Abstract
Although there are various echocardiographic measurements that can determine the hemodynamic significance of a patent ductus arteriosus (PDA), there are few quantifiable measures to determine if there is a vascular steal secondary to a large PDA from the abdominal organs. If the PDA is as large as the branch pulmonary arteries, then effectively one-third of the blood that should go to the abdomen is pushed back toward the lungs. This suggests a vascular steal from the abdomen, causing potential ischemia to the bowel and kidneys. Our original research resulted in a diagnostic marker (PDAM) to assist clinicians with the management of PDAs. The superior mesenteric artery and renal artery spectral Doppler waveforms were added to the PDAM score to evaluate perfusion in infants with PDA. The absence of diastolic flow or staccato flow, in which there is a loss of end-systolic flow, is prognostic of a vascular steal phenomenon.
Introduction
In utero, the ductus arteriosus (DA) allows the blood flow of the fetus to be diverted from the pulmonary circulation to the descending aorta and placental bed. Most of the blood flow exiting from the right ventricle bypasses the pulmonary arteries in a right-to-left shunt across the DA. Spontaneous closure of the DA occurs in most full-term healthy infants within 1 to 5 days after birth. The spontaneous closure of the DA is less likely in premature infants (<30 weeks’ gestation), particularly among infants with low birth weight (<1000 g), whose lungs have not yet fully developed.1–4
Numerous studies have demonstrated that a persistently large and hemodynamically significant patent ductus arteriosus (PDA) in premature neonatal infants is associated with a worsening of cardiopulmonary status, particularly during the recovery period from respiratory stress syndrome.5–8 Specifically, premature neonatal infants with moderate to large left-to-right shunts across the DA have a greater mortality rate than those without a PDA. Furthermore, many complications are associated with a hemodynamically significant PDA, including chronic lung disease (pulmonary hypertension, bronchopulmonary dysplasia, increased pulmonary hyperemia and edema); decreased renal, mesenteric, and cerebral perfusion; intraventricular hemorrhage; periventricular leukomalacia; bacterial necrotizing endocarditis; and cerebral palsy.
For more than a decade, echocardiography has been the gold standard for the diagnosis of a PDA in neonates.9–12 However, the decision to select an infant with a hemodynamically significant PDA for treatment, based only on echocardiographic criteria, presents a challenge to clinicians. It is prudent to individualize the decision to treat a hemodynamically significant PDA by selecting only the most fragile infants with the highest risk of morbidity and mortality, based on objective measurements, including not only echocardiographic criteria but also birth weight, respiratory status, and other clinical evidence. Mitra et al. 13 suggested, however, that no generally accepted guidelines exist for the diagnosis of PDA to provide a proper direction for attending clinicians to decide on appropriate treatment. The clinical evidence to diagnose and treat a hemodynamically significant PDA varies between different neonatal care centers, and furthermore, the comparability and repeatability of different types of echocardiographic diagnostic evidence have not yet been optimized or harmonized across different centers.
Clinicians often choose to treat a PDA in an attempt to minimize the risk of morbidity and mortality; however, because of the uncertainty about diagnosis and treatment modalities, there is substantial variability in clinical practice. 14 Current practice is primarily focused on PDA closure using pharmacologic and mechanical (surgical or nonsurgical ligation) treatments.1,5,7,11,13,15–18 The more recent trend toward less aggressive management is supported by Clyman et al., 19 who determined that in preterm infants less than 28 weeks with moderate to large PDAs who were receiving respiratory support after the first week, early routine medical treatment did not reduce PDA ligations. In fact, their research suggested that early treatment delayed full feeding and was associated with higher rates of late-onset sepsis and death in infants >26 weeks of gestation. Conversely, current research by Hagadorn et al. 20 concluded that neonatal intensive care unit–specific outcomes of preterm birth may be improved by PDA closure in infants weighing between 400 and 749 g at birth.
Guthrie 21 developed a novel score or diagnostic marker (PDAM) to assist neonatal clinicians at a western regional medical center. The PDAM score was applied in practice to select at-risk premature infants with a hemodynamically significant PDA for treatment. PDAM is computed by a mathematical combination of echocardiographic criteria, specifically the ratio of the left atrial diameter (LA) to the aortic root diameter (Ao), the ratio of the diameter of the PDA to the diameter of the left pulmonary artery (LPA), and the diameter of the patent foramen ovale (PFO). Figure 1 illustrates the components of the PDAM score. Cases with a very hemodynamically significant PDA that needed treatment were identified by a cutoff PDAM score of ≥5.1. The sensitivity and specificity of using PDAM ≥5.1 (0.933 and 0.756, respectively) were better than using a PDA diameter of only >4.1 mm/kg (0.856 and 0.751, respectively). Other researchers, in contrast, have suggested that only the PDA diameter and/or the LA/Lo ratio should be used to define a hemodynamically significant PDA.9,22
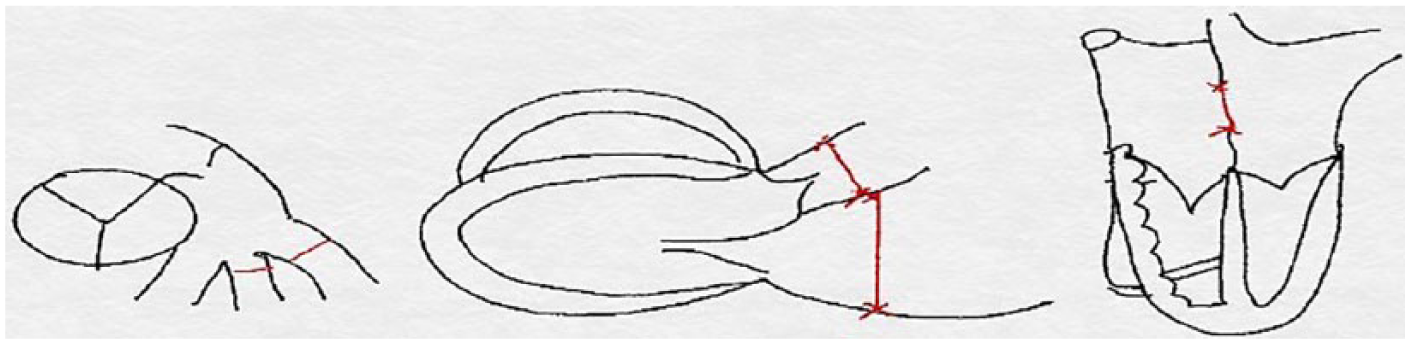
PDAM score = patent ductus arteriosus/left pulmonary artery + left atrial diameter/aortic root diameter ratio + patent foramen ovale/kilograms. The PDAM combination score is used to detect hemodynamically significant PDAs in preterm infants.
