Abstract
Echocardiography is an excellent method for evaluating cardiac morphology and dynamic function. It has a long history of innovative thinking mixed with some degree of serendipity. Its early applications were as a tool to evaluate the mitral valve, left ventricular characteristics, and pericardial effusion. Today it has evolved into a robust modality that allows for a very wide range cardiac interrogation, able to evaluate the valves, chambers, myocardium, and pericardium. The practice of echocardiography also is often separate from that of general or vascular sonography. The objective of this article is to provide the non–cardiac sonographer with an overview and appreciation of the basic principles and practices of echocardiography. It is not meant as a guide to scanning but rather, as a vehicle to spark the interest and imagination of the reader.
Introduction
Echocardiography provides the ability to evaluate the morphology and function of the heart and can yield valuable information regarding the cardiac status of the patient. Prior to the development of echocardiography, cardiac catheterization was the primary method of visualization of the heart. Interest in echocardiography in the 1950s led to the use of this noninvasive technique as the first-line study of choice for the evaluation of cardiac anatomy and pathology. 1 Modern echocardiography is able to evaluate the valves, chambers, myocardium, and pericardium of the heart in order to detect a wide range of pathologies utilizing M-mode, two-dimensional (2D), and color and spectral Doppler.
As new technology emerges, the ability of echocardiography to detect, quantify, and treat cardiac pathology and dysfunction will continue to advance. Present and future advances in acquisition methodologies such as three-dimensional instantaneous full volume imaging, speckle tracking, contrast echocardiography, and miniaturization of scanning equipment and intracardiac probes present exciting new paths for the evolution of echocardiography.
Historical Overview
The history of echocardiography is an interesting one filled with innovation, ingenuity, and serendipity. 1 The use of sonography as a diagnostic tool in the detection of heart disease was pioneered by cardiologist Inge Edler and physicist Carl Helmuth Hertz. Elder was disappointed in the ability to detect mitral valve disease using cardiac catheterization techniques. He imagined the possibility of a noninvasive alternative. In the early 1950s, he was referred to Hertz, whose father was the director of Siemens Research Laboratory. Through him, he discovered that Siemens had developed a reflectoscope that was being used for nondestructive metal testing at a local shipyard. Hertz made a visit to the shipyard to see the equipment. During his visit, he placed the transducer on his chest. The first A-mode display of the heart was made that day. 1 Edler and Hertz were able to obtain a modified reflectoscope from Siemens, and they went on to develop M-mode capabilities.
It wouldn’t be until the 1960s that interest in echocardiography came to the United States where cardiologist Harvey Feigenbaum would emerge as one of the leading proponents for the use of echocardiography. The term
In addition to introducing echocardiography’s utility as a method of measuring ventricular dimensions and detecting pericardial effusions, Feigenbaum is credited with advancing standardization and education within the discipline. He established educational programs to train not only physicians but also non-physician operators. He was the first to hire a non-physician to perform echocardiography, and it was his opinion that a productive, dedicated echocardiographer could produce more reliable results than the occasional physician operator. The physician would then interpret the resulting study. In fact, he asserted that “many sonographers become extremely skilled and are almost artists as they create their ultrasonic pictures.” 1
The next major advance in echocardiography was two-dimensional imaging. When a mechanical sector scanner was finally developed, two-dimensional echocardiography (2DE) in the clinical environment became feasible. With the addition of spectral Doppler and color Doppler technology, modern echocardiography was born.
Cardiac Anatomy Review
Understanding the principles of echocardiography must begin with at least a brief understanding of basic cardiac anatomy. The heart has an oblique lie within the thoracic cavity, with two-thirds of its mass residing to the left of the median plane. The apex is formed by the left ventricle at the level of the fifth intercostal space. The atria are the major components of the base of the heart, which projects superiorly, posteriorly, and medial to the apex. The heart has three surfaces: the base or posterior surface, the anterior sternocostal surface, and the diaphragmatic surface. It also has four borders: the right, left, inferior, and superior borders. The heart wall consists of three layers. The outermost of these layers is the epicardium, which is also the visceral layer of the pericardium. The cardiac muscle layer is the myocardium. The endocardium is the innermost endothelial lining. The heart also consists of four chambers and valves, which along with their associated vessels are depicted in Figure 1.
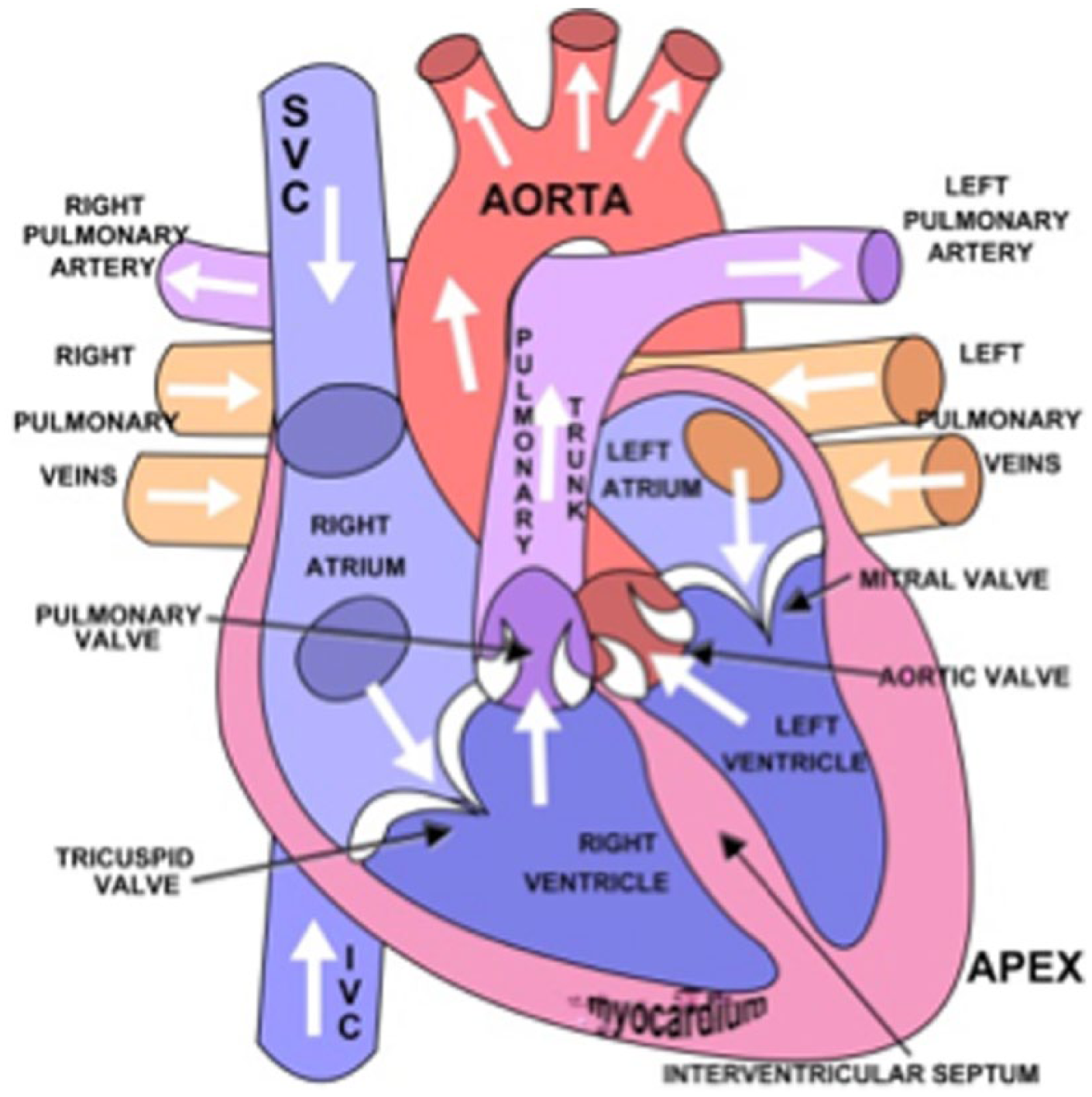
Artist’s drawing of the human heart showing the four chambers and their associated valves, as well as the major inflow and outflow vessels. (Used with paid permission from Shutterstock.com).
