Abstract
Four echocardiographic markers—left ventricular ejection fraction (LVEF), the ratio of the left atrial diameter to the aortic root diameter (LA/Ao), the ratio of the patent ductus arteriosus (PDA) diameter to the left pulmonary arterial diameter (PDA/LPA), and the patent foramen ovale (PFO) diameter—were explored to predict a PDA in 221 premature neonates (53% male). There were 182 cases with PDA (mean diameter = 2.49 mm); 39 control subjects were observed with a closed ductus arteriosus. Although 104 of the cases (57.1 %) were diagnosed when 0 to 3 days old (median age = 1 day), 78 (42.9%) were diagnosed when 4 to 86 days old (median age = 13 days). Birth weights of the neonates ranged from 0.5 to 4.8 kg (mean = 1.68 kg). After controlling for age and birth weight, the mean LVEF (%) was not significantly different in the cases versus controls (P > .05); the mean LA/Ao and mean PFO diameter were significantly larger (P < .001) in the cases. The mean PDA/LPA was consistently ≥ .7 in the cases. The sensitivity of the LA/Ao ratio as a positive test for PDA (cutoff = 1.5) varied with age but not birth weight. When the neonatal age was 0 to 3 days, the sensitivity was 56% with a specificity of 88%; when the neonatal age was > 3 days, the sensitivity of LA/Ao was 82% with a specificity of 63%. The sensitivity of PFO (cutoff = 3.0 mm) was 61% with a specificity of 48%, irrespective of age or birth weight. These data suggest that LVEF is not a significant predictor of the presence of PDA, but a combination of the LA/Ao ratio, PDA/LPA ratio, and PFO diameter is predictive of the presence of PDA.
Keywords
Introduction
The ductus arteriosus (DA) is a blood vessel in the developing fetus connecting the pulmonary artery to the proximal descending aorta. It permits the output from the right ventricle to be diverted away from the fetal pulmonary circulation and bypass the fluid-filled nonfunctioning lungs. In normal full-term infants, an abrupt contraction causes the functional closure of the DA to occur spontaneously within 12 to 24 hours of birth. 1 Patent ductus arteriosus (PDA) is a congenital disorder in which the infant’s DA does not close spontaneously after birth but remains patent (open). In full-term infants, the incidence of PDA is approximately 1 in 2000 births. 2 Patent ductus arteriosus is more frequent in infants with a gestational age of less than 28 weeks due to physiologic factors associated with prematurity. 3
For more than 30 years, the diagnosis of PDA has traditionally been confirmed by two-dimensional (2D) echocardiography with Doppler assessment to assess cardiac geometry and function. Typically, direct PDA imaging with color flow and pulsed wave Doppler analysis reveals a measurable connection between the junction of the main and left pulmonary arteries and the aorta, just below and opposite the left subclavian artery, resulting in irregular transmission of blood between the aorta and the pulmonary artery. Even a very small PDA can be detected by a color flow signal entering the pulmonary artery. 4 An example of an echocardiographic image (2D gray-scale and color Doppler imaging) of a PDA is presented in Figure 1.
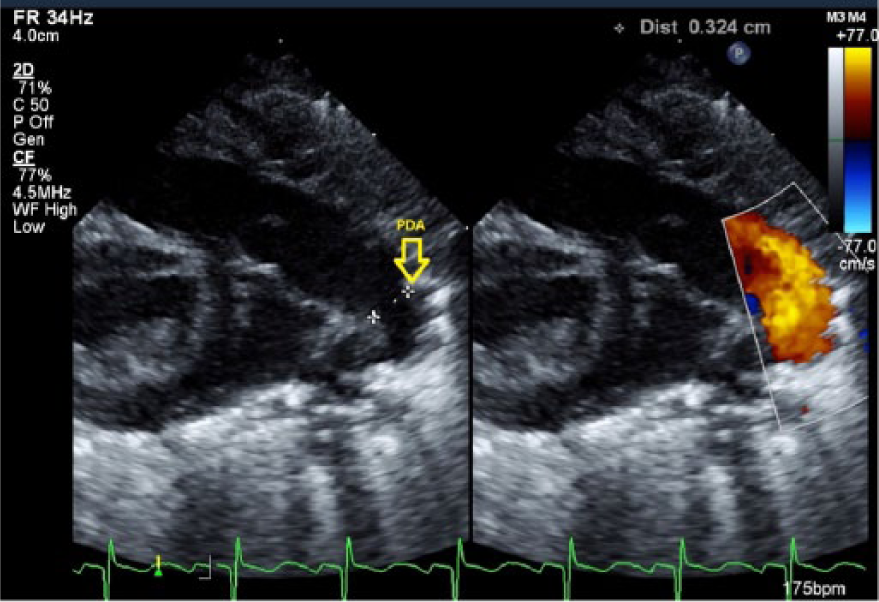
Side-by-side gray-scale and color Doppler echocardiographic image of a patent ductus arteriosus.