The purpose of the current study was to extend the utility of the PDAM score by the addition of markers that indicated the possibility of abnormal blood flow to the abdominal organs. The PDAM score determines whether the PDA is significant at the heart and lung level but does not answer what the overall effect of a large PDA is on the entire body. We hypothesized that a hemodynamically significant PDA could be associated with a vascular steal from the abdominal organs (i.e., that a large PDA causes the blood that should be going to the abdomen to return back to the lungs). A vascular steal occurs when flow that should normally be directed to an organ is redirected. This is seen in other conditions such as large tumors, arteriovenous malformations, or subclavian steal in the vertebrobasilar system. This hypothesis was underpinned by previous studies using Doppler sonography, revealing that large PDAs may possibly be associated with abnormal flow to the abdominal organs. For example, holodiastolic reversal in the abdominal aorta may result in abnormal blood flow patterns to the intestines 23 and kidneys. 24 These abnormal flow patterns may predispose premature infants to subsequent impairment of bowel and renal function. The purpose of the current study was consistent with the recommendation of Kindler et al. 12 that the diagnosis of a hemodynamically significant PDA, and treatment decisions, should ideally be made using a standardized scoring system, based not only on a summary of specified echocardiographic criteria but also on objective evidence of clinical deterioration.
To determine whether a PDA in premature infants was possibly associated with a vascular steal from the abdominal organs, we conducted an observational study using spectral Doppler sonography to determine the extent to which the flow in the supermesenteric artery (SMA) and the renal artery (RA) was statistically related to the PDAM scores. The statistical significance and strengths of the partial regression coefficients between the PDAM scores, body weights, respiratory status, and RA and SMA arterial Doppler waveforms provided research-based evidence to extend the utility of the PDAM score by the addition of sonographic markers that indicated abnormal diastolic blood flow to the abdominal organs.
The overall purpose of this study is not to determine the clinical management but rather to provide additional clinical markers to assist physicians in the management and treatment of PDAs.
Characteristics of the Patient Population
The target population consisted of N = 205 premature infants (<30 weeks’ gestation) born in the Community Regional Medical Center, Fresno, California. The infants’ body weights at birth were classified using an ordinal scale as 0 = 500 to 750 g, 1 = 751 to 1000 g, 2 = 1001 to 1500 g, and 3 = >1500 g. The respiratory status of each infant was classified using an ordinal scale as 0 = room air, 1 = nasal cannula, 2 = continuous positive airway pressure (CPAP), or 3 = intubated. Table 1 presents a cross-tabulation of the frequencies of body weight versus respiratory status. There was a statistically and clinically significant negative correlation between body weight and respiratory status (Spearman rank correlation coefficient = −0.806, P < .001). This correlation arose because a high proportion of infants (n = 121, 59.0%) with very low body weights (<1000 g) experienced a high level of respiratory distress and received CPAP or were intubated, whereas only a small proportion of infants (n = 17, 8.2%) with higher body weights (>1000 g) received CPAP or were intubated.
Cross-tabulation of Body Weight Versus Respiratory Status.
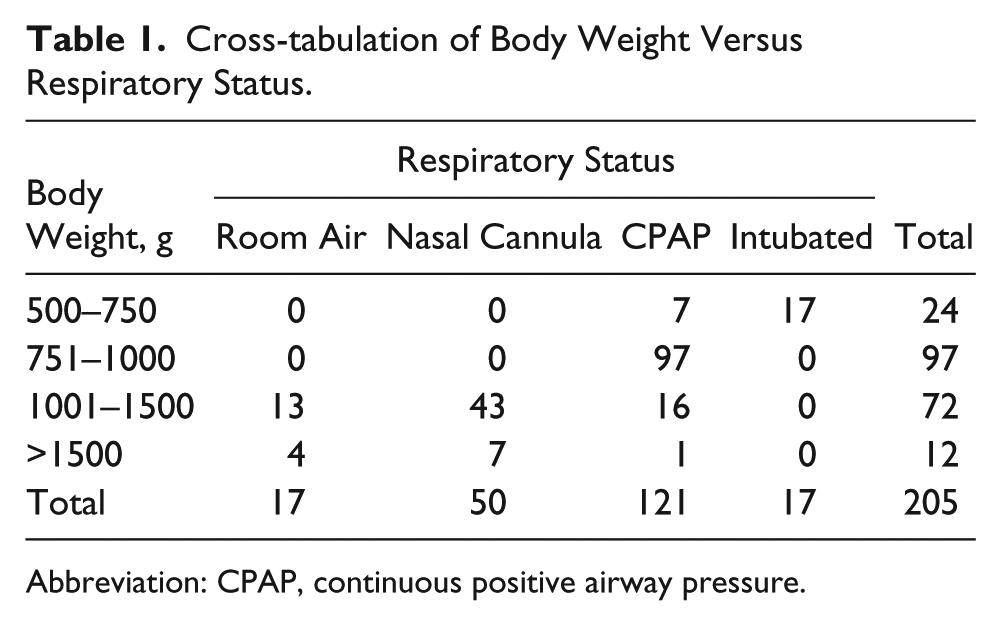
Abbreviation: CPAP, continuous positive airway pressure.
