Abstract
Pancreatic tuberculosis is very rare, especially in immunocompetent patients, and represents a diagnostic challenge. The clinical features in patients with pancreatic tuberculosis are usually nonspecific, and the radiologic features may mimic pancreatic malignancy or pancreatitis. We describe four cases with pancreatic tuberculosis mimicking carcinoma or pancreatic cyst on imaging. None of these patients was immunocompromised. Endoscopic ultrasound–guided fine needle aspiration demonstrated necrotizing granulomatous inflammation in these cases and acid-fast bacilli in two of them. We suggest that pancreatic tuberculosis should be included in the differential diagnosis of a pancreatic cystic or solid-cystic mass, especially in patients from a region where tuberculosis is endemic.
Introduction
Abdominal tuberculosis, which today is rare in Western countries, is a diagnostic challenge to the physician, because it mimics a broad spectrum of miscellaneous diseases. In most instances, it occurs in the setting of disseminated or miliary tuberculosis. The diagnosis of pancreatic tuberculosis (TB) has often been made at exploratory laparotomy, although currently, abdominal tuberculosis is treated medically by various effective drug regimens. Therefore, early diagnosis is important to avoid unnecessary diagnostic or therapeutic procedures.1,2 We present four cases of pancreatic tuberculosis in otherwise healthy adults. Diagnosis of pancreatic tuberculosis was made using endoscopic ultrasonography–guided fine needle aspiration (EUS-FNA) of the lesion.
Case Reports
Case 1
A 66-year-old man who presented with fever, abdominal pain, and progressively worsening general condition for two weeks was referred to our hospital. His mother had a history of treated pulmonary tuberculosis. Abdominal computed tomography (CT) revealed a multiloculated solid-cystic lesion within the uncinate process of the pancreas (Figure 1). The patient had had negative tuberculin tests for many years. The chest x-ray was normal. Laboratory findings showed the white blood cell count to be 8400 cell/microL 4, with mild normochromic-normocytic anemia (Hgb = 10.9). Erythrocyte sedimentation rate was elevated (87 mm/h), and serum amylase was elevated as well at 346 u/100 mL. Endoscopic ultrasonography–guided fine needle aspiration of the solid-cystic lesion was performed (Figure 2), and reddish, purulent samples were taken. Ziehl-Neelsen staining demonstrated acid-fast bacilli, which were biochemically confirmed to be Mycobacterium tuberculosis. Histology revealed a necrotizing granuloma. The patient remains in good condition at nine months status post-medical therapy.
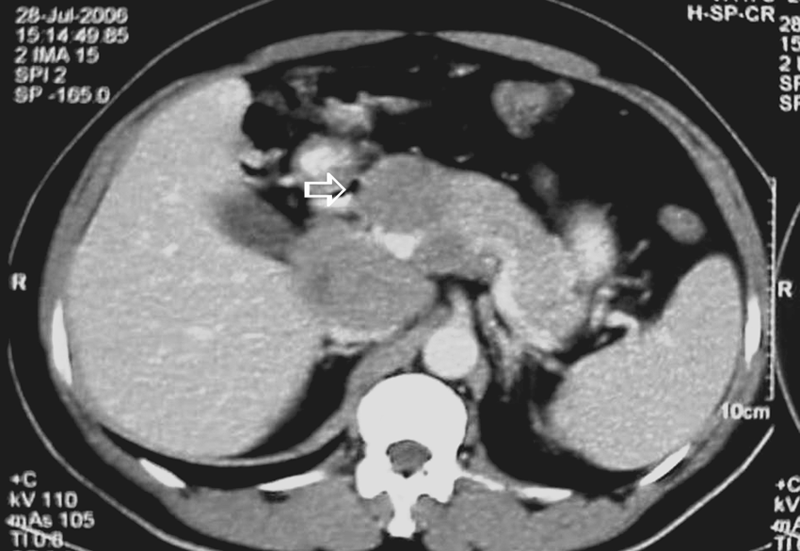
Case 1: Computed tomography scan showing solid-cystic lesion (tuberculosis) in pancreas head and neck (arrow).
