Abstract
Persistent hemodynamically significant patent ductus arteriosus (PDA) in preterm infants may lead to significant morbidity and increased mortality. This report proposes a new composite marker to predict a hemodynamically significant PDA, termed the PDA marker (PDAM) score, which incorporates the diameter of the patent foramen ovale (PFO) in addition to the ratio of the diameter of the left atrium to the diameter of the root of the aorta (LA/Ao) and ratio of the size of the patent ductus arteriosus to the left pulmonary artery (PDA/LPA) diameter. PDA was suspected in 191 infants between 2009 and 2013. Analysis of the data indicated that there was no significant difference between the accuracy of the PDA diameter and the PDAM score as markers of either a hemodynamically significant PDA or a very hemodynamically significant PDA that needed ligation. These data did show that the PDAM score provided slightly higher sensitivity (93.3%) and specificity (75.6%) than the PDA diameter to identify a PDA that needed ligation at the specified cutoff level of PDAM = 5.1. Consequently, clinicians may be more confident using the PDAM score, rather than the PDA diameter alone, when selecting PDA cases for surgical intervention.
Hemodynamically significant persistent patent ductus arteriosus (PDA) in preterm infants often leads to increased oxygen demand, poor feeding, and failure to thrive and is associated with increased morbidity and mortality. 1 Therefore, early treatment and intervention are strongly advised. 2 Several echocardiographic markers to predict a hemodynamically significant PDA that requires ligation have been described in the literature. These include (a) the PDA diameter is ≥1.4 mm/kg body weight, 1 (b) the ratio between the left atrial (LA) diameter and the aortic root (Ao) diameter is 1.4 to 1.6,2–4 (c) the PDA diameter is large relative to the left pulmonary artery (LPA) diameter, 4 (d) celiac arterial flow to left ventricular output (CAF:LVO), 5 and (e) retrograde diastolic flow in renal and mesenteric arteries. 6 While these methods have been previously reported, there is still a need to further develop methods for diagnosing and quantifying hemodynamically significant shunts to optimize outcomes for neonates with PDA. Although PDA and the patent foramen ovale (PFO) are associated with frequent comorbidities if left untreated, 7 the combination of these methods to determine a hemodynamically significant PDA has not been previously reported.
This study proposes a new composite marker to predict a hemodynamically significant PDA, termed the PDA marker (PDAM©) score, which incorporates the diameter of the PFO, the LA/Ao ratio, and the PDA/LPA ratio. Although PDA and PFO are frequent comorbidities, 8 the use of the PFO diameter as a marker for a hemodynamically significant PDA has not previously been reported. The data below compare a well-established marker (PDA diameter) with this new marker—specifically, PDAM = LA/Ao + PDA/LPA + PFO diameter—to evaluate the hypothesis that PDAM may be more effective than the PDA diameter alone to indicate a hemodynamically significant PDA and also to better detect cases that may need ligation.
Materials and Methods
A retrospective data analysis on pediatric echocardiograms performed in the neonatal intensive care unit (NICU) of a western US regional medical center from 2009 to 2014 was done. Institutional review board approval was obtained prior to any data collection. A total of 191 cases with a suspected PDA were identified and reviewed. The data were blindly collected in a retrospective manner. All pediatric echocardiograms were performed using a Philips IE33 ultrasound machine (Philips Ultrasound, Bothel, WA) equipped with a phased sector array S12-4 MHz transducer. All echocardiographic data were uploaded from the machine and stored on the hospital electronic data server. All echocardiograms were performed by American Registry for Diagnostic Medical Sonography (ARDMS)–credentialed sonographers trained in pediatric and neonatal echocardiography. All patients were monitored for PDA closure with repeated echocardiograms according to clinical finding (continuous machinery murmur, systolic murmur, heart murmur, pulmonary hypertension) or routine follow-up. Some patients with PDAs were under medical treatment, and others underwent PDA ligation.
Following hospital imaging protocol, the following measurements were obtained as part of the pediatric echocardiogram: the PDA was measured on 2D gray-scale images taken from parasternal long-axis, short-axis, and suprasternal notch views. The PDA diameter recorded was the measurement closest to the aortic end of the PDA (Figure 1). The left pulmonary artery (LPA) was measured in the high parasternal short view (Figure 2). The LA/Ao ratio was obtained from the parasternal short-axis view (Figure 3). The PFO diameter was measured in subcostal long- and short-axis views; the recorded measurement was the largest measurement of the two views (Figure 4). All measurements were done at the time of study and recorded from the hospital electronic server; only the PDA/LPA ratio was retrospectively calculated.
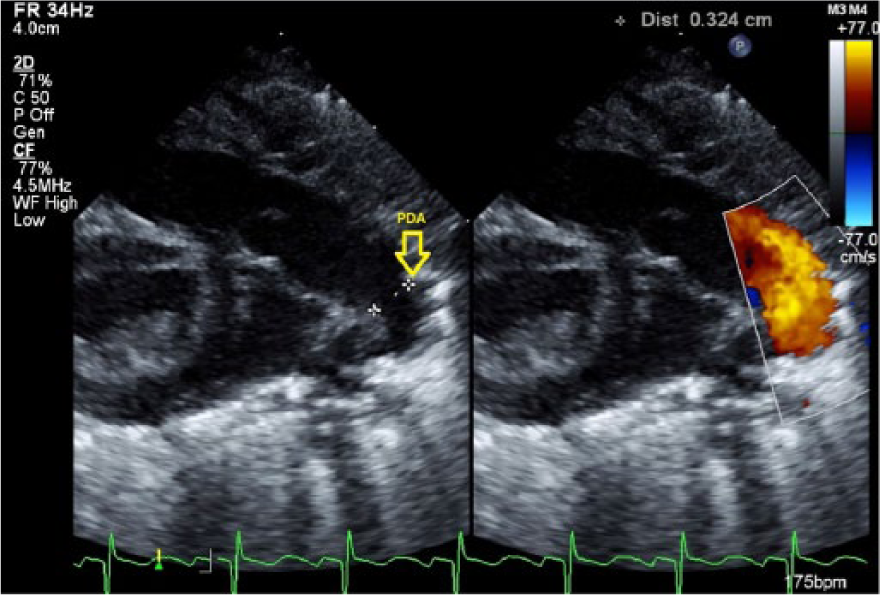
Side-by-side gray-scale and color Doppler images showing the patent ductus arteriosus (PDA) diameter measurement taken from a right ventricular outflow tract view.