The clinical management of PDA in preterm infants is a controversial topic, and several major issues have not yet been clarified. To date, there are no universally accepted guidelines for the diagnosis of a hemodynamically significant PDA, and there is no objective system of reliable markers to predict the need for therapeutic closure of a PDA.5,6 The purpose of this case-control study was to explore the potential of using four markers to predict the presence versus absence of PDA in premature neonates. The markers were (a) LVEF: the left ventricular ejection fraction, the percentage of blood leaving the left ventricle within each cardiac cycle; (b) LA/Ao: the ratio of the left atrial diameter (LA) to the aortic root diameter (Ao); (c) PDA/LPA: the ratio of the diameter of the PDA to the diameter of the left pulmonary arterial diameter (LPA); and (d) PFO: the diameter of the patent foramen ovale (mm).
The rationale for using LVEF was that the presence of a PDA causes left-to-right ductal shunting, resulting in pulmonary overcirculation and left heart volume overload. 7 The magnitude of shunting depends on the flow resistance of the PDA, related to its length, diameter, shape, and elasticity. 8 The LVEF of a healthy fetus is normally about 65%. 9 Consequently, we hypothesized that excessive cardiac output (LVEF ≥ 66%) may potentially predict the presence of PDA.
The ratio of the left atrial diameter to the aortic root diameter was historically one of the first markers used to diagnose PDA, because its measurement requires only basic ultrasound technology. 10 When left heart volume overload dilates the left atrium, the aortic root is relatively unaffected; consequently, an elevated LA/Ao may predict a clinically significant PDA. Older studies disputed the use of LA/Ao as a positive test for PDA, apparently because the gold standards used to estimate sensitivity and specificity were inaccurate.11,12 More recent research, using Doppler echocardiography assessment as the gold standard, has confirmed the utility of LA/Ao. A study to re-evaluate the LA/Ao ratio as a marker of PDA reported that among 56 preterm infants (mean gestational age 27 weeks, weight < 1.5 kg), the mean LA/Ao was 1.17 and 1.21 when the DA was respectively closed or restricted, compared to 1.61 when the DA was wide open. 13 An LA/Ao of 1.5 was proposed as a cutoff for diagnosis of PDA after the first postnatal day, providing a sensitivity of 88% with a specificity of 95%. (However, it was recommended that this cutoff not be used on the first day, due to a reduced sensitivity of only 79%.) Based on these findings, we hypothesized that an LA/Ao cutoff of 1.5 could predict the presence of a PDA in older neonates.
The rationale for using PDA/LPA as a marker was that if the PDA is large, relative to the LPA, then an excessive blood volume may flow from the lung, overloading the left atrium, potentially marking the effect of PDA; however, to our knowledge, no previous studies have proposed a cutoff for the PDA/LPA ratio. We hypothesized that the LPA should be more than twice the size of the PDA (cutoff = 0.5) to predict the presence of a PDA. The foramen ovale is a hole in the atrial septum of the fetal heart that allows oxygenated blood to flow from the right to left atrium. Increased blood pressure in the left atrium usually forces the hole to close within 36 hours of birth; however, a PFO occurs if the atrial septum fails to close properly, allowing blood to flow from the left to the right atrium. Patent foramen ovales vary considerably in diameter from 1 to 19 mm. 14 To our knowledge, no previous studies have related the presence or sizes of PFOs to PDAs. We hypothesized that a relatively small PFO (cutoff = 3.0 mm) could potentially predict the presence of a PDA. Birth weight and age were considered as confounders of the four markers, and for purposes of statistical analysis, they needed to be controlled because (a) a significantly higher incidence of PDA (40%) has been reported to occur among premature infants with extremely low birth weights (< 1 kg) compared to a 20% incidence among those with higher birth weights, 15 and (b) the presence of PDA beyond the third day of life is considered to be abnormal, irrespective of gestational age. 16
Materials and Methods
A retrospective data analysis of pediatric echocardiograms performed from 2009 to 2014 in the neonatal intensive care unit of a western US regional medical center was completed. A total of 221 cases with a patent ductus arteriosus were reviewed. Hospital institutional review board approval was obtained prior to the data collection. All patients were monitored for PDA closure with repeated echocardiograms according to clinical findings (continuous machinery murmur, systolic murmur, heart murmur, pulmonary hypertension) or routine follow-up. Some patients with PDAs were under medical treatment and others underwent PDA ligation.