The right atrium receives de-oxygenated blood from systemic and coronary veins via the inferior vena cava, superior vena cava, and coronary sinus. The blood then enters the right ventricle and is pumped into the pulmonary trunk for oxygenation during transit through the lungs. The left atrium, which forms most of the base of the heart, receives oxygenated blood from the right and left pulmonary veins. The left atrium empties into the left ventricle, which pumps the oxygenated blood into the ascending aorta. The ventricles are separated by an interventricular septum.
There are four heart valves that consist of fibrous tissue with an endothelial covering. The valves consist of leaflets or cusps that allow them to open and close. There are two basic types of valves: semilunar and atrioventricular. The semilunar valves are found between the ventricles and the great vessels, the aortic valve between the left ventricle and the aorta, and the pulmonic valve between the right ventricle and the pulmonary trunk. The semilunar valves have three cusps that are cup shaped and open during ventricular contraction. They close during ventricular relaxation in order to prevent reflux of blood into the ventricles.2,3
The atrioventricular valves allow blood to flow from the atria to the ventricles. The mitral valve has two cusps and communicates between the left atrium and ventricle. The tricuspid valve has three cusps and communicates between the right atrium and ventricle. The walls of the atrioventricular valves are thinner than those of the semilunar valves, and they readily move in the direction of blood flow. The atrioventricular valves are attached to the ventricular walls by chordae tendineae, cordlike tendons, which attach to the papillary muscles that project from the ventricular walls. 3 They have a fibrous ring, called an annulus, that encircles the opening and provides stability. Ventricular contraction forces the valves closed to prevent reflux of blood into the atria. 2
Blood is supplied to the heart muscle itself by the coronary arteries. The right and left coronary arteries arise from the ascending aorta. They then branch to provide blood supply to the entire cardiac muscle. Coronary venous drainage is achieved primarily through the coronary sinus, a thin-walled venous channel on the posterior surface of the heart that empties into the right atrium. 2 The pericardial sac encloses the heart muscle. It is comprised of two layers, the visceral and parietal layers. The visceral layer forms the outer layer of the cardiac wall and is alternately referred to as the epicardium. A small amount of serous fluid lubricates the space between the two layers.
Echocardiography Basics
Traditional echocardiography incorporates a variety of acquisition methodologies, including M-mode, 2D echo, pulsed and continuous wave Doppler, and color flow Doppler. M-mode continuously interrogates along a single scan line and is useful for timing cardiac events and providing dimensional measurements. The 2D echo evaluates cardiac anatomy, real-time ventricular and valvular motion, and is used to position the M-mode and spectral Doppler cursors. Spectral Doppler provides velocity measurements. Pulsed wave Doppler primarily evaluates valvular flow patterns, left ventricular diastolic function, stroke volume, and cardiac output. Continuous wave Doppler, which does not suffer from the aliasing artifact, is useful in high flow velocity situations, such as evaluation for valvular stenosis and regurgitation, as well as providing information on the velocity of flow in shunts. Color flow Doppler superimposes a color map of the presence and direction of flow over a 2D gray-scale image and provides a rapid, qualitative means of detecting regurgitation. 4
Transthoracic Echocardiography
The typical transthoracic echocardiogram is obtained with the patient in either a supine or left lateral decubitus position. The echocardiographer may scan the patient using either the left or right hand, depending on environmental ergonomics or echocardiographer preference. It is advantageous for the echocardiographer to gain experience with both approaches so as to minimize the potential for repetitive stress injuries. 5
Scanning Windows and Views
There are several scanning positions, or windows, that are helpful in obtaining a complete transthoracic echocardiographic evaluation of the heart: the left parasternal, apical, subcostal, and suprasternal notch (Figure 2). The right parasternal position, though not a part of the normal routine echocardiogram, may also be used. Several views are utilized in the course of a typical examination in order to visualize structures within the heart, as shown in Table 1 and in Figures 3, 4, and 5.