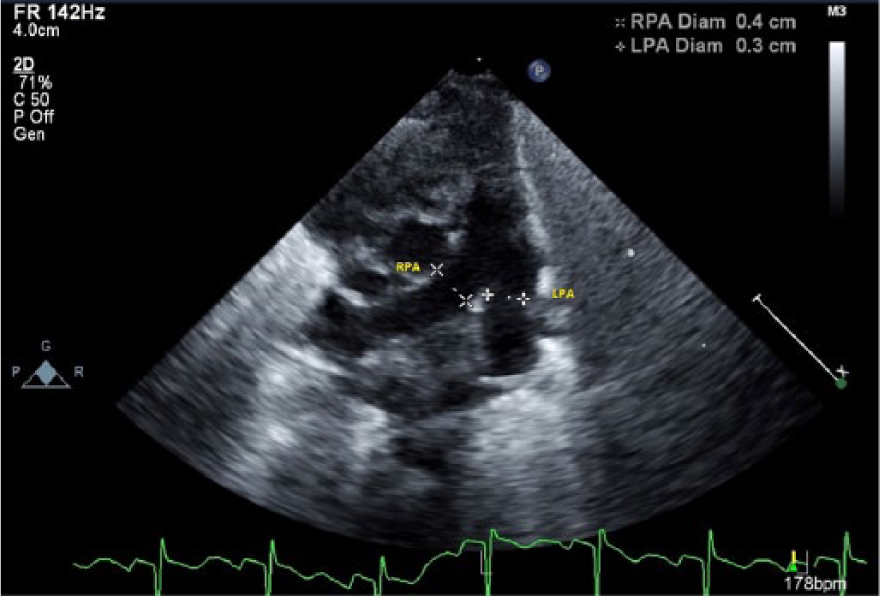
Gray-scale image showing the left pulmonary artery diameter measurement taken from a high parasternal short-axis view. LPA, left pulmonary artery; RPA, right pulmonary artery.
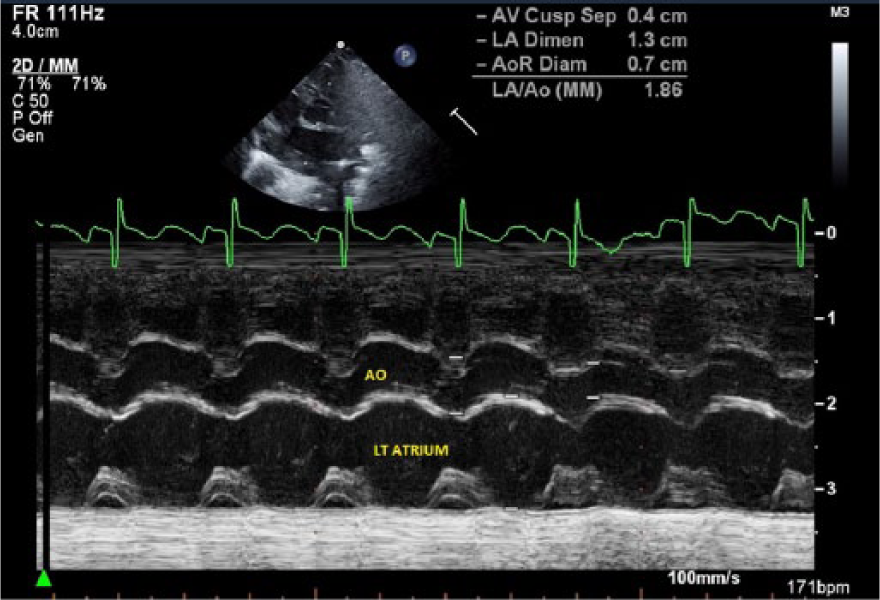
M-mode echocardiographic image showing the left atrial (LA) and aortic root diameter (Ao) measurements used to form the LA/Ao ratio, taken from a parasternal short-axis view.
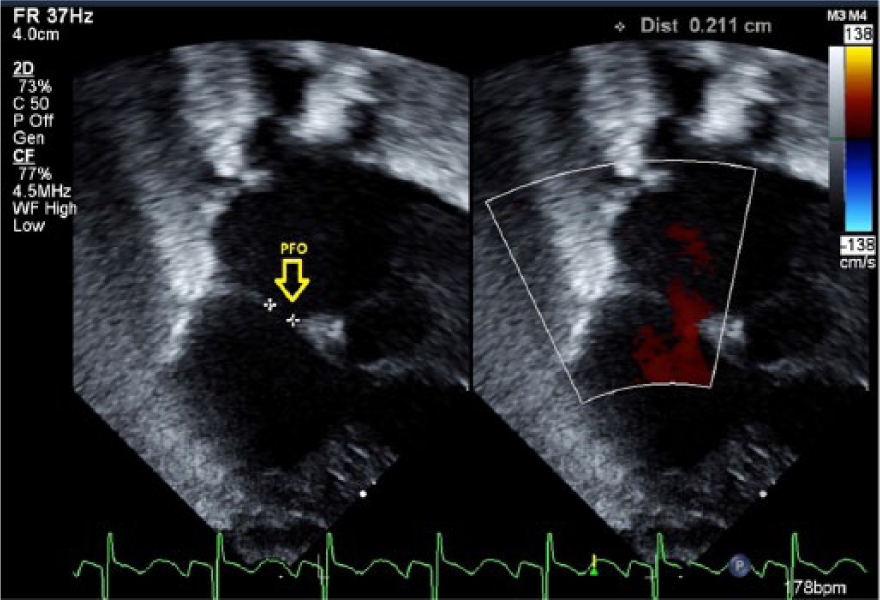
Side-by-side gray-scale and color Doppler images showing the measurement of the patent foramen ovale, taken from a subcostal sagittal view.