The data were blindly collected in a retrospective manner. All pediatric echocardiograms were performed using a Philips IE33 ultrasound machine (Philips Ultrasound, Bothel, Washington, USA) equipped with a phased sector array S12-4 MHz transducer. All echocardiograms were performed by ARDMS-credentialed sonographers, and all echocardiographic data were uploaded from the machine and stored on the hospital electronic data server.
Following hospital imaging protocol, the following measurements were obtained as part of the pediatric echocardiogram: The patent ductus arteriosus diameter was measured on 2D gray-scale imaging, from parasternal long, short, and suprasternal notch views. The PDA diameter recorded was the measurement closest to the aortic end of the PDA. The LPA was measured in the high parasternal short view. The ratio of the left atrial diameter to the aortic root diameter was obtained from the parasternal short axis view. Patent foramen ovale diameter was measured in subcostal long and short views; the recorded measurement was the largest measurement of the two views. The LVEF was determined from M-mode analysis in the parasternal short axis view. Only the PDA/LPA ratio was retrospectively calculated.
All clinical information was retrieved from the electronic medical record including age, sex, and ethnicity. The weight was recorded at the time of the echocardiographic study. To de-identify data, a key was made for all patient health information and a numerical code was generated accordingly for each patient.
Patients
The characteristics of the resulting purposive sample are outlined in Table 1. The total sample consisted of 221 premature infants, with an approximately equal proportion of males (53%) and females (47%). The majority (182, 82.3%) were diagnosed with PDA (diameter = 1.0-9.0 mm; mean = 2.49 mm; 95% confidence interval [CI], 2.32-2.65). The remaining patients (39, 17.7%) were observed to have a completely closed DA and were classified as controls. Although 104 of the cases (57.1%) were diagnosed with PDA when 0 to 3 days old (median age = 1 day), 78 patients (42.8%) were diagnosed when 4 to 86 days old (median age = 13 days). The birth weight of the neonates ranged from 0.5 to 4.8 kg (mean = 1.68 kg; 95% CI, 1.53-1.84). The majority of cases (116, 63.7%) had a very low birth weight (≤ 1 kg). Nearly half of the patients (94, 42.5%) consisted of white/non-Hispanic cases, with about one third (67, 30.3%) Hispanic cases. The remainder were Asian or black/non-Hispanic.
Characteristics of the Patients with (Case) and without (Control) Patent Ductus Arteriosus. a
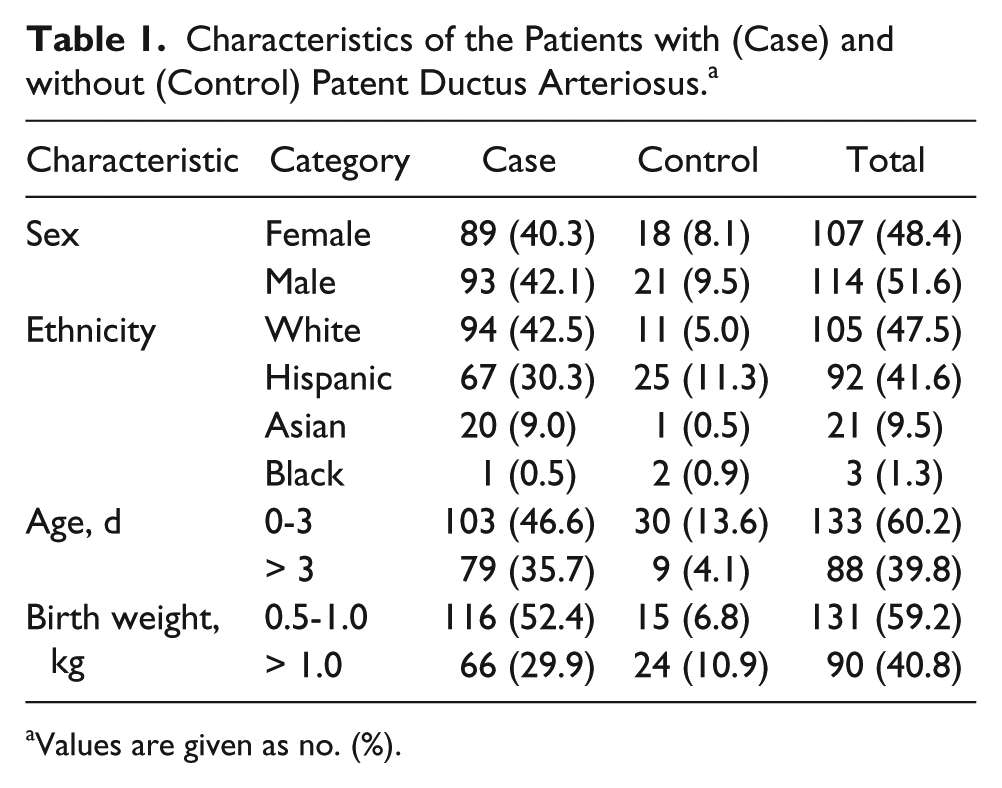
Values are given as no. (%).
