Abstract
To determine if socioeconomic status is another risk factor in the development and treatment of breast cancer, the records of 250 women, equally divided between those with and without breast cancer, were studied at an outpatient clinic. The frequency of sonographically detected breast cancer increased systematically with respect to age, from 0% at age <30 years to 70.5% at age ≥50 years. The frequency of detection was higher among white (non-Hispanic) women, 69.4%, compared with nonwhite (Hispanic, Black, Asian) women, 42.3%. Breast cancer detection was higher (55.3%) among the lowest income group (<$40,345) than among higher income groups (45.6%) and higher among patients with Medicare (89.2%) and Medicaid (50%) compared with private, other, or no medical insurance (38.1%–44.4%). Detection was higher among women with a family history of breast cancer (55.0%) compared with no family history (46.7%) and higher among nulliparous women (100%) than nonnulliparous women (29.1%). These data suggest that aspects of socioeconomic status may be as predictive of sonographically detected breast cancer as age, ethnicity, and nulliparity.
Socioeconomic status (SES) has always played a role in the overall well-being and health status of adult women. 1 Poverty has been shown to have a profound impact on stress levels, heart disease, dietary control, and hygiene. There are also well-known risk factors for the development of breast cancer, notably family history, advanced maternal age, nulliparity, and obesity. 2 Sonography, with or without mammography, has long been used in the detection of breast masses, especially palpable breast lesions (Figures 1 and 2). In fact, sonography is sometimes the only modality available to those without insurance to detect breast masses. 3 Sonography, however, can be extremely operator dependent. In order for it to be an effective technology for screening patients at risk for breast cancer, the scans must be performed by highly competent sonographers and interpreted by highly competent physicians. The goal of this study was to evaluate whether socioeconomic factors were as predictive as family history, age, and ethnicity for sonographically detected breast cancer.
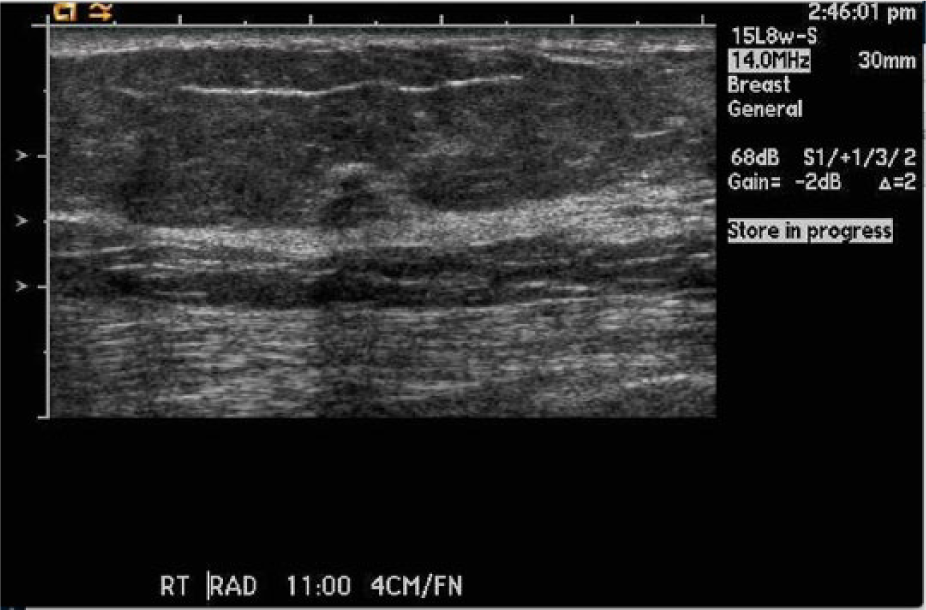
Gray-scale image of a breast mass that had characteristics of a malignant neoplasm that was not found on initial physical examination but was identified by a credentialed breast sonographer.
