Abstract
Agenesis of the ductus venosus is a rare anomaly that was first reported in literature within the past decade. Color/Doppler sonography is an important tool for mapping blood flow in determining this aberrant anastomosis. Careful observation of the fetus for signs of congestive heart failure can lead to a positive outcome. In the following case, cardiomegaly was detected and digoxin was introduced, which arrested progression into congestive heart failure.
Embryological development of the ductus venosus occurs between 21 and 35 days after conception, when the embryo is 4 to 9 mm in length. This process involves three veins: the cardinal veins, the umbilical veins, and the vitelline veins. The common cardinal veins drain blood from the embryo and direct it to the developing heart. The right anterior cardinal vein becomes the superior vena cava and the right posterior cardinal vein becomes the azygous. The umbilical veins are directed through the hepatic sinusoids, at which time the right atrophies. The left umbilical vein unites with the ductus venosus and returns oxygenated blood from the primitive placenta. As the liver develops, the vitelline veins become the portal and hepatic veins and return poorly oxygenated blood from the yolk sac. The ductus venosus is then formed from the vitelline vessel lying within the liver. 1 The ductus venosus connects the intra-abdominal umbilical vein to the inferior vena cava and is an important regulator of blood flow.
Absence of the ductus venosus in utero results in aberrant connections of the umbilical venous return. Four different types of abnormal circulation have been reported in literature: (1) The umbilical vein bypasses the liver and assumes a suprahepatic or infrahepatic connection directly into the inferior vena cava. (2) The umbilical vein bypasses the liver and drains directly into the renal veins or iliac veins. (3) The umbilical vein drains directly into the portal vein. (4) The umbilical vein bypasses the liver and drains directly into the right atrium. 2 This type of connection into the right atrium associated with absent ductus venosus (ADV) makes up 46% of anomalous venous connections and is the most common. 3 Connections into the right atrium that also bypass the liver generally carry the worst prognosis due to congestive heart failure.4,5
Incorporating the cardiovascular profile (CVP) into an ultrasound scan proves to be a valuable tool to assess fetal cardiac well-being. Starting with 10 points, scoring the CVP involves subtracting up to 2 points in each of the following five categories:
Reversal in the ductus venosus subtracts 1 point; pulsatility in the umbilical vein subtracts 2 points.
The presence of hydrops subtracts 1 point for ascities or effusions and subtracts 2 points for skin edema.
Calculate the cardiothoracic ratio (heart area to chest area) with a normal cardiothoracic ratio less than 0.35. A cardiothoracic ratio of 0.35 to 0.50 subtracts 1 point. If the cardiothoracic ratio exceeds 0.50, 2 points are deducted.
Assess cardiac function by documenting the presence or absence of regurgitation in the heart valves. If mild regurgitation is present, 1 point is subtracted and if severe regurgitation is visualized, 2 points are deducted.
Umbilical artery Doppler is assessed with absent of end diastolic flow minus 1 point, reversed end diastolic flow minus 2 points.
Overall, the lower the CVP score, the worse the prognosis and the greater risk for cardiac failure.6,7
Case Report
A 25-year-old Caucasian female, gravida 2, para 0-0-1-0, was referred to our center for an abnormal second trimester multimarker screen positive for Down syndrome. The pregnancy was affected by numerous episodes of vaginal bleeding during the first trimester. Review of analytes revealed elevated hCG of 5.6 MoM, increased maternal serum inhibin of 6.32 MoM, and decreased maternal serum estriol of .49 MoM.
Detailed fetal sonography (Philips IU22 with 5.0 curvilinear probe) revealed a single intrauterine pregnancy, biometrics consistent with 18 weeks, 5 days gestation. The umbilical vein, measuring 3.3 mm in diameter, was noted to enter the fetal abdomen and assume a suprahepatic course, connecting directly into the right atrium of the heart. There was nonvisualization of the ductus venosus (Figures 1 and 2).
