Abstract
Prenatal diagnosis of congenital heart disease (CHD) during routine obstetric sonography has been aptly named the sonographer’s Achilles heel. Although CHD occurs more commonly than any other major congenital abnormality, the detection rate remains low. The goal of this study was to improve the prenatal diagnosis of CHD during routine obstetric sonography through the development and implementation of a simple and effective screening protocol.
Keywords
Introduction
Congenital heart disease (CHD) comprises a wide variety of abnormalities, from relatively benign ventricular septal defects to life-threatening complex malformations of the outflow tracts or the heart chambers. Cardiac abnormalities are the most common of all congenital malformations, with an incidence of 75 cases per 1000 live births. 1
Despite improvements in treatment and surgical advances that have resulted in decreased mortality, CHD is still a major cause of death in infancy. 2 Heart defects comprise one quarter to one third of all birth defects and are a large contributor to infant mortality. 3 Not all forms of major CHD will be evident at birth or in the neonatal period.4,5 Even complex CHDs commonly go undetected until after the infant is discharged home, leading to avoidable morbidity and mortality.6,7 Kuehl et al. 8 reported that during the first week of life, one quarter of deaths due to CHD occurs before the diagnosis of CHD is made, and occasionally the cardiac lesion is never recognized.
Prenatal detection of CHD is beneficial as it may make it possible to improve preoperative conditions, postoperative survival, and neurological outcomes. It also allows for perinatal preparation for both families and care providers. Most major CHD cases can be detected prenatally in experienced hands.9,10 However, despite the wide use of prenatal ultrasound and the fact that screening for CHD has been well established for the past 25 years, the proportion of CHD cases diagnosed prenatally remains low.1,11–12
Many attempts have been made to address this shortcoming. In 1977, Lee et al. 14 described the use of sonography to identify fetal cardiac structure. The literature since that time is replete with articles on the failure of prenatal sonographic screening for detection of CHD, with a reported detection rate that varies widely from 4.5% to 63%.15,16 Continuing education for the frontline screener has been suggested by many to be an effective means of improving detection rates; however, each article cites the expense of such a program as a limiting factor.17,18
The approach described in this study differed from others in that it focused the sonographer’s attention on the fundamentals of scanning to determine if a fetal heart was normal or abnormal. This study describes the process of creating a new fetal heart screening protocol that has been successfully implemented at the population level. Sonographers along with team members from three disciplines developed an educational intervention that was delivered to all sonographers in the province. The focus of the study was the development of a protocol that allows sonographers of every skill level to identify normal cardiac anatomy consistently and confidently. This approach of seeking 100% specificity in identifying a short target list of complex congenital cardiac abnormalities is not described in the literature to date.
Materials and Methods
A-four part plan was devised to achieve the goals of the study:
Development of a simple but highly focused fetal heart screen protocol to identify the critical anatomical markers that would enable the sonographer to decide if the heart was normal or abnormal.
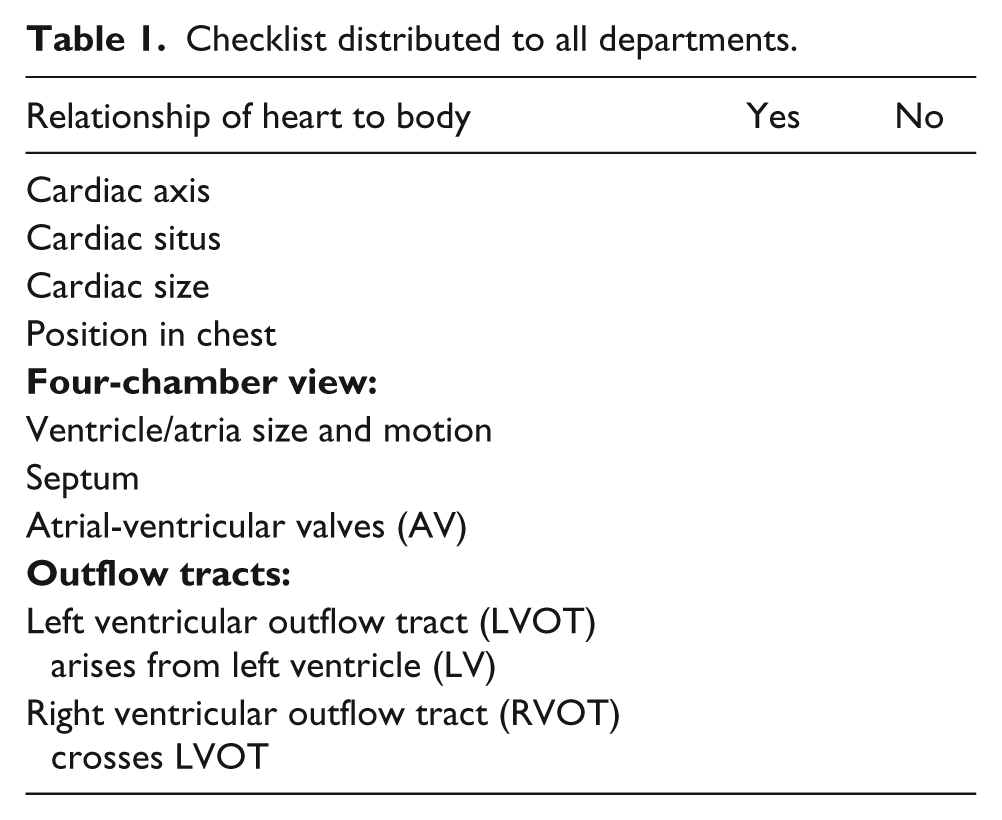
Creation of a checklist that sonographers were asked to use on every examination.
