Abstract
Extranodal marginal zone lymphomas comprise approximately 5% of all non-Hodgkin lymphomas. These tumors may arise in any epithelial tissue, including the bowel lining. A report is presented of a patient who presented to the emergency department with epigastric abdominal pain and vomiting. An abdominal sonogram showed a 3.1 cm dilated loop of small bowel near midline just above the umbilicus as well as an abrupt transition zone to normal bowel suggestive of small bowel obstruction, confirmed by subsequent computed tomography. Small bowel resection found an intraluminal tumor, and biopsy showed extranodal marginal zone lymphoma of the mucosa-associated lymphoid tissue (MALT lymphoma).
Introduction
Mucosa-associated lymphoid tissue (MALT) lymphoma is a common subtype of non-Hodgkin lymphoma, which is characterized by local recurrence and has the potential for systemic spread. It frequently involves the stomach, although it may affect any mucosal site (thyroid, breast, skin, etc.), and it is therefore amenable to detection via sonography.1,2 The American Joint Committee on Cancer proposes the further use of ultrasound endoscopy for TNM staging of MALT lymphomas. 3 The TNM staging system is based on the size and/or extent (reach) of the primary tumor (T), whether cancer cells have spread to nearby (regional) lymph nodes (N), and whether metastasis (M), or the spread of the cancer to other parts of the body, has occurred. Presented in the following is a case of a MALT lymphoma within the small bowel discovered by ultrasonography. The sonographic and computed tomography (CT) findings are described.
Case Report
A previously healthy 81-year-old female presented to the emergency room with intermittent and increasing epigastric pain; she had visited the emergency department two months previously for a similar episode. Computed tomography done at that initial visit had shown a partial small bowel obstruction that was felt to be resolved. She had no personal history of cancer; her family history indicated one sister with colon cancer and one brother with lung cancer. At the time of this emergency department evaluation, her white blood cell count was normal, hemoglobin stable, and other than hypokalemia, with a potassium of 3.4, her laboratory values were unremarkable. Upon asking the patient where she felt the worst pain, she pointed to one location just cephalad to the umbilicus. Abdominal sonography was ordered and done using a Philips IU22 ultrasound system (Philips Healthcare Ultrasound, Bothel, Washington, USA) with a C9-4 curved array probe. A dilated 3.1 cm loop of bowel was seen with a transition point to the right showing a hypoechoic wall (Figures 1 and 2), then normal collapsed bowel more distally, highly suggestive of recurrent small bowel obstruction. A subsequent CT scan was done within three hours of the sonogram to confirm the obstruction, with the transition point seen in the anterior abdomen just posterior to the umbilicus (Figure 3). A small bowel resection with primary anastomoses was done; during the procedure, an intraluminal tumor with a small area of fibrosis on the antimesenteric border was seen with no adenopathy. This was sent to pathology and was determined to be MALT lymphoma. The patient was informed of the diagnosis and referred to hematology-oncology for follow-up. She underwent positron emission tomography (PET) scanning, which showed diffuse distant involvement of the neck, spine, and pelvis. She was offered further staging and chemotherapy but ultimately declined.
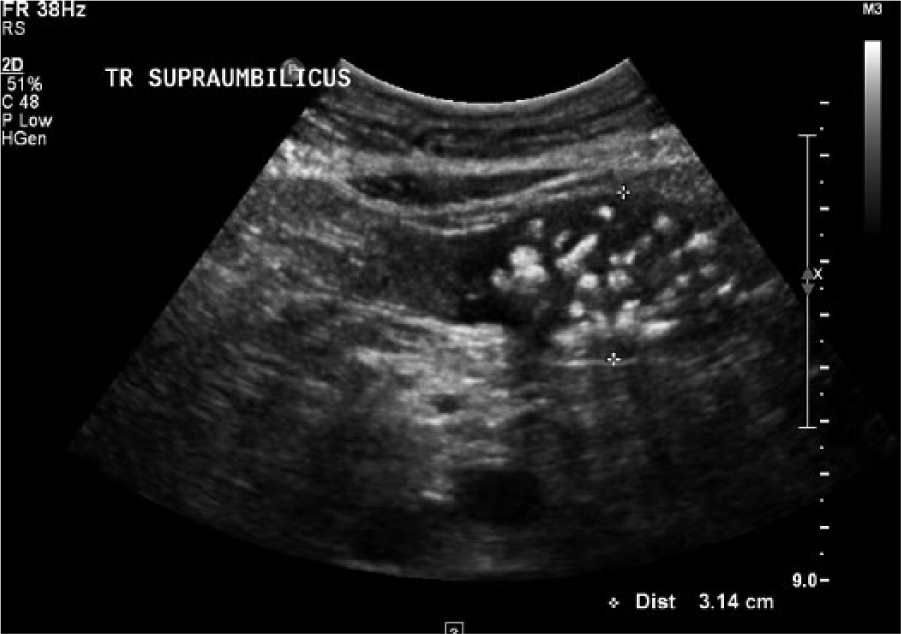
Transverse abdominal gray-scale image at the approximate level of the umbilicus showing a dilated loop of bowel and adjacent hypoechoic bowel wall.