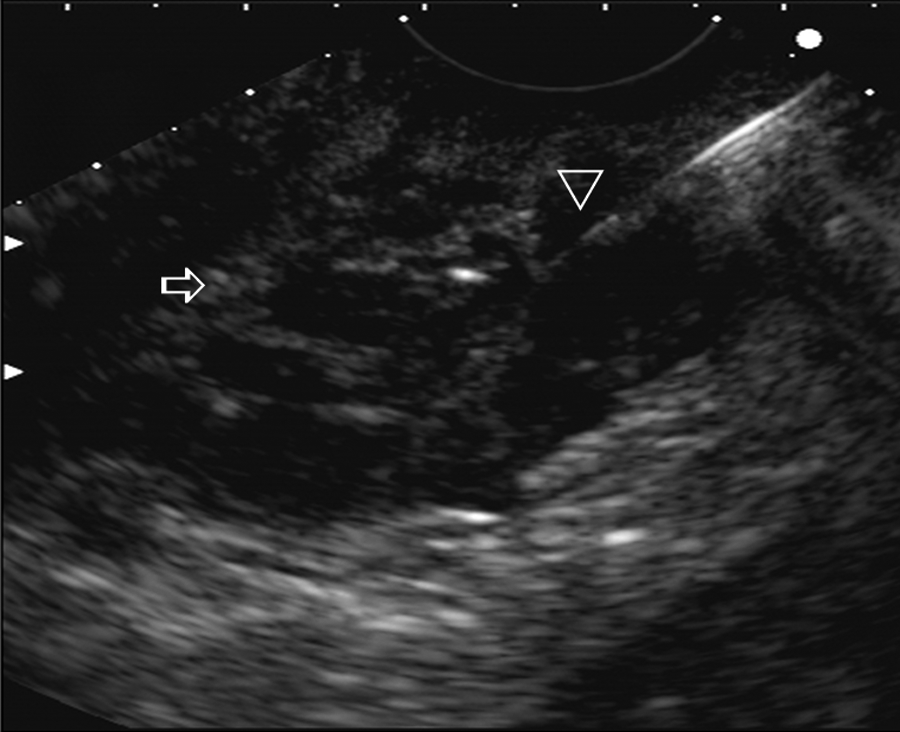
Case 1: Endoscopic ultrasonography-guided fine needle aspiration of the solid-cystic lesion (arrow) noted in Figure 1. Note the fine needle aspiration needle tip (arrowhead).
Case 2
A 25-year-old woman presented with a two-week history of epigastric pain. The tuberculin (tuberculin purified protein derivative, or PPD) skin test was positive (15 mm). The chest x-ray was normal. Abdominal CT scan detected a 20 × 25 mm hypodense lesion within the pancreatic head. The laboratory findings indicated hypochromic microcytic anemia (Hgb = 10.1). The patient underwent EUS-FNA (Figure 3), and histopathology revealed caseating (necrotizing) granuloma. No acid-fast bacillus was noted. She underwent medical therapy and remains asymptomatic after nine months.
Case 2: Endoscopic ultrasonography of the lesion (arrows) found in patient 2.