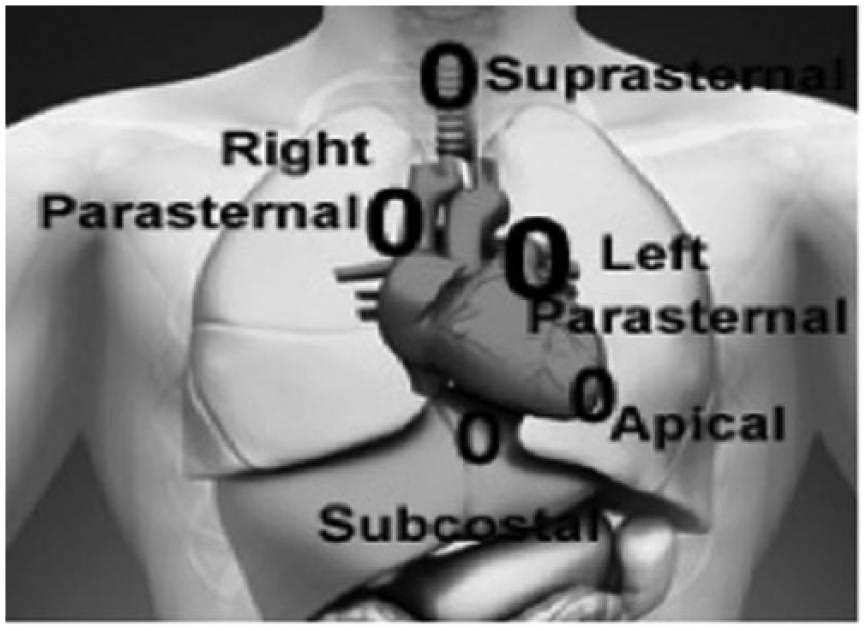
The basic scanning windows for transthoracic echocardiography. (Used with paid permission from Shutterstock.com).
Standard Transthoracic Echocardiographic Anatomic Windows and the Cardiac Data Derived From Each (adapted from Kaddoura 4 and Armstrong and Ryan 5 ).
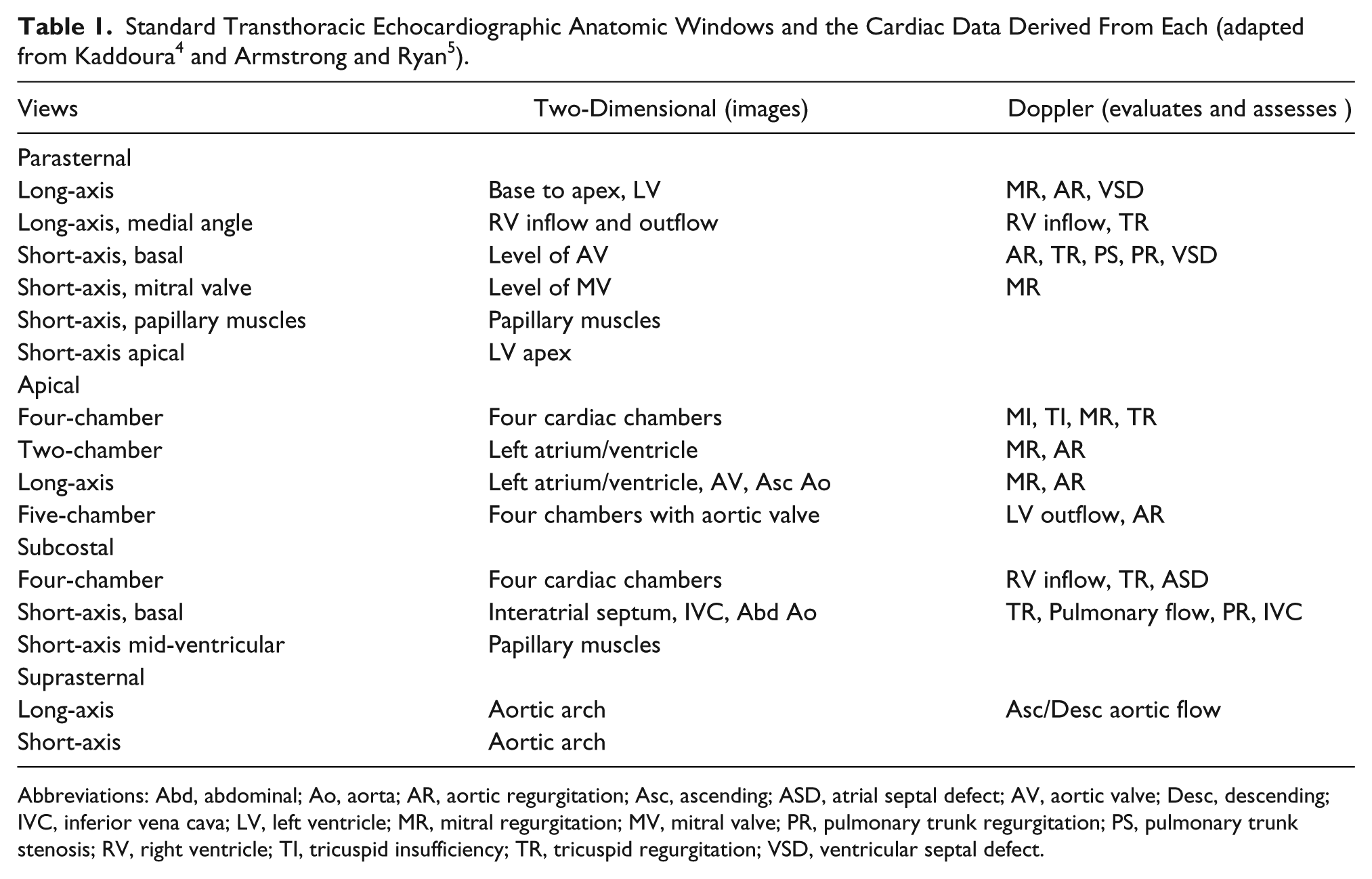
Abbreviations: Abd, abdominal; Ao, aorta; AR, aortic regurgitation; Asc, ascending; ASD, atrial septal defect; AV, aortic valve; Desc, descending; IVC, inferior vena cava; LV, left ventricle; MR, mitral regurgitation; MV, mitral valve; PR, pulmonary trunk regurgitation; PS, pulmonary trunk stenosis; RV, right ventricle; TI, tricuspid insufficiency; TR, tricuspid regurgitation; VSD, ventricular septal defect.