A continuing education program that provided several educational events to teach sonographers how to follow the new protocol, which included education on how to perform outflow tract views.
Initiation of a feedback loop to the original sonographer of both positive and negative findings. In 2004 a survey was sent to all obstetric sonographers in the province (16 sonography departments, including urban and rural hospitals, plus two fetal assessment units). Current practice relating to prenatal cardiac sonography was evaluated, including the views of the fetal heart obtained, the sonographer level of confidence in performing a cardiac screen, and the perceived success rate in obtaining these views. The survey asked sonographers about the proportion of cases in which they routinely evaluated the fetal heart by looking at the four-chamber view and if they included assessment of the outflow tracts.
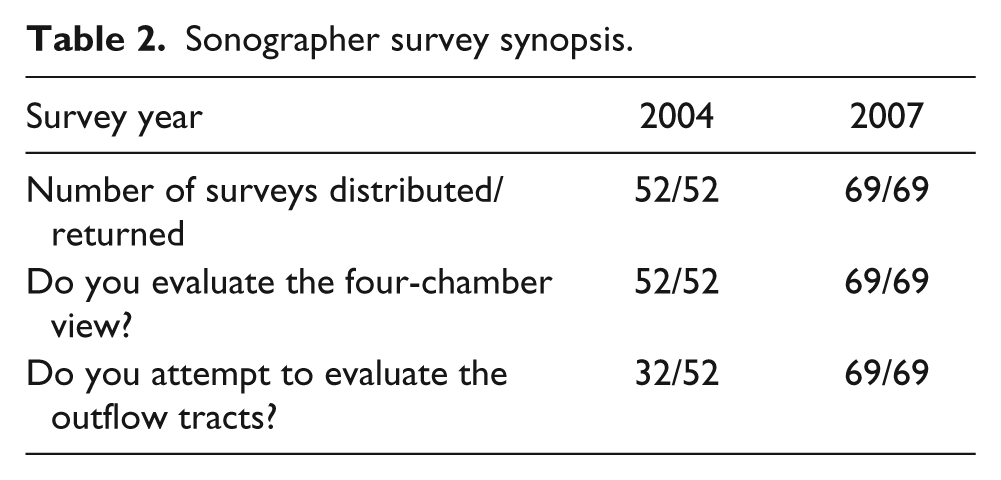
Information gathered in the survey indicated that less than half of the sonographers in Manitoba were routinely evaluating outflow tracts during their fetal heart screen. This information was used to create and deliver an educational intervention that focused on teaching sonographers how to evaluate outflow tracts and how to improve their evaluation of the four chambers. The education was geared toward the sonographers but also included all radiologists and obstetric perinatologists, with the anticipated outcome being a change in the practice of fetal heart screening across our province. The survey was readministered in 2007 following the educational intervention, which was delivered to all practicing sonographers in the province. Chi-square analysis (Fisher’s exact test) was used to test the difference in the distribution of cases receiving a complete evaluation (i.e., four-chamber view with outflow tracts) before and after the educational intervention.
The intervention included instruction on the critical aspects of a fetal heart screen, the four-chamber view, and outflow tract evaluation. Sonographers were asked to use the developed guidelines when attempting the four-chamber view and to evaluate outflow tracts in all cases. The protocol included that each screen should include video clips of both views.
