Abstract
Cor triatriatum sinister is a rare congenital condition in which the left atrium is partitioned into two chambers by a fibromuscular membrane. Two-dimensional transthoracic echocardiography and 2D transesophageal echocardiography are the standard tests of choice for the assessment of function and physiology, although with complex pathology, evaluation of specific structures may be limited. We are reporting the additional use of 3D transesophageal echocardiography to delineate the anatomical features of the membrane in a comprehensive way not possible by conventional 2D imaging.
Cor triatriatum sinister is a rare congenital condition in which the left atrium is partitioned into two chambers by a fibromuscular membrane. The role of echocardiography is to determine whether the membrane is found to be obstructive or nonobstructive, which, in addition to patient symptoms, will determine surgical intervention. Although 2D transthoracic echocardiography (2DTTE) and 2D transesophageal echocardiography (2DTEE) are the tests of choice for the assessment of function and physiology, evaluation of specific structures may be limited. With the use of 3D transesophageal echocardiography (3DTEE), we were able to delineate the anatomical features of the membrane in a comprehensive way not possible by conventional 2D imaging. In addition, cross-modality comparison with cardiac computed tomography (CT) demonstrated a linear structure located in the left atrium consistent with the echocardiographic findings. To our knowledge, this is the first report of two cases of nonobstructive and obstructive cor triatriatum sinister evaluated by multiple imaging modalities, including 3DTEE.
Case 1 (Nonobstructive Cor Triatriatum Sinister)
A 66-year-old man with known nonobstructive cor triatriatum sinister and a history of recent biological aortic valve replacement presented to the echocardiography laboratory for postoperative evaluation of a suspected prosthetic perivalvular leak. The 2DTTE examination confirmed a solid linear membrane dividing the left atrium into two chambers (Figure 1). Continuous-wave Doppler demonstrated no significant pressure gradient across the membrane (Figure 2). A 2DTEE was ordered to further evaluate the severity of a suspected perivalvular leak.
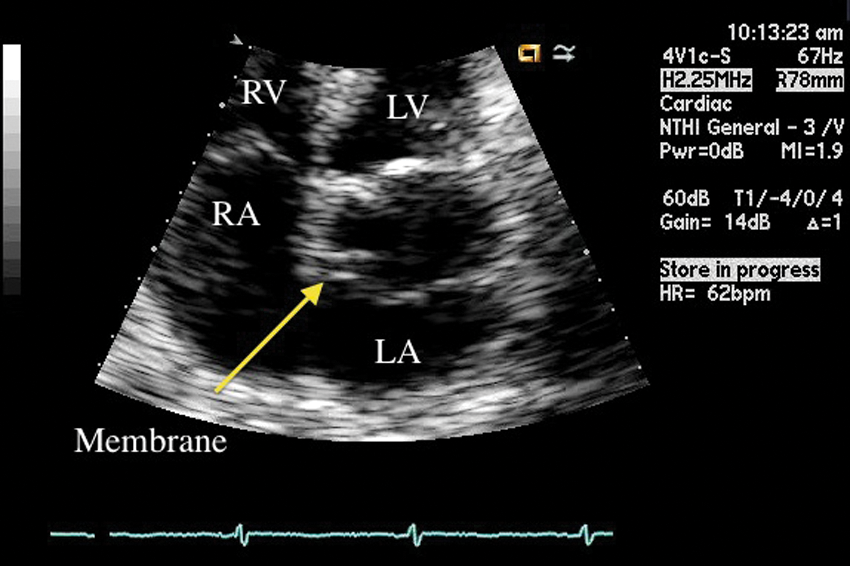
A 2D transthoracic echocardiography magnified image of the LA and RA obtained from the apical four-chamber view. A solid linear membrane is seen partitioning the LA into two chambers displaying a third atrium (arrow). LA, left atrium; RA, right atrium; RV, right ventricle; LV, left ventricle.