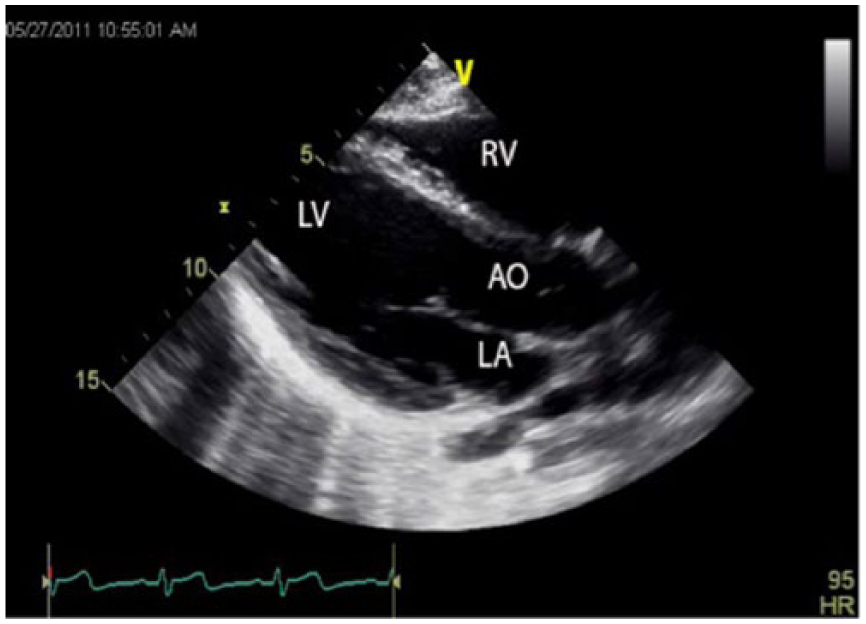
Gray-scale echocardiographic image of the heart taken using the parasternal long-axis view, showing both the left (LV) and right (RV) ventricles, the left atrium (LA), and the aortic outflow tract (AO).
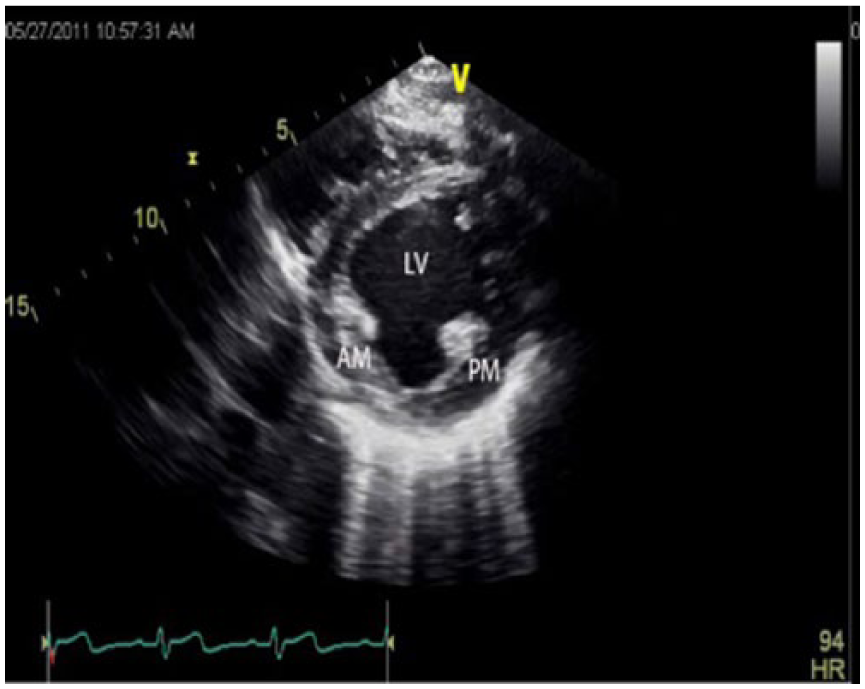
Gray-scale echocardiographic image of the heart taken at the approximate midlevel of the left ventricle (LV) using the parasternal short-axis view, showing the anterolateral (AM) and posteromedial (PM) papillary muscles as well.
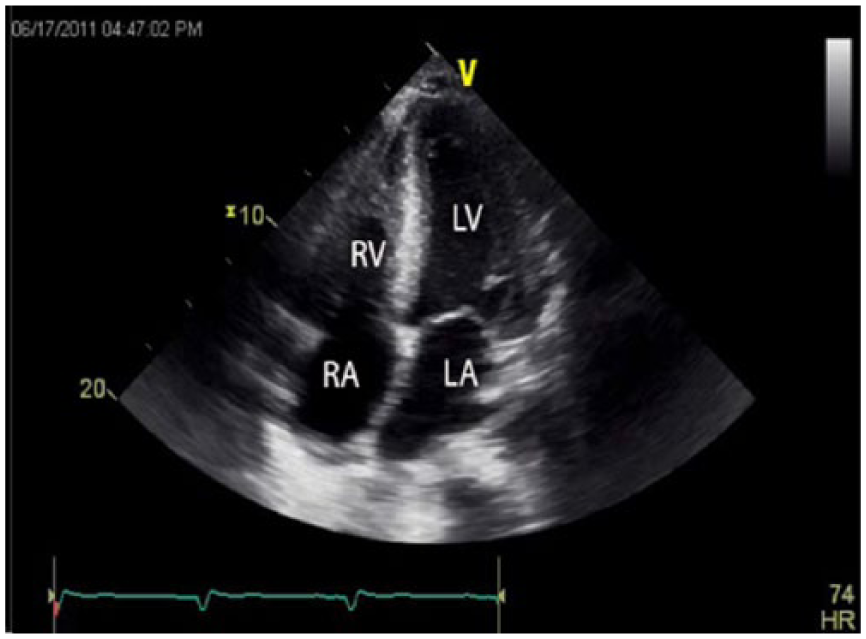
Gray-scale echocardiographic image of the heart using the apical four-chamber view showing all four chambers. LA, left atrium; LV, left ventricle; RA, right atrium; RV, right ventricle.
Transesophageal Echocardiography
Transesophageal echocardiography (TEE) is an invasive procedure that involves inserting a flexible echocardiography probe into the esophagus. It has the advantage of an anatomically closer visualization of the heart, which allows better resolution through the use of higher probe frequencies, with less artifact interference. TEE also provides visualization of cardiac structures that are not accessible by the transthoracic method due to their posterior location and provides a superior method for the evaluation of cardiac tumors and thrombi. Its disadvantage is its invasive nature, which carries a small potential risk. It also may be limited in patients with dysphagia, esophageal disease, cervical instability, and severe pulmonary disease.