Methods
All 205 infants underwent consecutive pediatric echocardiograms after birth to diagnose the existence of a PDA and to measure the ratio of the LA diameter to the Ao diameter (LA/Ao), the ratio of the diameter of the PDA to the diameter of the LPA, and the diameter of the PFO. All echocardiograms were performed by experienced RDCS-credentialed sonographers using the Philips IE 33 echocardiographic unit (Philips, Andover, MA). The institutionally accepted PDA echocardiographic assessment was performed on all cases. The image quality was considered good in l98 of 205 cases and adequate in the other 7 cases. The cases deemed adequate were due to lung artifact in the very premature infants, where the PDA was visualized only in a low parasternal window.
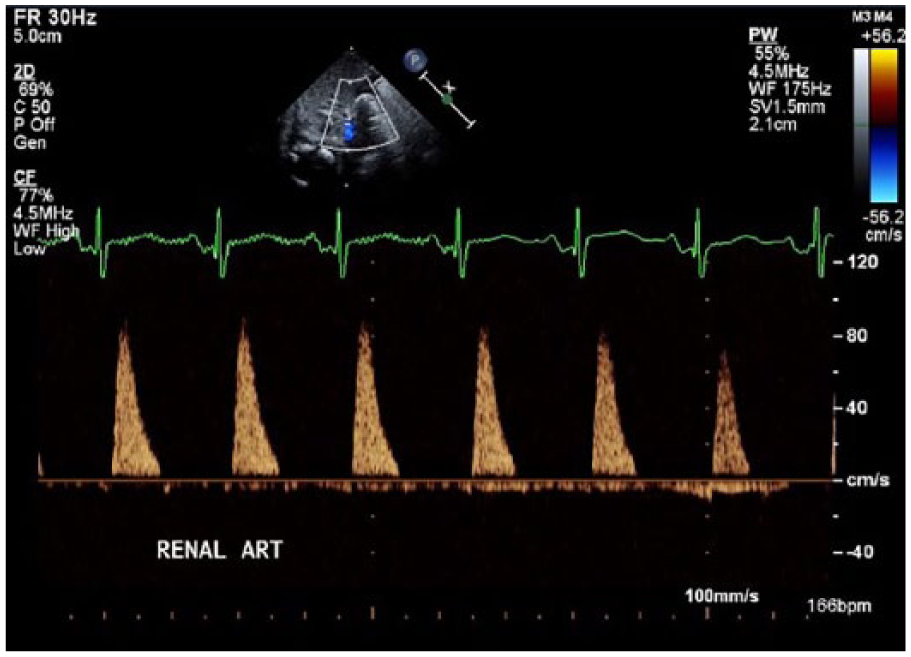
The PDAM scores were coded dichotomously by 0 < 5.1, or 1 ≥ 5.1, where 0 = not hemodynamically significant PDA or 1 = hemodynamically significant PDA. The additional two abdominal vascular Doppler waveforms included a pulsed-wave spectral Doppler waveform of the SMA and RA aortic ostia. The Doppler waveforms were classified using an ordinal scale as 0 = normal, 1 = reduced diastolic flow, 2 = absent diastolic flow, or 3 = staccato and/or absent (Figures 2–7).
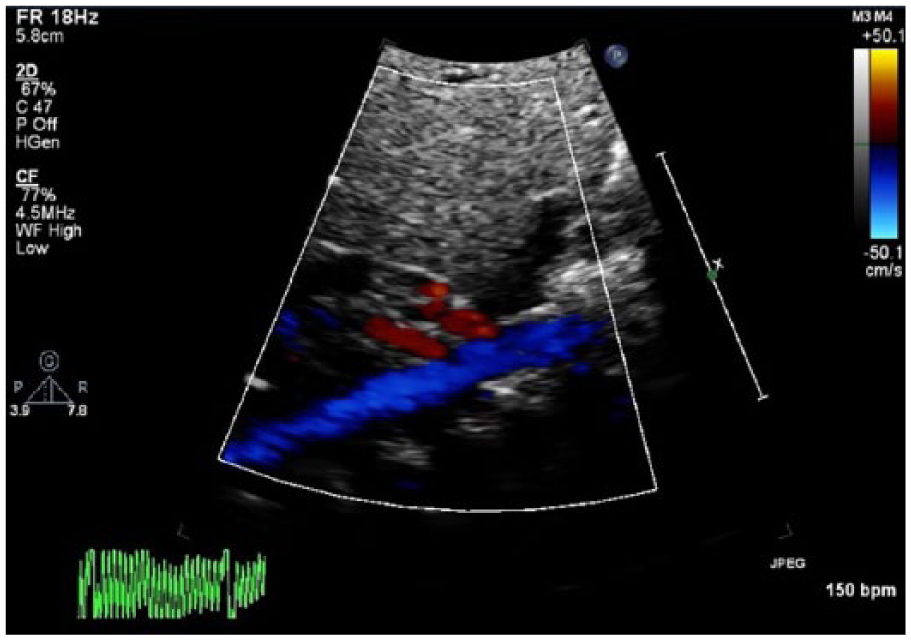
The addition of the supermesenteric artery (denoted by arrow) arising from the abdominal aorta (echocardiographic view) to determine if there is a vascular steal from the patent ductus arteriosus.
