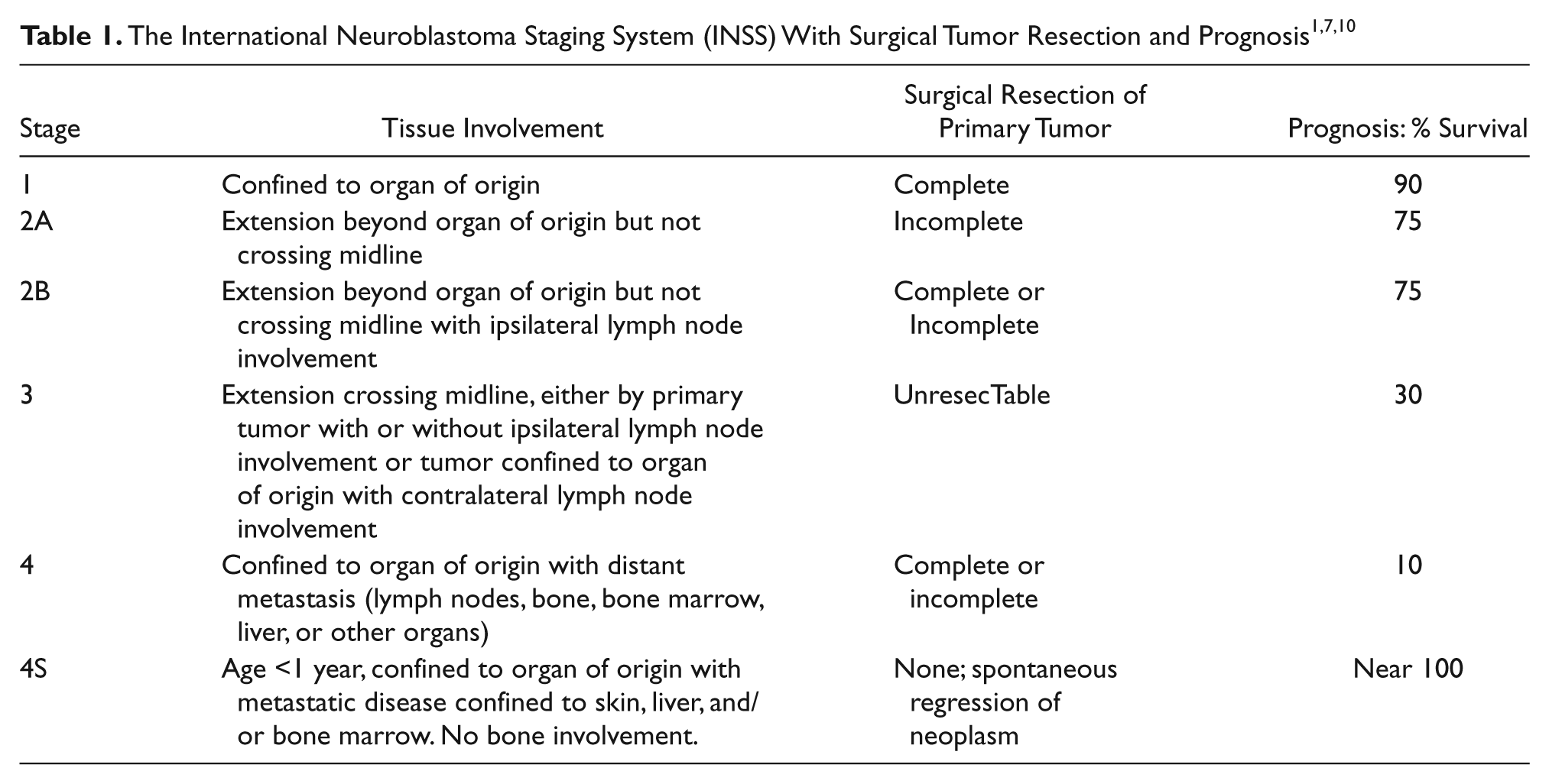
Abstract
Neuroblastoma is a common malignancy of infancy and early childhood. The difference in the outcomes for patients with these neoplasms is striking. Infants younger than one year have a good prognosis, even in the presence of metastatic disease, whereas older patients with metastatic disease fare poorly even when treated with aggressive therapy. The noninvasive nature of diagnostic sonography has allowed it to play a major role in the early detection and treatment of neuroblastoma. This early sonographic detection, in conjunction with early postnatal surgical intervention, has greatly improved the prognosis of infants born with neuroblastoma.