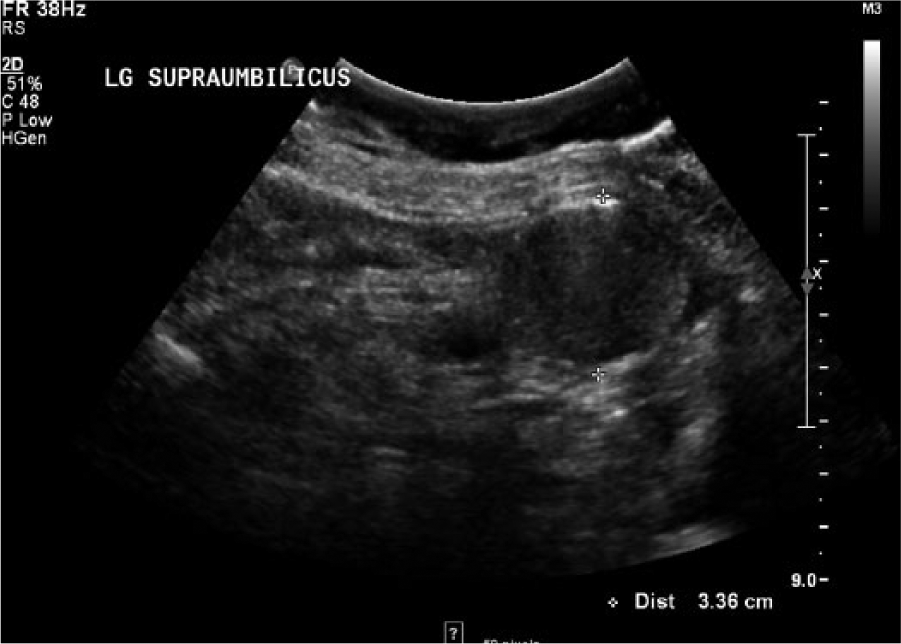
Longitudinal abdominal gray-scale image at the approximate level of the umbilicus showing the dilated loop of bowel.
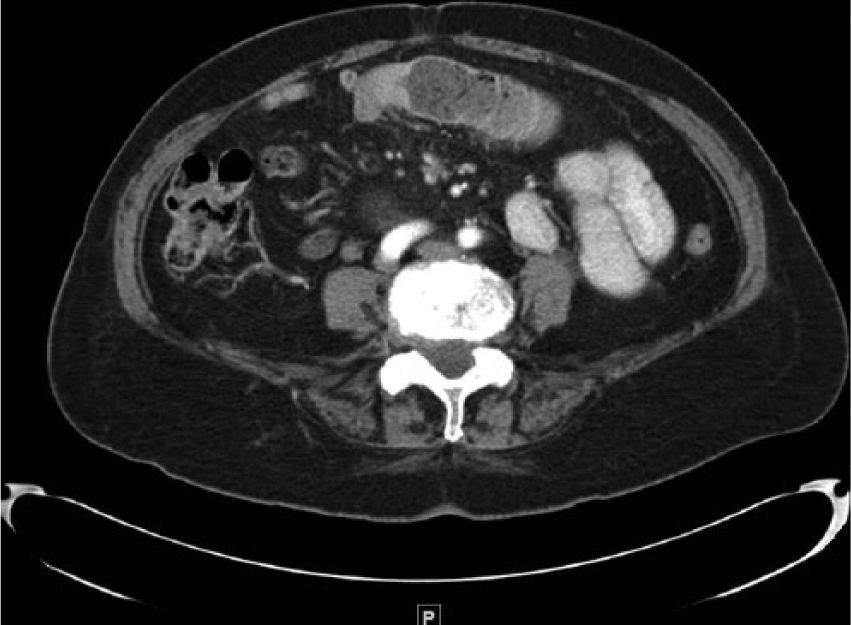
Corresponding cross-sectional abdominal computed tomographic scan showing the dilated bowel at the level of the umbilicus with the transition point slightly to the patient’s right.