Statistical Analysis
Statistical analysis was conducted using IBM SPSS version 20.0 (IBM Software Support Group, Rochester, New York, USA). The mean LVEF (%), LA/Ao, PDA/LPA, and PFO diameter (mm) for the cases versus the controls, controlling for birth weight and age, were compared using factorial analysis of variance (ANOVA), assuming homogeneity of variance and residual normality. Sensitivity and specificity were calculated based on the hypothesized cutoff values for LVEF (66%), LA/Ao (1.5), PDA/LPA (0.5), and PFO (3.0 mm), assuming that diagnosis of PDA with 2D echocardiography was the gold standard.
Results
LVEF
The estimates of LVEF (mean ± 95% CI) classified by cases versus controls, and controlling for birth weight and age, are compared in Figure 2. Analysis of variance indicated that the presence of a PDA had no significant effect on LVEF (F = 0.85, P = .357). The mean LVEF did not differ significantly between the cases (mean = 66.2%; 95% CI, 65.1-67.4) and the controls (mean = 65.6%; 95% CI, 63.5-67.6). This finding, and the relatively low sensitivity (61%) and specificity (48%) using a 66% cutoff as a positive test for the presence of PDA (Table 2), indicated that the LVEF was not a useful marker to predict the presence of a PDA.
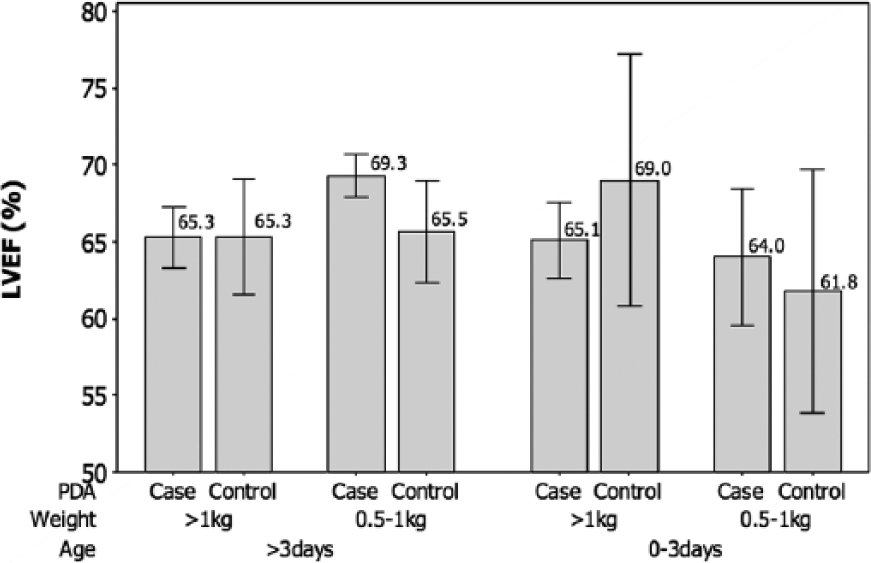
Graphic presentation of mean left ventricular ejection fraction (LVEF) ± 95% confidence intervals in cases of patent ductus arteriosus (PDA) versus controls, controlling for weight and age [PDA (F = 0.85, P = .357); weight (F = 2.23, P = .137); age (F = 1.73, P = .190)].
Sensitivity and Specificity for LVEF as a Marker of PDA.