All clinical information was retrieved from the electronic medical record: age, sex, and ethnicity. The weight was recorded at the time of the echocardiographic study. To de-identify all patient data, a key was made at the time of data collection, and a numerical code was generated accordingly for each patient. Statistical analysis was done using SPSS version 20.0 (SPSS, Inc., an IBM Company, Chicago, IL).
Results
Participants
The sample consisted of 191 preterm infants, of whom 154 (80.6%) were male. The ethnic groups were white/non-Hispanic (81 [42.4%]), white/Hispanic (89 [46.6%]), Asian (16 [8.4%]), and African American (5 [2.6%]). At the time of their first echocardiogram, the infants ranged in age from 0 to 102 days (mean [SD], 13.2 [18.3] days), with a correspondingly wide range of body weights from 0.450 to 5.808 kg (mean [SD], 1.66 [1.08] kg). Most infants (119 [62.3%]) had a low body weight (<1.5 kg). All cases with a PDA also had a PFO. Among the PDA cases, 27 were aggressively followed up with multiple echocardiograms (from two to seven) because they were considered by the attending clinicians to be potentially in need of surgical intervention.
Classification of Controls and Cases
On the basis of the analysis of echocardiographic measurements (the PDA diameter, the ratio between the LA/Ao diameter, the ratio between the PDA/LPA diameter, and the PFO diameter), the sample was classified into four mutually exclusive groups: (1) 38 infants without a PDA (controls), (2) 126 cases with a PDA that was not considered by the clinicians to be hemodynamically significant at the first measurement (–HS), (3) 12 cases with a PDA that was considered by the clinicians to be hemodynamically significant at the last measurement but in whom the PDA was not ligated (+HS), and (4) 15 cases with a PDA that was considered by the clinicians to be very hemodynamically significant at the last measurement and the PDA was ligated (L-HS). Some cases that were considered hemodynamically significant were also recognized by other clinical factors, including poor weight gain, increased oxygen requirement, prolonged ventilation days, and increased systolic PDA shunt velocity.
PDA Diameter
The PDA diameters (mean ± 95% confidence interval [CI]) across the groups are compared in Figure 5. Due to the wide variability in the body weights of the infants at the time of the echocardiography, the PDA diameters are compared in units of mm/kg body weight, consistent with El Hajjar et al. 1 A progressive systematic increase in the mean PDA diameter (mm/kg) was observed across all three groups of cases. Cases that were considered not hemodynamically significant (–HS) had a mean PDA diameter of 1.76 mm/kg. Cases that were considered hemodynamically significant (+HS) had a mean PDA diameter of 2.44 mm/kg. The cases that required ligation (L-HS) had a mean PDA diameter of 3.69 mm/kg. Analysis of variance (ANOVA) indicated that the PDA diameters were significantly different between the three groups of cases (F = 21.37, P < .001).
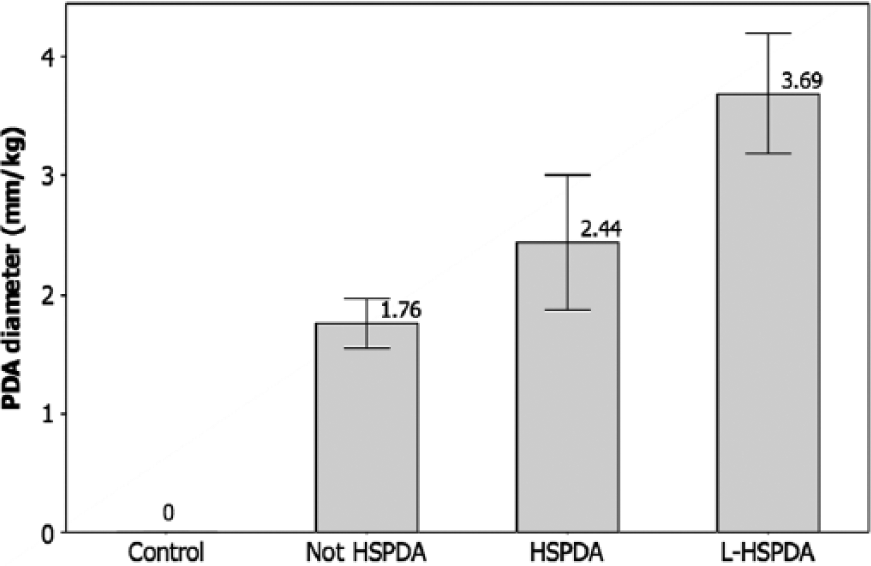
Patent ductus arteriosus (PDA) diameter (mm) data, normalized by weight (kg), for the four classes of infants, showing the mean ± 95% confidence interval values. Control, no PDA; Not HSPDA, not hemodynamically significant PDA; HSPDA, hemodynamically significant PDA; L-HSPDA, hemodynamically significant PDA requiring ligation.
LA/Ao
The LA/Ao ratios are compared in Figure 6. Across the four groups, the mean LA/Ao increased systematically, from a minimum of 1.38 in the control group, through 1.66 in the –HS group and 1.86 in the +HS group, to a maximum of 1.98 in L-HS group. ANOVA indicated that the mean LA/Ao ratios were significantly different between the four groups (F = 12.74, P < .001).