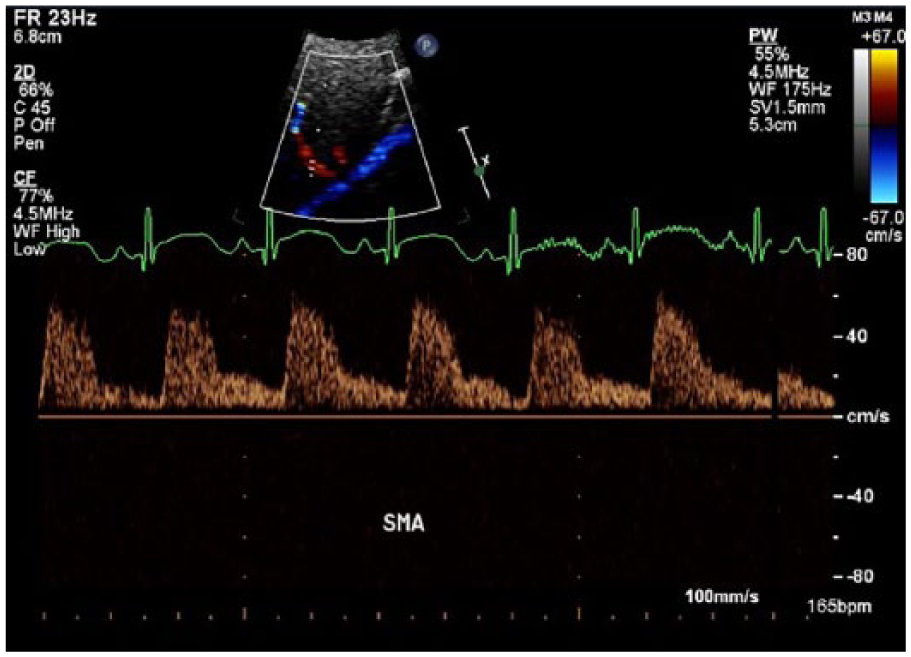
This image demonstrates normal postprandial flow in the superior mesenteric artery.
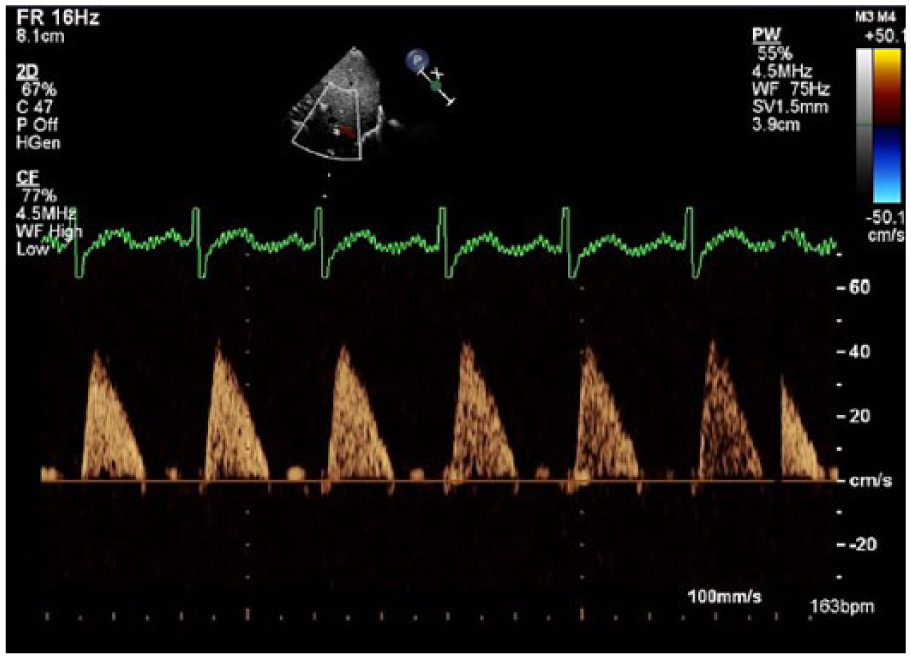
This waveform represents the absence of diastolic flow in the supermesenteric artery in a premature infant with a hemodynamically significant patent ductus arteriosus.