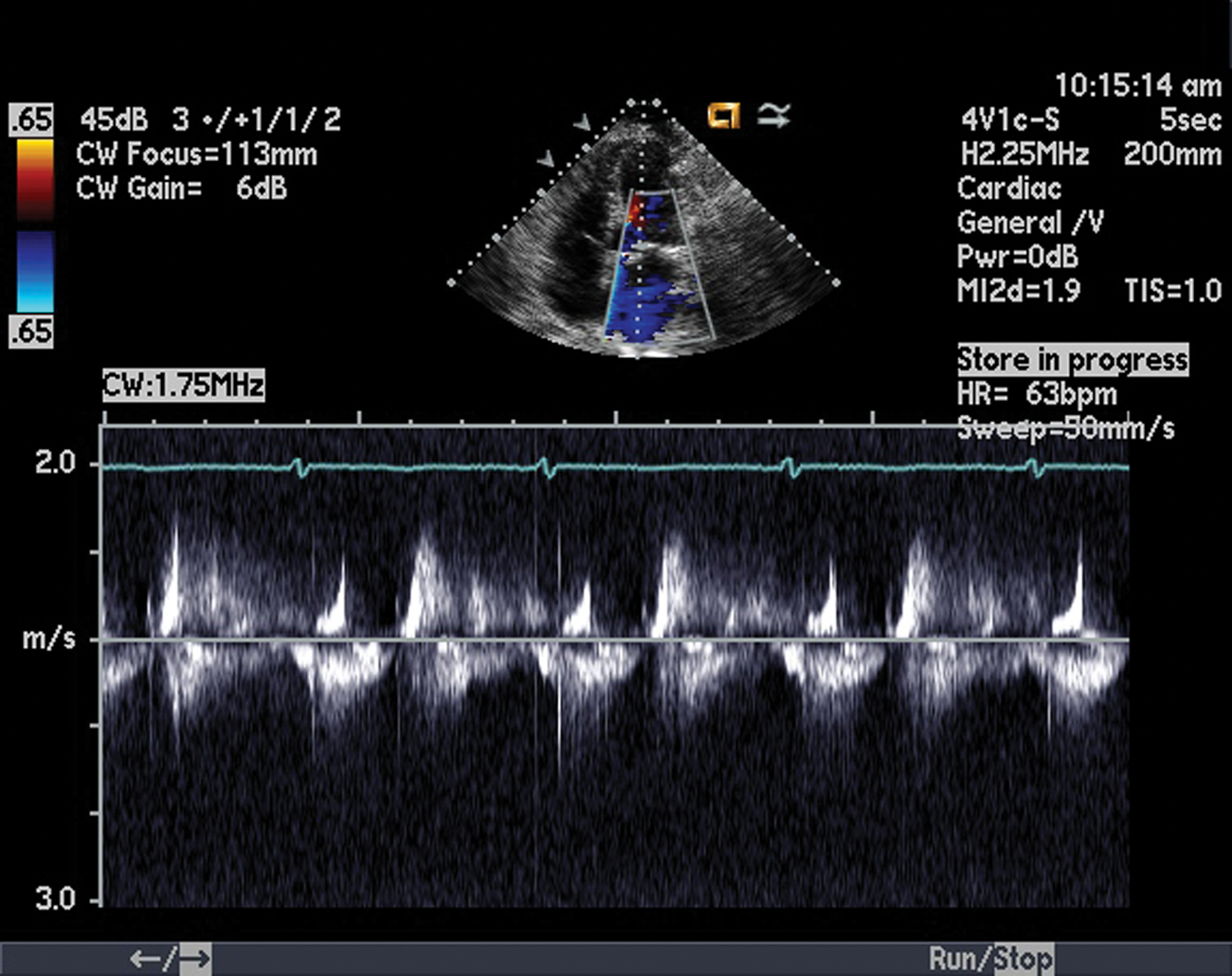
Continuous-wave spectral Doppler obtained from the apical four-chamber view. The entire spectrum of the membrane was interrogated and no significant Doppler gradient was demonstrated across the membrane.
As expected, 2DTEE imaging of the left atrium showed a continuous linear hyperechoic structure partitioning the left atrium into two discrete chambers (Figure 3A). Because 2DTEE angulation does not enable “en face” or direct frontal viewing of the membrane, we were limited in obtaining accurate delineation of the anatomical features of the perforations. Doppler color flow imaging demonstrated laminar flow and no significant aliasing consistent with the absence of obstruction (Figure 3B). This was confirmed with mitral and pulmonary venous inflow evaluation (not shown).
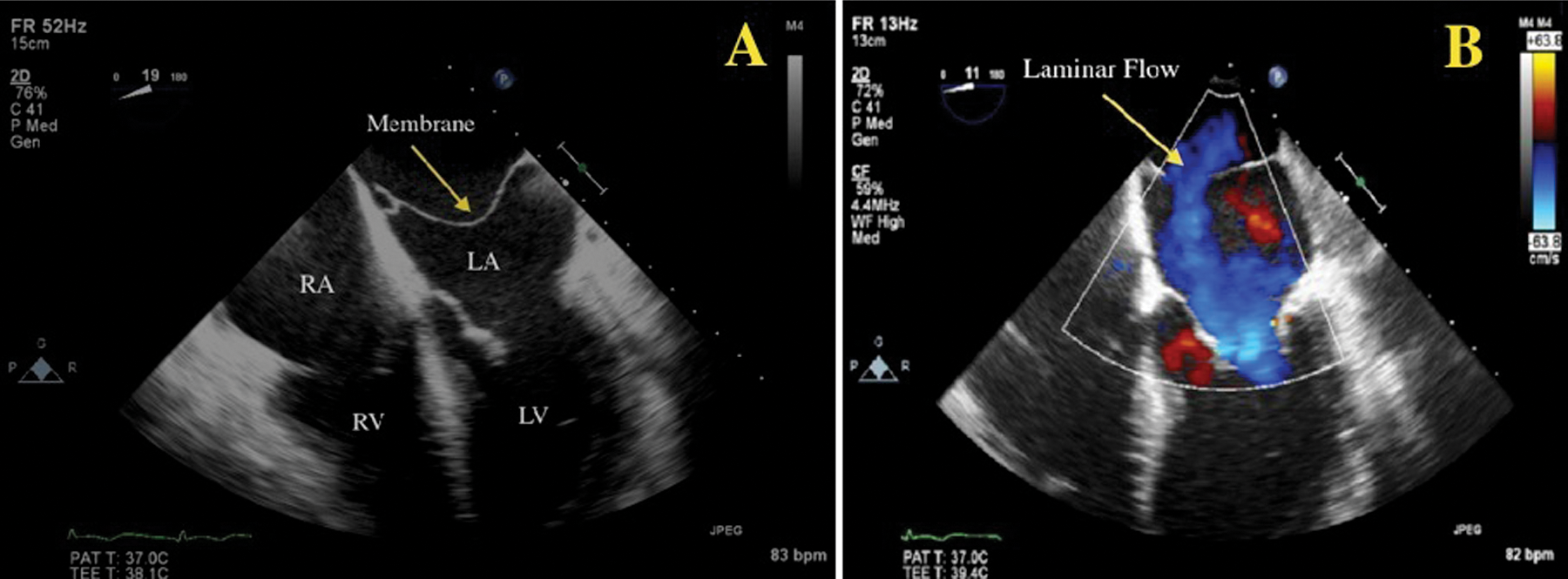
A 2D transesophageal echocardiography four-chamber view. (A) Notice the appearance of a solid linear membrane (arrow) dividing the LA. (B) Color Doppler imaging suggests laminar, nonobstructive flow across the membrane (arrow). LV, left ventricle; RV, right ventricle; LA, left atrium; RA, right atrium.