One out of 7000 children born will have a neuroblastoma, which translates into about 600 to 650 new cases in the United States each year.1,2 Neuroblastoma is the most common abdominal malignant neoplasm in neonates and early childhood. This condition most commonly affects children ages five or younger, with the majority occurring at two to three years old.3–5 A study done by Maris et al. 6 found a familial tendency of neuroblastoma in 1% to 2% of cases, although it most commonly occurs sporadically. 1 It has a predisposition to affect males more often than females.5,7 According to Lee and Cho, 8 50% of neonatal malignancies and 14% of childhood malignancies are neuroblastomas. Fatalities associated with neuroblastoma usually occur within two years of diagnosis. 5
Neuroblastoma
Neuroblastomas can be found in various locations in the body where nerve cells exist but are most commonly found in the abdomen (Figure 1). Approximately 65% to 70% of abdominal tumors are located in the adrenal medulla.1,8 In a literature review of 59 cases of prenatally diagnosed neuroblastoma, right-sided adrenal neuroblastomas were more common, occurring in 49% of reported cases. Left-sided adrenal neuroblastomas accounted for 34% of cases and bilateral neuroblastomas just 2% of cases. The remaining 15% of neuroblastomas were undetermined. 8 This is an aggressive neoplasm with extensive metastatic deposits that can occur in the liver, lung, bone, brain, and skin. Neuroblastoma is most likely to spread to the liver and bone, but it is least likely to spread to the lung.
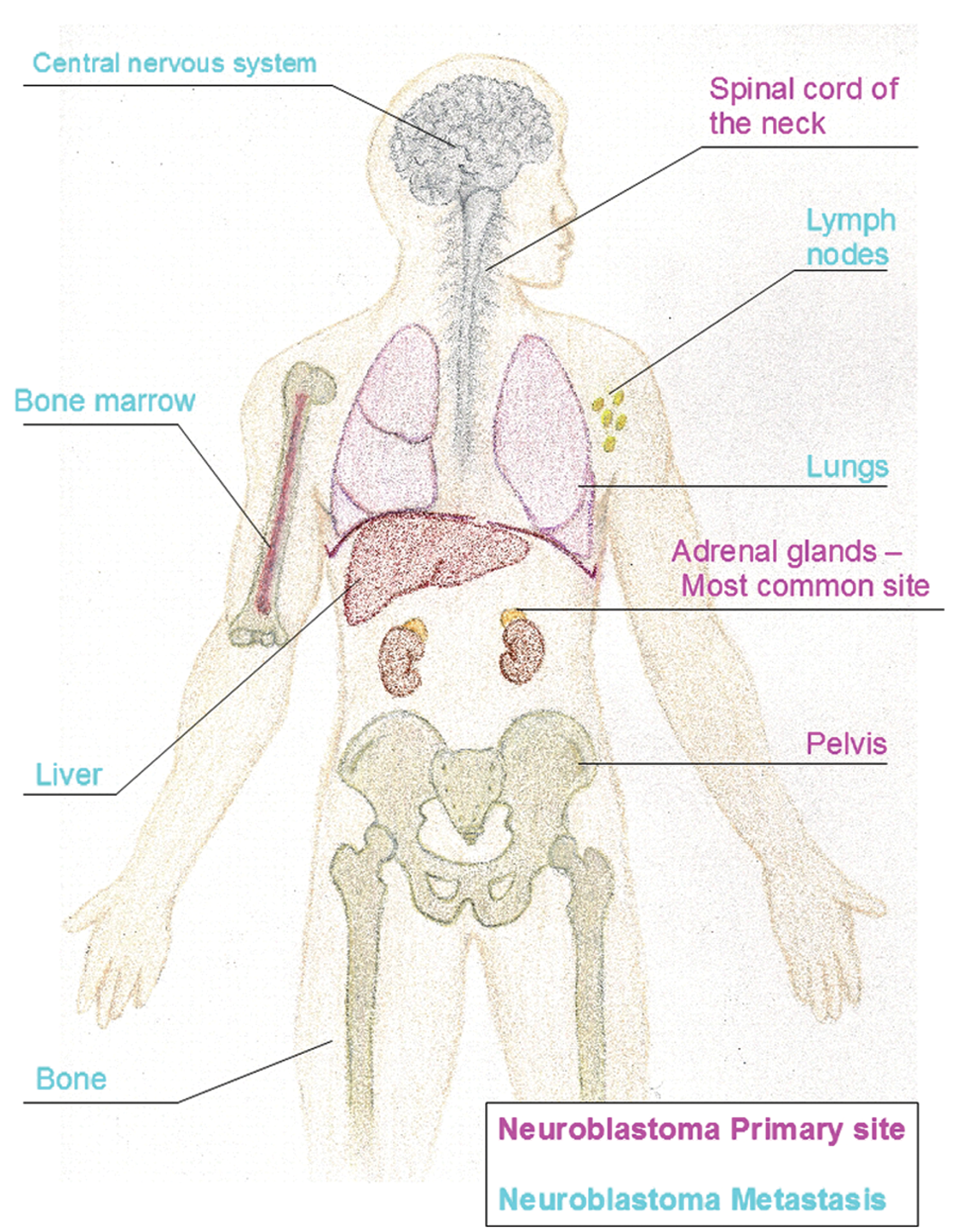
Sites of neuroblastoma primary tumors and metastasis.
A neuroblastoma originates from the primitive cells of the neural crest that form the sympathetic nervous system in embryologic development. 8 The failure of these neural crest stem cells to differentiate or to give rise to other types of cells leads to a neuroblastoma.3,9 The tumors, therefore, can occur anywhere along the sympathetic chain of the nervous system. Malignant cells form in the nerve tissue of the adrenal glands, neck, chest, or spinal cord. Neuroblastomas, like most cancers, begin with a genetic mutation. The exact genetic source, however, remains unclear. 3