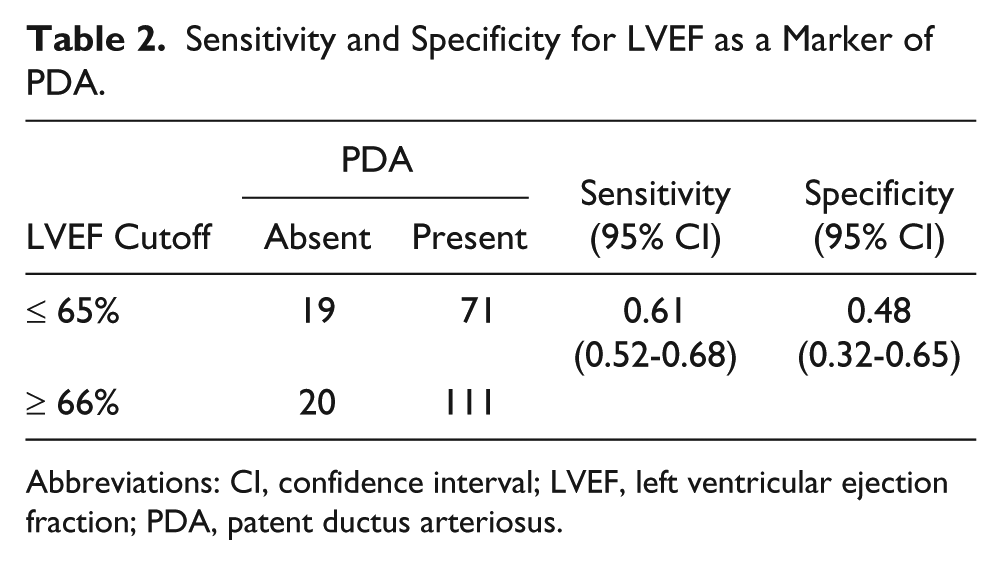
Abbreviations: CI, confidence interval; LVEF, left ventricular ejection fraction; PDA, patent ductus arteriosus.
LA/Ao
The estimates of LA/Ao (mean ± 95% CI) classified by cases versus controls, controlling for birth weight and age, are compared in Figure 3. Analysis of variance indicated that the presence of PDA (F = 30.48, P < .001) and the age of the infant (F = 13.38, P < .001) had a significant effect on the mean LA/Ao; however, birth weight had no significant effect (F = .001, P = .974). The mean LA/Ao was significantly higher (F = 30.48, P < .001) in cases (mean = 1.68; 95% CI, 1.62-1.73) compared to controls (mean = 1.36; 95% CI, 1.27-1.46).
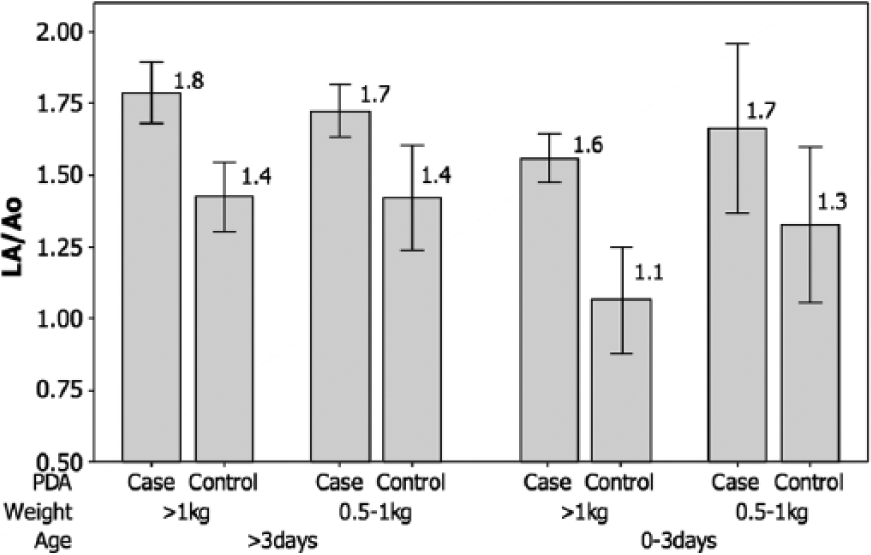
Graphic presentation of mean ratio of left atrial diameter to aortic root diameter (LA/Ao) ± 95% confidence intervals in cases of patent ductus arteriosus (PDA) versus controls, controlling for weight and age [PDA (F = 30.48, P < .001); weight (F = 0.001, P = .974); age (F = 13.38, P < .001)].
The cross-tabulation and sensitivity and specificity estimates for LA/Ao, classified by age, are presented in Table 3. When the neonatal age was > 3 days, the sensitivity of LA/Ao using 1.5 cutoff as a positive test for the presence of PDA was high (82%); however, the specificity was low (63%). The sensitivity was lower (56%) when the age was 0 to 3 days but the specificity was higher (88%). These findings were consistent with previous studies indicating that LA/Ao is a useful marker to predict a clinically significant PDA, particularly in older neonates.
