Abstract
Schizencephaly is a rare developmental disorder of the brain that is characterized by abnormal clefts within the cerebral hemispheres. Infants with this disorder commonly have developmental delay, delay in speech and language, partial or complete paralysis, poor muscle tone, and seizures. The manifestations and severity of the disease are related to the size and location of the clefts. The authors report a case of type II (open-lipped) schizencephaly diagnosed in the second trimester by sonography. Early prenatal diagnosis of fetal type II (open-lipped) schizencephaly allows time for patient counseling and discussion of options and preparation.
Schizencephaly is an extremely rare condition, and the incidence is currently unknown. Most cases are thought to be due to generalized or localized brain ischemia leading to the characteristic cerebrospinal fluid–filled clefts within the brain. 1 Several processes that cause the ischemia have been proposed, including hypotension, vascular agenesis, or overwhelming antenatal infection. 2 Possible teratogens connected with schizencephaly include congenital infections such as cytomegalovirus, warfarin, and cocaine.3–5 Most cases are thought to be sporadic, although a few familial cases have been identified. Schizencephaly can be identified by prenatal sonography. Amniotic fluid volume, placenta, and fetal growth are usually within normal limits. Sonographic findings include midline cerebrospinal fluid–filled clefts.
Case Study
A 14-year-old gravid 1 presented for evaluation of an abnormal sonogram finding in the fetal brain. The patient’s past obstetrical, gynecological, medical, and surgical histories were unremarkable. Her social history was negative for any tobacco, alcohol, or drug abuse. Her family history was negative for any mental retardation, birth defects, or genetic abnormalities. Routine prenatal labs were normal. The sonogram, performed with a Phillips IU22 (Philips, Seattle, Washington) and a C5-1 transducer, revealed an intrauterine pregnancy in cephalic presentation at 27 weeks and 2 days’ gestation. Estimated fetal weight was 1099 grams, putting the fetus in the 50th percentile for growth. Evaluation of the brain parenchymal revealed a unilateral fluid-filled defect extending from the inner aspect of the skull to the temporal horn of the lateral ventricle (Figure 1). The choroid plexus was observed to be dangling within the fluid collection. The defect was noted to be open and communicated with the intraventricular fluid and extended from just posterior to the thalamus laterally to the inner table of the skull. Approximately 45% of the surface gray matter lateral to the third ventricle was absent. The calvarium overlying the defect appeared normal, and the remainder of the anatomic survey was unremarkable.
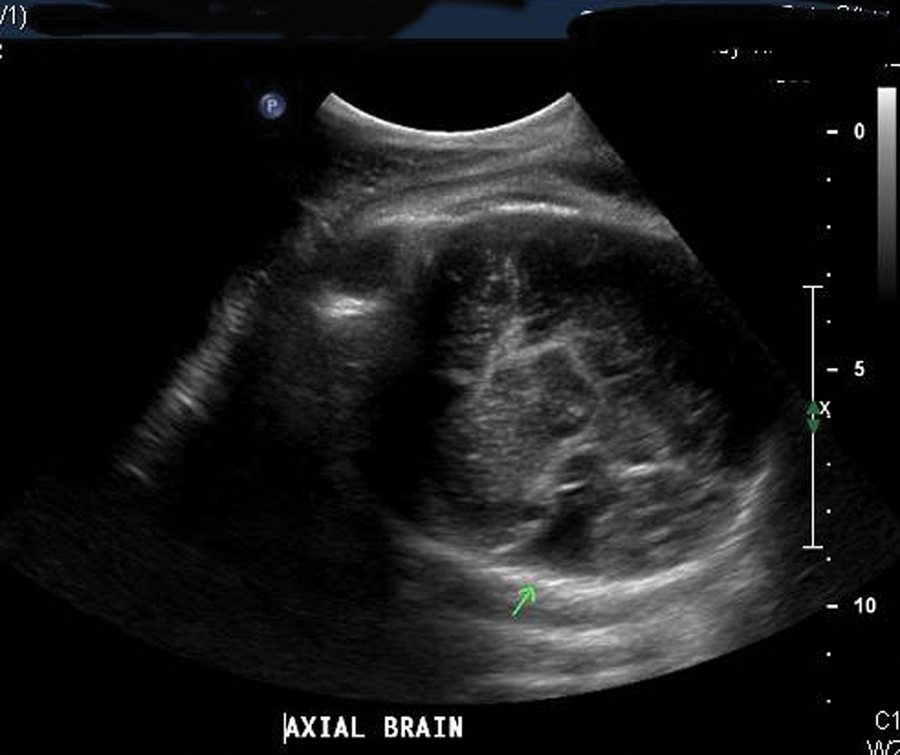
Fetal head at 27 weeks showing fluid-filled defect (arrow).