The Bannatyne Campus Research Ethics Board of the University of Manitoba (the equivalent to the Institutional Review Board, or IRB, in the United States) granted ethical approval for this study prior to its initiation.
Fetal Heart Screen
The fetal heart screen protocol is focused on ruling out four-chamber and conotruncal lesions, particularly those that are ductal dependent. This includes the two categories of lesions identified by the pediatric cardiologist as causes of the highest neonatal morbidity and mortality: single ventricle lesions such as hypoplastic left heart syndrome and transposition of the great arteries. The protocol focused on accurately detecting or excluding these two abnormalities in every examination. The first step in the protocol emphasized the importance of the fetal position. This principle is based on a review of previously missed cases, which identified that the majority of “misses” occurred when the fetus was in a “bad” position (i.e., lying prone) during the fetal heart screen. Sonographers now begin the obstetric examination by looking at the fetal position, so that if the fetus is in a position that makes the heart screen effective (that is, lying supine) they take advantage of this (Figure 1). If the fetus is lying prone, the noncardiac components of the fetal scan are performed first, giving the fetus time to roll into a better position. The education materials provided included comparison images and videos of the same CHD, where the abnormality is seen quite easily when the fetus is lying supine but missed when the fetal back is up.
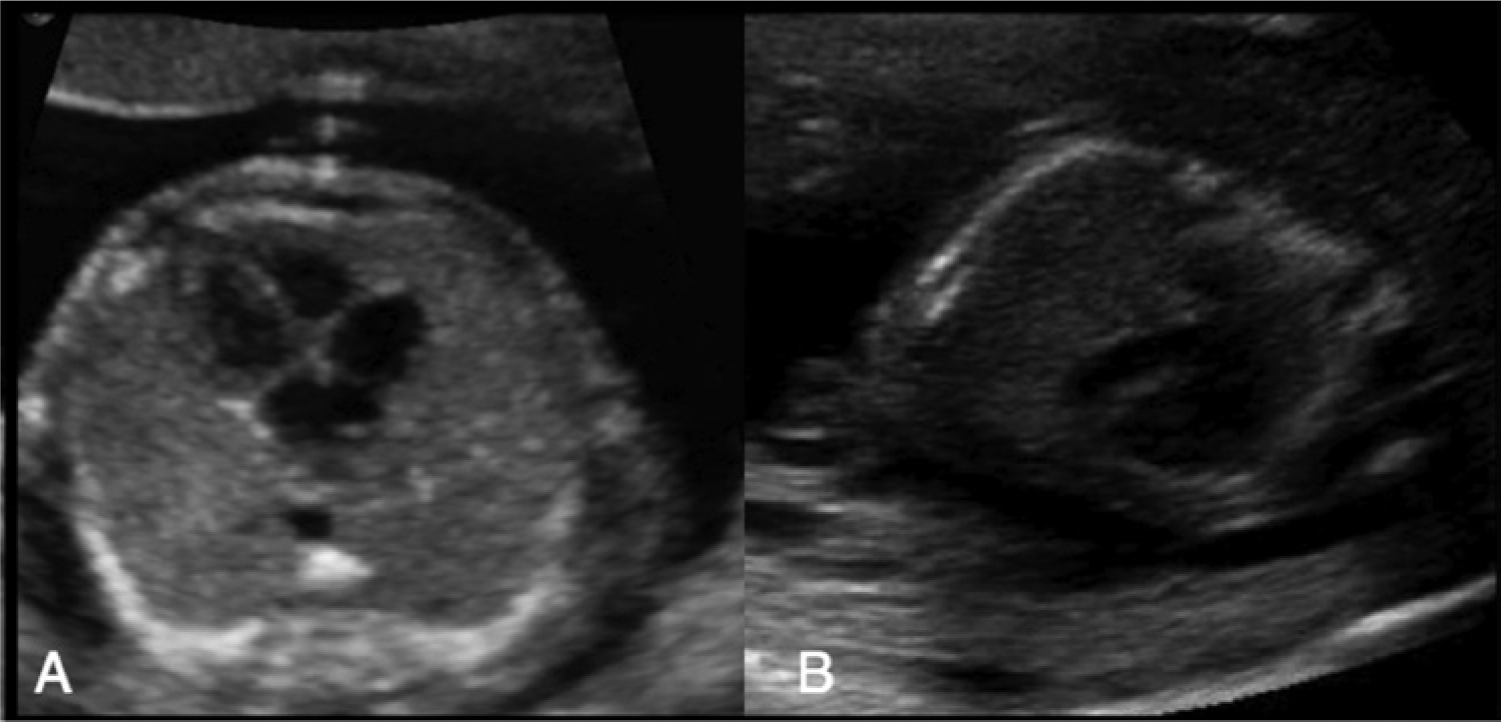
Two images of a normal heart. In (A), the fetus is lying supine and the heart can be clearly seen as normal. In (B), the fetus is lying prone, compromising evaluation of the heart.
