Abstract
A 41-year-old man was admitted to our department with a 7-day history of jaundice of the skin. He was misdiagnosed with carcinoma because imaging tests showed a space-occupying lesion in the pancreatic head, and laboratory examinations showed elevated liver enzymes, and elevated serum bilirubin, alpha-fetoprotein, carbohydrate antigen 19-9, and ferroprotein levels. However, there was slight calcification in the lesion and a subsequent T-Spot test result was positive. The patient then underwent endoscopic retrograde cholangiopancreatography for biopsy and bile drainage. Histologically, the pancreatic mass showed granulomatosis, and the pathologic diagnosis of the isolated pancreatic neoplasm was tuberculosis. The patient accordingly received anti-tuberculosis agents, resulting in a significant decrease in the size of the pancreatic mass. The patient recovered well. Pancreatic tuberculosis can masquerade as malignancy; however, careful attention to a differential diagnosis can prevent the need for laparotomy.
Introduction
Tuberculosis (TB), which is caused by Mycobacterium tuberculosis, remains the top infectious killer worldwide, claiming close to 4,000 lives per day. 1 Although most TB cases are pulmonary, approximately 20% are extrapulmonary,2,3 with abdominal TB accounting for 10% of cases.4,5 Abdominal TB can involve various intra-abdominal organs, including the peritoneum, gastrointestinal tract, urinary tract, genital tract, or solid viscera, such as the liver, spleen, and pancreas.4,6–8 Pancreatic involvement is a relatively rare phenomenon worldwide; however, TB isolated around the pancreas can easily be misdiagnosed as pancreatic carcinoma.9,10 It is therefore important to make an early and accurate diagnosis to determine the appropriate course of treatment, and minimize unnecessary surgical procedures and health-care costs for patients. Here, we report the clinical and imaging findings in a 41-year-old man with isolated pancreatic TB, with an emphasis on the chronological changes in multimodality imaging findings using endoscopic ultrasound (EUS), computed tomography (CT), and magnetic resonance imaging (MRI).
Case report
A 41-year-old male patient was admitted to our department with a 7-day history of jaundice of the skin. An abdominal CT scan performed at another hospital had revealed a space-occupying lesion in the pancreatic head, and radical pancreaticoduodenectomy had been recommended by the hospital. The patient was then admitted to our hospital for further evaluation and treatment. His medical history included no previous illness and he denied any personal history of alcohol or cigarette consumption. His family history was also unremarkable, with no chronic pancreatitis or other related diseases. Physical examination revealed scleral icterus and cutaneous jaundice, with no palpable abdominal tenderness or mass.
Blood analysis revealed a normal leukocyte count of 6.7 × 109/L with mildly increased neutrophils (76%), and normal hematocrit and platelet counts. Prothrombin and partial thromboplastin times, d-dimers, and serum C-reactive protein were all normal. Blood biochemistry yielded the following results: alanine aminotransferase 440 U/L (reference 9–50 U/L), aspartate aminotransferase 107 U/L (reference 15–40 U/L), gamma-glutamyltransferase 2734 U/L (reference 1060 U/L), alkaline phosphatase 435 U/L (reference 40–150 U/L), total bilirubin 53 μmol/L (reference < 21 μmol/L), and direct bilirubin 28 μmol/L (reference < 5 μmol/L). Additional laboratory analysis revealed increased levels of alpha-fetoprotein (AFP) (9.13 ng/mL, reference < 7 ng/mL), carbohydrate antigen 19-9 (CA19-9) (108.8 U/mL, reference < 43 U/mL), and ferroprotein (Fer) (1584.7 ng/mL, reference 27–375 ng/mL). Fecal test and urine analysis were normal. Electrocardiogram, chest X-ray, and arterial blood gas were also normal.