Case 3
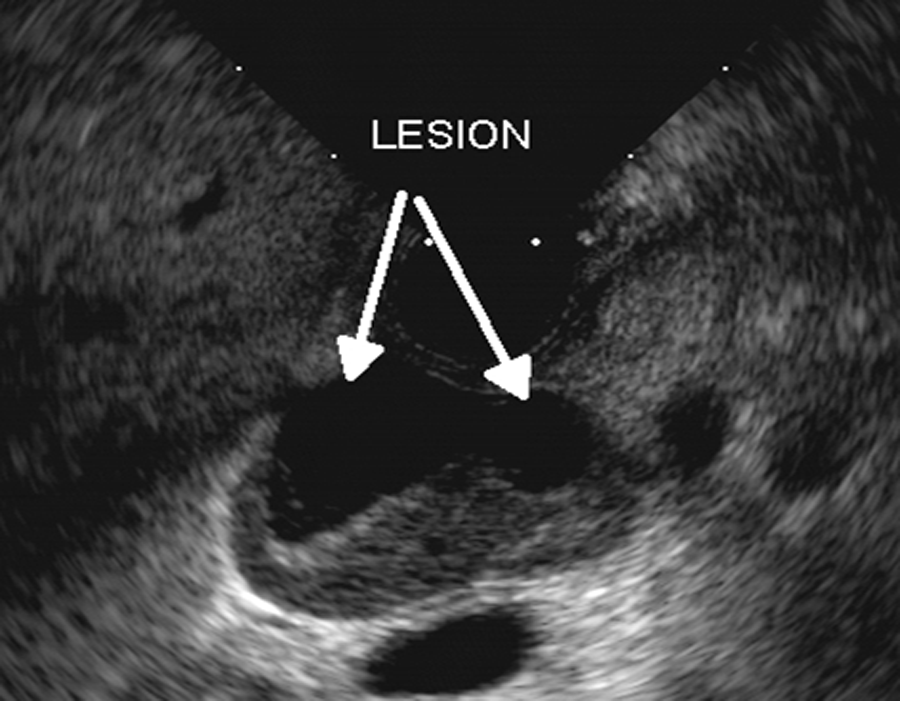
A 38-year-old woman presented with back pain and anorexia, which had begun a few weeks prior to admission. Transabdominal ultrasonography revealed a 20 × 17 mm pancreatic head cyst. Endoscopic ultrasonography confirmed the size and location of the cystic lesion (Figure 4), and FNA showed caseating granuloma.
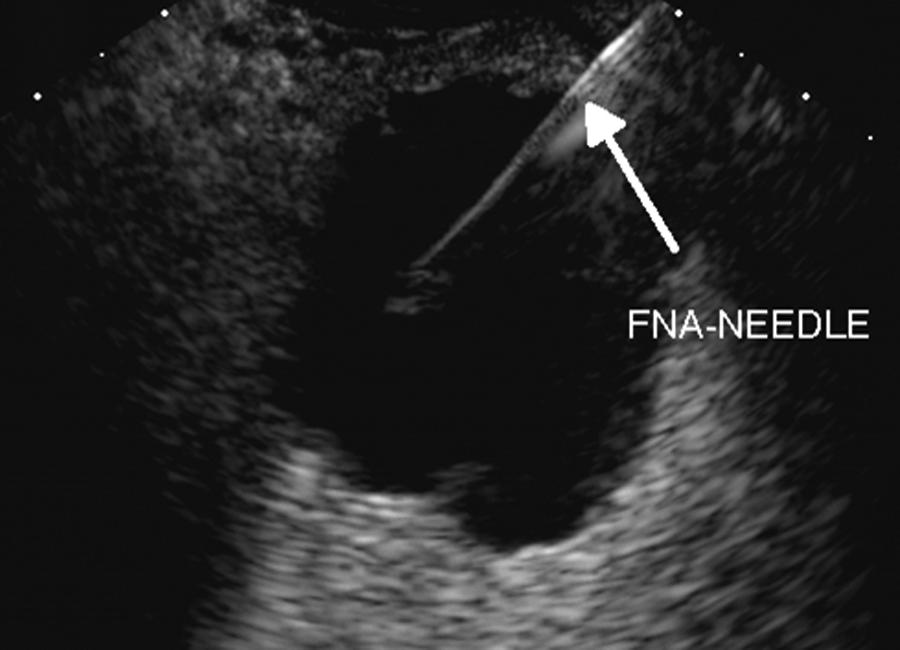
Case 3: Endoscopic ultrasonography–guided fine needle aspiration of the solid-cystic mass in patient 3, showing the fine needle aspiration needle (arrow).
Case 4
A 75-year-old man presented with a two-month history of abdominal pain and mild weight loss. Physical examination revealed mild tenderness in the epigastrium and no fever. Laboratory tests were remarkable for mild anemia and showed an erythrocyte sedimentation rate of 99 mm/hour. Abdominal CT scan showed a heterogeneous lesion in the pancreatic head and a second lesion in the porta hepatis region.