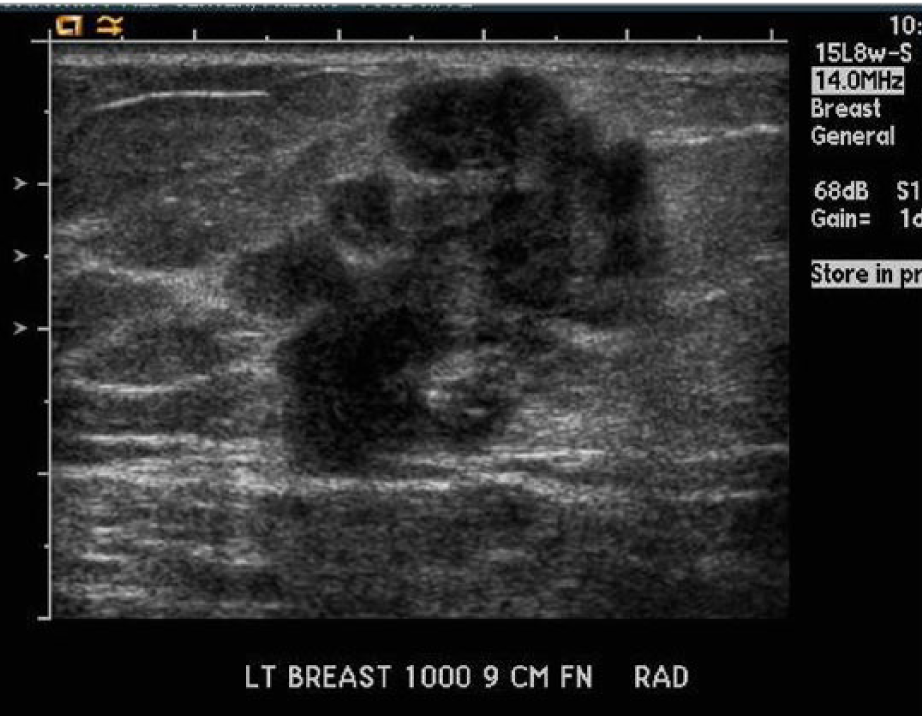
Gray-scale image of a malignant BI-RADS 4 (Breast Imaging–Reporting and Data System) mass that was identified in an advanced stage.
Methods
A retrospective data analysis was performed on women with and without breast cancer. Institutional review board approval was obtained prior to any data collection. Included as possible participants were all patients with and without sonographically detected breast masses reported at an outpatient imaging center in the western United States between March 2011 and October 2013. Pregnant women were excluded from the data sample. The sample ultimately consisted of 250 women between the ages of 20 and 83 years, equally divided into 125 with sonographically detected breast cancer and 125 with no observed cancer. The frequency distributions of women with and without detected breast cancer were classified with respect to their age, ethnicity, income, insurance, family history of cancer, and parity. Several of the already known risk factors for women diagnosed with breast cancer were compared with those of the participants, as well as the average yearly income based on the region in which they lived in, as classified by zip code. 4 The available data did not allow any classification based on the participants’ citizenship or immigration status. All sonographic examinations were performed by American Registry for Diagnostic Medical Sonographers–credentialed breast sonographers, and data were collected retrospectively from breast sonograms performed at a single location. A binary logistic regression model was constructed to predict the sonographic detection of breast cancer (coded by 1) relative to no detection (coded by 0) using age, ethnicity, income, insurance, and family history as independent variables. The model excluded nulliparity because binary logistic regression cannot include categorical variables that contain zero frequencies.5–9 All raw data were recorded on a Microsoft Excel spreadsheet prior to statistical analysis by using SPSS version 17 (SPSS, Inc., an IBM Company, Chicago, Illinois).