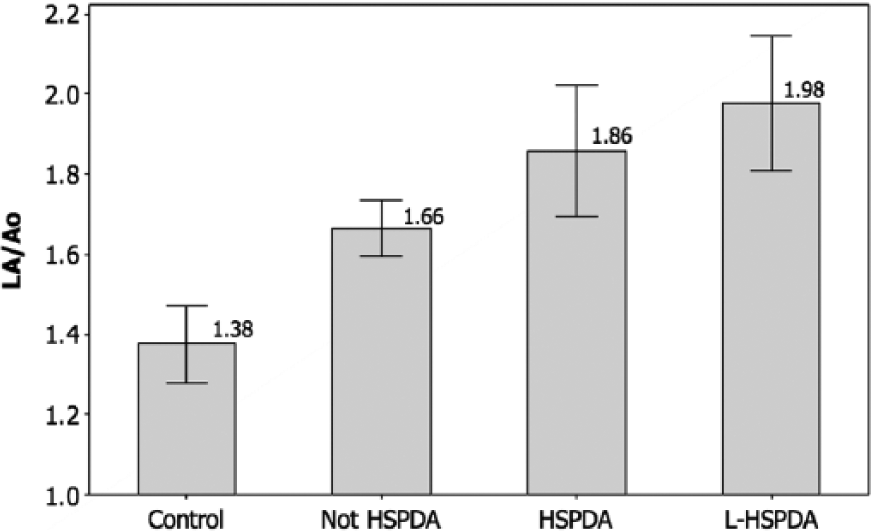
Left atrial (LA) diameter to aortic root (Ao) diameter ratio data for the four classes of infants, showing the mean ± 95% confidence interval values. Control, no PDA; Not HSPDA, not hemodynamically significant PDA; HSPDA, hemodynamically significant PDA; L-HSPDA, hemodynamically significant PDA requiring ligation.
PDA/LPA
The PDA/LPA ratios are compared in Figure 7. Across the three groups of cases, the mean PDA/LPA increased systematically from a minimum of 0.69 in the –HS group to 0.75 in the +HS group and to a maximum of 0.87 in L-HS group. ANOVA indicated that the mean PDA/LPA ratios were significantly different between the three groups (F = 3.53, P = .031).
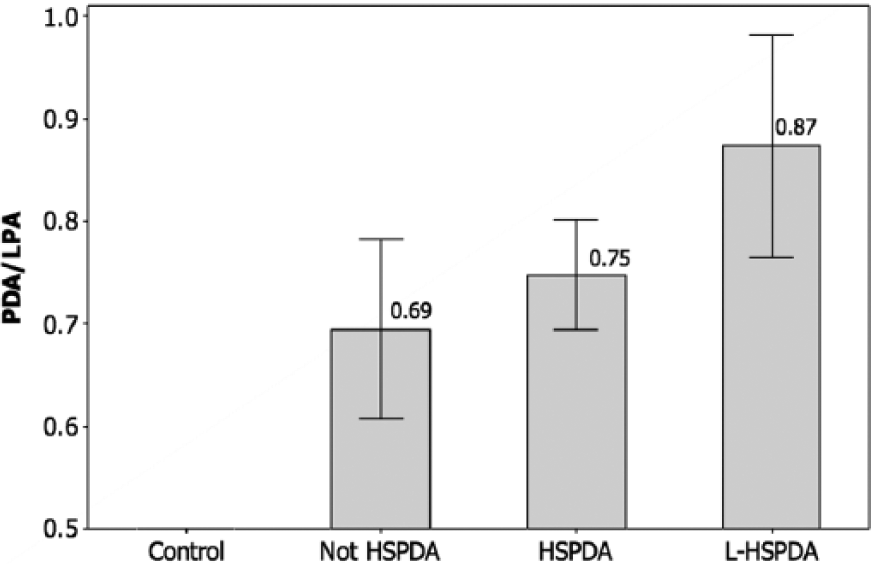
Patent ductus arteriosus (PDA) diameter to left pulmonary artery (LPA) diameter ratio for the four classes of infants, showing the mean ± 95% confidence interval values. Control, no PDA; Not HSPDA, not hemodynamically significant PDA; HSPDA, hemodynamically significant PDA; L-HSPDA, hemodynamically significant PDA requiring ligation.
PFO Diameter
The PFO diameters are compared in Figure 8. Due to the wide variability in the body weight of the infants at the time of the echocardiograms, the PFO diameters were compared in units of mm/kg body weight, similar to the PDA diameter data. Across the four groups, the mean PFO diameter increased systematically from a minimum of 1.81 mm/kg in the control group, to 2.03 mm/kg in the –HS group and 2.24 mm/kg in the +HS group, to a maximum of 3.11 mm/kg in the L-HS group. ANOVA showed that the mean PFO diameters were significantly different between the four groups (F = 14.43, P < .001).