Cardiac Pathology
Valvular Diseases
Evaluation of the heart valves, specifically the mitral valve, was one of the earliest applications of echocardiography. The mitral valve was historically imaged using M-mode to record valve cusp motion and to evaluate how the valve functioned over a period of time. Today, 2DE is considered the reference method for evaluation of mitral valve abnormalities, which may include stenosis, regurgitation, and/or prolapse. Advances in technology and technique have expanded the ability of echocardiography to include evaluation of all four valves by M-mode, 2DE, and Doppler. The most common cause of valvular dysfunction is a history of rheumatic heart disease. 5
Stenosis can occur in any of the four valves to a greater or lesser degree, and it generally is a result of a thickening of the valve cusps that restricts their ability to open fully. While M-mode can provide information regarding reduction in valvular motion, 2DE allows for direct visualization of the orifice and the associated valve anatomy. Color and spectral Doppler have proven to be the most useful methods of evaluation of valvular stenosis, as they can provide both flow direction and pressure gradient information and allow quantification of the area of stenosis. 6
In cases of mitral and tricuspid valve stenosis, the thin leaflet tips typically become stiff and may calcify. Mitral stenosis is far more common a condition than tricuspid stenosis.7–9 When tricuspid stenosis does occur, it is usually in conjunction with mitral stenosis.10–12
The aortic and pulmonary valves have thicker leaflets than the mitral and tricuspid valves, and stenosis in these valves may be at any one of three levels, valvular, subvalvular, or supravalvular, with the most common location the valvular level. 5 Both aortic and pulmonary stenosis may be congenital or acquired. Pulmonary stenosis is more often congenital, whereas aortic stenosis is often acquired. 13 Aortic stenosis is most commonly the result of rheumatic heart disease, age-related degenerative changes, and congenital bicuspid valve.14–16
Regurgitation is a reflux of blood flow through the valve during the cardiac cycle. Mitral and tricuspid regurgitation occur during systole and result in backflow from the corresponding ventricle to the atrium. It may be caused by damage to the valves and supporting structures due to rheumatic disease and age-related deterioration. It may also result from rupture of the papillary muscle or chordae tendineae. 5 Tricuspid regurgitation is a relatively common finding in adult patients and usually results from right ventricular dilation. Continuous wave (CW) Doppler velocity measurements of tricuspid regurgitation are useful in the estimation of pulmonary artery systolic pressure.17–20
Aortic and pulmonary regurgitation occur during diastole and result in a backflow of blood from the great vessels into the ventricles (Figure 6). It is frequently caused by abnormalities of the aorta or the pulmonary artery. It may also be caused by leaflet abnormalities. M-mode and 2DE are both effective methods for evaluating the aortic valve for regurgitation but cannot directly assess pulmonary regurgitation. Doppler remains the method of choice for definitive diagnosis of pulmonary regurgitation.21–26
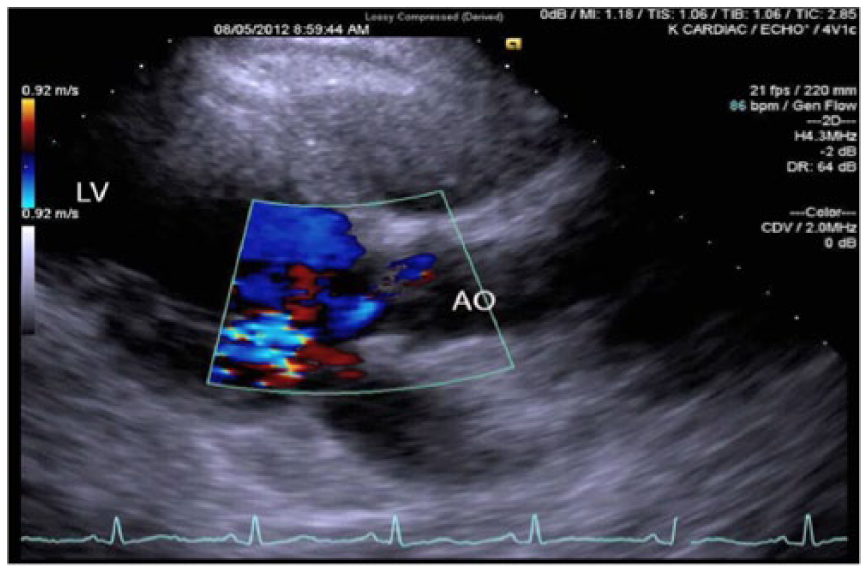
Gray-scale and color Doppler echocardiographic image of the heart taken using the parasternal long-axis view, showing reflux flow secondary to aortic insufficiency in the aortic outflow tract (AO) at the level of the aortic valve.
Improper or abnormal valve closure, or prolapse, occurs most commonly in the mitral valve. The systolic protrusion of one or both of the valve leaflets into the left atrium secondary to mitral valve prolapse may be mild or it may be severe enough to cause regurgitation during systole. It is a common condition that affects up to 5% of the population 4 and may be identified by 2DE when either of the leaflets protrudes above the level of the mitral annulus during systole.27–30
Chamber Size and Function
The cardiac chambers are examined during a routine transthoracic echocardiogram for both function and morphology. The left ventricle, in particular, is measured both in diameter and volume to allow the calculation of functional information such as stroke volume and ejection fraction. These are clinically relevant particularly when ischemic changes secondary to myocardial infarct are suspected.31–33 The left atrium can offer important information relevant to several disease states such as dilation, thrombus, and diastolic dysfunction.34–37 The right atrium may become dilated in the case of right ventricular pressure and volume overload.38,39 The right ventricle is not usually well seen on echocardiography due to its location behind the sternum. It can, however, be grossly measured and a visual comparison made with the left ventricle.40,41 The normal right ventricle should be approximately two-thirds the size of the normal left ventricle. The interatrial septum should be evaluated for any defects, 42 particularly a significant patent foramen ovale (PFO). PFO is common, occurring in 25% to 30% of adults, though only a small fraction of these are clinically significant. 5 The interventricular septum should also be evaluated for ventricular septal defects or rupture.43–45
Cardiomyopathy
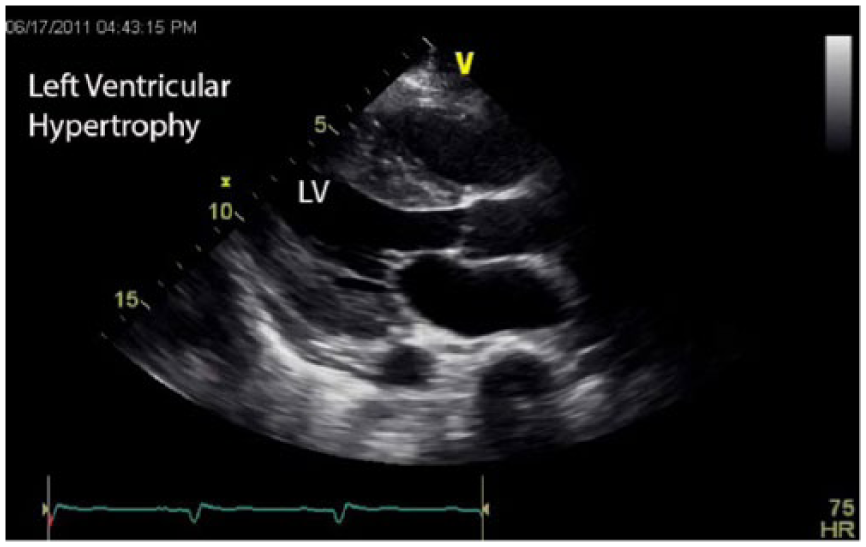
Gray-scale echocardiographic image of the heart taken using the parasternal long-axis view showing thickening of the muscular layers of the left ventricular (LV) wall characteristic of ventricular hypertrophy.
