Abstract
A prospective single-site survey was performed to gauge perceived pain during automated breast ultrasound (ABUS) and digital x-ray mammography (XRM). Patients also described differences in duration of the exams, provided their bra size, and indicated whether they would recommend ABUS to a friend. Pain was measured on a Likert-type visual analog scale (1–10). Participants were 199 female volunteers who also participated in the prospective, multicenter, nationwide clinical research study SOMO•INSIGHT. There was a mean pain and/or discomfort score of 6.41 (± 2.41) for XRM and a mean score of 1.86 (± 1.24) for ABUS (P = .001). Bra size and perceived examination duration played no role in determining patient pain and/or discomfort. One hundred percent of patients who participated said that they would recommend ABUS to a friend. These data suggest that if ABUS is proven to be a valuable adjunctive screening test for breast cancer among women with dense breast tissue, its routine use will be well tolerated by patients.
Introduction
In the United States, breast cancer ranks second only to lung cancer in incidence; in 2010 there were 207,090 new cases in women. 1 Digital x-ray mammography (XRM) is an invaluable tool in screening for breast cancer. Unfortunately, the sensitivity of mammography decreases in the 40%–60% of patients with increased breast tissue density. 2 The decreased sensitivity of XRM can lead to delays in detection and potentially, delays in lifesaving intervention. This issue is particularly important in women under the age of 50 who have denser breast tissue and a longer life expectancy. In addition, mammography is a source of anxiety and pain for patients as a result of breast compression during imaging. It has been shown that an unpleasant experience during a previous XRM can deter women from returning for additional XRM screenings. 3
New technologies such as automated breast ultrasound (ABUS) have been developed to improve breast cancer detection in patients with dense breast tissue. Previous studies have shown ultrasound is able to detect cancerous lesions not present on XRM. 4 When compared to the gold standard (contrast magnetic resonance imaging [MRI]), ABUS has been shown to have high interobserver agreement in breast cancer detection by a diagnostic radiologist with experience in breast imaging. 1 These results indicate that ABUS is a cost-effective alternative to contrast MRI when considering breast cancer screening protocols.
A prospective, nationwide, multicenter clinical research study, SOMO•INSIGHT, is currently underway to determine whether ABUS, performed concurrently with screening XRM, is more accurate than screening XRM alone in detecting breast cancer in asymptomatic women with dense breast tissue. 5 We are reporting the results of a survey of SOMO•INSIGHT study participants taken at a single institution to determine the level of comfort during the ABUS examination. Survey results were reviewed for patient satisfaction and comfort during ABUS examination versus mammography and correlated with bra size and the perceived duration of the examination.
Materials and Methods
Approval for this study was first obtained from the local Institutional Review Board. A prospective single-site (Henry Ford West Bloomfield Hospital, West Bloomfield, Michigan) investigational survey was performed to gauge patient comfort during ABUS examination compared to digital screening mammogram for women enrolled in the SOMO•INSIGHT clinical study. The women were asked to volunteer to complete an anonymous survey immediately after completion of XRM and ABUS examinations.
The SOMO•INSIGHT clinical study was designed to evaluate the sensitivity and specificity of using ABUS and XRM together as a screening method for women with > 50% parenchymal density. 5 These data are then compared with XRM alone. Patient eligibility criteria are based upon asymptomatic women with a history of breast density on mammography, if available. Further inclusion criteria include being at least age 25, not currently pregnant or breastfeeding, and having no history of breast interventions or cancer diagnosis in the past 12 months.
This clinical study is being performed across the nation at 11 clinical sites certified under the Mammography Quality Standards Act (MQSA), with a minimum patient volume of 5000 screening XRM procedures and 1000 breast ultrasounds. Each site has at least one MQSA–certified radiologist. Qualified radiologists and auxiliary staff were trained by U-Systems expert trainers to determine preliminary density assessments on XRM.
A single Somo-V Automated Breast Ultrasound system (U-Systems, Inc, Sunnyvale, CA) with a 10-MHz transducer was used. Upon meeting criteria to be a participant in the SOMO•INSIGHT study, the patient’s XRM was assessed to determine final parenchymal density. The Breast Imaging-Reporting and Data System score (BI-RADS) of I through IV was determined, and patients were assigned to one of four BI-RADS assessment categories (0, 1, 2, or 3). Using only this information, an initial BI-RADS management recommendation was made, which was then locked. With the XRM images still available, the same radiologist then reviewed the ABUS images.