Symptoms and Clinical Factors
Neuroblastoma is usually detected at a mean age of 22 months old. 10 The most common finding upon a physical examination is a nontender, firm, irregular abdominal mass that crosses the midline, which presents as abdominal fullness or distension. Renal hypertension from renal artery compression or dyspnea from elevation of the diaphragm can occur with a large neoplasm. 4 Typically, children with localized disease are asymptomatic, whereas children with disseminated neuroblastoma are generally sick and may have a systemic manifestation. Symptoms include unexplained fever, weight loss, anorexia, failure to thrive, general malaise, irritability, and bone pain. 7 Catecholamine-induced hypertension or an autoimmune response may cause encephalopathic symptoms, which are nonspecific.4,5
Prenatal neuroblastomas rarely present with fetal hydrops or any maternal symptoms. Maternal symptoms include preeclampsia caused by placental vascular metastasis or hypertension from increased catecholamines within the maternal circulation.5,11 Any of these prenatal findings suggest a very poor prognosis, often fatal for the fetus/neonate.
Multimodality Imaging
Prenatal neuroblastoma is usually detected by sonography in the third trimester with a mean gestational age of 35 weeks, but it can be seen as early as 19 weeks’ gestation.4,8 The evaluation of a child with an abdominal mass commonly involves multimodality imaging to aid in the diagnosis of possible neoplastic disease and to assist in the clinical decision-making process. 12 Imaging may include sonography, computed tomography (CT) or magnetic resonance imaging (MRI), and general radiography. Any combination of these imaging modalities will be made specific to the individual patient based on age, symptoms, site of the neoplasm, suspected metastasis, and current treatment protocols. 5 CT, MRI, and sonography are typically used for the initial evaluation of abdominal masses. If the tumor is large, these imaging modalities may have difficulty in delineating the neoplasm’s site of origin.