Pancreatic ultrasound showed a hypoechoic round mass with an unclear border located in the pancreatic head, measuring approximately 4.4 cm × 3.0 cm × 2.4 cm, with some punctate strong echoes (Figure 1). Abdominal contrast-enhanced CT revealed an irregular medium-density mass measuring approximately 4.0 cm × 3.0 cm × 1.7 cm, which was incrementally enhanced after intravenous administration of contrast material. It also revealed some punctate calcifications in the mass (Figure 2). The pancreatic duct, extrahepatic bile duct, and intrahepatic ducts upstream of the obstruction were dilated (Figure 2). Abdominal contrast-enhanced MRI revealed an irregular bulky region in the head of the pancreas with low T1-weighted and relatively high T2-weighted images, which was incrementally honeycomb-enhanced after administration of contrast material (Figure 3). Calcification is unusual in pancreatic carcinomas and we therefore carried out a T-Spot assay, which was positive. An enzyme-linked immunosorbent assay for TB antigens gave a result of 292.5 pg/mL (reference < 14 pg/mL), further confirming TB infection. The patient accordingly also underwent a chest CT scan, which revealed no classic abnormal imaging changes, apart from some pulmonary bullae in the upper lobes of both the right and left lungs (Figure 4).

Ultrasound (2020-07-29) showed a hypoechoic round mass with unclear border measuring approximately 4.4 cm × 3.0 cm × 2.4 cm in the pancreatic head, with some punctate strong echoes.

Abdominal contrast-enhanced computed tomography (CT) images (2020-07-30). Abdominal contrast-enhanced CT revealed an irregular medium-density mass (red arrows) measuring approximately 4.0 cm × 3.0 cm × 1.7 cm (a), which was incrementally enhanced in the arterial phase (b) and faded in the venous phase (c) and delay phase (d), with some punctate calcifications in the mass.
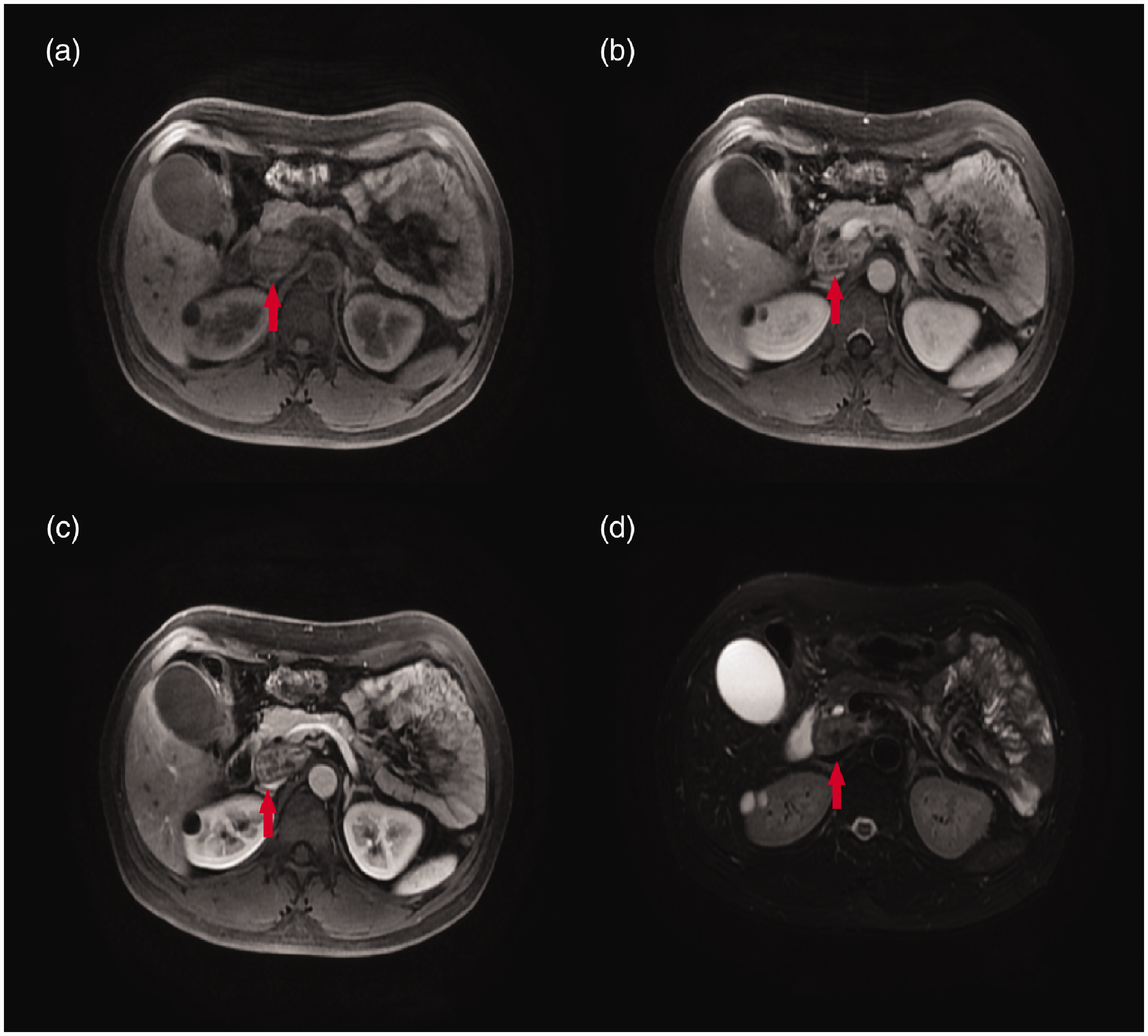
Abdominal contrast-enhanced magnetic resonance imaging (MRI) (2020-07-30). Abdominal contrast-enhanced MRI revealed an irregular bulky region (red arrows) in the head of the pancreas with low T1-weighted images (a), enhanced in the arterial phase (b) and faded in the venous phase (c), and relatively high T2-weighted images (d).

Chest computed tomography scan (2020-07-30) showed no abnormal imaging changes, apart from some pulmonary bullae in the upper lobes of both the right and left lungs