A BI-RADS category (0, 1, 2, or 3) was assigned to the ABUS images, and then a final management recommendation was assigned that was collectively representative of an assessment of both XRM and ABUS combined. Management of either BI-RADS assessment was consistent with universally accepted standards of care. Sensitivity and specificity calculations were based on the initial and final management recommendations for each case once the truth was confirmed by pathology or confirmed by at least 12 months of benign or negative clinical status, including the next routine mammogram exam.
During this investigation, a routine screening digital mammography evaluation was used to determine whether the women had > 50% parenchymal density. All women with < 50% parenchymal density were ineligible under the study criteria and were given no further evaluations using the study protocol. All subsequent automated breast sonography examinations for this study were performed by two investigators working on the SOMO•INSIGHT study who were extensively trained by a U-Systems clinical applications specialist. Study participants were then asked to volunteer to complete an anonymous survey immediately after completion of XRM and ABUS examinations.
An example of the survey tool used is shown in Figure 1. Survey questions measured patient perceptions of pain for both ABUS and mammography on a Likert-type visual analog scale of 1–10. Patients also described their perception of the differences in duration of the examinations, their bra size, and if they would recommend ABUS to a friend. A Wilcoxon matched-pairs test was used to analyze the pain and/or discomfort questions and the time questions. A Kruskal-Wallis test for group differences was used to compare outcomes with bra sizes. A P value of < .05 was taken to indicate a significant difference.

Survey tool used to compare automated breast ultrasound to digital x-ray mammography.
Results
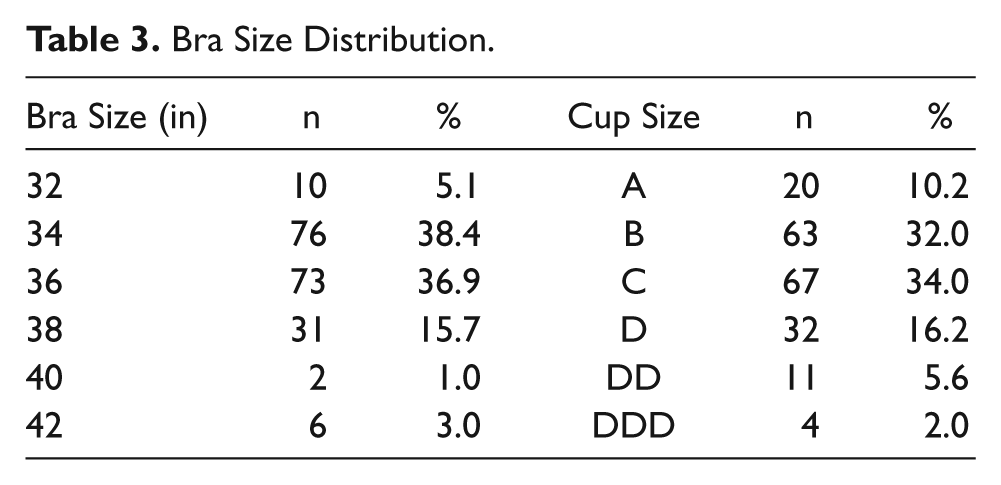
The survey was completed by 199 women. All data are shown in Tables 1–4. There was a mean pain and/or discomfort score of 6.41 (± 2.41) for XRM and a mean score of 1.86 (± 1.24) for ABUS (P = .001). Bra size played no role in determining patient pain and/or discomfort. There was also no significant difference in patient time estimations, with > 90% of both mammograms and ABUS estimated at < 20 minutes. Also, 100% of patients who participated in this study said that they would recommend ABUS to a friend.