A 3DTEE full-volume data set was obtained of the left atrial structure, including surrounding valves. Using examination-specific orientation and cropping techniques, the base of the left atrium was cropped away and the membrane was isolated and fully appreciated. This permitted improved visualization of the membrane’s attachments and area of perforations (Figure 4A,B). In Video 1A,B (available online at http://jdm.sagepub.com/supplemental), the floppy movement of the membrane is appreciated in addition to the transparencies in the fibrous tissue where flow likely would pass through, as seen by the color Doppler imaging.
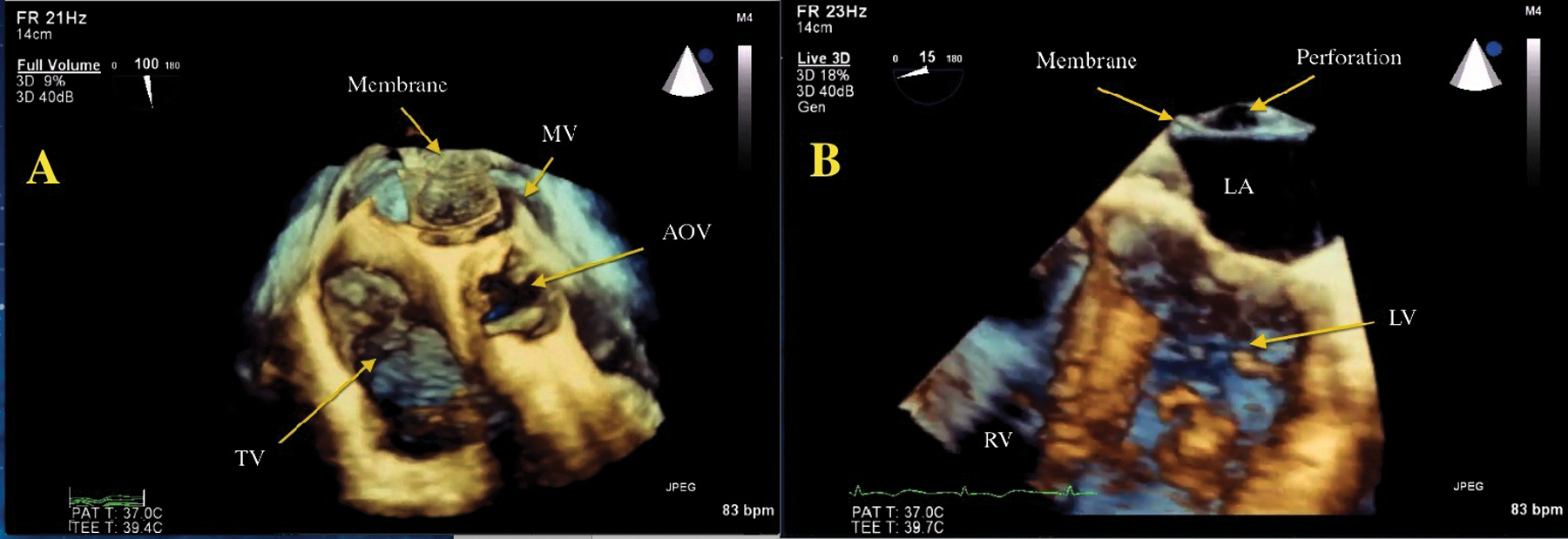
A 3D transesophageal echocardiography four-beat, full-volume and Live 3D data set. (A) The 3D volume data set is cropped, or cut in from the apex to the left atrial level, demonstrating the “en face” or frontal view of the membrane (arrow). Notice the fenestrations (lines) seen throughout the membrane suggestive of a nonobstructive membrane. (B) The Live 3D data set is cropped from the lateral aspect for visualization of the four-chamber view. The image demonstrated a small opening superiorly consistent with a large perforation (arrow). MV, mitral valve; TV, tricuspid valve; AOV, aortic valve; LA, left atrium; RV, right ventricle; LV, left ventricle.