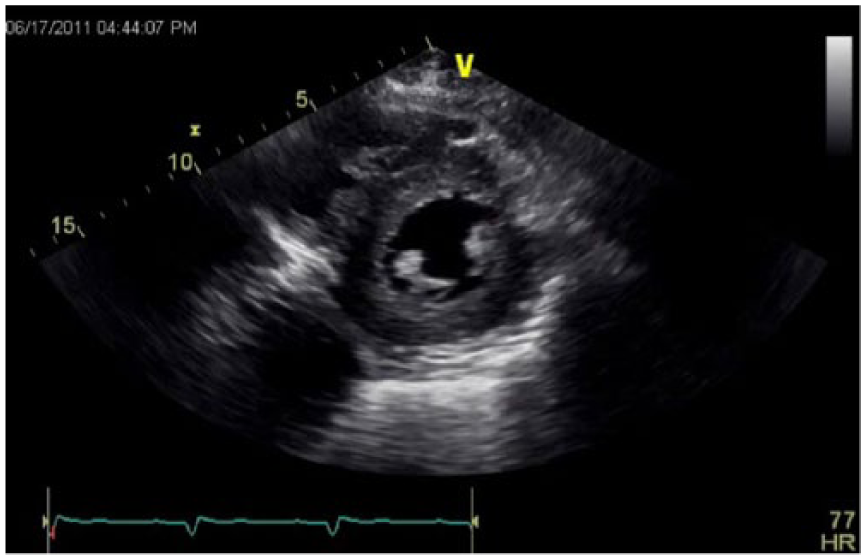
Gray-scale echocardiographic image of the heart taken using the parasternal short-axis image view showing concentric thickening of the left ventricular wall characteristic of ventricular hypertrophy.
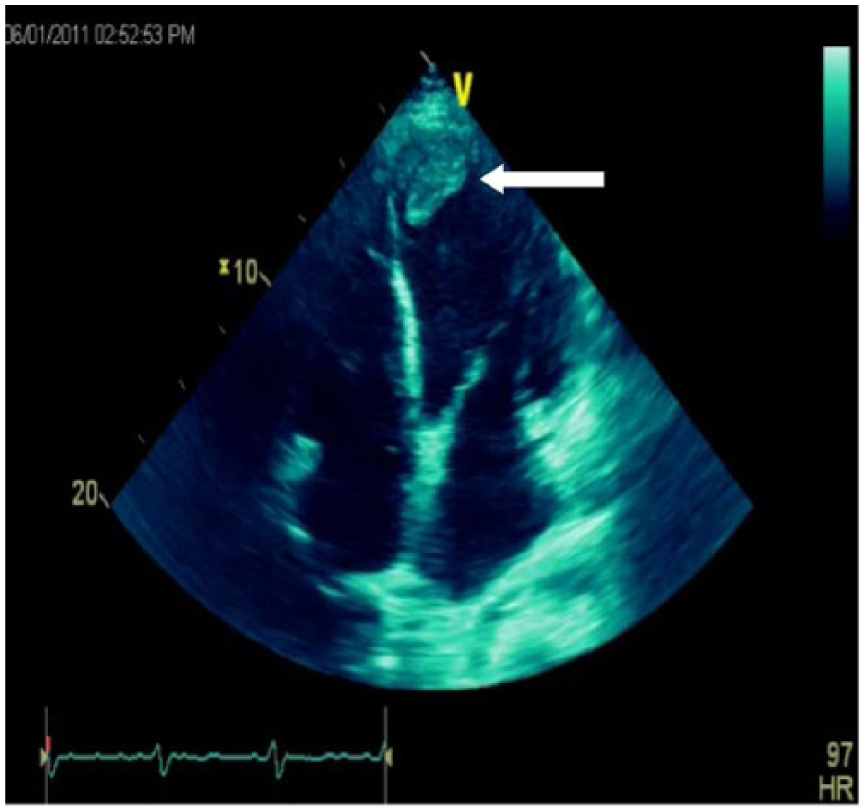
Colorized gray-scale echocardiographic image of the heart using the apical four-chamber view showing a thrombus in the apex of the left ventricle (arrow).
Pericardium
The pericardium, the lining that surrounds the heart, can also become diseased and cause complications. The three major categories of pericardial complications include pericardial effusion, constrictive pericarditis, and cardiac tamponade.5,53,54 Pericardial effusion is the accumulation of fluid between the layers of the pericardium (Figure 10). Its evaluation was one of the first clinical applications of echocardiography. It may be secondary to an inflammatory or an infectious process. It is possible to detect an effusion with M-mode, as was originally done, but it is much better assessed with 2DE.55,56
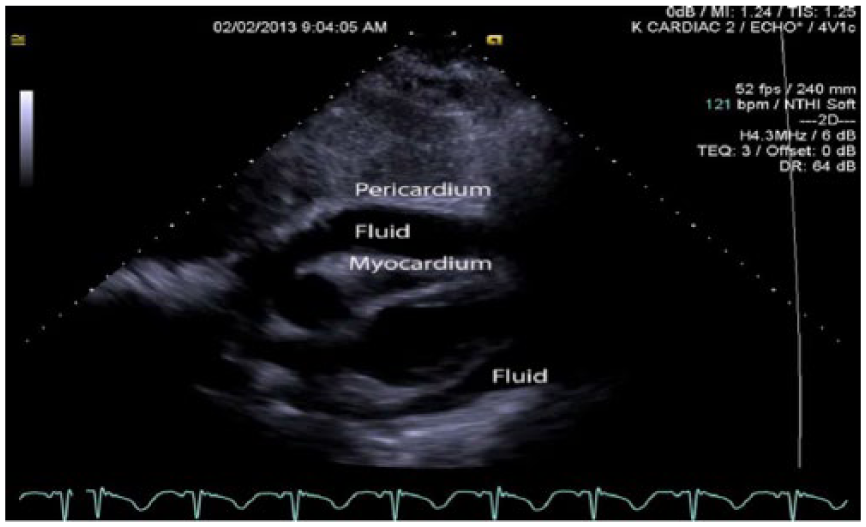
Gray-scale echocardiographic image of the heart showing fluid between the pericardium and myocardium (pericardial effusion) causing extrinsic compression of the right ventricle.