Sonography improves the diagnostic evaluation and is probably the single best imaging modality. It is noninvasive and provides relevant information regarding the location, appearance, and size of the mass. Sonography can detect tumor thrombi and survey surrounding structures. The most beneficial aspect of sonography is the ability to image in utero without any harm to the fetus. Because of the very low biologic effects of sonography, it is the ideal modality to monitor neuroblastomas before and after resection, as well as neuroblastomas treated nonsurgically. It also does not require any contrast or sedation, can be performed at the bedside, and is relatively inexpensive in comparison to other imaging modalities. 12
Although neuroblastoma is a suprarenal mass that arises from the adrenal medulla, it generally does not invade the kidney but inferiorly displaces the ipsilateral kidney (Figure 2).5,10 This “mass effect” usually displaces the aorta and the inferior vena cava (IVC) anteriorly. 12 The sonographic appearances of adrenal neuroblastomas are variable. They can be masses with increased echogenicity or varying levels of echogenicity with areas of necrosis/hemorrhage. 10 Neuroblastomas found postnatally are usually echogenic solid masses. Most fetal neuroblastomas are cystic or complex but can uncommonly be solid (Figure 3). These masses can also contain calcifications that demonstrate posterior shadowing (Figure 4).10,12 Liver metastases will be visualized as hypoechoic lesions or diffused liver enlargement. 10
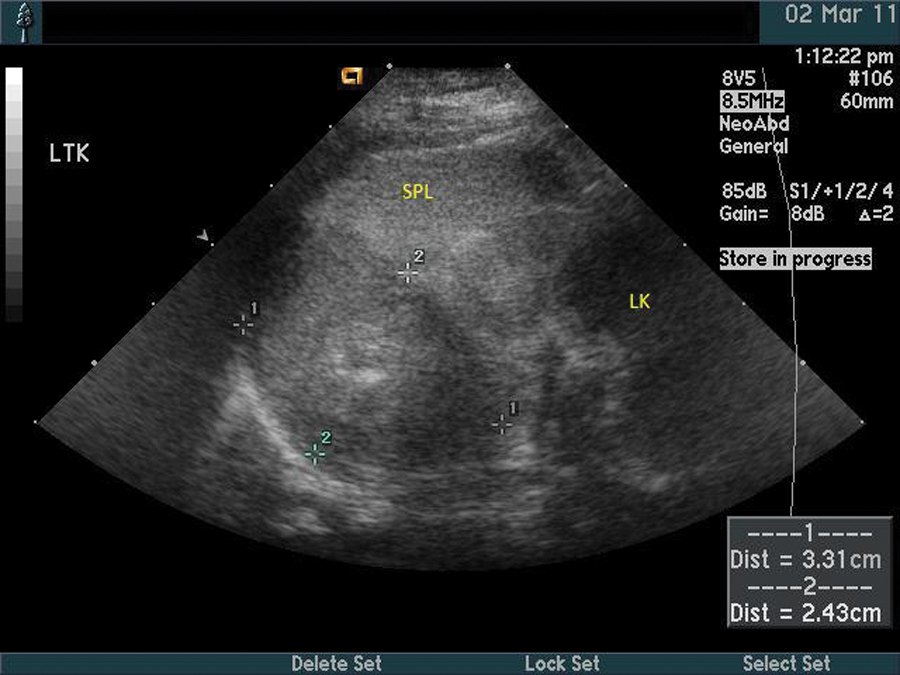
Postnatal scan of a four-month-old neonate illustrating the left upper quadrant in a sagittal view from the coronal plane demonstrating a left adrenal neuroblastoma (marked between the calipers), the spleen (SPL), and the left kidney (LK). The adrenal neuroblastoma displaces the kidney.