Case 2 (Obstructive Cor Triatriatum)
A 27-year-old woman with a past history of transient symptoms of asthma presented with increased dyspnea on exertion. An electrocardiogram (ECG) revealed atrial fibrillation; upon clinical exam, auscultation detected a significant holodiastolic murmur (3/6 Levine’s scale) heard at the cardiac apex. A 2DTTE was ordered to assess potential origins of these findings. The echo findings showed a linear hyperechoic region across the left atrium most consistent with cor triatriatum sinister (Figure 5A). Color Doppler imaging demonstrated an obstructed appearance with aliasing across the membrane (Figure 5B). Using an off-axis apical long-axis view, spectral Doppler was aligned parallel to membranous flow demonstrating a mean gradient of 17 mmHg across the membrane consistent with obstruction (Figure 6). Because of the patient’s symptoms and identification of obstruction, surgery was performed to remove the membrane. In the operating room, transesophageal echocardiography was performed using both 2D and 3D imaging techniques.
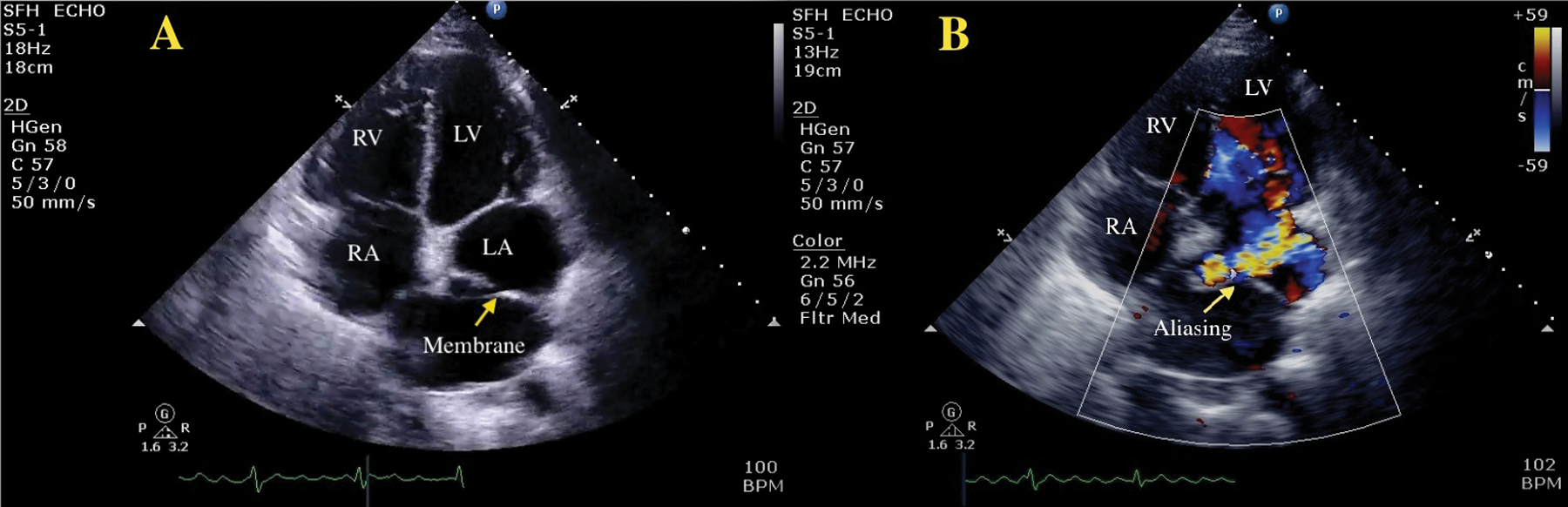
A 2D transthoracic echocardiography apical four-chamber view. (A) There is a solid structure traversing the LA consistent with a membrane (arrow). (B) Color Doppler imaging demonstrated aliasing of color flow (arrow) suggestive of obstruction. LV, left ventricle; RV, right ventricle; LA, left atrium; RA, right atrium.