Constrictive pericarditis is a condition in which the pericardium becomes rigid and may even become calcified.50,51,57 It may be the result of disease or injury, or it may have an idiopathic origin. As the name would imply, it restricts ventricular movement, limiting expansion, and filling of the ventricles. Doppler can detect abnormal flow patterns of the mitral valve as a result of abnormal diastolic ventricular filling.50,51
Cardiac tamponade results from external pressure on the heart that is significant enough to decrease cardiac function. Even small effusions may cause tamponade if they occur suddenly, as the pericardial sac may not have time to adjust and stretch to accommodate the excess fluid. 58 Tamponade is an emergency situation that may require the performance of therapeutic echocardiographic-guided pericardiocentesis to relieve the fluid pressure.59–61
Stress Echocardiography
Stress echocardiography provides information about the effects of exercise on the heart and can detect changes in morphology and function of the stressed heart as compared with the resting heart. Some ischemic regional wall motion and thickness abnormalities may only become apparent when the heart is stressed.4,5,62 The physiologic stress may be induced chemically or by physical exercise by the patient. Stress echocardiography may be particularly helpful in women with complaints of chest pain, as ECG stress testing alone has been shown to be less reliable in this demographic.63–65
Current Trends and Future Advances
Three-Dimensional Echocardiography
Until very recently, three-dimensional echocardiography (3DE) systems depended on time-consuming and cumbersome reconstruction of 2DE images that had to be synchronized with the electrocardiogram and respiration. Not surprisingly, it was not a widely popular acquisition method and was used primarily in research facilities. Advances in transducer and software technology have allowed for real-time acquisition of 3D images over several cardiac cycles. Automated reconstruction software makes the images available for offline analysis (Figure 11). Even with these advances, however, this method is still only useful as an adjunct to 2DE and does not replace it.
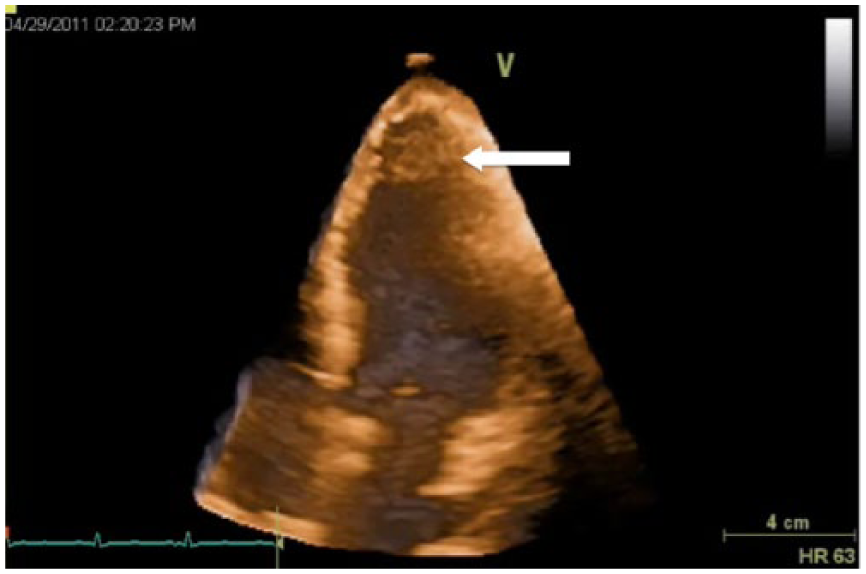
Reconstructed colorized three-dimensional echocardiographic image of the left ventricular showing an apical thrombus (arrow).
With the development of instantaneous full-volume echocardiography, acquisition can now be obtained in a single heartbeat and analyzed in real time. 66 Instantaneous full-volume imaging has overcome some of the temporal resolution issues present with earlier 3DE methods. It has been successful in accurately identifying valvular pathologies and can precisely recognize prolapsed segments. It has also shown to be promising in the assessment of mitral regurgitation and mitral stenosis. Conventional 2DE is considered to be the reference method for evaluating mitral stenosis, but it can lead to overestimation of the mitral valve area. 3DE allows for the manipulation of image orientation in real time, thus allowing for capture of the most optimal plane from which to measure. The 3DE is able to image the aortic and tricuspid valves in ways that allow better visualization of anatomic features. The potential of 3DE to evaluate the origins of the coronary arteries is also proving to be very promising. 67
Speckle Tracking
Speckle tracking echocardiography (STE) quantifies myocardial function by tracking the speckle pattern with the 2DE image. Imaging software merges the individual speckles into functional units called kernels. Each kernel has a unique pattern of speckles, much like a fingerprint, that makes them readily identifiable by the tracking software.