Pain Analysis
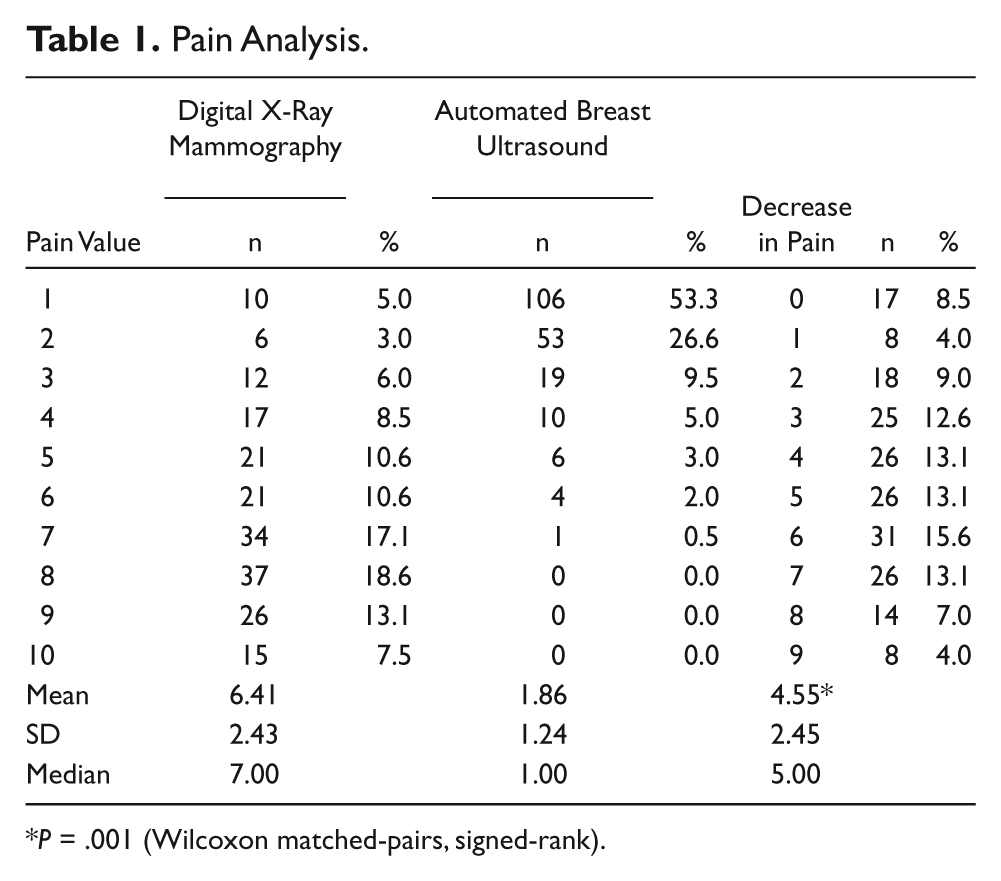
P = .001 (Wilcoxon matched-pairs, signed-rank).
Time Analysis
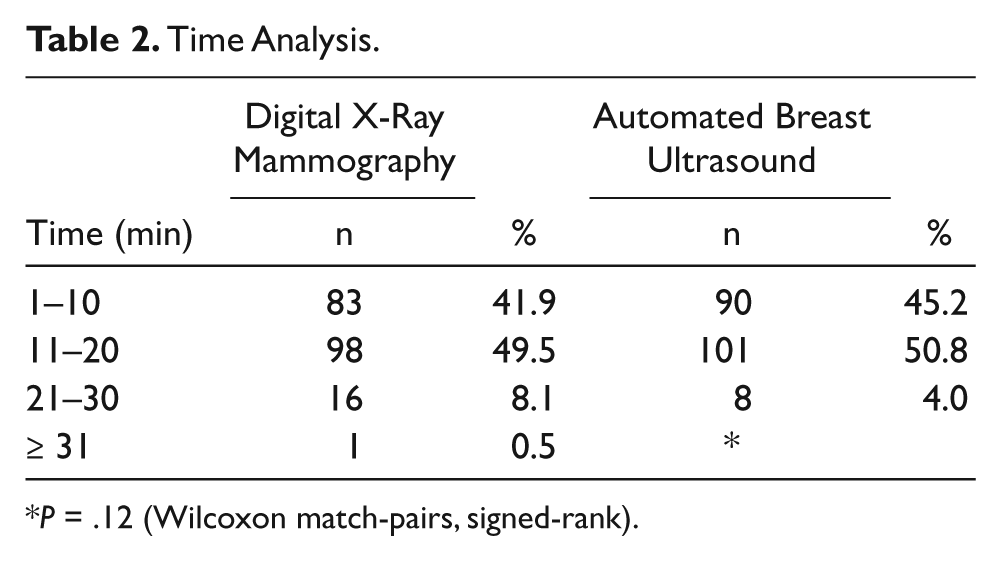
P = .12 (Wilcoxon match-pairs, signed-rank).