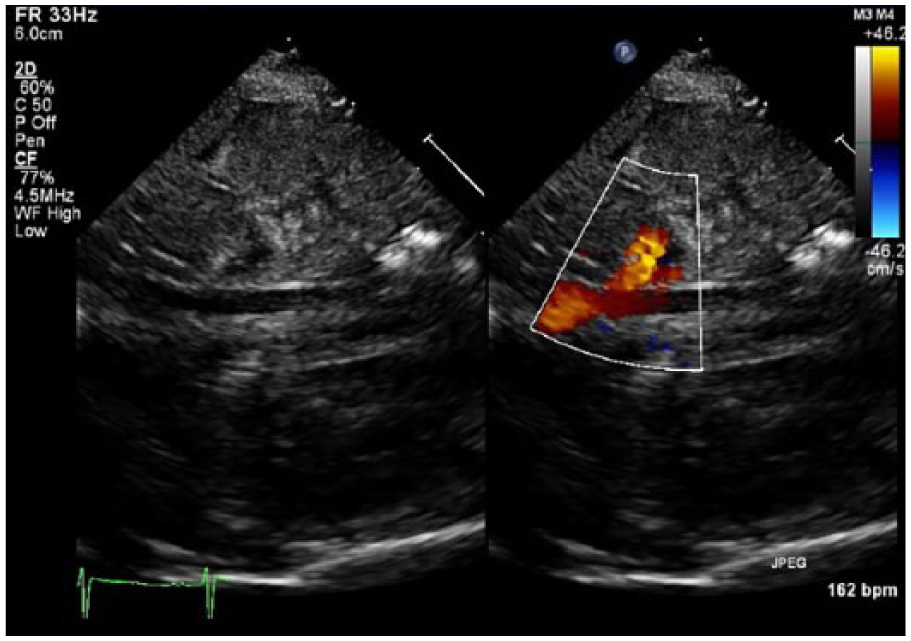
The kidney in a healthy infant has preserved systolic and diastolic flow. This is a color image of the renal artery added to the pediatric echocardiogram.
This image demonstrates staccato flow in the renal artery with absent end-systolic flow and absent diastolic flow. Loss of diastolic flow is seen in renal dysfunction, but loss of systolic flow is reserved for a loss of volume, as seen in a vascular steal from a large patent ductus arteriosus.
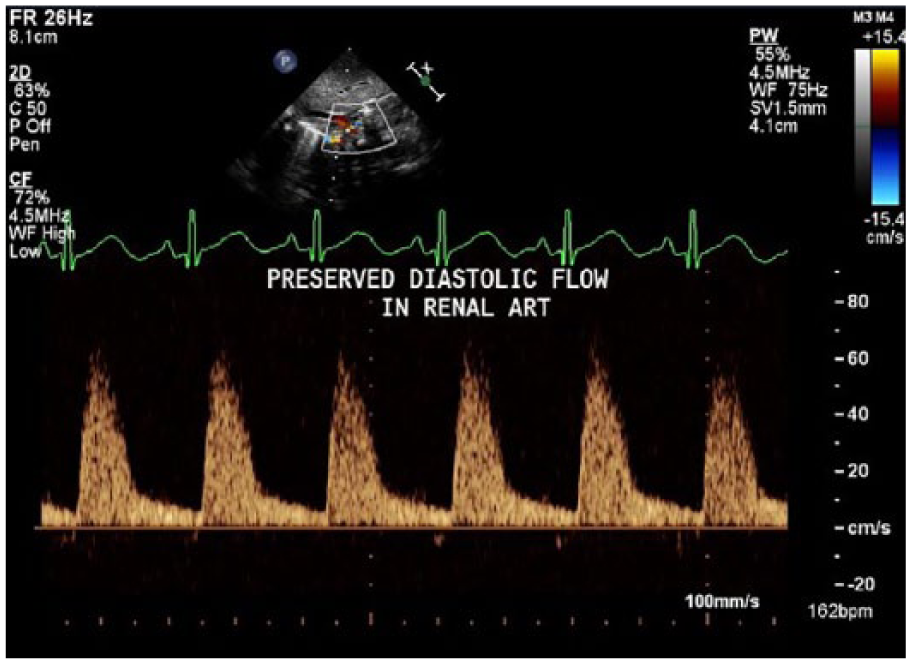
The kidney is a viable organ that requires blood throughout systole and diastole. This represents a normal renal arterial Doppler waveform.
All cases with hemodynamically significant PDAs by echocardiographic criteria and either absence of diastolic flow in the SMA and RA or staccato flow were treated and followed up at 48 hours to reassess ventricular function and the flow to the bowel and kidney. Staccato flow is defined as a Doppler waveform that drops to the baseline prior to the end of the systolic phase of the cardiac cycle.
Path analysis was conducted using SmartPLS v. 2.0 software to measure the magnitudes and statistical significance of the standardized path coefficients between the dichotomized PDAM scores, the four body weight categories, the respiratory status categories, and the RA and SMA Doppler waveform scores. A nonparametric path analysis using partial least squares (PLS) was applied instead of a parametric analysis such as regression analysis using ordinary least squares because all of the measurements were at the ordinal level and deviated from normality.
The statistical significance of the standardized path coefficients was indicated by two-tailed t-tests after bootstrapping the data with 5000 random subsamples to estimate the standard errors. If P < .05, then the path coefficient was assumed to be statistically different from zero. The effect sizes were indicated by R2, representing the proportions of the variance explained. A substantial effect size, irrespective of the P value, was assumed to indicate practical significance, implying that the results of the path analysis were meaningful and had practical implications in the context of medical research.25–27
Results
Figure 8 illustrates the sampling tree, indicating the results of echocardiography prior to treatment.