Sensitivity and Specificity for LA/Ao as a Marker of PDA, Stratified by Age.
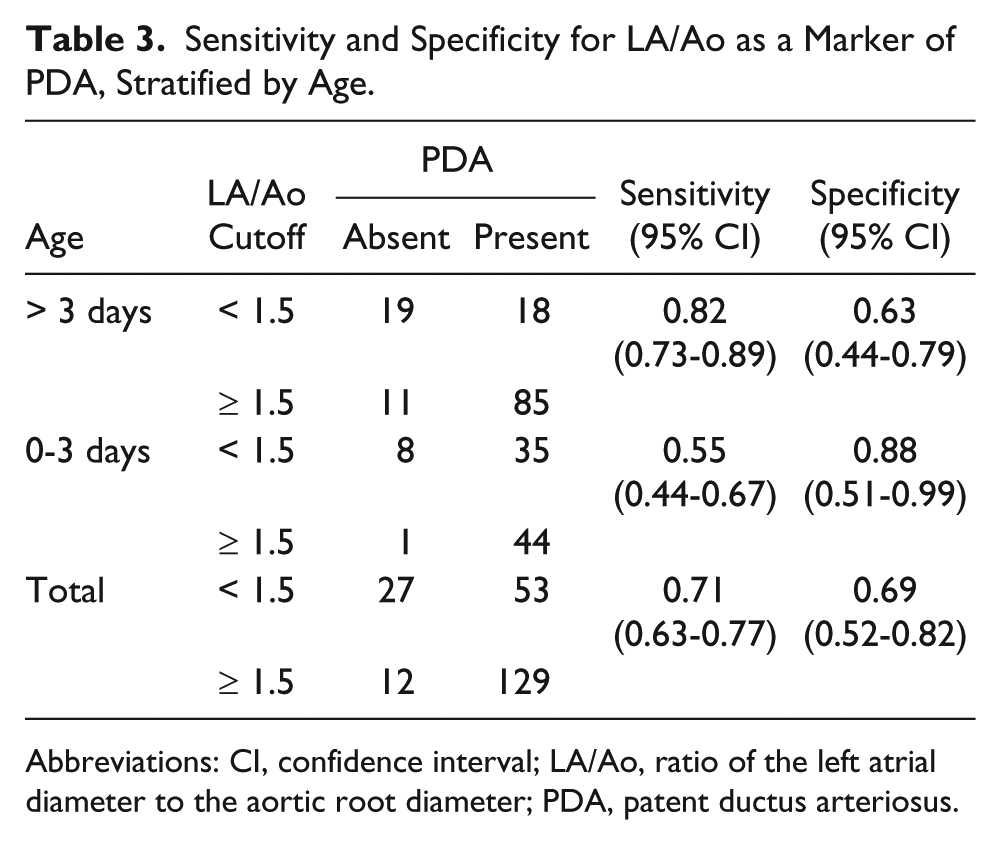
Abbreviations: CI, confidence interval; LA/Ao, ratio of the left atrial diameter to the aortic root diameter; PDA, patent ductus arteriosus.
PDA/LPA
The estimates of PDA/LPA (mean ± 95% CI) in the cases, controlling for birth weight and age, are compared in Figure 4. The PDA/LPA was consistently ≥ 0.7 among the cases (mean = 0.73; 95% CI, 0.68-0.77) but was zero in the controls (because the DA was closed). Analysis of variance indicated that birth weight (F = 0.218, P = .641) and age (F = 0.963, P = .327) had no significant effect on PDA/LPA. Sensitivity and specificity estimates for PDA/LPA were not justified, because of the zero frequencies in the cross-tabulation. These findings were consistent with the hypothesis that a PDA/LPA > 0.5 is associated with the presence of a PDA irrespective of age or birth weight.