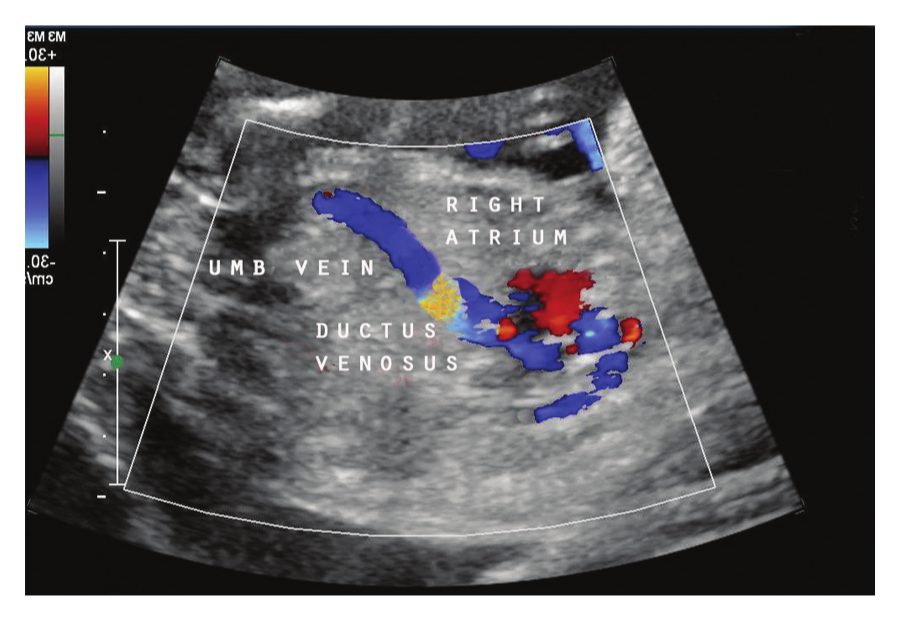
Color mapping of normal ductus venosus.
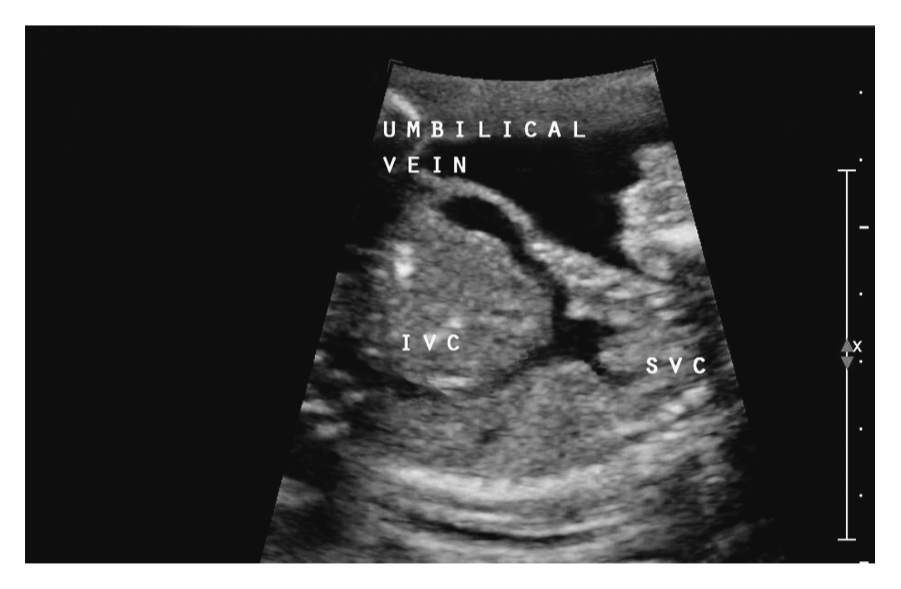
Aberrant connection of umbilical vein into right atrium.
A small stomach was visualized in the left abdomen. An echogenic focus was noted in the anterior/left upper quadrant at the level of the diaphragm. The remainder of the fetal structural survey was unremarkable. Genetic ultrasound, targeting markers that would increase the risk of aneuploidy, failed to reveal signs of chromosomal abnormality. TORCH serology, defined as Toxoplasmosis, Other (syphilis or parvovirus), Rubella, Cytomegalovirus, and Herpes simplex virus, was performed. The patient was remarkable only for toxoplasmosis antibodies, positive IgG, and positive IgM.
The patient was referred to a pediatric cardiology center for a fetal echocardiogram at 21 weeks, 5 days. No evidence of congenital heart disease or congestive heart failure was seen. The cardiothoracic ratio was noted to be 0.34. The cardiovascular profile scored 10 out of 10. The inferior vena cava did exhibit an increased a-wave. Again, confirmation of the umbilical vein into the right atrium bypassing the inferior vena cava was documented.