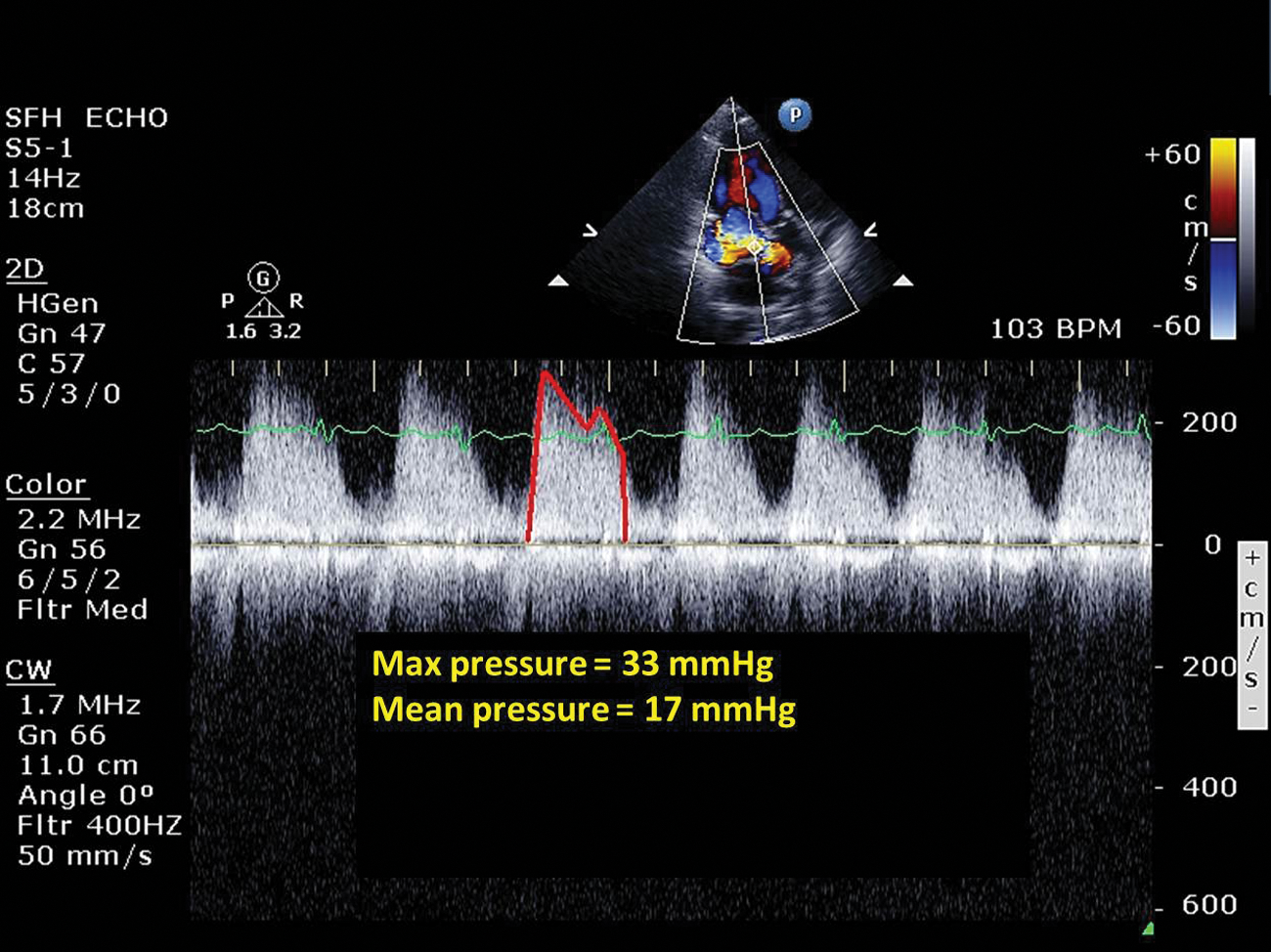
Continuous-wave spectral Doppler was measured across the aliasing color flow from the apical three-chamber view. Max and mean pressures were estimated at 33 mmHg and 17 mmHg, respectively.
Similar to the previous case described as nonobstructive, the membrane appeared as a continuous, linear structure transecting the left atrium (Figure 7A). Evidence of color Doppler aliasing typical of turbulent flow was noted laterally and adjacent to the left atrial appendage (Figure 7B). An echolucent gap or opening was seen superior to the aortic valve, which indicated further obstruction from an extended fibrous linear structure. Color Doppler imaging demonstrated aliasing of color flow through the opening of the membrane (Figure 8).