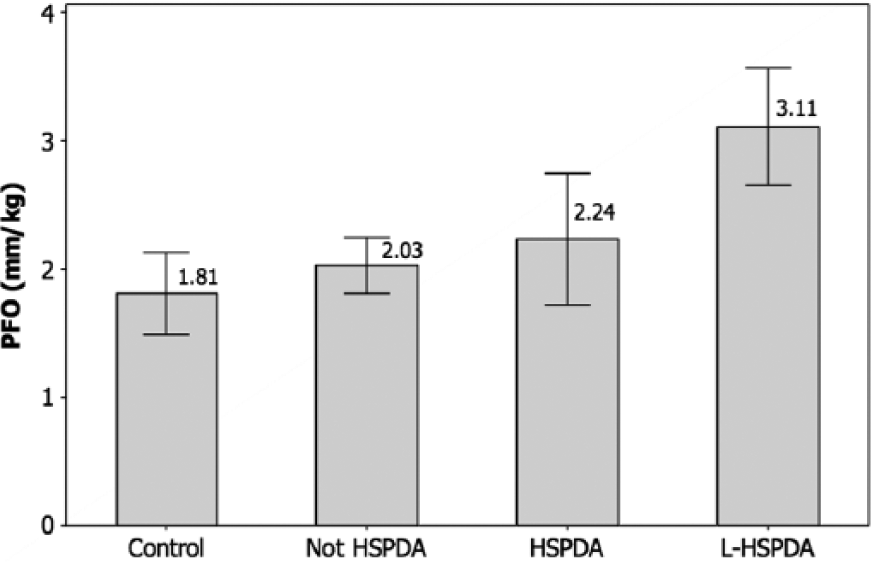
Patent foramen ovale (PFO) diameter data (mm), normalized by weight (kg), for the four classes of infants, showing the mean ± 95% confidence interval values. Control, no PDA; Not HSPDA, not hemodynamically significant PDA; HSPDA, hemodynamically significant PDA; L-HSPDA, hemodynamically significant PDA requiring ligation.
PDAM© Score
The scores for the new marker (PDAM) were calculated as the sum of LA/Ao + PDA/LPA + PFO diameter (mm/kg). The PDAM scores are compared in Figure 9. Across the four groups, the mean PDAM score increased systematically from a minimum of 3.19 mm/kg in the control group, to 4.44 mm/kg in the –HS group and 4.79 mm/kg in the +HS group, to a maximum of 5.96 mm/kg in the L-HS group. ANOVA showed the mean PDAM scores were significantly different between the four groups (F = 17.76, P < .001).
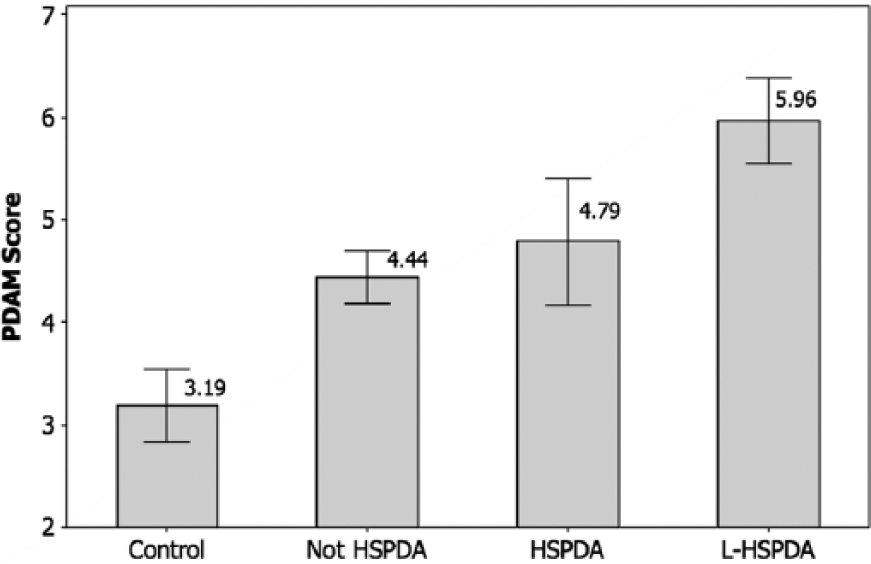
Patent ductus arteriosus marker (PDAM) scores for the four classes of infants, showing the mean ± 95% confidence interval values. Control, no PDA; Not HSPDA, not hemodynamically significant PDA; HSPDA, hemodynamically significant PDA; L-HSPDA, hemodynamically significant PDA requiring ligation.
ROC Curves
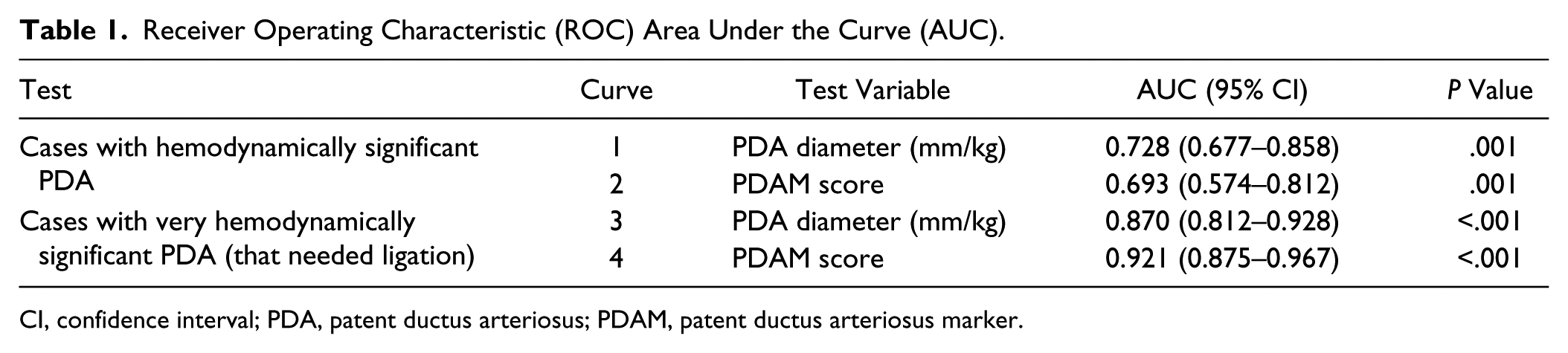
Four receiver operating characteristic (ROC) curves are presented in Figure 10 to test for (1) cases with a hemodynamically significant PDA (+HS) as the state variable, using the PDA diameter as the test variable; (2) cases with a hemodynamically significant PDA (+HS) as the state variable, using the PDAM score as the test variable; (3) cases that needed ligation as the state variable (L-HS), using the PDA diameter as the test variable; and (4) cases that needed ligation (L-HS) as the state variable, using the PDAM score as the test variable. The statistics presented in Table 1 indicate that the areas under the four ROC curves (AUCs) were significantly >0.5 at P ≤ .001, implying that markers consistently discriminated between the cases and the controls.