On the basis of these findings, the fetus was diagnosed with type II schizencephaly. The patient was informed of the overall poor prognosis and was referred to genetic counseling and neonatology. During the course of her initial evaluation, the patient was offered chromosomal analysis via amniocentesis, which she declined. Another sonogram was performed at 35 weeks and 4 days’ gestation (Figure 2), which showed a head circumference of 29 cm. This measurement was 3 weeks behind the expected head circumference for gestational age, and there was an overall drop in estimated fetal weight to the 21st percentile. Anatomically, there was continued irregular dilatation of the left cerebral ventricle with a wedge-shaped defect in the parietotemporal region. An axial scan performed at the same time did not reveal evidence of absent cavum septum pellucidum (Figure 3). The antenatal course was complicated by gestational hypertension, and she ultimately delivered vaginally at term. She had a spontaneous vaginal delivery of a viable infant weighing 5 lbs 13 oz with no evidence of immediate compromise. A sonographic encephalogram on day of life 1 revealed a cleft-like defect in the left hemisphere that appeared to communicate with the left lateral ventricle extending to the cortical surface (Figure 4).
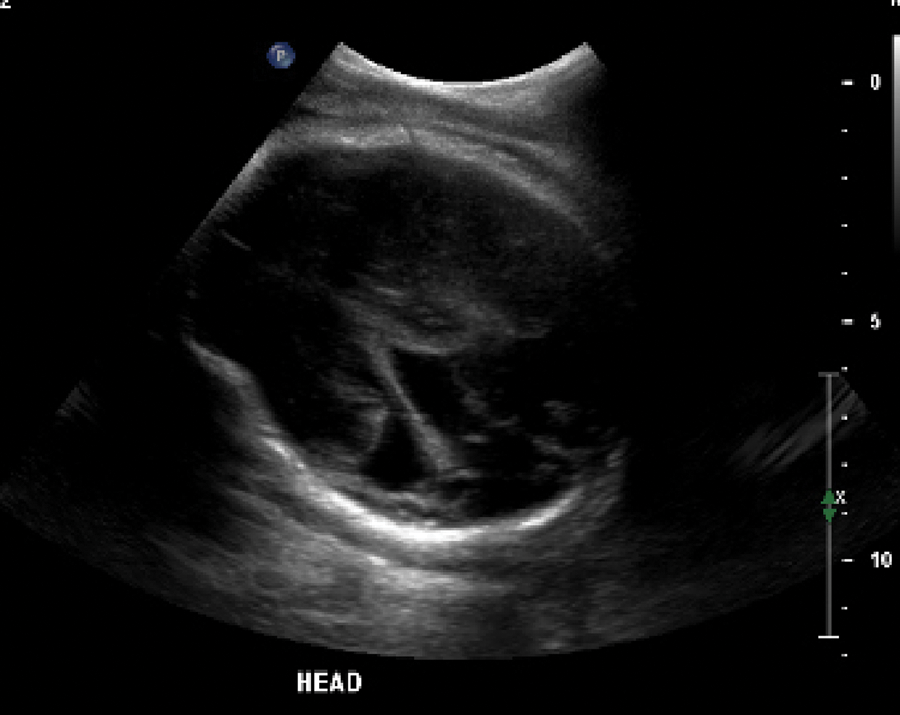
Fetal head scan at 35 weeks at the level of the biparietal diameter.
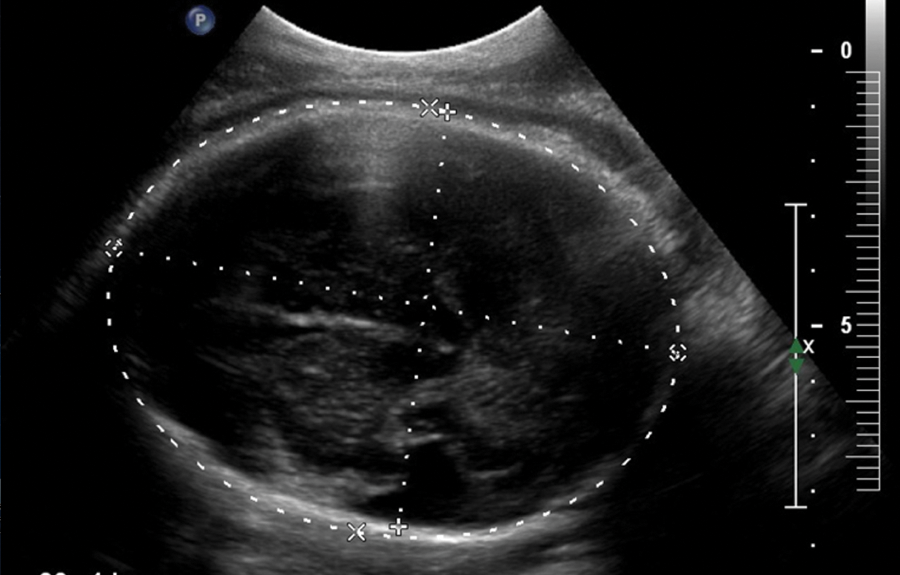
Fetal head at 35 weeks. Head circumference is 3 weeks behind the expected measurement for gestational age.