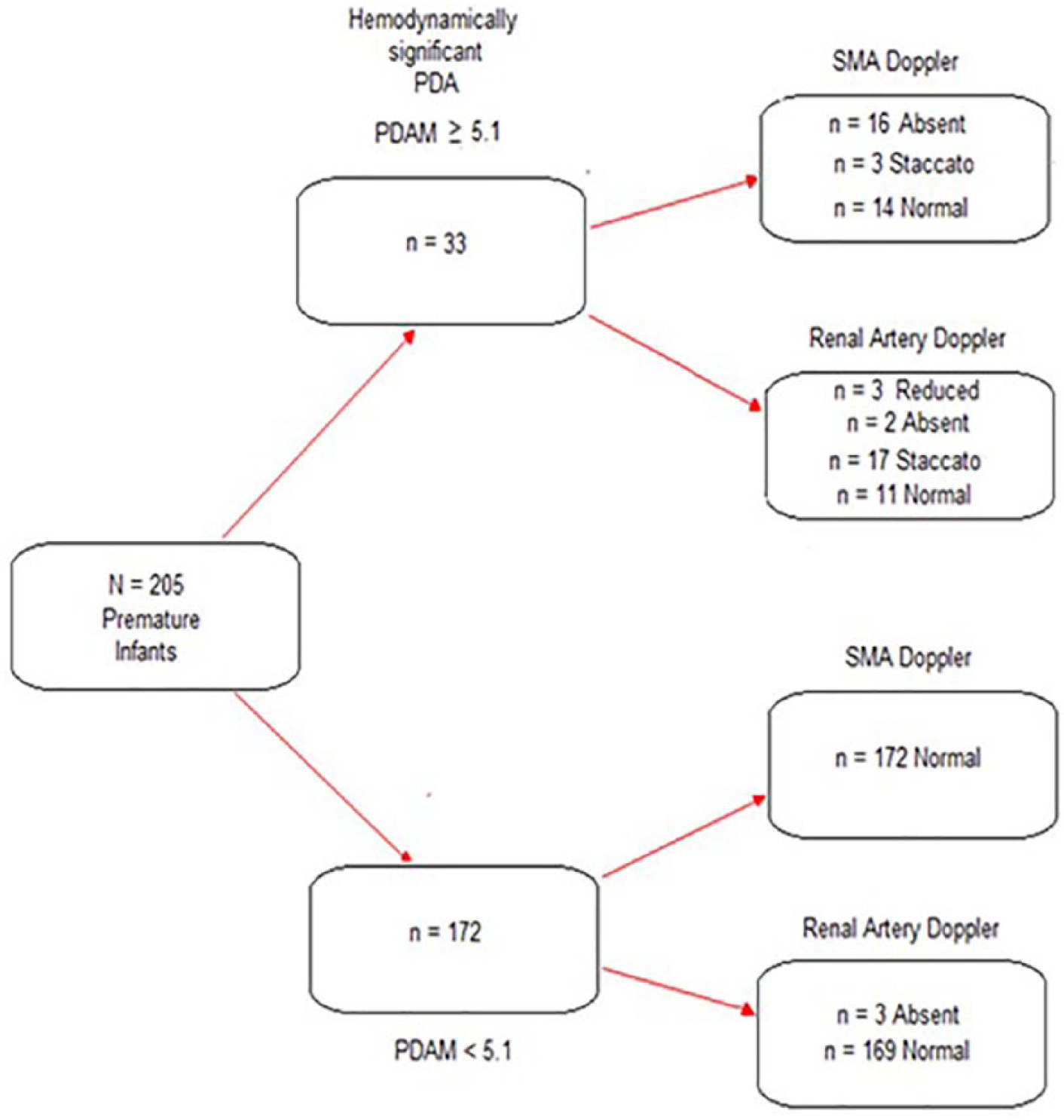
Sampling tree.
Of the target population of N = 205 premature infants, a small proportion (n = 33, 16.1%) were categorized as having a hemodynamically significant PDA (PDAM score ≥5.1), whereas the remainder (n = 172, 83.9%) had a PDAM score <5.1. Among the 33 infants with a PDAM score ≥5.1, most (n = 19, 57.6%) had staccato or absent SMA Doppler waveforms. Most (n = 19, 57.6%) also had staccato or absent RA Doppler waveforms. All of the 172 infants with a PDAM score <5.1 had normal SMA Doppler waveforms, and only a small proportion (n = 3, 1.7%) had absent RA Doppler waveforms. The 3 patients with absent diastolic flow were further worked up with renal Duplex Doppler examinations. The renal sonogram showed echogenic kidneys, decreased bladder volume, and increased resistive indices in the renal Doppler waveforms. All three patients had decreased renal function and decreased urine output that was deemed to be due to medical renal disease rather than a vascular issue. There was normalized flow to the kidneys following management of the medical renal disease.
Even in the setting of severe urosepsis, there is usually some diastolic flow to the kidneys. The resistive indices are high, but there is some diastolic flow, and in no case should there be staccato (absence of late systolic flow). This separates staccato flow to be exhibited only where there is a loss of volume, such as a vascular steal phenomenon described in this study. Diastole is a function problem, and systole is a volume problem.
Figure 9 illustrates the decision tree, indicating the outcomes of treatment for the 33 infants with PDAM score ≥5.1.