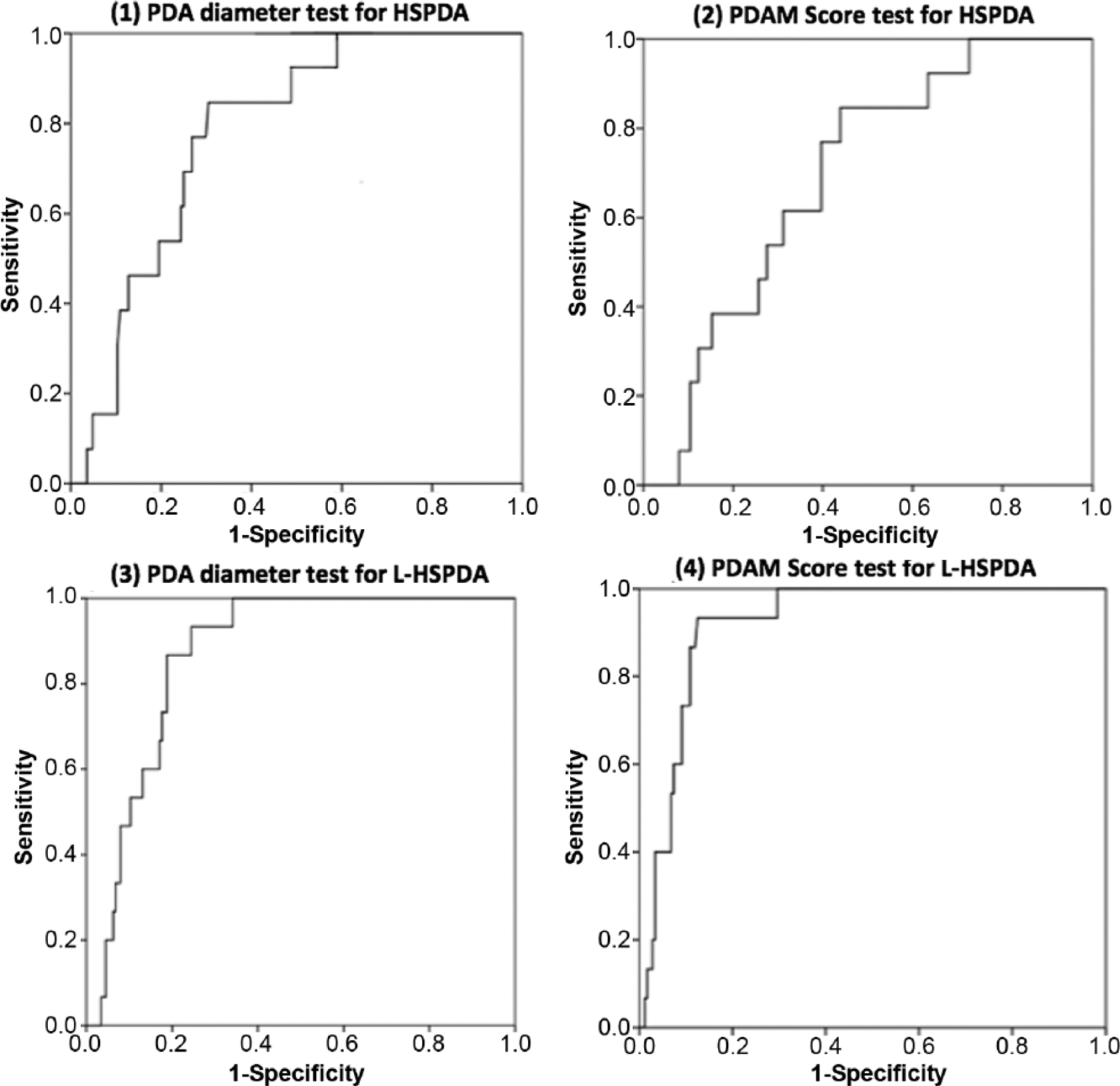
Receiver operating characteristic (ROC) curves for (1) sensitivity and specificity of the patent ductus arteriosus (PDA) diameter to detect a hemodynamically significant PDA (HSPDA); area under the curve (AUC) = 0.728. (2) Sensitivity and specificity of the patent ductus arteriosus marker (PDAM) score to detect an HSPDA; AUC = 0.693. (3) Sensitivity and specificity of the PDA diameter to detect a hemodynamically significant PDA requiring ligation (L-HSPDA); AUC = 0.870. (4) Sensitivity and specificity of the PDAM score to detect an L-HSPDA; AUC = 0.921.
Receiver Operating Characteristic (ROC) Area Under the Curve (AUC).
CI, confidence interval; PDA, patent ductus arteriosus; PDAM, patent ductus arteriosus marker.
The AUCs to test for a hemodynamically significant PDA using the PDA diameter (0.728) and the PDAM score as the test variable (0.693) were not significantly different. The AUCs to test for a PDA that needed ligation using the PDA diameter as the test variable (0.870) and using the PDAM score as the test variable (0.921) were also not significantly different.