The second step in the protocol was to ensure that the heart is only assessed when the chest is round (i.e., ribs create a circular shape, not oval). The highest yield occurs when the heart is assessed by scanning directly through the front of the fetal chest; however, views between the 2 o’clock to 10 o’clock positions are acceptable (Figure 2). Once the fetus is lying supine and the chest is displayed as round, the sonographer is then able to assess the relationship of the heart to the body for cardiac size, axis, situs, and position in the chest. The third step was the evaluation of the heart for atria and ventricle size and symmetry, the position and motion of the valves, and the crux of the heart. If the chest as imaged is round, and the sonographer is scanning through the front of the fetus, the comparison of ventricle and atria size will be accurate. If the chest view is not round, it is possible for abnormal hearts to appear normal (Figure 3).
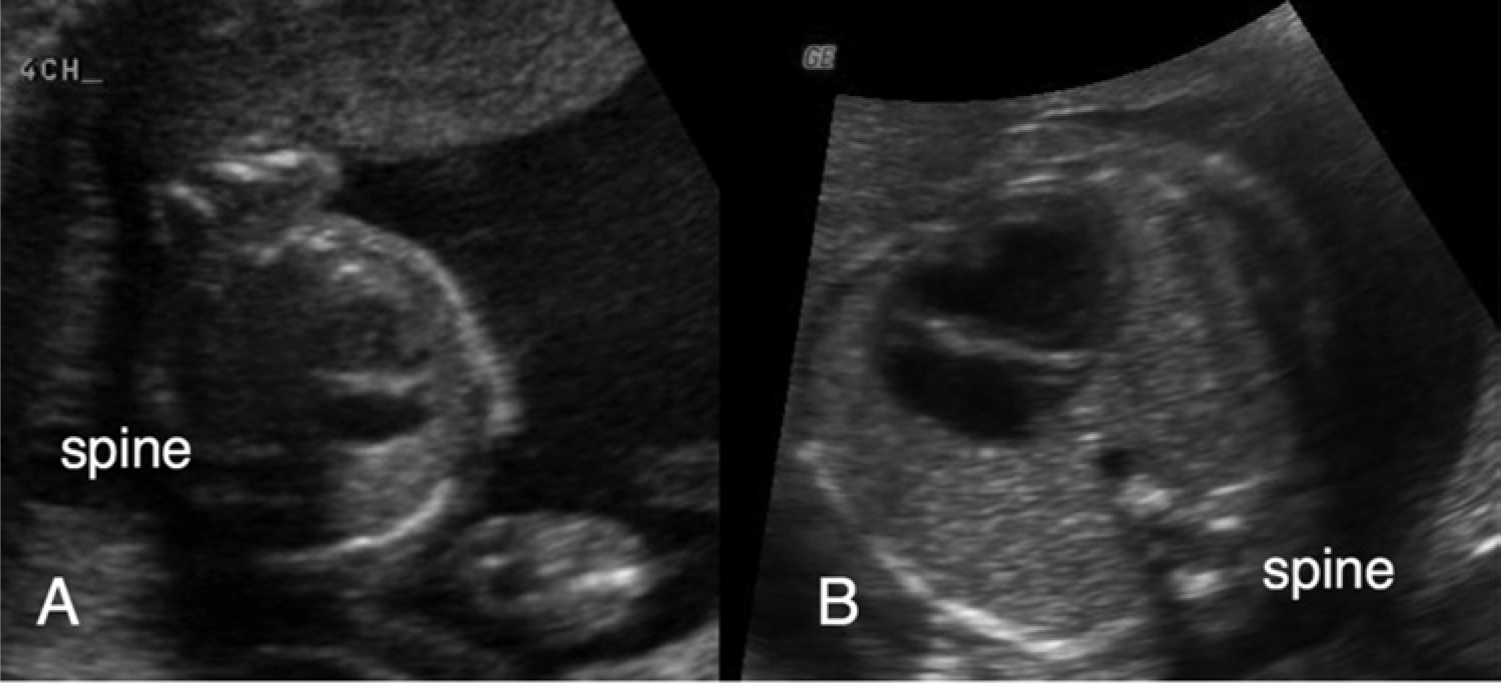
Two images of the same fetal heart. In (A) the fetus is lying on his/her left side limiting evaluation of the heart. In (B), the fetus is now supine, turned in the 10 o’clock position, allowing the sonographer a clear view of the heart, which can now be identified as abnormal.