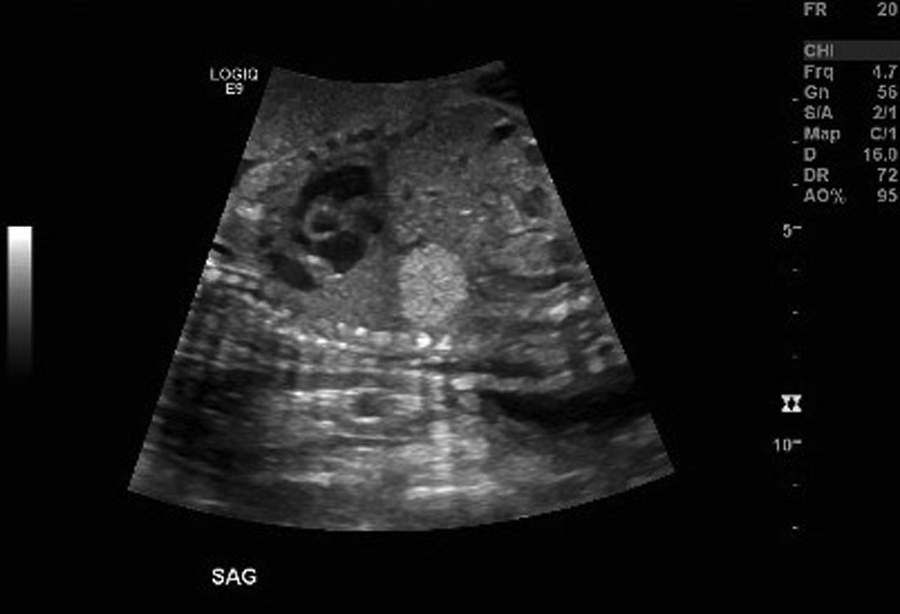
Prenatal scan of a fetus at 27.2 weeks’ gestation in a sagittal view with a solid echogenic mass arising from the left kidney.
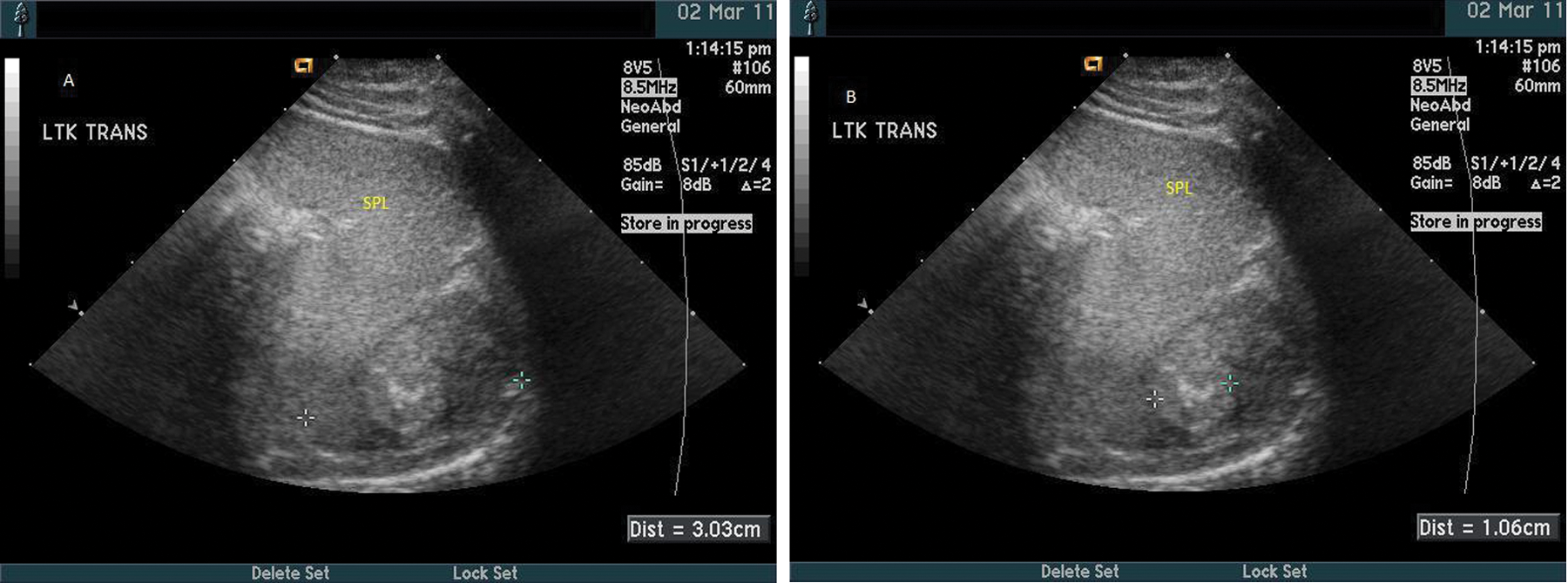
Postnatal scan of a four-month-old neonate illustrating the left upper quadrant in the transverse plane. (A) Measurement of a left adrenal neuroblastoma and the relational anatomy of the spleen (SPL). (B) An image similar to A, with measurement of a calcification within the left adrenal neuroblastoma.