Results
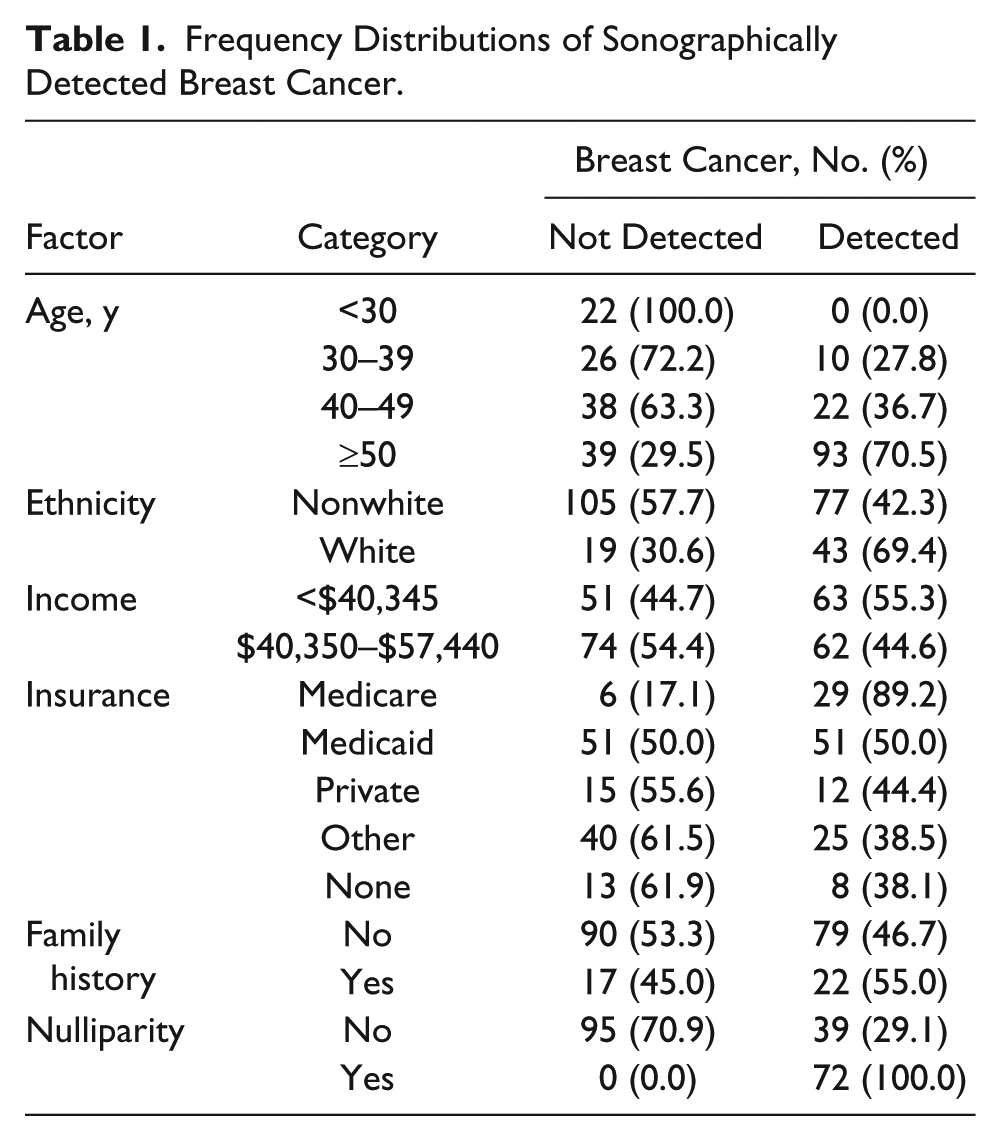
The frequency distributions of women with and without detected breast cancer are summarized in Table 1. The frequency of sonographic detection of breast cancer increased systematically with respect age, from 0% at age <30 years to 70.5% at age ≥50 years. In addition, the frequency of detection was (a) higher among white (non-Hispanic) women (69.4%) than among nonwhite (Hispanic, black, Asian) women (42.3%); (b) higher in the lowest income (<$40,345) group (55.3%) than in higher income groups (45.6%); (c) higher among patients who had Medicare (89.2%) or Medicaid (50%) compared with those with private, other, or no medical insurance (range, 38.1%–44.4%); (d) higher for women with a family history of breast cancer (55.0%) compared with those with no family history (46.7%); and (e) higher for women who were nulliparous (100%) compared with those who had given birth (29.1%). None of the women negative for malignancy were nulliparous. The binary logistic regression model computed to predict the sonographic detection of breast cancer is presented in Table 2.
Frequency Distributions of Sonographically Detected Breast Cancer.
Logistic Regression Model to Predict Sonographically Detected Breast Cancer.