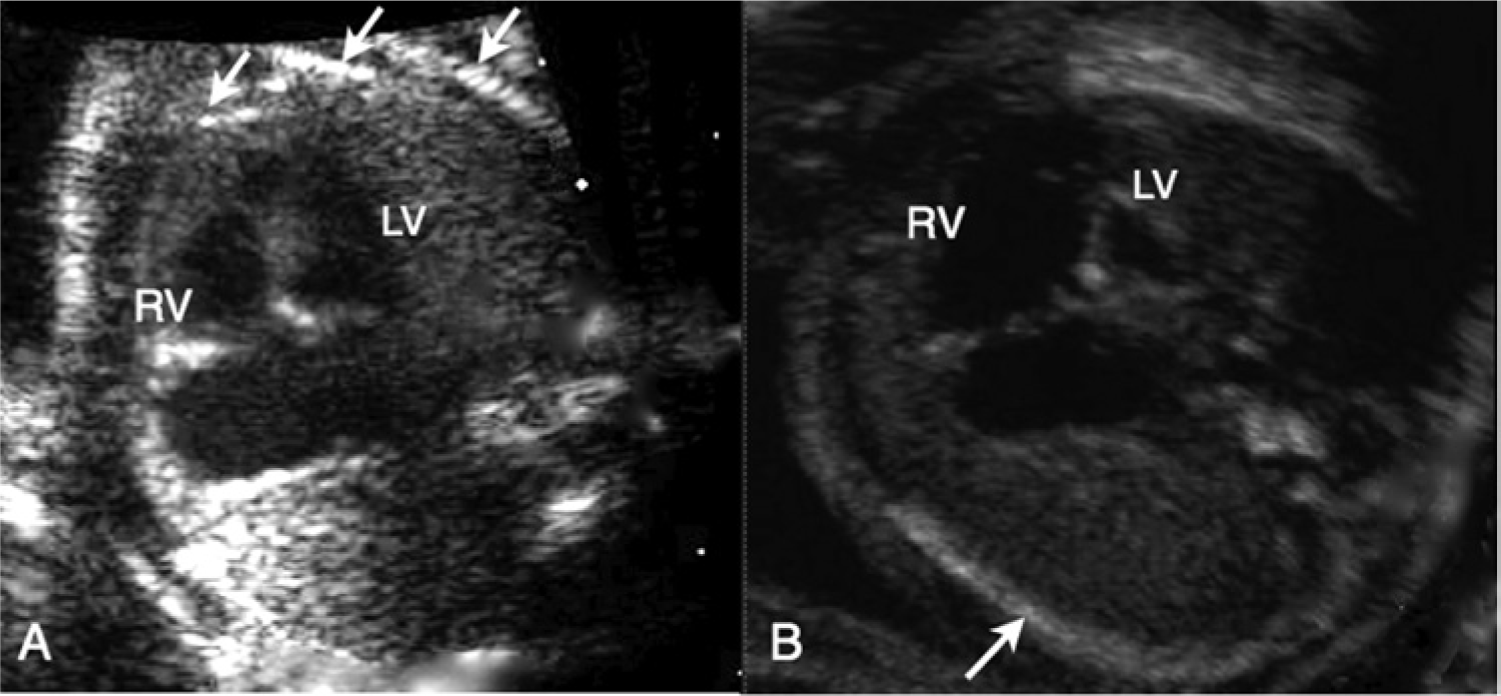
An example of a fetus with hypoplastic left heart syndrome (HLHS). (A) and (B) are images of the same fetal heart, taken from different planes. In (A), the ventricles appear to be of equal size and the left ventricle (LV) appears normal. However, the arrows point to two ribs providing evidence that the chest as imaged is not round. In (B), the fetal chest is now round and the arrow points to a single long rib demonstrating that the chest is round in this plane. The same heart now appears abnormal, with the left ventricle (LV) clearly smaller than the right ventricle (RV).