Another advantage of sonography is the ability to use power Doppler and color Doppler imaging. It is extremely effective at evaluating blood flow in real time. Power Doppler imaging is more sensitive to the presence of flow and is not dependent on angle and direction of flow. 13 Another sonographic finding of neuroblastoma and other neoplasms is hyperemia seen on color Doppler.10,13 According to Metha and Lim-Dunham, 3 several case reports have indicated vascular invasion by neuroblastomas. Neuroblastomas most often surround regional blood vessels. Although vascular invasion is rare, it is important to be aware of this unusual manifestation. Color Doppler and power Doppler are valued in the accurate detection and posttreatment follow-up of tumor thrombi. They are used to differentiate between a bland thrombus and a tumor thrombus. The visualization of vascular structures within a thrombus suggests that it is composed of viable tumor cells.3,9 A neuroblastoma thrombus can enter the circulatory system via the adrenal vein or the renal vein, and from there, it can travel to the IVC and enter the right atrium of the heart. Doppler imaging is the most accurate examination for detecting tumor extension to the right atrium because it may be missed on CT. Metha and Lim-Dunham, 3 using color Doppler in a study of 34 pediatric patients with solid intra-abdominal malignancies, found no false-negative diagnoses of tumor thrombi. The diagnoses were confirmed by two other imaging modalities or surgery. The accuracy of prenatal and postnatal diagnosis is improved by the use of Doppler by detecting any hemodynamic activity within a suprarenal mass. 9
Because sonography is unable to penetrate into or beyond very dense structures such as bone or significant calcifications, it is much less helpful in evaluating boney lesions or identifying neoplasms after chemotherapy due to overlying calicfications. 5 Sonography’s narrow field of view can cause difficulties in exact tumor size, location, and relational anatomy. A study done by Acharya et al. 11 points out that sonography is not as sensitive as intravenous (IV) contrast CT and MRI at detecting liver lesions in some cases.