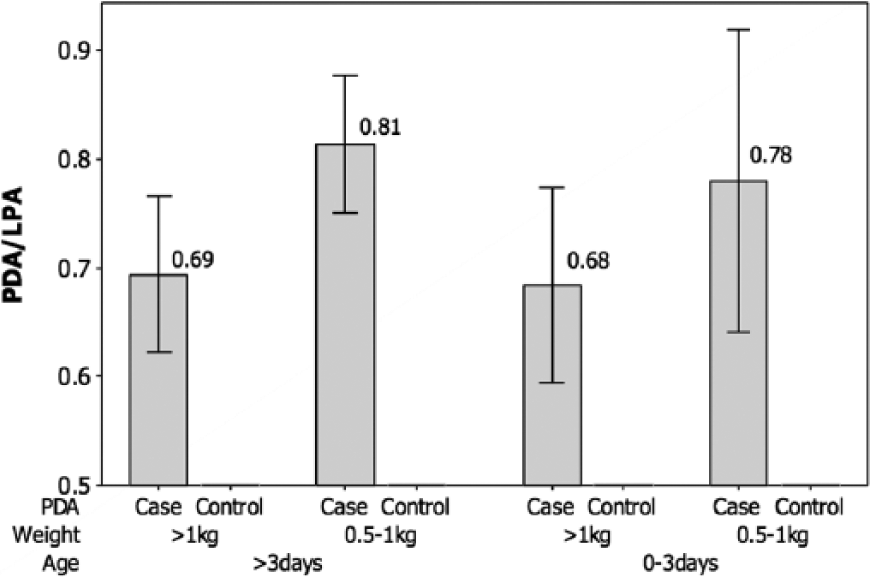
Graphic presentation of mean ratio of the diameter of the patent ductus arteriosus to the diameter of the left pulmonary artery (PDA/LPA) ± 95% confidence intervals in cases versus controls, controlling for weight and age [weight (F = 0.218, P = .641); age (F = 0.963, P = .327)].
PFO
The estimates of mean PFO diameter (mm) ± 95% CI classified by cases versus controls, controlling for birth weight and age, are compared in Figure 5. Analysis of variance indicated that the mean PFO diameter was significantly higher (F = 28.89, P < .001) in the cases (mean = 2.94; 95% CI, 2.75-3.12) compared to the controls (mean = 1.85; 95% CI, 1.61-2.10), but birth weight (F = 1.79, P = .183) and age (F = 2.35, P = .127) had no significant effect on the PFO diameter. The cross-tabulation in Table 4 indicated a relatively low sensitivity (61%) and specificity (48%), assuming a 3.0 mm cutoff. These findings were consistent with the hypothesis that a relatively small PFO is indicative of the presence of a PDA, irrespective of age or birth weight.
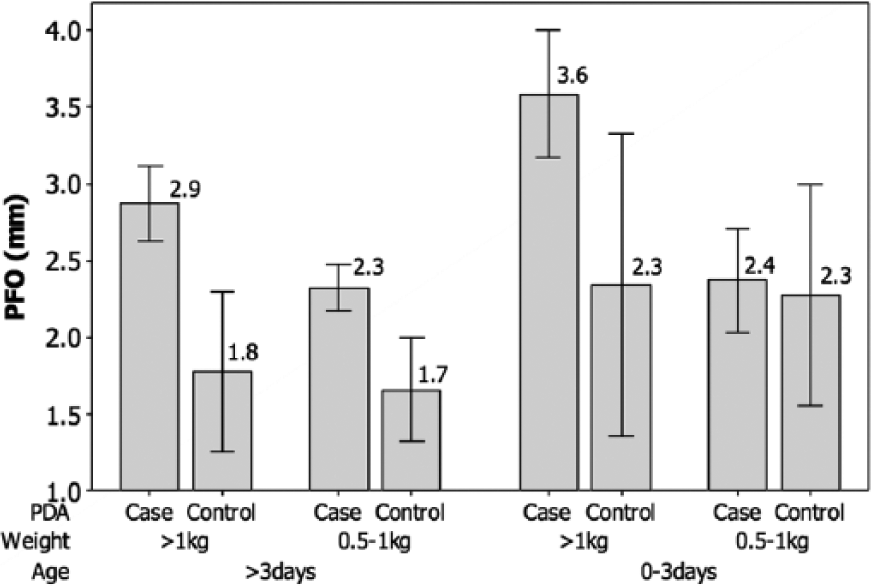
Graphic presentation of mean diameter (mm) of the patent foramen ovale (PFO) ± 95% confidence intervals in cases of patent ductus arteriosus (PDA) versus controls, controlling for weight and age [PDA (F = 28.89, P < .001); weight (F = 1.79, P = .183); age (F = 2.35, P = .127)].
Sensitivity and Specificity for PFO Diameter as a Marker of PDA.