Follow-up sonography at 22 weeks, 1 day revealed the umbilical vein diameter increasing, measuring 5.9 mm at the point of entrance into the right atrium. The right atrium was enlarged. Color mapping of the liver revealed prominent hepatic arteries (Figure 3). The splenic artery connected into the celiac axis, which was seen branching off the aorta. The splenic vein was identified connecting into the inferior vena cava. Small portal veins were visualized throughout the right lobe of the liver. Color mapping of the liver vessels is important to evaluate the circulatory connections for possible alterations.
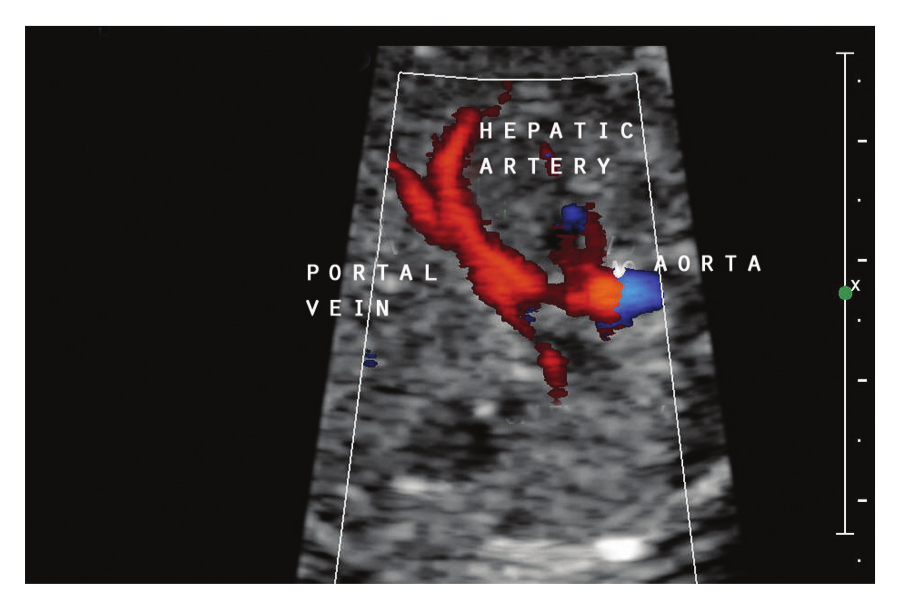
Color mapping of prominent hepatic arteries.
At 24 weeks, 6 days, follow-up fetal echocardiography again revealed agenesis of the ductus venosus with direct connection of the umbilical vein into the right atrium (Figure 4).
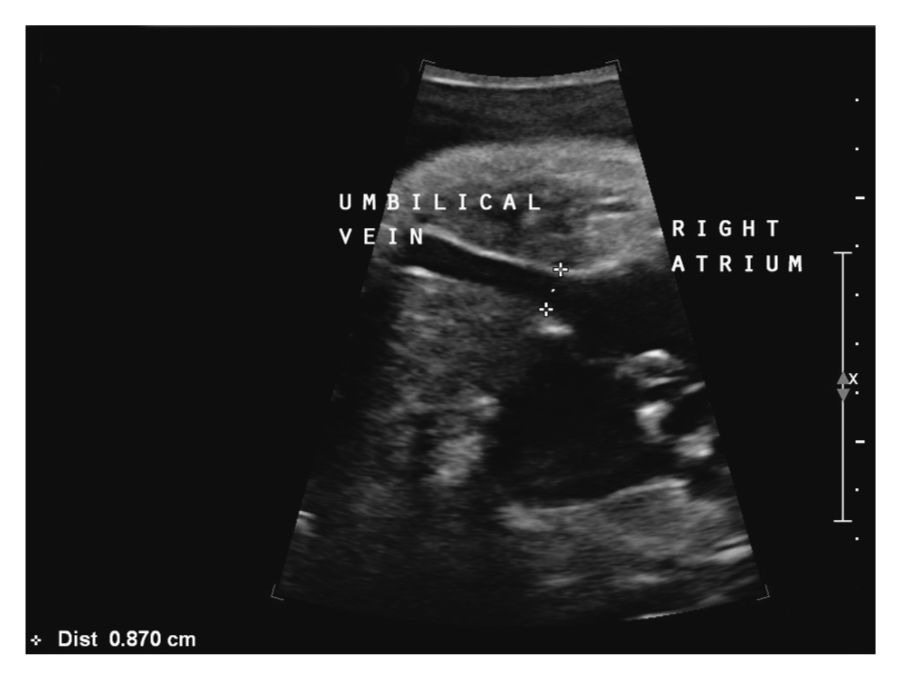
Umbilical vein connection into right atrium. Note the size of the anastomosis.