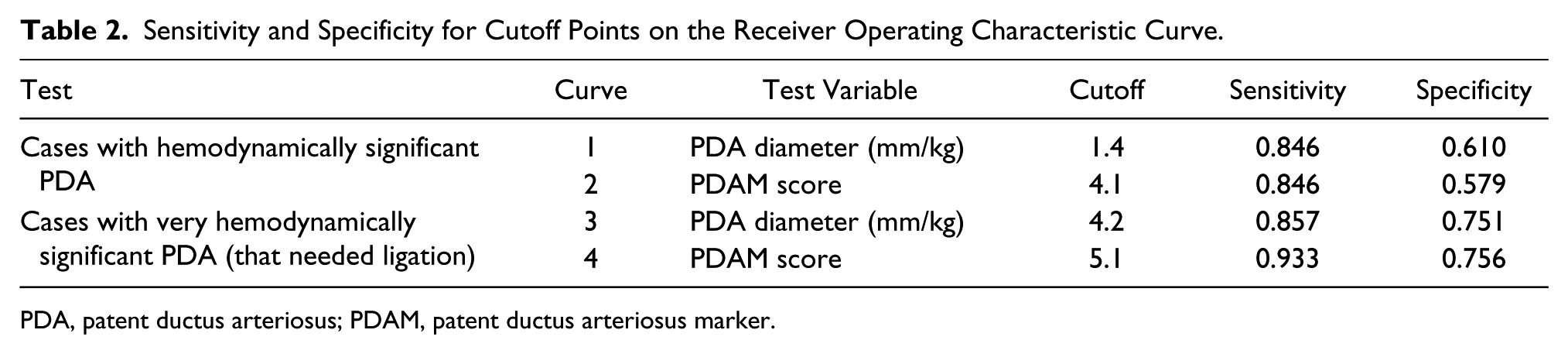
Table 2 presents the sensitivity and specificity estimates at the cutoff levels on the ROC curves nearest to the top left-hand corners. To test for cases with a hemodynamically significant PDA, a PDA diameter cutoff at 1.4 mm/kg and a PDAM score of 4.1 provided the same sensitivity (0.846) and similar specificities (0.610 and 0.579, respectively). To test for cases with a very hemodynamically significant PDA that needed ligation, however, the PDA diameter cutoff at 4.2 mm/kg provided lower sensitivity (0.857) and specificity (0.751) than did the PDAM score cutoff at 5.1 (0.933 and 0.756, respectively). A PDAM score cutoff of 4.0 still exhibited a high sensitivity and specificity for PDA when stratified by both age and weight (Table 3).
Sensitivity and Specificity for Cutoff Points on the Receiver Operating Characteristic Curve.
PDA, patent ductus arteriosus; PDAM, patent ductus arteriosus marker.
Sensitivity and Specificity for PDAM (Additive Combination of LA/Ao + PDA/LPA + PFO) as a Marker of PDA, Stratified by Age and Weight.
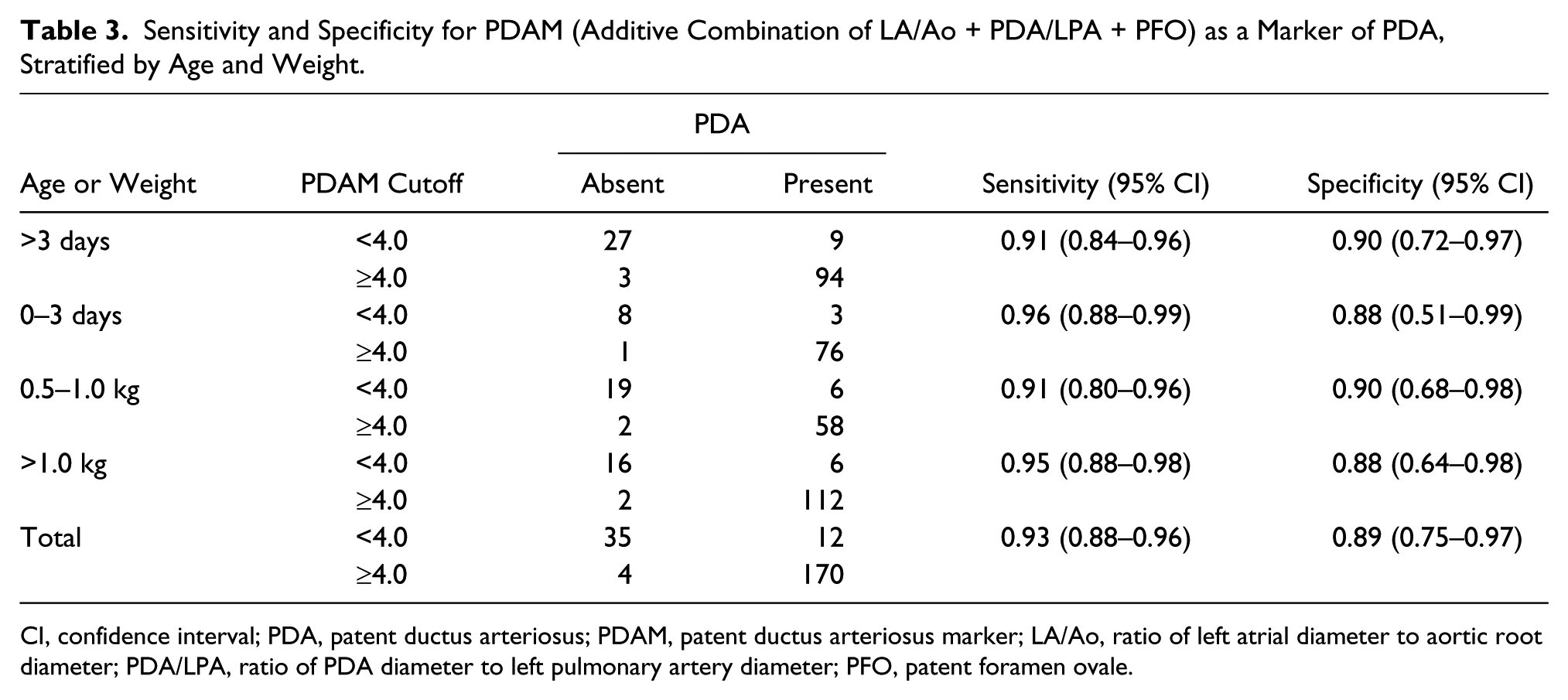
CI, confidence interval; PDA, patent ductus arteriosus; PDAM, patent ductus arteriosus marker; LA/Ao, ratio of left atrial diameter to aortic root diameter; PDA/LPA, ratio of PDA diameter to left pulmonary artery diameter; PFO, patent foramen ovale.