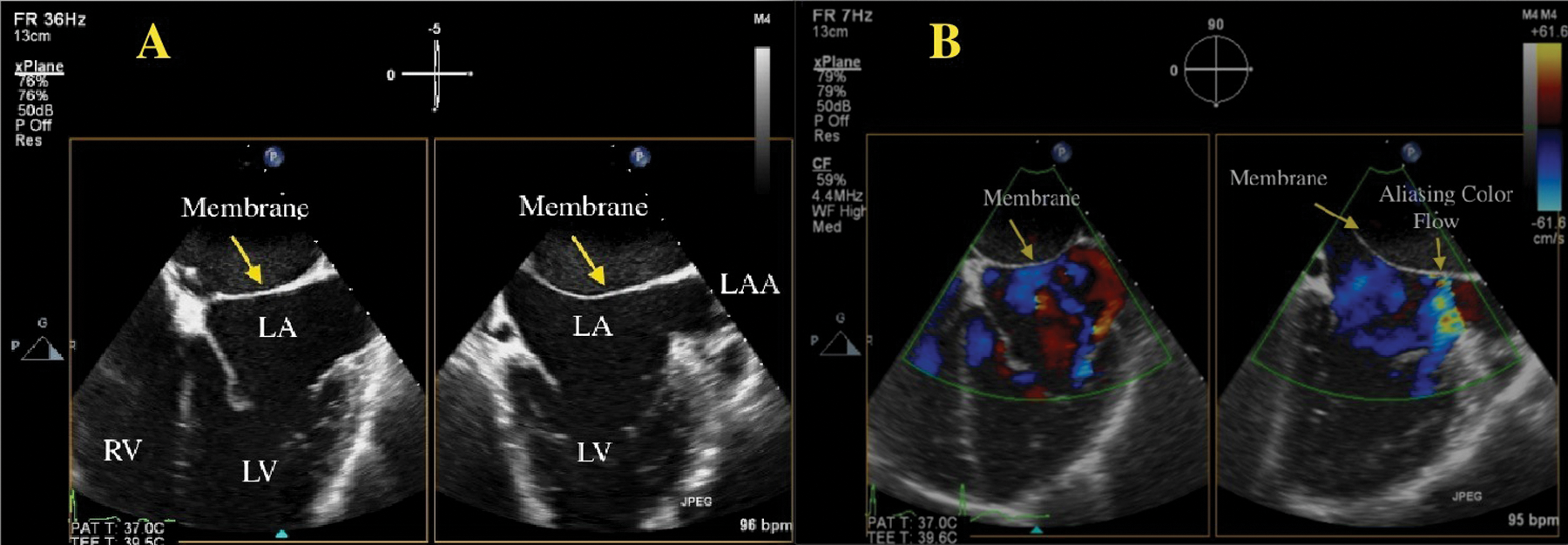
The 2D transesophageal echocardiography X-plane imaging demonstrating a four-chamber (left) and two-chamber (right) view. (A) Notice the solid linear structure traversing the LA beyond the LAA in the two-chamber view. (B) Aliasing of color flow is seen next to the LAA. This is suggestive of obstruction across the membrane, although the exact point of obstruction is not clear. LV, left ventricle; RV, right ventricle; LA, left atrium; LAA, left atrial appendage.
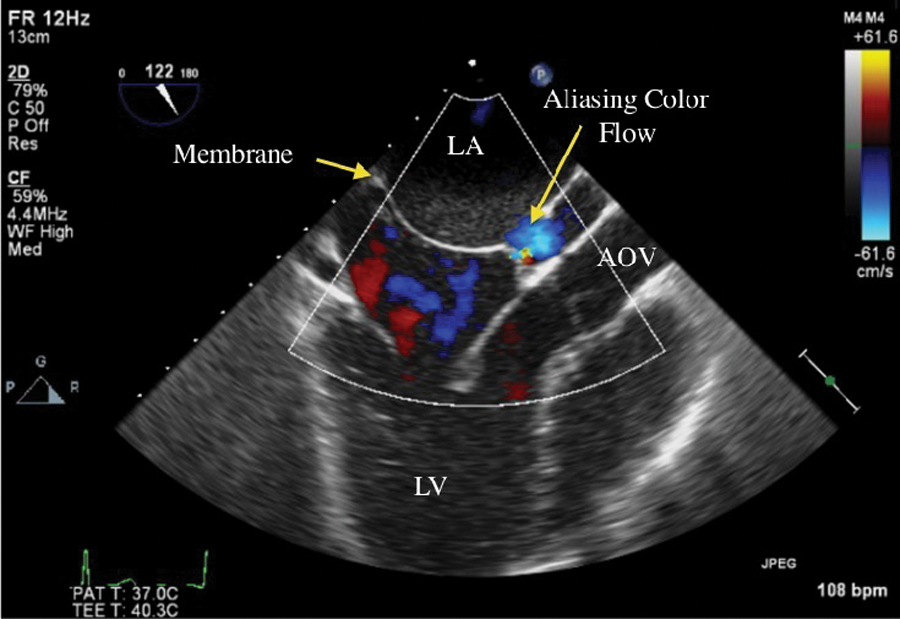
A 2D transesophageal echocardiography apical long-axis view with color flow Doppler across the membrane. Color flow aliasing is seen just above the AOV, which is suggestive of obstruction. LA, left atrium; LV, left ventricle; AOV, aortic valve.
In contrast to the nonobstructive case, 3DTEE delineated the triatriatum membrane with improved accuracy in anatomical appearance compared with conventional 2DTEE imaging, including the en face view of the membrane visualized from the left atrium toward the left ventricle (Video 2, available online). The movement and echolucent properties seen in the middle of the membrane were not evident, as was seen in the nonobstructive images (see Video 1A,B). The appearance of an opening or “tunnel” at the end of the membrane proximal to the aortic valve was noted. With the ability to orient the 3D image, the orifice was visualized through the membrane (Video 3, available online). The postoperative surgical report described the membrane as highly calcified with an opening about the size of a nickel (approximately 20 mm), correlating well with the 3DTEE images (Figure 9A,B).