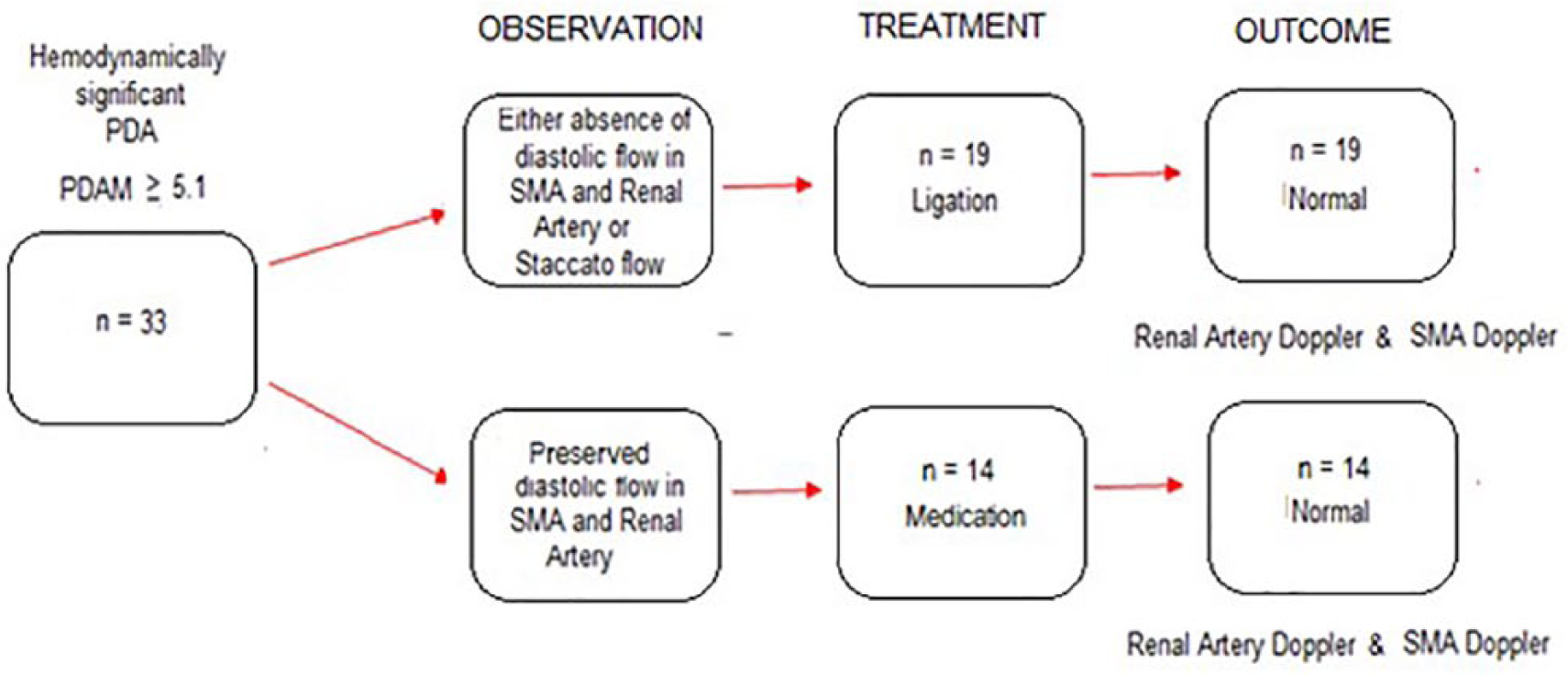
Decision tree.
More than half of the infants with PDAM ≥5.1 (n = 19, 57.6%) were observed to have either absence of diastolic flow in the SMA and RA or had staccato flow and were subsequently treated by ligation. The remainder (n = 14, 42.4%) were observed to have preserved diastolic flow in the SMA and RA and were subsequently treated by medication. The 48-hour follow-up sonographic observations indicated that the RA and SMA Doppler waveforms had returned to normal after treatment.
The results of the PLS–structural equation modeling path analysis are illustrated in Figure 10. The clinical significance of the PLS path model was reflected by substantial effect sizes. Most of the variance in the RA and SMA Doppler waveforms were explained (R2 = 0.577 and 0.530, respectively) by the PDAM scores, body weights, and respiratory status of the infants. A statistically significant (P < .05) positive path coefficient (β = .720) indicated that infants with a hemodynamically significant PDA (PDAM score ≥5.1) tended to have an abnormally high renal Doppler waveform (where 0 = normal, 1 = RDF, 2 = ADF, 3 = staccato and/or absent). A statistically significant path coefficient (β = .687) indicated that infants with a hemodynamically significant PDA also tended to have an abnormal high SMA Doppler waveform (where 0 = normal, 1 = RDF, 2 = ADF, 3 = staccato and/or absent). A statistically significant (P < .05) negative path coefficient (β = −.190, –.136, and –.115) indicated that infants with a low body weight tended to have a hemodynamically significant PDA (PDAM score ≥5.1) as well as an abnormal RA and SMA Doppler waveform. There were, however, no statistically significant path coefficients related to respiratory distress. This was indicated by the small path coefficients that were not different from zero (β = .080, –.019, and .008, respectively) between the PDAM scores, the RA, and SMA Doppler waveforms versus the four levels of respiratory status (where 0 = room air, 1 = nasal cannula, 2 = CPAP, and 4 = intubated).
