Abstract
Dysgerminomas are rare ovarian malignancies that primarily affect young women and can occur during pregnancy. Clinically, these tumors present with lower quadrant pain and adnexal mass, and laboratory values may include elevated beta human chorionic gonadotropin (β-hCG) and lactate dehydrogenase (LDH). These symptoms, along with the sonographic findings of a large, solid lobulated adnexal mass in young women, strongly suggest an ovarian dysgerminoma in the differential diagnosis. The majority of these tumors will be diagnosed at Stage IA and will respond well to fertility-sparing surgery. This case report demonstrates both the clinical and sonographic findings unique to this rare type of ovarian malignancy.
Introduction
An ovarian dysgerminoma is a rare, malignant tumor occurring in young women, accounting for 1% to 2% of all primary ovarian neoplasms. 1 The clinical presentation of pelvic pain, adnexal mass, and an elevated beta human chorionic gonadotropin (β-HCG) more commonly points to the possibility of an ectopic pregnancy rather than an ovarian malignancy. Sonographic findings of a solid, multi-lobulated adnexal mass with prominent internal vascularity in a young patient should raise the possible differential diagnosis of an ovarian dysgerminoma. 2 The majority of dysgerminomas are diagnosed early, Stage IA, and respond well to conservative fertility-sparing treatment of a unilateral salpingo-oophorectomy.3–6
Case Report
A female in her early 20s presented to her physician with complaints of right lower quadrant pain for approximately two weeks. The pain was accompanied with nausea, constipation, and urinary frequency. Menstrual cycles were regular with a last menstrual period of three weeks prior. On physical examination, the patient was thought to have a 16-week sized uterus. Quantitative β-HCG was 228 mIU/mL (normal, <3 mIU/mL).
Transabdominal and transvaginal sonography showed a normal-sized uterus measuring 7.02 cm in length. No gestational sac was identified within the endometrium. A large, heterogeneous, solid mass was detected anterior to the uterus measuring 14.8 × 12.8 × 8.7 cm (Figures 1 and 2). It did not appear to arise from the uterus. Evaluation with color and spectral Doppler demonstrated high velocity, low resistance arterial flow within the mass (Figure 3). The left ovary was normal in size and appearance. A small amount of pelvic fluid was present. The right ovary was not visualized; therefore, the mass was thought to have arisen from the right adnexal region, but definite origin was indeterminate. Additional laboratory studies were drawn at that time. The tumor marker β-HCG was elevated at 214 IU/L (normal, 0-5IU/L), as was the marker lactate dehydrogenase (LDH), significantly elevated to 1257 ng/mL (normal, 98-192 ng/mL). Alpha-fetoprotein (AFP) was normal. Based on the patient’s age, laboratory values, and sonographic findings, the most likely diagnosis was ovarian dysgerminoma. Two days later, the patient underwent exploratory laparotomy, right salpingo-oophorectomy with staging and right pelvic and peri-aortic lymph node dissection. The pathologic diagnosis was returned as dysgerminoma of the right ovary, Stage IA. Maximum gross dimension of the tumor was 15 cm.