Endoscopic retrograde cholangiopancreatography (ERCP) and EUS-guided fine needle aspiration (FNA) were planned for biopsy and drainage. Under EUS guidance (Figure 5), the pancreatic mass was pierced with 22G biopsy needles and sent for cytological and histological examination. An 8.5F 7-cm Boston biliary plastic stent was placed to facilitate bile drainage. Histologically, the pancreatic mass showed granulomatosis (Figure 6).

Endoscopic ultrasonography (2020-07-31) showed a hypoechoic round mass with an unclear border measuring approximately 3.8 cm × 3.1 cm in the pancreatic head.

Lymphocyte neutrophil epithelial cells and multinucleated giant cells were observed (endoscopic ultrasound liquid-based cytology + smear, hematoxylin and eosin staining), which was considered to indicate a granulomatous lesion (2020-08-03).
According to the blood analysis, imaging examinations, and biopsy results, the nodal mass was highly suspicious for a tuberculous mass and the patient was accordingly given anti-TB medication. Initial treatment included rifampin 10 mg/kg/day, isoniazid 5 mg/kg/day, pyrazinamide 25 mg/kg/day, and ethambutol 15 mg/kg/day, given daily for 2 months, followed by rifampin and isoniazid for 9 months. Therapeutic ERCP was planned as needed.
After treatment for 3 months, the patient’s jaundice symptoms were significantly relieved and routine blood examination showed normal results. His liver and renal functions also improved significantly, although gamma-glutamyltransferase (84 U/L), total bilirubin (24 μmol/L), and direct bilirubin (9 μmol/L) remained slightly higher than normal. Levels of tumor markers had decreased: AFP from 9.13 to 7.36 ng/mL, CA19-9 from 108.8 to 54.1 U/mL, and Fer from 1584.7 to 406.1 ng/mL. Contrast-enhanced CT also revealed that the pancreatic mass was significantly reduced to approximately 3.1 cm × 1.2 cm × 2.2 cm (Figure 7). The patient was accordingly ultimately diagnosed with isolated pancreatic TB.

Contrast-enhanced computed tomography (CT) (2020-11-14). Contrast-enhanced CT revealed that the pancreatic mass (red arrow) was significantly reduced to approximately 3.1 cm × 1.2 cm × 2.2 cm (a), which was slightly enhanced at the arterial phase (b), and faded in the venous phase (c) and delay phase (d). Biliary stents could also be seen. Biliary stents could also be seen.
The patient’s postoperative course was uneventful. At 3 months of follow-up, the pancreatic mass was reduced but still compressed the common bile duct. Therapeutic ERCP was therefore repeated to place a new biliary stent. Anti-TB medicines were sustained for the treatment of pancreatic TB.