The benefit of CT and MRI is that it enhances the sonographic findings by providing information about tumor size and location, regional lymph nodes, vessel invasion, and distant metastatic disease. 3 Along with all these advantages, the wider field of view of these modalities allows for relational anatomy involvement in assessing possible tumor resection. 5 MRI is a more sensitive test for detecting liver lesions in comparison to IV contrast CT and sonography, but it may not be warranted in some cases. 14 Patient prognosis or treatment will not be compromised in cases of isolated liver metastasis if MRI imaging is not obtained. 11
CT evaluation is more invasive because it uses ionizing radiation and requires general sedation for young children since CT image quality is dependent on a reduction in patient or visceral motion for the length of the examination. 15 Visceral motion is decreased by breath holding during image acquisition, which is impossible to do if the patient is sedated. It may also require oral contrast and/or IV contrast to be administered to the patient. Other disadvantages of CT include its inability to be performed at the bedside and the cost of the examination relative to sonography. 15 A review by Ilias et al. 14 states that the size of adrenal tumors can be underestimated on CT. The typical neuroblastoma will appear as a soft tissue mass that is lobulated and heterogeneous with incongruent or little enhancement (Figure 5). 5 The areas of low attenuation within the neoplasm represent necrosis or hemorrhage. On CT, the liver will demonstrate lesions of low attenuation or diffused heterogeneous enlargement. Bone tissue will have radiolucent, sclerotic, or mixed lesions, indicating the destruction of the bone by metastasis. 10
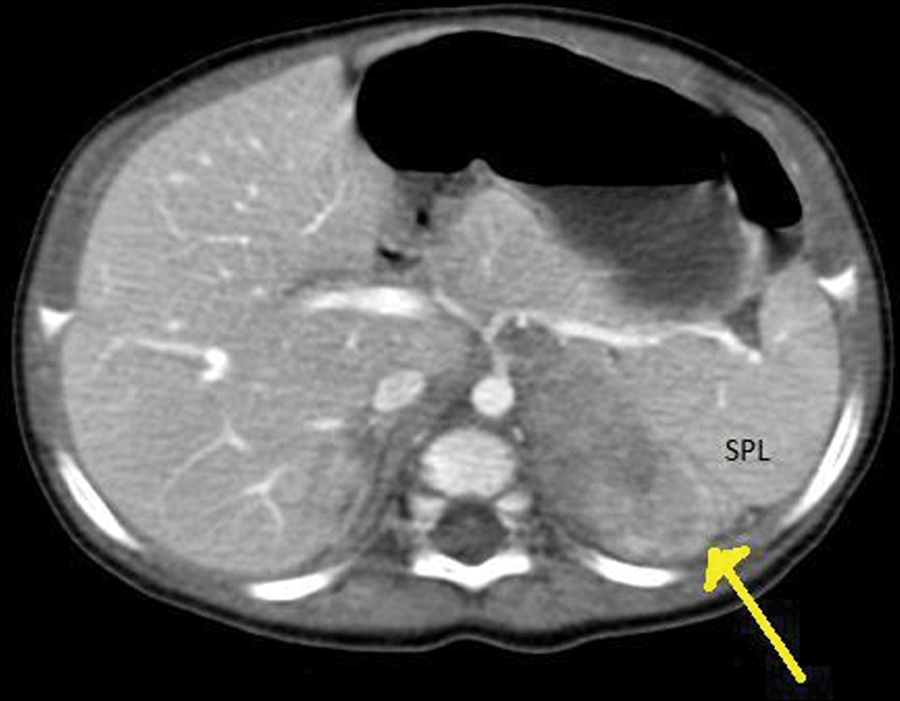
Postnatal computed tomography (CT) scan of a five-month-old neonate demonstrating a left adrenal neuroblastoma (arrow) next to the spleen (SPL).
McHugh 15 considers neuroblastoma staging by MRI evaluation to be the most useful, although Papaioannou and McHugh 5 argue that this claim is debaTable. MRI is more beneficial than CT when evaluating bone marrow invasion and intraspinal extension of the neoplasm because it has an overall better resolution of anatomic detail. 4 Prenatal MRI can be used when maternal body habitus, amniotic fluid volume, or fetal lie decreases the quality of sonographic images. A challenge to fetal MRI imaging is inaccuracy and degradation of the image during acquisition due to variation in fetal motion and fetal lie. This results in blurred images or images in the incorrect imaging plane. To reduce fetal motion, sedatives must be injected into the umbilical vein. More recently in Europe, maternal oral administration of a fetal sedative has been used. 16 MRI must be performed in the third trimester to produce satisfactory images due to smaller fetal size if done earlier. Fetal MRI is still in its infancy; the biologic affects are unknown, and teratogenesis and acoustic damage to the fetus are of concern. MRI imaging of the fetus is a secondary diagnostic examination and should be done only if the results are likely to alter treatment or delivery. 16
MRI has downfalls similar to CT, with the exception of no ionizing radiation exposure or use of oral contrast. The MRI characteristic appearance of neuroblastoma is usually heterogeneous with inconsistent enhancement. Papaioannou and McHugh 5 indicate that the “prolonged T1 and T2 relaxation times with low and high signal intensity on T1W and T2W . . . show little to no enhancement.” Calcifications cannot be seen, but there is good resolution of cystic and hemorrhagic areas in the tumor. Metastasis to the bone will appear as diffuse infiltrate or areas of low T1-weighted and high T2-weighted signal intensities with a nodular pattern.