Discussion
The role of echocardiographic markers to predict the need for therapeutic closure of a hemodynamically significant PDA is uncertain.5,9–11 A study by Jhaveri et al 8 in 2010 demonstrated that there are nearly equal outcomes using early surgical ligation versus a conservative approach. Although many studies have demonstrated that surgical intervention (ligation) is effective in closing the PDA,11,12 there are no generally accepted guidelines for the diagnosis of a hemodynamically significant PDA. In 2008, El-Kfuffash et al 5 performed a quantitative analysis of the degree of ductal steal using celiac artery blood flow (CAF) to left ventricular output (LVO) ratio. Their results were promising, concluding that a CAF:LVO ratio of 0.10 had a sensitivity of 93% and a specificity of 78% for the presence of a hemodynamically significant PDA. The need for intervention/ligation was again stated by Sehgal et al 6 in 2011. Their study showed that in the setting of a hemodynamically significant PDA, there is a significant “steal” of blood from the mesenteric vessels. This was an objective finding based on the postprandial diastolic reversal of flow in the superior mesenteric artery Doppler waveform. Their study suggested that it is the PDA itself, rather than the medical treatment, that contributes to the complication of neonatal necrotizing enterocolitis. These findings also confirmed previously published evidence recommending the use of the PDA diameter, the LA/Ao ratio, and the PDA/LPA as echocardiographic markers of a hemodynamically significant PDA.1,3,4,11,12
In an earlier study at our institution, a case-control evaluation was made using four individual markers to predict the presence versus absence of a hemodynamically significant PDA in premature neonates. 13 These markers were (a) left ventricular ejection fraction (LVEF), (b) LA/Ao ratio, (c) PDA/LPA ratio, and (d) PFO diameter. The results showed that the LVEF did not differ significantly between the cases (mean, 66.2; 95% CI, 65.1–67.4) and the controls (mean, 65.6; 95% CI, 63.5–67.6). The sensitivity of a LA/Ao cutoff of 1.5 as a positive test for the presence of PDA was high (82%); however, the specificity was low (63%). The sensitivity was lower (56%) when the age was zero to three days, but the specificity was higher in that age group (88%). A PDA/LPA cutoff of 0.5 or higher was associated with the presence of a hemodynamically significant PDA, and PDA was significantly associated with an increase in the mean PFO diameter above 3.0 mm, but age and birth weight had no significant effect. These individual markers did not produce adequate sensitivity and specificity for clinical management decisions and predicting which PDAs would require intervention and/or ligation.
Statistical comparisons of the data from this study using ANOVA indicated that the mean values of the PDA diameter, the LA/Ao ratio, the PDA/LPA ratio, the PFO diameter, and the PDAM score all increased systematically across the four groups of infants, classified in terms of increasingly hemodynamic significance of the PDA. This was the rationale for exploring a combination marker to predict a hemodynamically significant PDA. The hypothesis was that when there is a large PDA, there is an increase in the volume of blood entering the pulmonary artery. In the premature infant, there is significantly increased pulmonary resistance; therefore, the blood would tend to dilate the left atrium before it would go back into the lungs. With an increased left atrial volume, there is a possibility that the foramen ovale will stretch. This was the basis of considering the additive affect of these three markers rather than considering them alone. The current study provides new evidence to support the existence of a link between a hemodynamically significant PDA and a large PFO diameter, as well as the clinical value of our proposed PDAM marker, specified as LA/Ao + PDA/LPA + PFO diameter.
Limitations
Due to the wide variability in the body weight of the infants at the time of the echocardiograms, the PFO diameters were compared in units of mm/kg body weight. This helped to control for the variable PFO size with increasing body weight. Correlation of the PDAM score in a multicenter study is pending and should assist in validating this novel score.
Conclusion
Using mean values and sensitivity and specificity estimates (ROC curves), this study compared a well-established marker (PDA diameter) versus a proposed new marker, PDAM© = LA/Ao + PDA/LPA + PFO diameter, with the hypothesis that PDAM may be more effective than the PDA diameter alone to indicate a hemodynamically significant PDA and to detect significant PDAs that may need ligation. Analysis of the ROC curves indicated that in the patient sample studied, the PDAM score was numerically a slightly better marker but that there was no statistically significant difference between the accuracy of the PDA diameter and the PDAM score as markers of either a hemodynamically significant PDA or a very hemodynamically significant PDA that needed ligation. Based on these data, it appears that clinicians may confidently use either of the two markers to identify a hemodynamically significant PDA. In addition, the PDAM score provided slightly higher sensitivity and specificity than did the PDA diameter to identify a PDA that needed ligation at the specified cutoff levels. Clinicians may be more confident using the composite PDAM score, rather than the PDA diameter alone, when selecting PDA cases for surgical intervention.
Footnotes
Acknowledgements
I thank Ms. Thuan Flood, BS, RDCS, for assisting in the data collection and image acquisition for this study, which is an extension of her previously completed Sonography Research Institute (SRI) project. I would also like to extend my sincere gratitude to Drs. Krish Rajani, Steven Elliott, Anand Rajani, and David Aguilar, our superb neonatologists at Community Regional Medical Center, who allowed this research in our NICU and have now fully adopted the PDAM score into their clinical practice.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of the article.
Funding
The author declared no financial support for the research, authorship, and/or publication of this article.