The elements of myocardial function that are analyzed by STE are strain, strain rate, twisting, torsion, and untwisting. Strain is the amount of deformation the myocardium undergoes during the cardiac cycle. Strain rate is a measure of the rate of myocardial deformation, or how quickly the myocardium deforms. 68 Twisting is a normal part of left ventricular systolic contraction and is the reciprocal of the rotation of the apex and base during systole. Torsion uses twisting values normalized with respect to the base to apex distance. Twisting and untwisting play an important role in diastolic filling. 69
Image acquisition for STE is currently performed using conventional 2DE and analyzing the information offline. The images are acquired using a breath holding technique and electrocardiographic (ECG) tracing. Analysis is performed at dedicated workstations with specific speckle tracking software. Potential clinical applications for STE include the cardiac evaluation of patients with hypertension, diabetes, coronary artery disease, valvular heart disease, heart failure, cardiomyopathies, and as a follow-up to resynchronization therapy. It may also prove to be useful in the evaluation of heart transplantation, left atrial function, and chemotherapy-induced subclinical dysfunction. 69 One future advantage of STE is that it provides a method for evaluating myocardial function in ways that were only previously possible by magnetic resonance imaging. New technology is allowing it to be merged with three-dimensional technology, which may yield even more sophisticated data sets. As the acquisition and analytic capabilities of STE improve, it may emerge as the most advanced method for the evaluation of myocardial function. 69
Contrast Echocardiography
Echocardiography, while an excellent diagnostic tool, does have technical limitations that result in suboptimal imaging on some patients. Obesity and lung disease, for instance, can significantly reduce the diagnostic quality of the routine transthoracic echocardiogram. 70 Contrast echocardiography (CE) has been able to overcome some of these limitations. Contrast agents used in echocardiography consist of microbubbles made from a high molecular weight gas. The microbubbles, slightly smaller in size than a red blood cell, are injected intravenously and freely diffuse throughout the blood pool. They enhance visualization of the left ventricular cavity and myometrium because the microbubbles have highly echogenic sonographic characteristics that differentiate them from the surrounding tissue and red blood cells. An important feature of microbubble design is the encapsulating shell surrounding the gas; it must be elastic enough to compress in an ultrasound field and allow for resonant oscillation but not be so stiff as to rupture too easily. 71
CE allows improved visualization of the left ventricular endocardial borders (Figure 12), and this is the only FDA-approved application thus far. It may also provide significant information regarding myocardial perfusion, but the FDA has not yet approved it for that use in the United States. It has proved useful for the presence or exclusion of apical hypertrophic cardiomyopathy, left ventricular thrombus, and complications of myocardial infarction and can be used for both routine and stress echocardiography.70,71
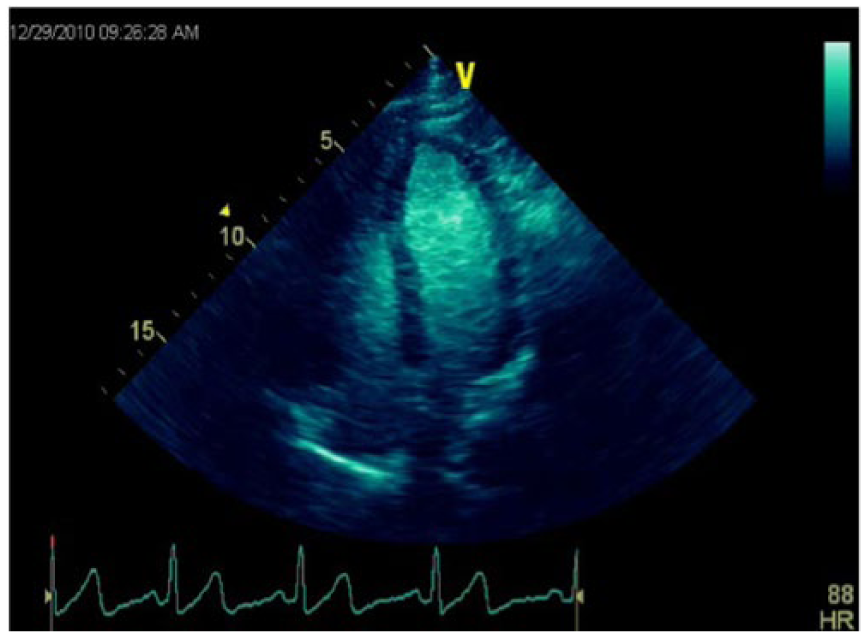
Contrast enhanced echocardiographic image showing opacification of the left ventricle with good visualization of the endocardial borders.
Certain limitations and safety concerns exist with the use of CE. Ultrasound power settings play significantly into these concerns. Cavitation, the interaction of ultrasound with gas, has long been a bioeffects consideration for diagnostic ultrasound. The introduction of contrast for echocardiography introduces gas containing bodies into the circulation and raises additional concerns that cavitation may result in damage to nearby cells. 72 Since the risk increases with increased power, the overall transmitted ultrasound power should be significantly reduced so as to control destruction of contrast microbubbles and to reduce the risk of tissue heating and cavitation. A recent prospective multicenter study of the contrast agent Definity (Lantheus Medical Imaging, North Billerica, Massachusetts, USA) 70 and a registry review of both Definity and Optison (GE Healthcare, Milwaukee, Wisconsin, USA) 73 have addressed this and other concerns. These studies showed no serious adverse events associated with contrast echocardiography.
Miniaturization
Miniaturization has had a huge impact on technology, and echocardiography is no exception. Echocardiography equipment 20 years ago was roughly the size of a refrigerator. Today’s fully functional echocardiography machines can be as small as a laptop computer. 74 Transesophageal echocardiography is used routinely in operating rooms to monitor cardiac hemodynamics during surgery, but until recently catheter size has been a significant limitation to more widespread use. New developments have led to probes that are approximately the size of nasogastric catheters, and these transducers are able to provide on-demand hemodynamic assessment for the intensive care patient. 75
Pocket-sized scanners that are the approximate size of a smartphone have also come onto the market. These scanners have full 2D and limited Doppler and color Doppler capabilities. Images may be stored to a standard SD card and uploaded to a computer for permanent storage or printing. Echocardiography capabilities of pocket-sized point-of-care scanners include bedside evaluation for pericardial and pleural effusions as well as ventricular and valvular function. It must be noted that appropriate training for all potential users must accompany the availability of pocket scanner for them to reach their full potential. 76 It must also be understood that they are not meant to replace conventional echocardiography but are meant as a first-line evaluation and triage tool. 77
Conclusion
Echocardiography has achieved significant advancements since its beginnings as an M-mode tool to assess mitral valve function and pericardial effusion. Sixty years of technological advances and innovative design features have made echocardiography an essential tool for the evaluation of cardiac morphology and function. The addition of sophisticated signal acquisition methods such as 3DE, speckle tracking, and contrast echocardiography have added new and exciting dimensions to the contribution that echocardiography can make to cardiology. The extent of the ability of echocardiography to interrogate the heart is still evolving today, and the promise of future innovations will keep the technology in the forefront in the battle against heart disease.
Footnotes
Acknowledgements
I would like to acknowledge Kene Osuno, RDCS, for his contribution of the cardiac images used and his review of this article.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