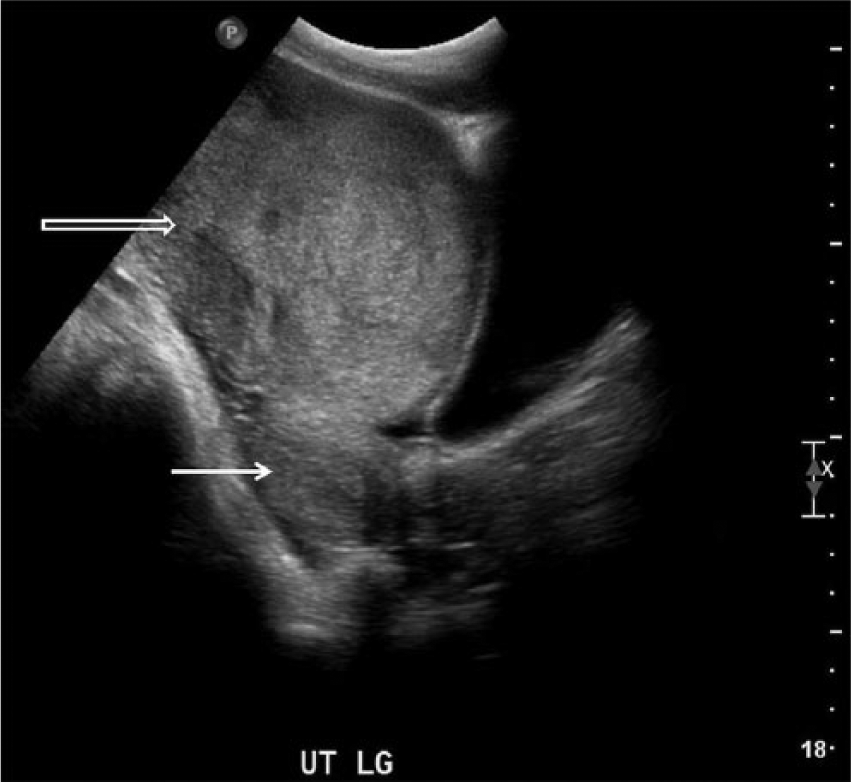
Transvaginal longitudinal gray-scale image of the pelvis showing a large solid heterogeneous mass (open arrow) arising anterior to the uterine fundus (solid arrow) but not appearing to arise from the uterus.
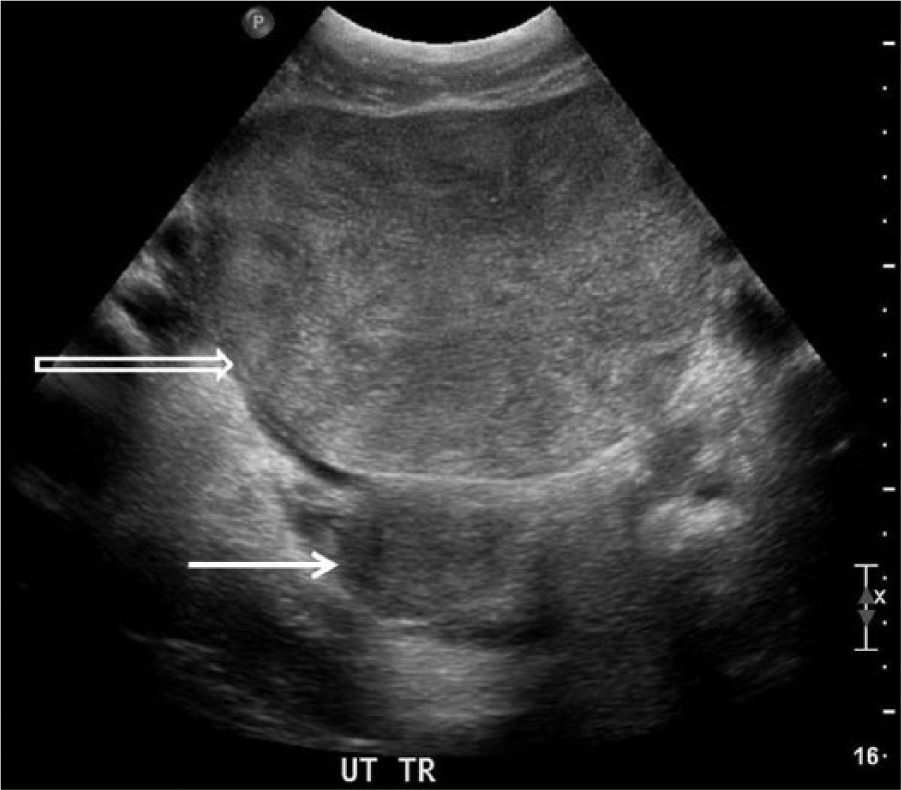
Transvaginal transverse gray-scale image of the pelvis showing the large solid heterogeneous mass (open arrow) arising anterior to the uterine fundus (solid arrow) but not appearing to arise from the uterus.