Endoscopic ultrasonography (Figure 5) demonstrated a 20 × 20 mm lesion with solid-cystic components in the pancreatic head, and a significantly enlarged lymph node measuring 40 × 39 mm in the porta hepatis region. Endoscopic ultrasonography–guided fine needle aspiration of the pancreatic head lesion and the lymph node was performed, which showed epithelioid histiocytes and granulomatous reaction. Polymerase chain reaction (PCR) on the FNA samples became positive for M. tuberculosis. Antituberculosis medical therapy was started, and the patient responded well and remains asymptomatic.
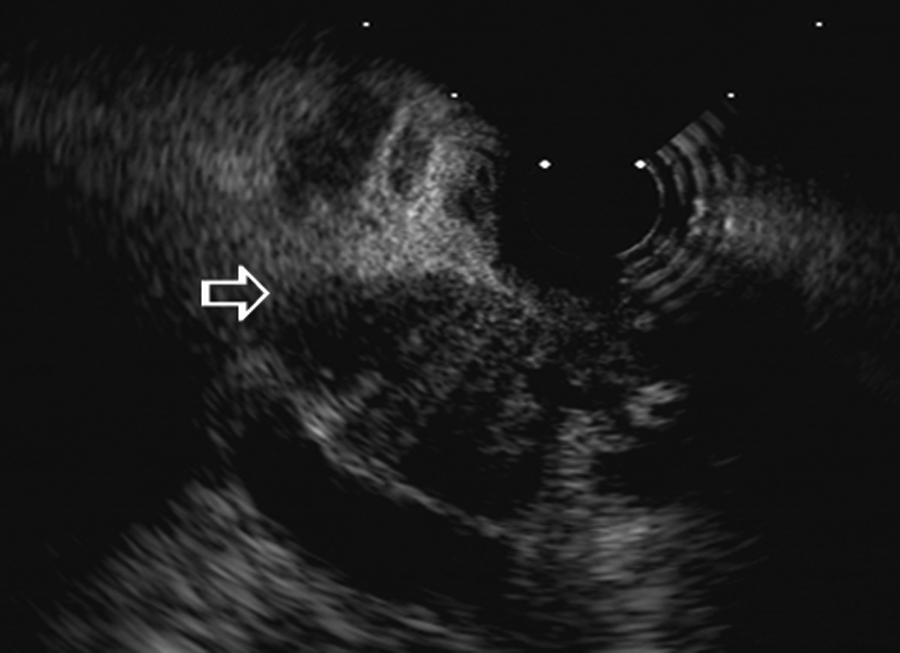
Case 4: Endoscopic ultrasonography of the lesion (arrow) found in patient 4.
Discussion
Abdominal tuberculosis usually affects the ileocecal region. Pancreatic tuberculosis is quite unusual. Auerbach did not find any cases of isolated pancreatic tuberculosis in 1656 autopsies performed on tuberculosis-infected patients. 3 Two-hundred ninety-seven patients (17.9%) in his series had acute generalized miliary tuberculosis, of whom 14 (4.7%) had tubercular involvement of the pancreas. We speculate that the tubercle bacilli reached the pancreas in our patients as a result of lymphohematogenous dissemination. However, it remains unclear why the pancreas would become a site of reactivation of tuberculosis. The pancreas is uniquely situated in the retroperitoneum, protected from direct environmental exposure. Purified lipases and pancreatic extracts appear to have antimycobacterial effects.4,5 Thus, the pancreas is relatively resistant to mycobacterial invasion, requiring a large intrapancreatic inoculum of M. tuberculosis to cause pancreatic lesions. 6 The two postulated routes of spread are directly, from involved peripancreatic lymph nodes, and more rarely, from hematogenous spread. Patients with pancreatic TB have presented with the following symptoms: anorexia, weight loss, low-grade fever, abdominal mass, pancreatic abscess, obstructive jaundice, acute or chronic pancreatitis, gastrointestinal hemorrhage, and splenic artery thrombosis.7-9 The patients in our series generally presented with abdominal pain, loss of appetite/weight loss, low-grade fever, and findings of an abdominal mass. These findings suggest that tuberculous involvement of the pancreas should at least be a consideration in any patient presenting with a mass in the pancreas in the setting of a febrile illness. Laboratory abnormalities including mild anemia, lymphocytopenia, elevated erythrocyte sedimentation rate, and elevated transaminases and alkaline phosphatase have been seen in approximately 50% of cases. 10
Radiographically, complex cystic lesions are reported more frequently than solid masses.11,12 Findings that may suggest mycobacterial infection include the presence of rim-enhanced lymph nodes in the peripancreatic region or the mesentery; ascites; and a thickened bowel wall in the ileocecal region.13,14 As the clinical and radiographic presentation mimics pancreatic cancer, a preoperative diagnosis of pancreatic tuberculosis is rare. Most reported cases have been diagnosed via laparoscopic biopsy or at laparotomy. Percutaneous (ultrasound- or CT-guided) FNA has more recently diagnosed pancreatic tuberculosis, although there are few reported cases worldwide,15,16 perhaps because acid-fast bacilli are rarely seen on FNA specimens, like our series, and culture of M. tuberculosis requires prolonged incubation. Additionally, as percutaneous FNA is usually performed if pancreatic cancer is suspected, the FNA sample is not routinely sent for mycobacterial stain and culture.