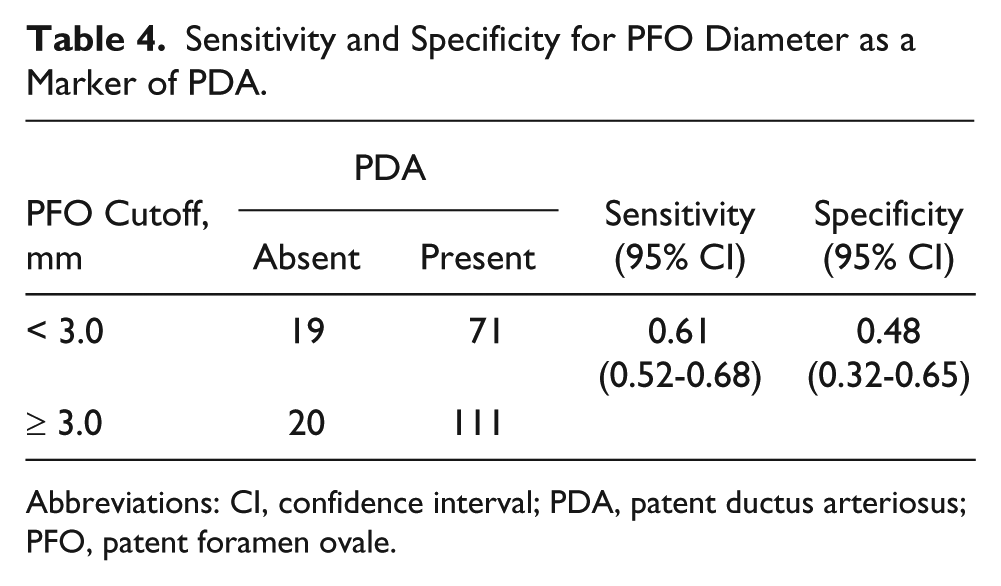
Abbreviations: CI, confidence interval; PDA, patent ductus arteriosus; PFO, patent foramen ovale.
Discussion
The findings above indicate that LVEF was not a significant predictor of the presence of PDA. Even with an abnormal ejection fraction, overall heart function may not be compromised, and excessive cardiac output does not necessarily correlate with LVEF. Previous studies on adults have also indicated that a normal ejection fraction does not always imply freedom from risk of cardiac pathology. 17
Our findings were consistent with other studies suggesting that two important echocardiographic markers have utility for predicting a PDA in premature neonates, specifically (a) a LA/Ao ratio of about 1.513,18,19 and (b) a PDA diameter that is large relative to the diameter of the left pulmonary artery. 20 A previously published study using a LA/Ao ratio of 1.5 as a cutoff for diagnosis of PDA in older neonates provided a sensitivity of 88% with a specificity of 95% 13 ; however, our estimates were lower (82% and 63%, respectively). Based on our findings, we suggest PFO as another potential marker for PDA (with a 3.0 mm cutoff), but PFO also has relatively low sensitivity (61%) and specificity (48%). Although PDA and PFO have been reported elsewhere as frequent comorbidities, 21 the use of PFO as a marker for PDA has not, to our knowledge, previously been reported in the literature.
Conclusion
The possible roles of echocardiographic markers to predict the need for therapeutic closure of a PDA are uncertain.6,20,22 The findings of this study suggest that when considered individually, the sensitivity and specificity of LA/Ao, PDA/LPA, and PFO diameter are not high enough to predict which PDAs are hemodynamically significant and/or which PDAs would require surgical intervention (ligation). We hypothesize that a mathematical combination of LA/Ao, PDA/LPA, and PFO may prove to be more accurate in determining which PDAs will not spontaneously close and may require ligation. This proposal is not unique, as other studies have shown that a combination of markers may have more utility than individual markers to predict a hemodynamically significant PDA. 19 In the report by Jaillard et al., 19 LA/Ao > 1.6 combined with PDA diameter > 3.0 mm provided evidence for ligation in 58 premature infants (< 28 weeks’ gestation; birth weight 630-1130 g). In the study by Hamrick and Hansmann, 5 LA/Ao ≥ 1.4 combined with PDA diameter ≥ 1.4 mm/kg body weight and with left ventricular enlargement has also been applied to mark a significant PDA shunt. Research is presently under way in the second stage of this project to determine the receiver operating characteristic curves for composite echocardiographic markers as positive tests for a hemodynamically significant PDA.
Footnotes
Acknowledgements
The first author would like to thank the SDMS for allowing her to participate in the Sonography Research Institute. She would also like to thank Dr. Joy Guthrie for serving as her guide and mentor for this project.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported in part by a grant from the Sonography Research Institute of the Society of Diagnostic Medical Sonography Foundation.