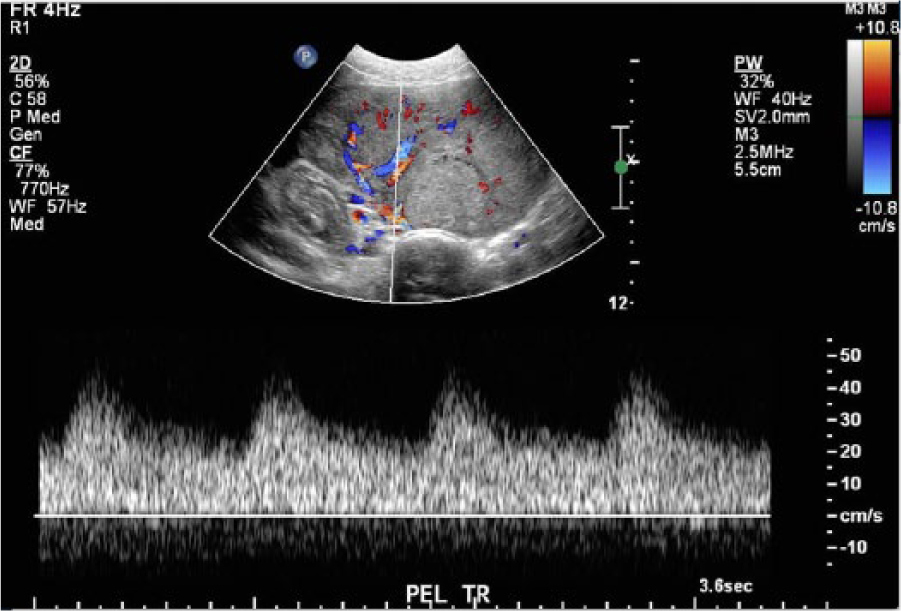
Color and spectral Doppler image showing a well-vascularized solid mass with flow in the hypoechoic septa characterized by hyperemic, low resistance arterial flow signals.
One month postoperatively, β-HCG and LDH tumor markers were within normal limits. Three months postoperatively, the patient returned to the clinic with severe nausea and vomiting. A pelvic sonogram revealed an intrauterine pregnancy at 5.9 weeks gestational age. The patient was referred to a high-risk obstetrics clinic for further treatment. No follow-up of her pregnancy or history of ovarian dysgerminoma has been documented in her notes.
Discussion
Malignant ovarian germ cell tumors account for less than 5% of all ovarian malignancies.1–11 Although rare, dysgerminomas are the most common malignant ovarian germ cell tumor, accounting for approximately 33% to 38% of all malignant ovarian germ cell tumors.1,4 Dysgerminomas originate from the primordial germ cells of the embryonic gonads and due to their origin are histologically equivalent to the testicular seminoma.1,2,4,5,7–11 Other non-dysgerminoma malignant germ cell tumors include immature teratomas, endodermal sinus (yolk sac) tumors, mixed germ cell tumors, embryonal carcinomas, polyembryomas, and choriocarcinomas.1,2,4,5,8,10
The most common clinical presentation of an ovarian dysgerminoma is a young patient with subacute, lower quadrant pain and a palpable pelvic mass by clinical examination.1–5,7,9,10,12 Symptom duration is short and associated with rapid tumor growth.1,4,9 Acute pain related to torsion or rupture is uncommon.1–3,5,10,11 Ninety percent of the tumors occur in patients less than 30 years of age2,3; the median age at diagnosis is 16 to 20 years.2,3,11 It is the most common ovarian malignancy in women younger than 20 years of age.4–7,11 With the age predilection for young women, 10% to 20% of dysgerminomas are found during pregnancy, 3 making it one of the most common ovarian malignancies to occur during pregnancy.9,10
Tumor markers may aid in the diagnosis and postoperative monitoring of ovarian dysgerminomas.1–5,7 Tumor marker elevation is dependent on the type of tissue components that comprise a mass. Elevation of β-HCG and serum LDH can be seen in association with a dysgerminoma, as documented in the case report. Beta human chorionic gonadotropin will be produced and levels elevated if syncytiotrophoblasts are present in the tumor, but less than 5% of patients will have increased HCG.1-5,7–10 Lactate dehydrogenase is an enzyme found in almost all body tissues, and levels rise with cell damage. Elevated LDH is noted in most cases of ovarian dysgerminomas.1–3,5,7,9,10 Alpha-fetoprotein values, which are elevated in some types of malignant ovarian germ cell tumors, are negative with dysgerminomas.1,3,5,10,11 Quantification of the tumor markers β-HCG and LDH at diagnosis allows for monitoring of tumor recurrence after treatment.