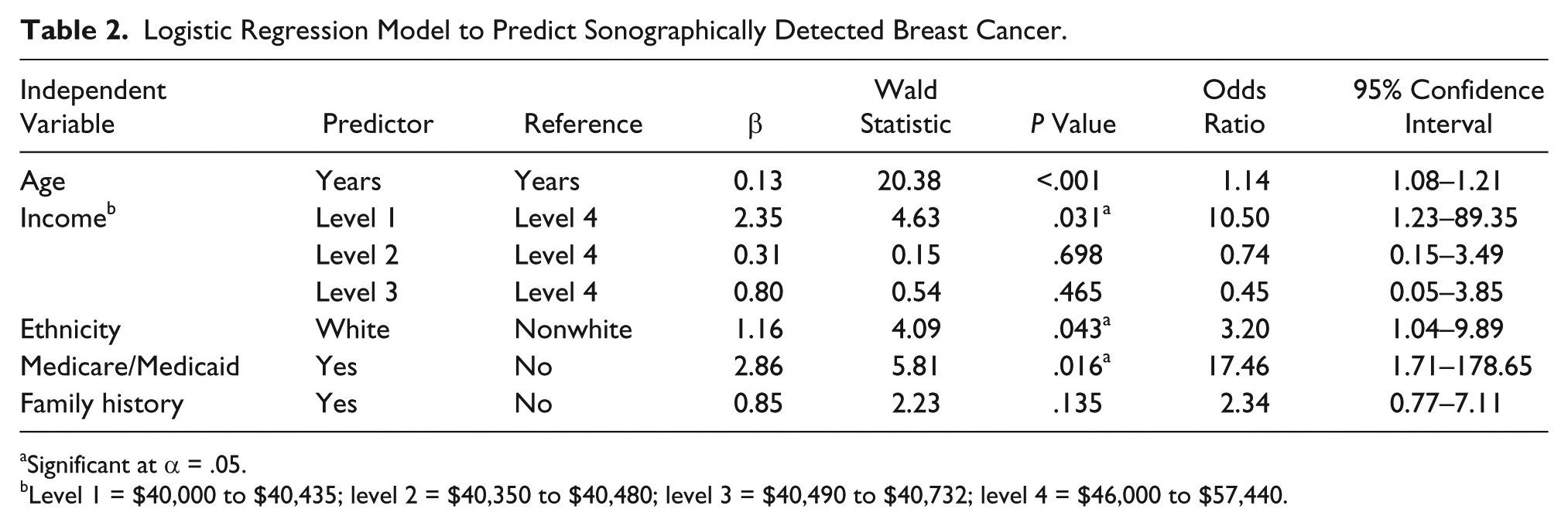
Significant at α = .05.
Level 1 = $40,000 to $40,435; level 2 = $40,350 to $40,480; level 3 = $40,490 to $40,732; level 4 = $46,000 to $57,440.
Family history was not a significant predictor at α = 0.05. The four independent variables that predicted the likelihood of sonographic detection of breast cancer at α = .05 were age, income, ethnicity, and receipt of Medicare/Medicaid. The calculated ratios (ORs) indicated that the likelihood of sonographic detection of breast cancer (a) increased by 1.14 for every one-year increase in age, (b) increased by 3.20 in white (non-Hispanic) women relative to nonwhite (Hispanic, black, or Asian) women; (c) increased by 10.50 in the lowest income group ($40,000–$40,435) relative to the highest income group ($46,000–$57,440); and (d) increased by 17.46 among women who received Medicare/Medicaid relative to those who did not.
Discussion
Socioeconomic status (SES) has always played a role in the overall well-being and health status of adult women. 1 Poverty has a profound impact on stress levels, heart disease, dietary control, and hygiene. Women who live in poverty may be nondiagnosed or underdiagnosed merely by their lack of access to health care. 10 If it is determined that poverty itself may be a risk factor and then affirmed by additional research studies, then a concerted effort to try and increase screening for this underserved population should be employed.
There are known risk factors for the development of breast cancer. At the top of the list are family history, advanced maternal age, nulliparity, and obesity. 2 Sonography, with or without mammography, has long been used for the detection of breast masses, especially palpable breast lesions; sonography may be the only modality available to those without insurance for such examinations. 3 Sonography, however, is extremely operator dependent. In order for it to be an effective technology for screening patients at risk for breast cancer, the scans must be performed by highly competent sonographers and interpreted by highly competent physicians.