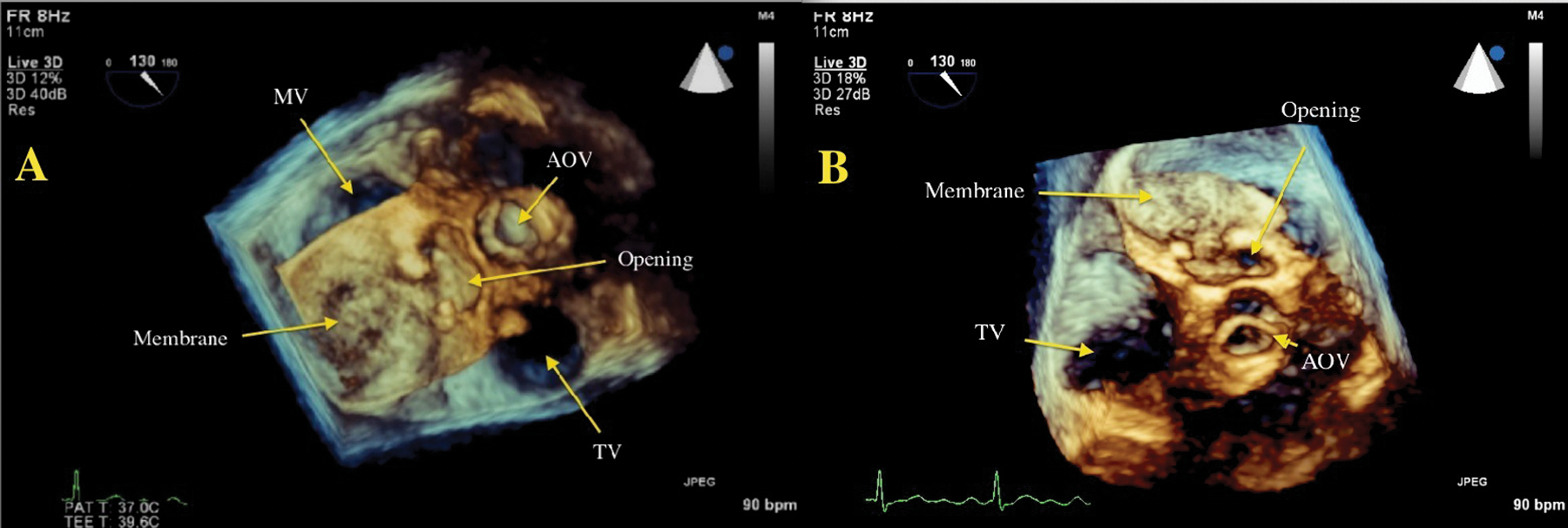
A 3D transesophageal echocardiography Live 3D Zoom dataset. (A) The 3D data set is cropped or cut in from the apex to the left atrial level, demonstrating the “en face” or frontal view of the membrane (arrow). Although not clearly seen, the MV would be visualized under the opaque membrane. The membrane appears to be a solid structure with the appearance of a “tunnel” or opening (arrow) just next to the AOV (arrow). (B) With the ability to rotate the 3D imaging in any plane, the exact pathway of the tunnel and only opening through the membrane was located (panel B arrow, top right). MV, mitral valve; AOV, aortic valve; TV, tricuspid valve.
Comparison With Cardiac Computed Tomography
For comparative assessment, contrast-enhanced CT was also performed in both cases (Figure 10A,B). The CT images confirmed the presence of cor triatriatum defects with a membrane crossing the left atrium and secondary atrial dilation. In the obstructed case, the perforation or hole was described as a highly calcified small fenestration between the membrane along the superior aspect of the left atrium (Figure 10B). No other structural abnormalities were noted.