Bra Size Distribution
Bra Size Analysis
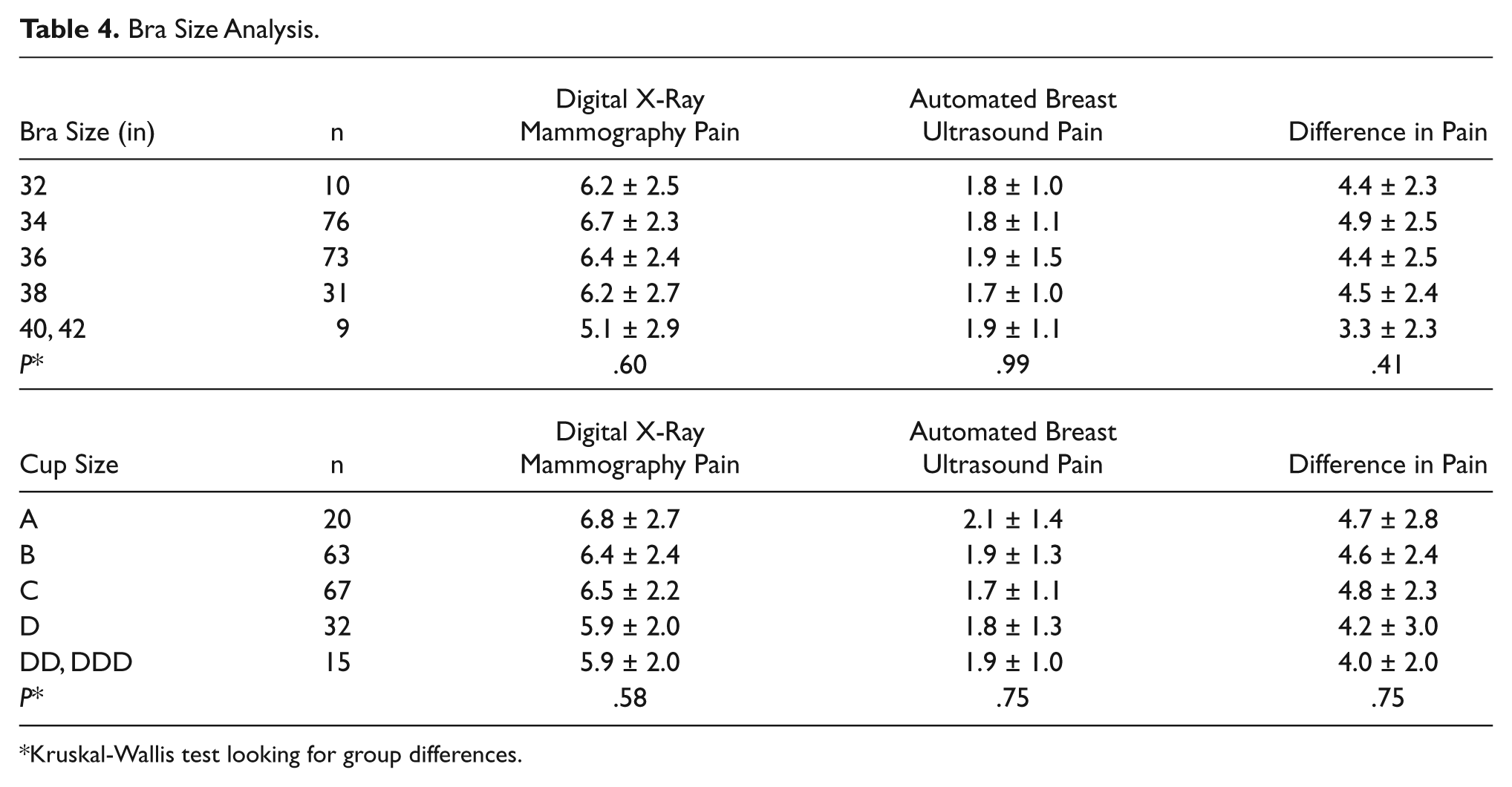
Kruskal-Wallis test looking for group differences.
Discussion
In the United States, automated breast ultrasound is a Food and Drug Administration–approved modality for breast cancer screening as an adjunct to mammography, but it has not yet been accepted for use in routine clinical screening. As new technology is developed for breast cancer screening, it is important to consider patient comfort and the impact it has on examination compliance.
Our survey results indicate that ABUS is perceived to be significantly less painful than digital screening mammography, regardless of breast size. Furthermore, all patients surveyed indicated that they would recommend an ABUS examination to a friend. One limitation of this study was the relatively small number of patients surveyed. The reason for the small number of subjects was the available period of time to recruit and to acquire data, caused by the completion of enrollment of new patients into the SOMO•INSIGHT clinical study at our institution. However, based on the number of patients surveyed, the data were shown to be statistically significant. This study was also conducted before the release of U-Systems’s ergonomic Reverse Curve Soft Touch transducer, which conforms to the anatomic curvature of a woman’s breast. This transducer was designed to provide greater patient comfort as well as improve image quality by minimizing refraction and reverberation artifacts at the tissue interface. A future study might be useful to determine whether there is a further drop in the mean pain and/or discomfort score compared to digital mammography with use of the Reverse Curve Soft Touch transducer.
Conclusions
Our findings indicate that an ABUS exam is very well tolerated by patients. If this result translates into improved patient compliance, ABUS may be more easily integrated into the breast imaging practice algorithm for breast cancer screening.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