The umbilical vein diameter increased to 8.6 mm upon entrance into the right atrium. Velocity through the ductus arteriosus was increased, measuring 260 cm/sec, elevated from a normal peak systolic velocity of 140 cm/sec.1,7 Holodiastolic pulmonary regurgitation was observed. Trace tricuspid and mitral valve regurgitation was visualized. The CVP scored 8 out of 10, minus 1 point for increased cardiothoracic ratio (0.37) and minus 1 point for pulmonary regurgitation.
At 25 weeks, 1 day, a small pericardial effusion was present. The patient was then started on digoxin 0.25 mg twice daily. Follow-up sonography at 27 weeks, 1 day demonstrated the ductus arteriosus velocity decreasing, measuring 200 cm/sec. Trace tricuspid regurgitation was visualized. The CVP scored 8 out of 10, minus 1 point for increased cardiothoracic ratio (0.42) and minus 1 point for mild tricuspid regurgitation. By 29 weeks, 5 days there was improvement in the CVP score (10 out of 10), but there was still elevated peak systolic velocity in the ductus arteriosus. The heart exhibited mild cardiac disproportion with the right side larger than the left; however, the cardiothoracic ratio remained in the normal range of 0.32. Repeat ultrasound at 34 weeks, 6 days demonstrated a normal cardiothoracic ratio and CVP. The peak systolic velocity in the ductus arteriosus was in the normal range for this gestational age, measuring 120 cm/sec.
The patient presented to the hospital at 37 weeks, 4 days in labor and spontaneously delivered a 3480 g neonate with apgars of 8 and 9 at 1 minute and 5 minutes. The neonate was then transferred to a children’s hospital for evaluation secondary to the prenatal diagnosis of agenesis of the ductus venosus. Other then mild cardiomegaly and the inability to visualize the splenic vein with Doppler, the neonate had an uneventful hospital stay and was released 9 days later. Subsequent follow-up revealed a normally developing infant without complications.
Discussion
The ductus venosus is a trumpet-shaped vascular shunt that links the umbilical vein to the inferior vena cava. Blood is oxygenated in the placenta and delivered to the fetus by way of the umbilical vein. Approximately 80% of the blood bypasses the microcirculation of the liver, travels superiorly and posteriorly, and delivers oxygenated blood to the inferior vena cava at the level of the portal sinus and ductus venosus confluence, just distal to the right atrium. 4 The high velocity of the oxygenated blood in the ductus venosus travels to the inferior vena cava and enters the right atrium. It is then preferentially shunted to the left atrium to be delivered, especially in early pregnancy, to the brain and fetal heart. The blood through the ductus venosus is regulated by a sphincter and can change if the pressure or resistance in the systemic circulation is altered. This fact makes the ductus venosus very important for the fetus as a means of protection from venous overload. After birth, the ductus venosus closes due to a decrease in pressure and flow. This process is completed by 1 to 3 months of age and results in the formation of the ligamentum venosum within the infant liver. 8
Conclusion
One of the effects of absent ductus venosus with direct connection of the umbilical vein into the right atrium is volume overload. This can progress into cardiomegaly and congestive heart failure. Close monitoring of the fetal heart with ultrasound can signal when introduction of digoxin is appropriate in an attempt to reverse signs of congestive heart failure. Equally important is color mapping the connection of the umbilical venous return and documenting liver circulation. Incorporate the cardiovascular profile into your exam to evaluate for signs of congestive heart failure. Check for anomalies in other systems such as gastric, genitourinary, cardiac, skeletal, or facial clefts. 9 Chromosomal defects may also occur, such as Turner syndrome or Noonan syndrome.2,4
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