Step four of the protocol was to evaluate the outflow tracts. Before the educational intervention, it was found that only a minority of sonographers were scanning the outflow tracts. This procedure can take months to master, but it will add only minutes to a scan once the sonographer is confident. To evaluate the outflow tracts, the sonographers were instructed to begin from a four-chamber view to identify the left ventricle. From the left ventricle, tilt the probe toward the top of the heart to visualize the left ventricular outflow tract (LVOT) and the aortic root, watching to be sure there is continuity of the interventricular septum. The LVOT will track toward the right side of the body. The right ventricular outflow tract (RVOT) can then be visualized originating in the right ventricle, crossing the LVOT at the crux of the heart. Emphasis was placed on being certain that the outflows cross at the crux of the heart. It is possible to capture images that give the impression of one hypoechoic structure aimed in one direction in the fetal chest, followed by another image of a hypoechoic structure pointing in the other direction in fetuses who have transposition of the great arteries (TGA) (Figure 4). This emphasized the importance of the sonographer integrating the four chamber features to the outflow tracts and the outflow tracts to each other. In Figure 5, for example, the LVOT and RVOT were correctly recognized as lying parallel in a case of TGA. The protocol emphatically recommended the use of a video clip to demonstrate these structures.
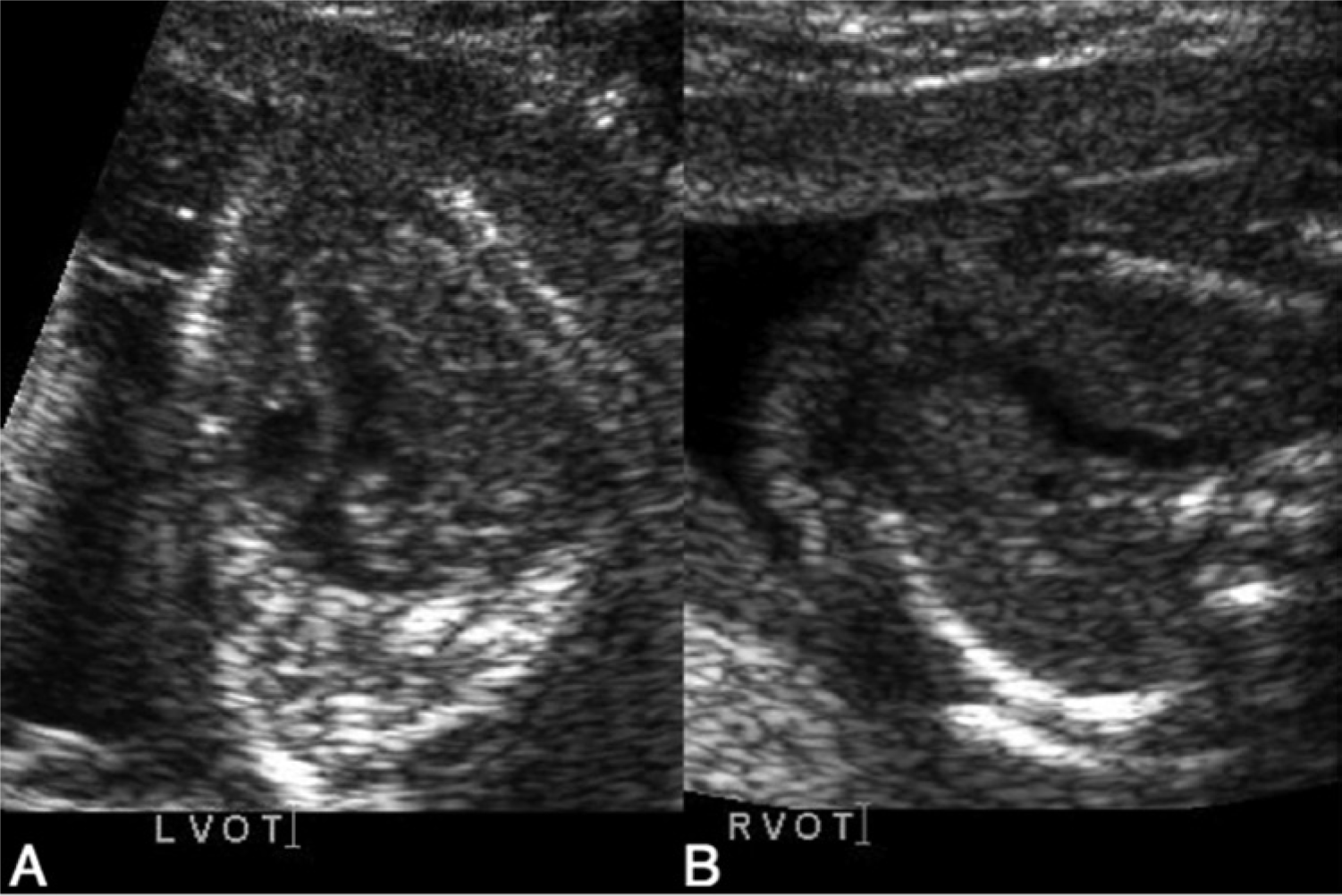
In (A) and (B), the LVOT and RVOT are labeled respectively, giving the appearance that the RVOT is crossing the LVOT, which led to the incorrect diagnosis of normally arranged vessels. This fetus was diagnosed at birth with dextro-transposition of the great arteries (d-TGA).