Three of the primary risk factors that may be used to predict the incidence of breast cancer in women are age, family history, and ethnicity.2,11 Increasing age is one of the strongest biological risk factors for breast cancer. Although breast cancer can occur early in life, in general, it is a disease of aging. For women in their 30s, the risk is approximately 1 in 250, whereas for a woman in her 70s, it is approximately 1 in 30. Most breast cancers are diagnosed after menopause; around 75% of breast cancers occur after 50 years of age. Age is considered a likely surrogate for DNA damage accumulated during life. 2
In 2012, the regional hospital used in this study published the Cancer Center Annual Report. 12 This report showed that age at breast cancer diagnosis was highest (28.4%) in women who were aged 50 to 59 years. The survival rate tended to be lower for women who were diagnosed before age 40 years. Cancer detected at a younger age tended to be a more aggressive form of cancer that did not respond well to treatment. Women who were diagnosed before age 40 years made up 6.8% of women diagnosed with breast cancer. 12
Overall in the United States, white women are slightly more likely to develop breast cancer than are African American women. However, in women younger than 45 years, breast cancer is more common in African American women. Asian, Hispanic, and Native American women appear to have a lower risk of developing and dying of breast cancer.13,14 Women with a sister or daughter with breast cancer are, on average, at twice the risk of those with no affected first-degree relative.2,15
Breast cancer death rates also vary by race and ethnicity.13,16–18 Between 2003 and 2007, Asian American/Pacific Island women accounted for the lowest death rate, with only 12.2 deaths per 100,000 women. African American women had the highest death rate due to breast cancer, with 32.4 deaths per 100,000 women, even though their incidence of breast cancer was lower compared with white (non-Hispanic) women. This is largely attributed to diagnosis at later stages of breast cancer and poorer stage-specific survival.16,17 Research further suggests that racial disparities in cancer mortality are driven in large part by differences in SES. Richardson et al 18 studied whites, African Americans, and Hispanics who were diagnosed with breast cancer. Conducted between 1977 and 1985, the study looked at the risk of late-stage diagnosis and long duration of symptoms before diagnosis with regard to ethnicity, SES, age, and year of diagnoses. They reported that race and SES were predictive for cancer detection at more advanced stages, and more of an effort should be made to reach this specific population.
With regard to survival after being diagnosed with breast cancer, Asian American/Pacific Island women have the highest 5-year survival rate (90.3%). African American women have the lowest 5-year survival rate (77.5%), indicative that they, more than any other ethnic group, have not only the highest likelihood of dying of breast cancer but of dying earlier. These survival statistics reflect all the differences in breast cancer stages at time of detection, type of breast cancer, and the amount of access and opportunity these women have to treatment. 13
The associations between lifestyle-related factors and breast cancer risk are more difficult to evaluate because, unlike age, family history, and ethnicity, lifestyles are not biologically or genetically fixed. A woman’s lifestyle may change dramatically over the course of her life. Some lifestyle factors, such as socioeconomic status, are not necessarily biologically or genetically linked to the root causes of a disease but may act as proxies for a variety of other causative factors. 1
Breast cancer is known to occur more frequently in affluent and Western populations and in subpopulations of higher socioeconomic status within individual countries. 11 Female breast cancer incidence has been shown to be strongly related to socioeconomic status and is one of the few cancers in which incidence rates are lower for more disadvantaged women. In the United Kingdom, there is a clear trend of decreasing rates from the least to the most deprived women. 19 This suggests that lifestyle factors related to affluence may be associated with increased risk of breast cancer; however, socioeconomic status may be confounded with ethnicity. For example, African American women are three times more likely to live in poverty than are white women. American Indian/Alaska Native, Hispanic, Pacific Island, and Native Hawaiian families are also more likely than white and Asian families to live in poverty. 20 Although breast cancer death rates are known to be higher among women with lower socioeconomic status, this is probably because screening rates tend to be lower among poor women. Breast cancer knowledge and screening behaviors appear to be lowest among low-income, elderly black women. 21
The reason why different races seek the help of medical facilities may have to do with not only their access to health care but also their cultural beliefs and trust or mistrust of physicians. A qualitative study conducted in 2002 looked into what factors may be limiting African Americans with cancer to seek medical help. 22 The study found that there were many cultural factors along with SES. The cultural factors included lack of education and misinformation about cancer, lack of trust of the medical community, privacy issues, religious beliefs, and fear of stigma associated with their disease. These worries, coupled with their lack of access to health care, only confounds the situation.