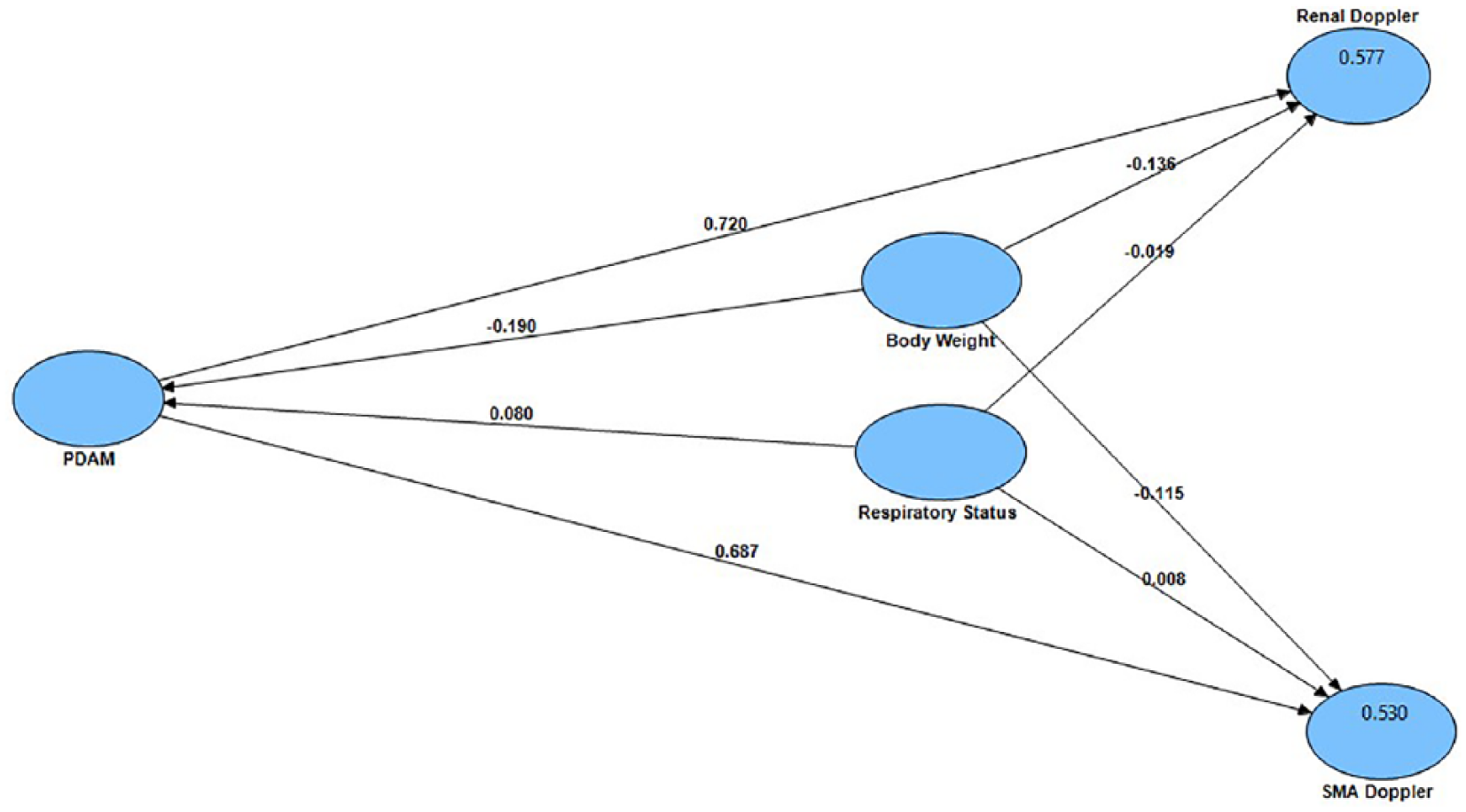
Partial least squares–structural equation modeling path analysis to evaluate the relationships between PDAM scores, body weight, respiratory status, renal Doppler waveform, and supermesenteric artery Doppler waveform.
Limitations to the Study
The SMA Doppler waveform changes with prandial state. The SMA Doppler was added as a conscious choice because of the very high incidence of necrotizing enterocolitis in preterm infants. The hypothesis that a vascular steal from a PDA may be a contributing factor to the incidence of necrotizing enterocolitis was the rationale for including the SMA Doppler. The SMA Doppler may need to be coordinated with the care team to be taken in the fasting state only to provide consistency.
Conclusion
The addition of subdiaphragmatic vessel interrogation (SMA and RA) in all infants with a PDA, and specifically a hemodynamically significant PDA, provides a quantitative way to determine whether a vascular steal is occurring secondary to the PDA. The spectral Doppler analysis of the abdominal vessels suggests that diastolic flow relates to function and systolic flow to the bowel and kidney volume. The addition of subdiaphragmatic vessel interrogation in conjunction with standard echocardiographic assessment of PDAs improves diagnostic accuracy and will assist physicians with clinical management of premature infants with PDAs.
Supplemental Material
How_to_measure_PDAM_plus_abd_Doppler – Supplemental material for Addition of Subdiaphragmatic Vessels in Evaluation of Hemodynamically Significant Patent Ductus Arteriosus in Premature Infants
Supplemental material, How_to_measure_PDAM_plus_abd_Doppler for Addition of Subdiaphragmatic Vessels in Evaluation of Hemodynamically Significant Patent Ductus Arteriosus in Premature Infants by Joy Guthrie in Journal of Diagnostic Medical Sonography
Supplemental Material
Visual_Abstract – Supplemental material for Addition of Subdiaphragmatic Vessels in Evaluation of Hemodynamically Significant Patent Ductus Arteriosus in Premature Infants
Supplemental material, Visual_Abstract for Addition of Subdiaphragmatic Vessels in Evaluation of Hemodynamically Significant Patent Ductus Arteriosus in Premature Infants by Joy Guthrie in Journal of Diagnostic Medical Sonography
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