Endoscopic ultrasound–guided fine needle aspiration has emerged as an excellent tool to both image and sample pancreatic lesions. It is the most sensitive and specific method to identify pancreatic masses, and it is recommended by many as the preferred diagnostic modality for pancreatic masses.17,18
Malignant pancreatic tumors account for approximately 5%–10% of the cases involving lymphomas, endocrine carcinomas, or metastases. All of these lesions can be also confirmed by EUS-FNA.19,20 The treatment and often the prognosis can differ markedly from those of adenocarcinomas, and thus it is important to obtain a histological confirmation. In addition, EUS-FNA can be helpful in cases of incidental findings of a solid mass in the pancreas, as they raise the question of benefit and risk of a surgical procedure, and the preoperative histological diagnosis of these often small lesions is essential in therapeutic decision making. The presence of a solid mass within the pancreas does not necessarily imply the diagnosis of pancreatic cancer. Findings such as pancreatic pseudotumor, chronic pancreatitis, and infectious pancreatic masses are sometimes difficult to differentiate from pancreatic cancer, even by means of all available imaging procedures including standard EUS; EUS-FNA can be the key determinant to solving these dilemmas. The overall clinical interest in EUS has recently been enhanced by the availability of these interventional procedures. Fine needle aspiration is the most important advancement, especially for the diagnosis (cytology and/or histology and/or molecular marker analysis) of pancreatic cancer and solid-cystic tumors, with an accuracy ranging from 85% to 90%. Endoscopic ultrasound–guided fine needle aspiration has been shown to be a safe and reliable technique to obtain tissue from pancreatic masses, with a low (1%-3%) risk of complications. 21
The management of patients whose cystic lesion cannot be diagnosed with certainty presents a much greater problem. Most experienced endocrinologists and pancreatic surgeons recommend resection for even moderately suspicious lesions in the body and tail of the pancreas, and for highly suspicious lesions in the head of the gland. An accurate preoperative diagnosis of tumor type was achieved in only 20%, 30%, and 29% of those ultimately diagnosed with serous cystadenomas, mucinous cystadenomas, and mucinous cystadenocarcinomas, respectively.22,23 Many authors recommended that surgical resection should be performed for all mucinous cystic neoplasms and cystic tumors that are not clearly defined. Our findings support the role of EUS-FNA for the prevention of unnecessary surgery in patients who present with cystic or cystic-solid mass lesions of the pancreas when there is a possibility that the lesion may be pancreatic tuberculosis. Therefore, in addition to conventional CT scan and ultrasonography of the abdomen, evaluation of a pancreatic mass in a susceptible patient (eg, in an endemic area) should also include a smear for acid-fast bacilli and culture of pancreatic tissue and aspirate obtained by EUS-FNA.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received financial support for the research, authorship, and/or publication of this article from the Digestive Disease Research Center, Tehran University of Medical Sciences, Iran.