Breast cancer death rates have been steadily declining since 1975, but the most notable and earliest decline was from women who were living in more affluent areas.13,14,21 However, in 1990, breast cancer death rates were lower in women living in poor areas compared with those living in affluent areas. This high mortality rate for women in affluent areas may reflect a higher breast cancer incidence of women from higher SES. From 1975 to 1977, cancer death rates for women living in poor areas where down by 7% compared with women from more affluent areas. From 2003 to 2007, the trend had reversed, and women in poor areas were 7% more likely to die of cancer than women in affluent areas. 21 Even with advances in medicine and technology, long-term survival rates are still low among women who live in less affluent areas, and not all segments of the population have benefited equally from medical advances, as reflected in survival disparities by SES. 13 Women in less affluent areas have less access to new medical care advances and less access to medical options. A study conducted showed that the cost for breast cancer treatment ranged from $16,226 to $39,305, relative to the type of care received. Mastectomy was the least expensive option, with breast-conserving surgery the most expensive since it involves multiple surgeries.14,15
Sprague et al 23 studied the survival rate after breast cancer detection across SES and showed that compared with college graduates, women who had no education beyond high school were 1.39 times more likely to die of breast cancer. Their multilevel analysis included the use of mammography screening, disease stage at diagnosis, lifestyle factors, education level, and decreased household income and showed the significant increase in breast cancer mortality with a 95% confidence interval of 1.10 to 1.76. A study by Farley and Flannery 24 from 1982 to 1985 found that lower SES women, compared with higher SES women, had a higher rate of expected deaths from breast cancer of 24.6 vs 19.7 per 100,000. Of those deaths, most were considered preventable by early detection. The percentage of preventable deaths for lower SES women was 2.5 times greater than for women with a higher SES.
The previously mentioned Cancer Center Annual Report also reviewed the breakdown of breast cancer by ethnicity. 12 The highest incidence of breast cancer detection was in white (non-Hispanic) women (60.9%); the next highest frequency was in Hispanic women (27.6%). These statistics suggest that white women, who are likely to come from more affluent backgrounds, can afford the cancer screening process, while other ethnicities may be underreported because of lack of access and/or health care insurance. 12 The Centers for Disease Control and Prevention found that the percentage of uninsured people between January and March 2008 was 30.4% Hispanic, 9.9% non-Hispanic white, and 17.0% non-Hispanic black. 25 According to the California poverty level, Latinos and African Americans are more likely to live in poverty compared with whites and Asians. Poverty levels by race in California, where this study was conducted, are reported as follows: Latino (23.6%), African Americans (24.2%), Asians (12.6%), and whites (9.8%). 26
Cancer screening has also shown to be relegated to a higher SES. A study by Hiatt et al 27 showed that 76% of women who were English speaking, white, and older than 40 years had at least one mammogram. In addition, 88% of the women also received a clinical breast examination. This is in stark contrast to non-English-speaking Latinas (56%) and Chinese women (32%). Maintenance screenings also varied from 7% of non-English-speaking Chinese to 53% of African Americans. African Americans are known to have less access to medical care and are less likely to undergo breast cancer screening. 17 This research found that the strongest predictor of a woman’s screening behavior was in direct correlation to the type of insurance the woman had. Women with private health insurance were the ones who sought maintenance screening and regular use of medical services. 27
In the state of California, counties are the health care providers of last resort for people who cannot afford health care. The counties insure the people who would not otherwise be covered by the statewide program Medi-Cal. At the time this retrospective study was conducted, there were several programs available for people who come from low socioeconomic backgrounds that allow them to apply for aid in health care. One program specifically through the community hospital where this study was conducted is the Medically Indigent Services Program (MISP). MISP allows people who do not qualify for Medicare or Medicaid to apply for this insurance. People who qualify for MISP may be undocumented, legal, or permanent residents or US citizens aged 21 to 65 years who meet the income/expenses requirements. Programs like these provide a vital health care safety net for the state’s underinsured and allow patients to get the care they need but to which they otherwise would not be privy. 28 It should be noted that this research was conducted prior to enactment of the Affordable Care Act. With the enactment of the Affordable Care Act, it may be worthwhile to repeat this study to see if this underserved population will receive the benefits they need from the new health care system.
Conclusion
The binary logistic regression model provided statistical evidence to confirm that white (non-Hispanic) ethnicity and increasing age are significant biological risk factors for breast cancer. The model also provided new evidence to conclude that socioeconomic status may be as predictive of sonographically detected breast cancer as age, ethnicity, and nulliparity. The highest odds ratios predicting the likelihood of sonographic detection of breast cancer were among the lowest income group relative to the highest income group (OR, 10.50) and among women who were in receipt of Medicare/Medicaid (OR, 17.46) compared with those with private, other, or no medical insurance. Because poverty appears to be a risk factor, a concerted effort to try and increase breast cancer screening for this underserved population should be employed.
Footnotes
Acknowledgements
The author thanks the sonographers and mammographers at the outpatient imaging clinic at our community regional medical center for all of their help. A special recognition to Pam Denton and Alma Luna for all their help.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported in part by a grant from the Sonography Research Institute of the Society of Diagnostic Medical Sonography Foundation.
