altered blood flow and increased marrow adiposity in osteoporotic bones may further impede the uniform distribution of nanomedicines. Furthermore, nanomedicines must be designed not only for potency but also for temporal control, possibly releasing anabolic and antiresorptive agents in a sequential manner to mimic the natural bone remodeling cycle.
Conclusion and prospects
Osteoporosis has received widespread attention because a large number of patients are afflicted by this condition and its serious consequences worldwide. Currently, the clinical treatment of osteoporosis mainly involves oral or intravenous drugs; however, traditional osteoporosis drugs face limitations such as insufficient targeting, low bioavailability, and potential side effects.
To overcome these problems, strategies such as the TOP–DOWN design have been applied to create various nanomedicines that can load and modify traditional drugs. Nanoscale design improves the delivery pathways of traditional drugs, such as local transdermal delivery, which provides a means to increase the local bone mineral density while avoiding issues such as first-pass effects, thereby increasing bioavailability. By coating with PEG, conjugating with TC, and other methods, the circulation time and bone-targeting ability of nanodrugs have also been improved. Furthermore, various nanomaterials have intrinsic abilities to treat osteoporosis themselves. For example, AuNPs can regulate the acidic microenvironment of OCs and inhibit excessive bone resorption. Fullerenol nanoparticles and IONPs can block OC fusion by inhibiting the actin ring structure of OCs.
The development of nanotechnology provides new prospects for the treatment of osteoporosis; nevertheless, potential issues in the design of nanomedicines should not be ignored. First, increased attention should be paid to the spatiotemporal coupling characteristics of OBs and OCs in terms of function and location. Because of this characteristic, separately improving the activity of OBs or arbitrarily inhibiting the activity of OCs is not satisfactory. Second, preclinical studies should not overlook patient heterogeneity in clinical treatment, which is currently highlighted by insufficient animal models of male osteoporosis. Moreover, because osteoporosis patients are primarily older adults, in addition to sex differences, future research should focus on complex models incorporating common underlying diseases of older adults and osteoporosis to better promote the use of nanomedicine in clinical treatment. Finally, the specific structure of bone presents a challenge for physical stimulus–responsive nanomedicines aimed at improving the local bone mineral density. The dense structure and depth of bone make most physical stimulation methods for nanomedicine intervention in the bone marrow cavity inefficient. Although ESW is promising for the efficient and safe mediation of intramedullary responsive nanomedicines owing to its excellent bone compatibility and conductivity, more precise dose–response relationships should be studied.
The future of nanotechnology in osteoporosis management is highly promising but depends on strategically overcoming the current challenges through innovative, multidisciplinary approaches. The key directions for the next decade include the following: 1. Nanomaterials can evolve into multifunctional “smart” systems. These may include nanoparticles with inherent therapeutic properties—such as osteogenic peptides, antioxidant cerium oxide, or Sr-substituted materials. Bioinspired strategies such as engineered exosomes or cell membrane–coated nanoparticles can improve targeting, biocompatibility, and immune evasion. 2. The field of controlled drug release will shift toward precision nanomedicine. Nanotheranostics that combine treatment with imaging (e.g. using gold or iron oxide nanoparticles) will enable the real-time monitoring of drug delivery and efficacy. Artificial intelligence and machine learning will aid in nanoparticle design, patient-specific pharmacokinetic prediction, and personalized dosing. 3. Combining nanomedicine with physical stimulation (e.g. shockwave-triggered release) and digital health tools (e.g. wearables for monitoring fracture risk) could enable holistic, site-specific treatment strategies.
In conclusion, the importance of clinical demands in the design of nanomedicine cannot be overstated. Therefore, a comprehensive understanding of the pathological process and clinical treatment of osteoporosis is essential for the design of nanomedicine. With a deep understanding of the pathological characteristics of diseases and clinical treatment needs, new and robust nanomedicines can be designed and developed to provide help for future clinical applications. The future of osteoporosis nanomedicine lies in intelligent, patient-tailored systems manufactured to high standards. Through collaboration across materials science, biology, medicine, and data science, nanotherapy may become a cornerstone of personalized osteoporosis care.
Footnotes
Acknowledgments
We would like to thank Editage (www.editage.cn) and Deepseek (![]() ) for English language editing.
) for English language editing.
Author contributions
Conceptualization, C.Y.; investigation, writing—original draft preparation, H.Q.; writing—review and editing, WK.L.; visualization and supervision, Y.H. All authors have read and agreed to the published version of the manuscript.
Data availability
This study did not generate or analyze any new data. Data sharing is not applicable to this article.
Declaration of conflicting interests
All the authors declare that they have no conflicts of interest related to this work.
Funding
This research was funded by the Fundamental Research Funds for the Central Universities (3332024146 and 3332024147).