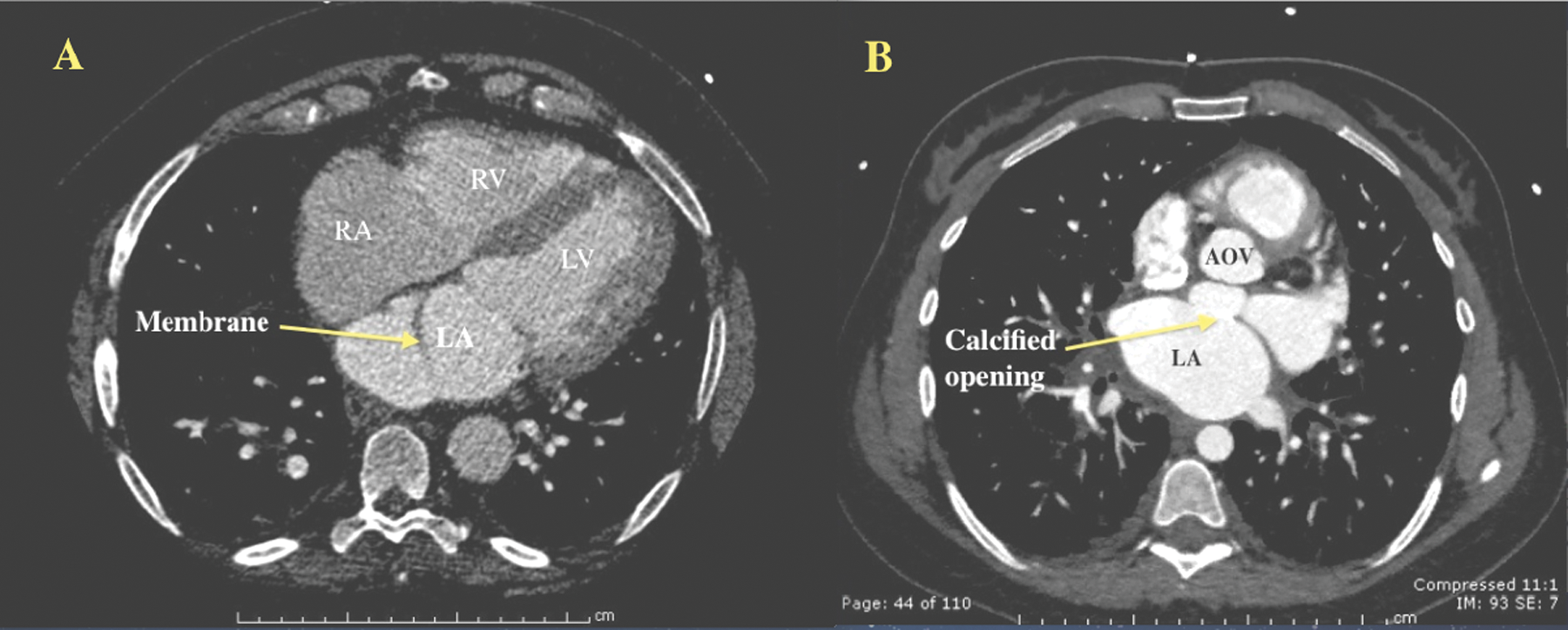
Contrast-enhanced cardiac CT in both nonobstructed (A) and obstructed (B) cases. (A) Long-axis view at the level of the membrane demonstrating a thin linear density. (B) Transverse view at the level of the membrane. The bright white area (arrow) represents the calcified opening that was seen in prior 3D transesophageal echocardiography images. CT, computed tomography; LV, left ventricle; RV, right ventricle; LA, left atrium; RA, right atrium; AOV, aortic valve.
Discussion
Cor triatriatum, first reported in 1868, was described as a congenital anomaly in which the left atrium (cor triatriatum sinister) or right atrium (cor triatriatum dextrum) is divided into two parts by a fibromuscular band. 1 This anomaly is rare, occurring in 0.1% to 0.4% of patients with congenital heart disease. 2 Although most often detected in children and young adults, individuals can present in adulthood.2–5 Both cases presented here involve cor triatriatum sinister, formed as a failure in the development of the common pulmonary veins, resulting in a left atrium divided by a fibromuscular diaphragm. 6 The right valve of the sinus venosus divides the right atrium. Its persistence can develop into a complete septation of the right atrium (cor triatriatum dextrum).6,7
Due to the complexity of this defect, several classifications have been suggested. In 1949, Loeffler 8 classified cor triatriatum into three groups: group 1 has no opening where the left atrium drains into the right atrium, group 2 has one or more small openings resulting in high obstruction, and group 3 has a single, large opening typically with little or no obstruction. Groups 2 and 3 best describe both obstructive and nonobstructive cases presented in this report.
When obstructive, surgical intervention is necessary as symptoms may mimic that of mitral stenosis, causing pulmonary venous congestion and pressure overload of the right heart. 5 Symptoms may include exertional dyspnea, arrhythmia, and fatigue. Cor triatriatum is often associated with additional cardiac anomalies, including atrial septal defects, persistent left superior vena cava, and unroofed coronary sinus. 3 Other associated but less common anomalies include aortic regurgitation with dissecting aneurysm and anomalous pulmonary venous connection. 3
Although both 2DTTE and 2DTEE imaging are often the initial imaging modalities of choice when assessing anatomy and physiology in congenital heart disease, adjunctive 3D imaging may improve diagnosis of cor triatriatum and visualization of the culprit membrane in a way not possible by standard 2D imaging. The addition of 3D imaging offers the ability to view both sides of the membrane. As 3D imaging is primarily used for anatomical assessment with limitations of color Doppler assessment, it is necessary to incorporate conventional spectral Doppler imaging to determine the presence and severity of obstructive disease across a restrictive orifice. Further testing with other imaging modalities such as magnetic resonance imaging and CT may be used to confirm the presence or absence of cardiac malformations.
Conclusion
Cor triatriatum is a rare congenital anomaly that should be considered by the sonographer when a linear hyperechoicity is seen traversing the left or right atrium. These two cases, although rare, highlight the spectrum of cor triatriatum that may be seen in clinical practice. The combined 2D and 3D imaging provided superior interrogation of this rare congenital anomaly and should be considered a standard approach in echocardiography laboratories that have these technological capabilities.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