At 6 months of follow-up, routine blood, liver, and renal function examinations all showed normal results. Abdominal ultrasound showed that the hypoechoic mass had reduced to 2.4 cm × 1.3 cm in the pancreatic head, and the shape and size of the gallbladder had returned to normal (Figure 8). The biliary stent was therefore removed by ERCP. Rifampin 10 mg/kg/day and isoniazid 5 mg/kg/day were maintained for anti-TB treatment.
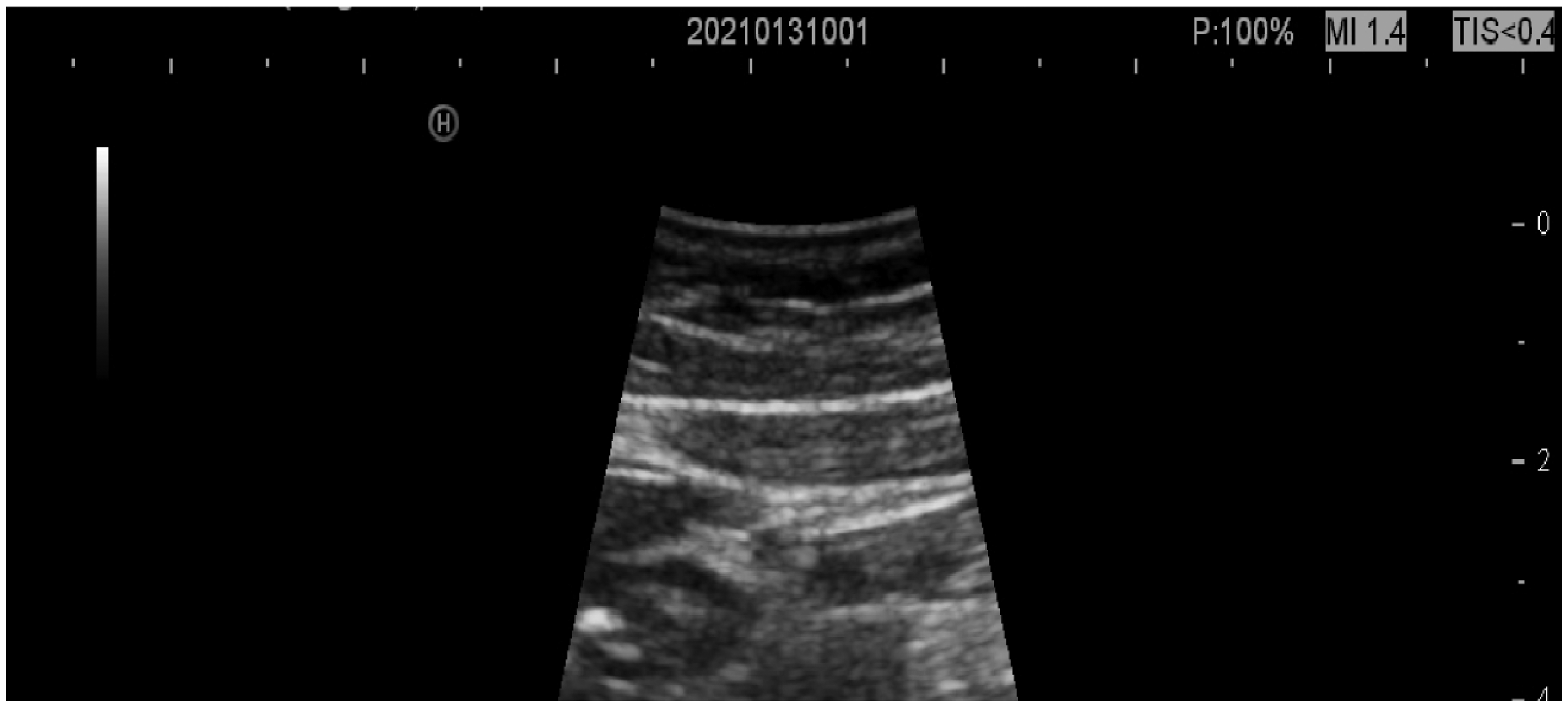
Abdominal ultrasound (2021-01-31) showed that the hypoechoic mass had decreased to 2.4 cm × 1.3 cm in the pancreatic head, and the shape and size of the gallbladder had returned to normal.