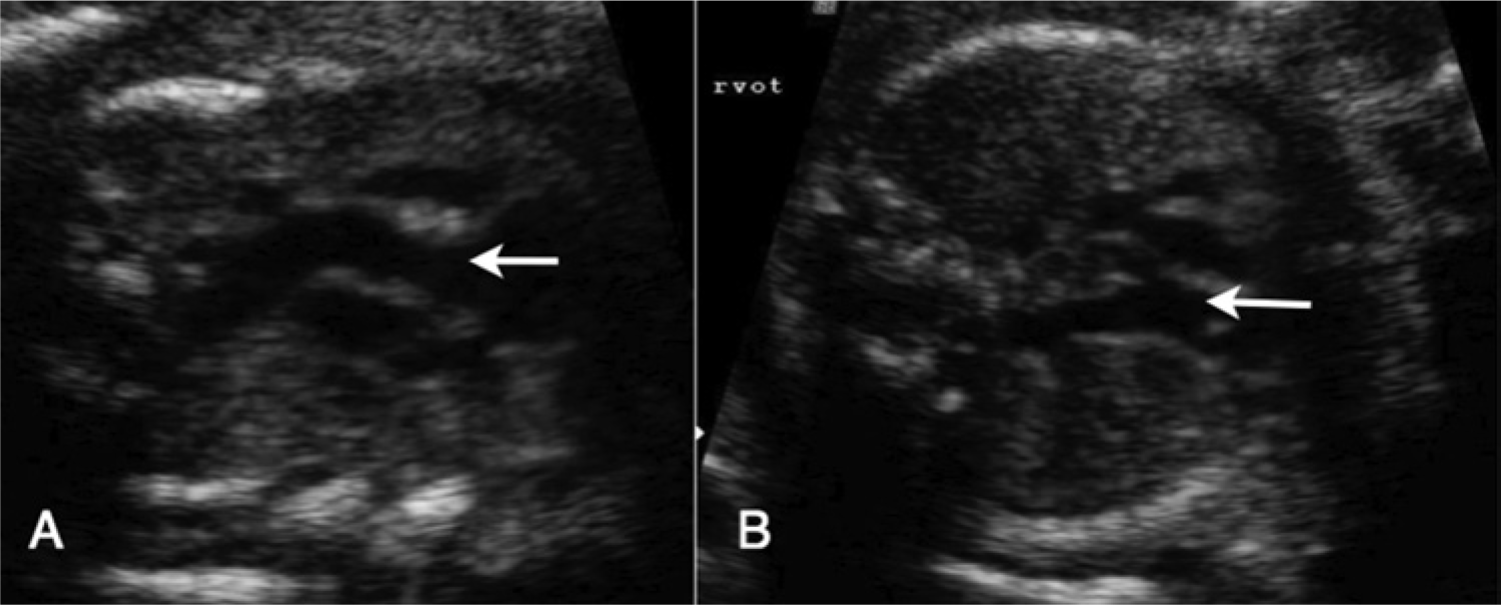
Sonogram of a fetal heart with d-TGA. In (A), the LVOT (arrow) is seen lying parallel to the RVOT (arrow) in (B). The postnatal diagnosis confirmed the neonate had d-TGA.
Included in the study were sonographers from the entire province of Manitoba, with all levels of experience, a mix of new and old equipment, and a variety of settings. Feedback was provided to the original sonographer and to the interpreting doctor. Departments were asked to sign on to the study to improve compliance. This required sonographer attendance at all educational events and adherence to the checklist (Table 1). All sonographers performing obstetric scans in Manitoba were invited, and all agreed to participate in the project.
Checklist distributed to all departments.
Results
The pre-intervention survey indicated first that all participants (n = 52) wanted additional training in fetal heart screening. Second, all sonographers were performing an evaluation of the heart in a four-chamber view. However, only 61% (32/52) were routinely attempting to evaluate the outflow tracts, and these had limited success. If the outflow tracts were not seen, a follow-up examination was not automatically performed, and reports would state “fetal heart not well seen.”