Discussion
Mucosa-associated lymphoid tissue lymphoma is a distinct neoplasm comprising 7.6% of non-Hodgkin lymphomas, 1 and it can uniquely involve tissues that are not primary organs of the immune system. 2 The two main types, characterized by their site of involvement, include the acquired type and the native type. 3 The acquired type is the more common of the two and develops in the setting of chronic inflammation from either infection or autoimmune disease. These are most commonly located in the stomach; 72% to 98% of gastric MALTomas are associated with chronic Helicobacter pylori gastritis. 4 Tumors can also be found in regions affected by autoimmune disease; examples include the lacrimal gland in Sjogren’s syndrome and the thyroid gland in Hashimoto’s thyroiditis. 2 The pathogenesis is thought to be attributed to chronic activation of lymphocytes in which after numerous replications a mutated clone eventually arises and replaces the normal population. With additional mutations, the tumor eventually becomes capable of spread beyond its original site. 3 The native type of MALToma arises within physiologic lymphoid tissue present in the gut. Several associated chromosomal translocations have been identified, including t(11;18) and t(14;18). These mutations cause activation of NF-kappa B, a potent stimulator of B-cell survival and tumor formation. 3
Mucosa-associated lymphoid tissue lymphomas involving the gut mucosa frequently present clinically as nonspecific epigastric pain and may be visualized via sonography, CT, or endoscopy. 2 After histopathologic diagnosis, staging should be performed. This usually includes some combination of endoscopic ultrasound (to determine tumor depth of invasion and nodal involvement), CT, positron emission tomography (PET)-CT, bone marrow biopsy, and testing for active H. pylori infection, if applicable. 2 Therapy is guided by tumor grade and the original site of involvement (gastric vs. nongastric). Gastric MALToma often responds to H. pylori eradication therapy alone; regression was seen in more than half of patients treated with antibiotics only. 5 Radiotherapy may be given in cases of inadequate response. 5 Limited stage nongastric MALT lymphoma, defined as a tumor involving a single extralymphatic site or a lymph node extending into contiguous extralymphatic tissue (stage I-II), may be treated with close monitoring alone or local radiotherapy. Surgery is reserved for tumor in areas that are not conducive to radiation therapy. With radiation therapy, rates of complete response and local control are greater than 90%. 6 Advanced nongastric MALToma is defined as an extranodal tumor plus multiple sites of nodal involvement (stage III-IV). It is treated with a chemo-immunotherapy regimen used for follicular lymphoma such as CHOP (C: Cyclophosphamide, H: Hydroxy doxorubicin, O: Oncovin, and P: Prednisone) plus rituximab. Five-year survival rate of higher-grade lesions is only approximately 50%; 10-year survival rate is approximately 20%. 7
The use of sonography to identify tumors within the breast, thyroid, prostate, and other organs has already been well established. The use of sonography to define tumors of the alimentary tract is less well understood. A study by Goerg et al. 8 suggests that sonography is useful in the staging and detection of patterns of involvement of gastrointestinal tumors. They found varying degrees of hypoechoic wall thickening in all 54 patients, with circumferential involvement of the bowel wall in the great majority (39/54, 72%). Four patients had bulky tumors, two had nodular extraluminal spread, eight were seen to have segmental infiltration, and a single patient was seen to have involvement of the mucosa.
Schwerk et al. 9 described 19 patients with gastrointestinal tumors, including colon tumors, that were diagnosed by sonography, suggesting that ultrasonographic examination may be a valuable screening tool as well. They also noted that sonography could identify mesenteric and/or liver metastases. However, both articles were in agreement that a normal abdominal sonogram does not exclude the possibility of malignancy, particularly for early stage lesions or tumors in regions that are difficult to visualize via ultrasonography.8,9 This limitation should diminish as advances in ultrasound technology continue. Computed tomography should be utilized in cases of high suspicion with a negative sonography result. Because of the small patient numbers in published series, the overall sensitivity and specificity for the use of sonography in the detection of abdominal lymphomas is not yet known. The patterns seen on both sonography and CT correlate with the type of lymphoma present. Gastrointestinal lymphoma may appear as thickened bowel segments, a superficial spreading lesion, a bulky mass, segments of narrowed bowel lumen, or mesenteric nodal masses. 10 After initial detection by CT or sonography, the definitive diagnosis of MALTomas, like other lymphomas, requires a tissue diagnosis with histopathological examination.
With appropriate clinical correlation, ultrasonography provided accurate and expedient information that guided the appropriate next steps in patient management for the case presented. Sonography led to prompt investigation into the source of the bowel obstruction, which was further elucidated by CT and managed surgically. It also is appropriate to utilize sonography whenever possible in an effort to minimize radiation exposure in patient evaluation, given that most cancer patients receive follow-up CT scans for surgical preparation, tumor staging, and monitoring for disease recurrence.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