At 9 months of follow-up, contrast-enhanced CT revealed that the pancreatic mass was approximately 3.0 cm × 1.1 cm × 2.1 cm, with no abnormality in the gallbladder or intrahepatic and extrahepatic bile ducts (Figure 9). However, the patient still had a positive T-Spot result (364.9 pg/mL, reference < 14 pg/mL).

Contrast-enhanced computed tomography (CT) (2021-4-27). Contrast-enhanced CT revealed that the pancreatic mass (red arrow) was approximately 3.0 cm × 1.1 cm × 2.1 cm (a), which was slightly enhanced in the arterial phase (b), and faded in the venous phase (c) and delay phase (d). There was no abnormality in the gallbladder, and the intrahepatic and extrahepatic bile ducts were seen without biliary stents.
The reporting of this study conforms to the CARE guidelines (for case reports). 11
Discussion
Isolated pancreatic TB is a relatively rare type of abdominal TB, even in endemic areas,2,5 possibly partly due to the anti-mycobacterial effects of pancreatic lipase and deoxyribonucleases. 12 Reports of pancreatic TB have increased slightly in recent years, in line with the increased availability of improved imaging modalities and the ability to obtain specimens from the pancreas. 9 However, the diagnosis of isolated pancreatic TB remains challenging because of its non-specific presentation, which mimics malignancy.
Panic et al. recently reviewed data for 166 patients with pancreatic TB. 10 This systematic review showed that pancreatic TB was most common in middle-aged and elderly patients, with a mean age of 41.61 years (range 16–82 years) and a male-to-female ratio of approximately 3:2. Asia had the highest rate of diagnoses (50.0%), followed by North America (22.9%) and Europe (20.5%). The clinical symptomatology was diverse, with abdominal pain being the most frequently reported symptom (74.8% of patients), followed by weight loss (51.6%) and fever (46.5%). 10
In the above review, 10 20.0% of the patients developed jaundice, compared with up to 75% of patients in other studies,13–15 possibly because these later studies mainly focused on isolated pancreatic TB. Pancreatic TB frequently presents as a pancreatic head mass (59.0%), followed by the body (18.2%), tail (13.4%), and neck (1.8%), 10 and jaundice thus commonly occurs due to biliary obstruction. However, cases in which jaundice is the only symptom, as in the current case, are extremely rare and make the diagnosis more difficult.
Although the above symptoms can be explained by TB infection, they may sometimes be a simple consequence of pancreatic infection or reflect other clinical entities involving the pancreas, such as tumors or cysts. 16 In addition, the laboratory results may also be non-specific, and can thus confuse or mislead the diagnosis. Although the present patient displayed a positive tuberculin test, we suspected malignancy in light of the elevated tumor indicators (AFP, CA19-9, Fer). Some diagnoses in previous studies were therefore only made after extensive surgeries.10,17,18
Diagnostic clues obtained from imaging features are important, to minimize unnecessary surgical procedures and health-care costs. In addition to TB, local autoimmune pancreatitis 19 and initial polyarteritis nodosa 20 are rare but can mimic pancreatic cancer, highlighting the need to pay more attention to the differential diagnosis of masses within or around the head of pancreas when CT or MRI images show atypical manifestations. The sensitivity of multidetector CT for pancreatic carcinoma was reported to be >85% to 90%,21,22 but its specificity was unsatisfactory at only 58%. EUS-FNA is therefore recommended for biopsy of atypical pancreatic masses, especially those located within the head of the pancreas, with a sensitivity for approximately 90%. 23 This procedure also has low incidences of about 1% to 2% for pain, acute pancreatitis, infection, and bleeding, and < 0.01% for needle-tract seeding. 24
Abdominal ultrasound is often used as the first-choice imaging modality because of its easy availability and absence of ionizing radiation risk. Most cases of pancreatic TB show hypoechoic lesions, but a few, including the current case, show mixed isoechoic–hypoechoic lesions. 9 CT is the primary imaging modality for evaluating solid and cystic pancreatic lesions, but is limited by being unable to detect features that can distinguish between pancreatic TB and pancreatic carcinoma. 9 MRI is occasionally used, and lesions usually appear as hypointense on T1-weighted images but may be hyperintense or a mixture of hypo- and hyperintense on T2-weighted sequences. 25 The lesions are sometimes multi-nodular and may show rim enhancement in post-contrast images. 26 The current case was atypical for cancer because of the calcification within the node and the mixing density of the mass on T2-weighted MRI.