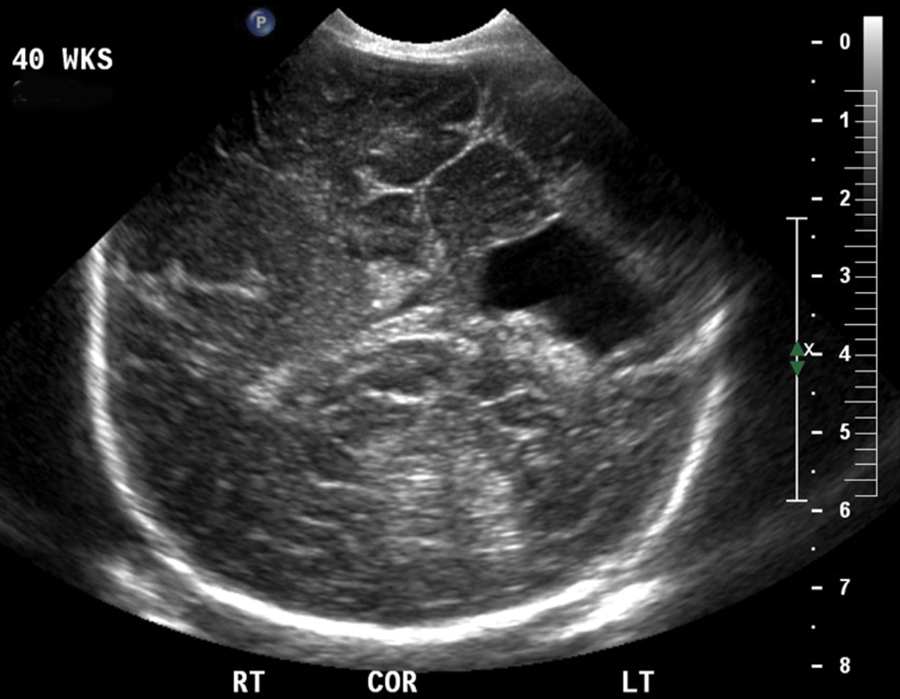
Neonatal head scan on post–delivery day 1.
The patient’s postpartum course was uncomplicated, and she was discharged home on postpartum day 2. The baby was found to have a head circumference in the 25th percentile. The infant had no evidence of seizure activity and no appreciable paralysis. A neonatal brain sonogram revealed a cleft-like defect in the left hemisphere that appeared to communicate with the left lateral ventricle. There was no evidence of hemorrhage. The mother and infant were discharged home at the same time with instructions to follow up with genetics and pediatric neurology.
Discussion
There are two types of schizencephaly. Type I (closed lip) is characterized by apposition of the cleft walls. It is typically unilateral, may fail to communicate with the lateral ventricle, and is characterized by acutely angled borders. In type II (open lip), the cleft walls are separate. The clefts commonly connect to the lateral ventricles, are bilateral, and have a round eccentric shape. There is some association between schizencephaly and other malformations of the brain, including ventriculomegaly, agenesis of the corpus callosum, and absence of the cavum septum pellucidum.6,7
Clinical manifestations vary greatly depending on disease severity. Unilateral clefts with closed lips usually manifest with mild hemiparesis and seizures. Bilateral clefts are associated with severe mental retardation as well as severe motor dysfunction, including spastic quadriparesis. Type II clefts are commonly related with more severe hemiparesis and developmental delays.8,9
Schizencephaly is an uncommon disorder. The etiology is unknown but may represent a primary destructive event within the first 16 to 20 weeks of gestation or an abnormality of neuronal migration during the early developmental period of the brain. 10 The final clinical manifestations are variable and appear to be related to the amount and location of tissue destruction. A recent report suggests that findings on imaging correlate with the ultimate clinical expression, and therefore imaging may be helpful in counseling parents what to expect in their child. 9 Unilateral defects have been associated with late-onset seizures that can be resistant to antiseizure medication. The condition may not shorten the life span.
Sonography is the most often used modality for the diagnosis of schizencephaly in the antenatal period. 11 Diagnosis is based on identification of a cleft within the brain and the determination if the walls of the cleft are open or closed. Magnetic resonance imaging (MRI) may also be used but is not absolutely necessary for the diagnosis. 12 MRI, however, has been shown useful to further characterize the lesion when discovered antenatally and also to rule out porencephaly. 13
Pregnancies complicated by fetal schizencephaly do not require increased monitoring, nor do most require delivery by cesarean section for the condition alone. If there are associated anomalies or the ventriculomegaly results in an expected head size dystocia, a cesarean delivery is indicated. The patient should be delivered at a hospital associated with a neonatal intensive care unit. In this case, after delivery, the infant had a sonographic echoencephalogram that confirmed the diagnosis.
Conclusion
Schizencephaly is a rare abnormality with distinct intracranial structural changes that frequently can be detected by prenatal sonography. Antenatal detection allows consultation with the appropriate team of specialists to refine and confirm the diagnosis, prepare for delivery, and give the patient and her family the options available. Schizencephaly may result in functional impairment of the child, and family expectations can be discussed.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