Considering that only 61% were attempting to evaluate the outflow tracts, coupled with the reported limited success in obtaining them, only approximately 25% of the fetal heart screenings performed included adequate four-chamber and outflow tract views. When asked why they did not perform outflow tract evaluation, 30% (16/52) stated that it was not required by the interpreting radiologist. Twenty-one percent responded that they did not know how to perform the outflow tract views.
In 2007, 69 sonographers were working in the obstetrical field (an increase from the 52 in 2004) and all 69 returned the postintervention survey. All 69 of the sonographers were routinely performing an evaluation of the four-chamber view. It is worth noting that at this point, outflow tract views were attempted by all sonographers (69/69), and their success rate in obtaining them increased as well. At a majority of the sites, the obstetric examination was not considered complete until the outflow tracts were well seen. If necessary, the patient was asked to wait until the fetus changed position to be able to see the heart well, and at times, the patient was brought back another day. This was not done immediately at every hospital, but this change in practice slowly became the new standard of practice in Manitoba. All sonographers reported that their interpreting radiologist now requested outflow tract evaluation. No sonographer stated that they did not know how to perform the outflow tract evaluation.
The results in Table 2 show that the distribution of cases on which sonographers reported performing outflow tract views improved significantly (P < .0001). Before the intervention, only 61% of sonographers attempted an outflow tract view, versus 100% after the intervention. (For the complete survey, see Appendix.)
Sonographer survey synopsis.
Discussion
A sonographer in a small rural hospital and a clinical instructor in an urban tertiary care center started this project. Every education session was aimed at the average sonographer. With the guidance of a sonologist, the fetal heart screen was pared down to one that is highly focused to find the most critical heart abnormalities yet was obtainable by any sonographer in any setting. The learning curve for each sonographer took many months; however, once the technique was mastered, this added only minutes to each examination. Bricker et al. 19 reported the time required to examine the fetal heart decreased as sonographers gained more experience and confidence. Focusing the fetal heart screen to key anatomical markers made the method attainable to all sonographers. By minimizing the diagnostic options to “normal or other,” sonographers were relieved from the burden of knowing a long list of pathologies.
With time, sonographers became more comfortable with the screen, and the detection rate on all heart abnormalities increased. Our unpublished findings indicate that there was an increase in the detection of cardiac lesions outside the target group, such as Tetralogy of Fallot. This was a consequence of the increased level of competence with the screening method and the increased attention to the fetal heart. In some cases, it made the obstetric scan more efficient as sonographers realized the efficacy of looking at the fetal position first, to see if the heart could be seen well. Previously, many sonographers would leave the heart scan until the end of the exam when the fetal position might have been suboptimal for cardiac imaging.
Examination of previous missed diagnoses with the pediatric cardiologist revealed that many of the abnormalities not diagnosed were CHDs that could have been identified on a four-chamber view. The same abnormality was picked up in some cases and missed in others. Comparison of these cases showed that in a remarkable number of “misses,” the fetal heart was examined when the fetus was in a position that made that evaluation less effective. Therefore, the decision was made to ask sonographers to wait until the fetus turned to a more optimal position before evaluating the heart. It was made clear to the sonographers the expectation was a screening protocol only, not a fetal echocardiogram. Sonographers were not taught the dozens of CHDs, focusing instead on the technical factors that contribute to a “good” fetal heart evaluation.
Even with major improvements in case detection rates, this new process did not increase scan times or wait times. Furthermore, caseloads for both the Fetal Assessment Units (FAU) and the pediatric cardiologist were not increased. On the contrary, the lead cardiologist for the study confirmed that knowing about a fetal diagnosis in advance saved time and expedited the entire process from diagnosis to counseling and to any eventual referral to cardiac surgery.
Conclusion
The sonographer’s role is vital in ensuring that no potentially fatal congenital heart lesion is missed. The four-step fetal heart screening protocol described in this study has been successfully implemented and well accepted among all sonographers in Manitoba, significantly improving prenatal cardiac screening of the population. This was accomplished at minimal expense, using resources already in place. Well over 95% of pregnant women undergo obstetric sonographic examination in the province, providing an opportunity for a fetal heart screen during the routine examination. Fetal echocardiography is performed on less than 1% of these pregnancies. Though a detailed fetal echo performed by a pediatric cardiologist would be the gold standard in prenatal diagnosis of CHD, our study indicates that a simplified, focused fetal heart screen can be effective and attainable by any sonographer, regardless of experience level, available equipment, and whether in a tertiary or community care center.
Footnotes
Appendix
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