Sonography is the imaging modality of choice in evaluating young females with a possible adnexal mass.1,3,4 Both transabdominal and transvaginal imaging should be used if possible. Transabdominal sonography allows surveillance of the entire pelvis and adnexa. Transvaginal ultrasound offers a more detailed evaluation with its higher frequency and closer proximity to the tissue of interest, but it is limited by the smaller field of view. Sonographically, the dysgerminoma in this case study is consistent with the limited findings reported in the literature. Findings include a large, solid, multi-lobulated, heterogeneous mass separate from the uterus.1,2,4,8,9,11 Tumor borders are smooth and well defined.1,9 The internal echotexture exhibits hypoechoic, fibrous septae running between the lobules, creating a slightly irregular pattern.2,4,8,9 Small anechoic areas of hemorrhage or necrosis may be present. 9 Dysgerminomas are well vascularized, and color Doppler will demonstrate flow in the hypoechoic septa; spectral Doppler analysis displays a low resistance arterial waveform.1,2,4,8,9,11 Cul de sac fluid may also be present.9,11 Pertinent to the sonographic examination, it should be noted that dysgerminomas are bilateral in 10% to 15% of cases.1–3,7,9
Patient management is focused on treating the tumor while preserving fertility in these young patients. Tumor staging based on the International Federation of Obstetrics and Gynecology (FIGO) finds 65% to 75% of patients are classified with Stage IA dysgerminoma with disease limited to one ovary and with no metastatic involvement at the time of diagnosis.2–6,7,10 The standard treatment for Stage IA dysgerminoma is a fertility-preserving surgical approach: unilateral salpingo-oophorectomy with staging biopsies and preservation of the uterus and contralateral ovary.1–3,5–7,9–11 The literature varies on whether a biopsy should be done of a normal appearing contralateral ovary. It may detect occult disease, but it may also affect future fertility due to adhesions or ovarian failure.1,3,5,9,10 Five-year survival rate for Stage 1A dysgerminoma is greater than 90% to 95%.1–7,10 Stage 1A disease treated surgically does require close clinical follow-up and monitoring of relevant laboratory values.1,3,5 Recurrence of Stage 1A dysgerminomas is rare (10%-15%),1,3,9 typically occurs during the first two years after diagnosis, 2,3,5,7,9,10 and responds well to chemotherapy.1-3,5–7,10,11,13
Conclusion
Ovarian dysgerminomas should be included in the differential diagnosis for a young female who presents with non-acute lower quadrant pain, palpable pelvic mass, and elevated β-HCG and LDH. Supporting sonographic findings include a solid, heterogeneous, lobulated adnexal mass with low resistance internal blood flow. The majority of tumors are Stage IA at the time of diagnosis and can be conservatively treated with a unilateral salpingo-oophorectomy to preserve fertility. This case study demonstrated the unique characteristics of this rare type of malignant ovarian germ cell tumor, including age of presentation, symptoms, elevated lab values, and sonographic characteristics.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