Empirically, the diameters of the common bile duct and pancreatic duct are typically normal in patients with pancreatic TB, even for lesions positioned centrally in the head of the pancreas, while they are often dilated in patients with pancreatic carcinoma.25,27,28 In the present case, abdominal contrast-enhanced CT revealed that the pancreatic duct, extrahepatic bile duct, and intrahepatic ducts upstream of the obstruction were all dilated, possibly due to the large size of the mass. Accordingly, no abnormalities in the gallbladder or intrahepatic and extrahepatic bile ducts were found when the pancreatic mass had reduced in size after anti-TB treatment.
The presence of calcifications may also indicate pancreatic TB; however, although some reports have indicated calcifications in up to 56% of patients, others reported no calcification in the masses.13,27,29,30 Positron emission tomography CT has also been used to evaluate pancreatic TB, but studies suggest that it is not reliable for distinguishing pancreatic TB from malignancy.31,32
A definitive diagnosis of pancreatic TB is based on pathologic findings obtained through image-guided biopsy. EUS-guided FNA is the ideal method for identifying pancreatic TB, 27 but CT- or US-guided FNA have also been performed when EUS-guided FNA was unavailable. 9 FNA provides an opportunity to sample the pancreatic lesions and obtain material for cytological and microbiological evaluations to establish a diagnosis. Polymerase chain reaction may also help in bacteriologically unconfirmed cases. 33 However, FNA increases the risk of dissemination in cases of carcinoma or TB, 34 and percutaneous FNA has a higher chance of needle-tract dissemination than EUS-guided FNA. 35 Anti-TB therapy should thus be administered following a diagnosis of TB, and endoscopy examinations should be repeated during several treatment cycles. In the current case, the patient also had common bile duct obstruction and ERCP was carried out after 3 months for stent replacement, and we therefore checked the whole stomach, especially around the puncture point. Moreover, the effectiveness of anti-TB treatment in the current patient provided confirmation of the diagnosis of pancreatic TB.
When a diagnosis is made, the pharmacological treatment of pancreatic TB is similar to that of other forms of TB, although surgery may still be needed in some cases. Previous studies reported excellent cure rates following standard anti-TB therapy for 6 to 12 months. 10 The current patient received rifampin (10 mg/kg/day), isoniazid (5 mg/kg/day), pyrazinamide (25 mg/kg/day), and ethambutol (15 mg/kg/day) for 2 months, followed by rifampin and isoniazid for 9 months. A recent review suggested that the drugs and dosages do not need to be altered because of obstructive jaundice; however, physicians should closely follow-up these patients to prevent drug-induced liver injury. 9 Because our patient already had abnormal renal and liver functions, we performed ERCP to relieve the biliary obstruction and delineate the ductal anatomy, and his liver and renal function examinations showed normal results after two therapeutic ERCP procedures.
Conclusion
We presented the case of a patient with isolated pancreatic TB and described the imaging and histologic features. Although the incidence of isolated pancreatic TB is relatively low, it should be considered a diagnostic possibility. Imaging scans and FNA can provide valuable information for making a differential diagnosis between pancreatic TB and carcinoma. The early diagnosis of pancreatic TB can lead to satisfactory treatment results and prevent the need for unnecessary surgical procedures.
Footnotes
Acknowledgements
The authors thank the anonymous reviewers and the editor for their valuable comments.
Authors’ contributions
YG contributed to the study concept and design, collected and analyzed the data, and participated in drafting the manuscript; MX contributed to data acquisition and assisted in drafting the manuscript; ZW contributed to data acquisition and critically revised the article; QL conceived the study, participated in the study design and coordination, and revised the manuscript critically; all authors read and approved the final manuscript.
Data availability statement
The data supporting the conclusions are included within the article.
Declarations of conflict of interest
The authors declared that they have no conflicts of interest regarding this work.
Ethics statement
The studies involving human participants were reviewed and approved by the research ethics committee of Shulan (Hangzhou) Hospital. Written informed consent for publication of their clinical details and clinical images was obtained from the patient.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