Papaioannou and McHugh 5 suggest that chest, abdominal, and skeletal radiographs may be required for evaluation of presenting symptoms such as dyspnea, abdominal distention, and bone pain. A large retroperitoneal shadow that causes disposition of adjacent structures and calcifications can be seen on plain films. Metastatic bone lesions will appear radiolucent, sclerotic, or mixed. 10 General radiography is a relatively short examination, which is a huge plus with neonatal or pediatric imaging, but these films may not be warranted because of the potential negative effect of radiation exposure, especially to children and infants.
Additional Tests
Along with imaging studies, additional tests must be performed to rule out a diagnosis of neuroblastoma. A preoperative urine sample for catecholamines can help indicate neuroblastoma. 1 Ninety-five percent of neuroblastoma cases had increased levels of catecholamines in the urine analysis. 10 Acharya et al. 11 state that in prenatally diagnosed neuroblastoma, about 33% had elevated vanillylmandelic acid (VMA) and homovanillic acid (HVA). This result could be due to relatively smaller tumor size, or the catecholamine levels may be falsely within normal limits in comparison to the high creatinine levels normally found in the first few days after birth. A ratio of <1 of early metabolite catecholamines, VMA and HVA, is associated with an aggressive neuroblastoma and corresponds to a shortened life span. Another indication of neuroblastomas is Myc-N (a proto-oncogene) amplification. 5 Roughly 20% of neuroblastomas have amplified Myc-N; this suggests the existence of metastasis and a poor prognosis. 2 To confirm the diagnosis of neuroblastoma, histological evidence of neural origin or differentiation is required.
Differential Diagnosis
A differential diagnosis can be narrowed down by the appearance of the tumor on imaging studies and/or histology, the pattern of invasion of regional viscera, and vascular involvement. A large palpable mass in the left or right upper quadrant of the abdomen in infants or children can be a neuroblastoma or a Wilms tumor. Wilms tumor is the number one differential for neuroblastoma. 3 Wilms tumor is a common malignant childhood neoplasm with a mean age of occurrence of three years. It is a complex mass that originates from the kidney and most commonly invades the vascular system by way of the renal vein and IVC, which differentiates it from neuroblastoma. 10 Although there have been reports of vascular invasion from neuroblastoma, it is a rare occurrence. Adrenal neuroblastomas more commonly encase and displace the regional blood vessels.
If a palpable mass occurs on the right side, a hepatoblastoma should also be considered. This is yet another pediatric tumor that typically invades the blood vessels. The sonographic appearance of this tumor is a solid large solitary mass with mixed echogenicity that contains small cysts and areas of calcification. Another differential for an echogenic solid neuroblastoma is perinephric fat, except it does not displace the kidney, or intra-abdominal pulmonary sequestration (Figure 6).5,8 An adrenal hemorrhage is the most important differential diagnosis for a cystic adrenal neuroblastoma in a neonate, but this is an unusual finding in a fetus.8,11 It will appear anechoic to hypoechoic in the early stages, mimicking a neuroblastoma, but can be followed on consecutive diagnostic sonograms as the appearance changes with each stage until resolution.8,13 Perinephric fat, pulmonary sequestration, and adrenal hemorrhage will all be avascular. Most neoplasms persist or enlarge over time and will demonstrate vascularization. 13 The differential diagnosis will depend on the sonographic appearance of the tumor and regional tumor involvement.
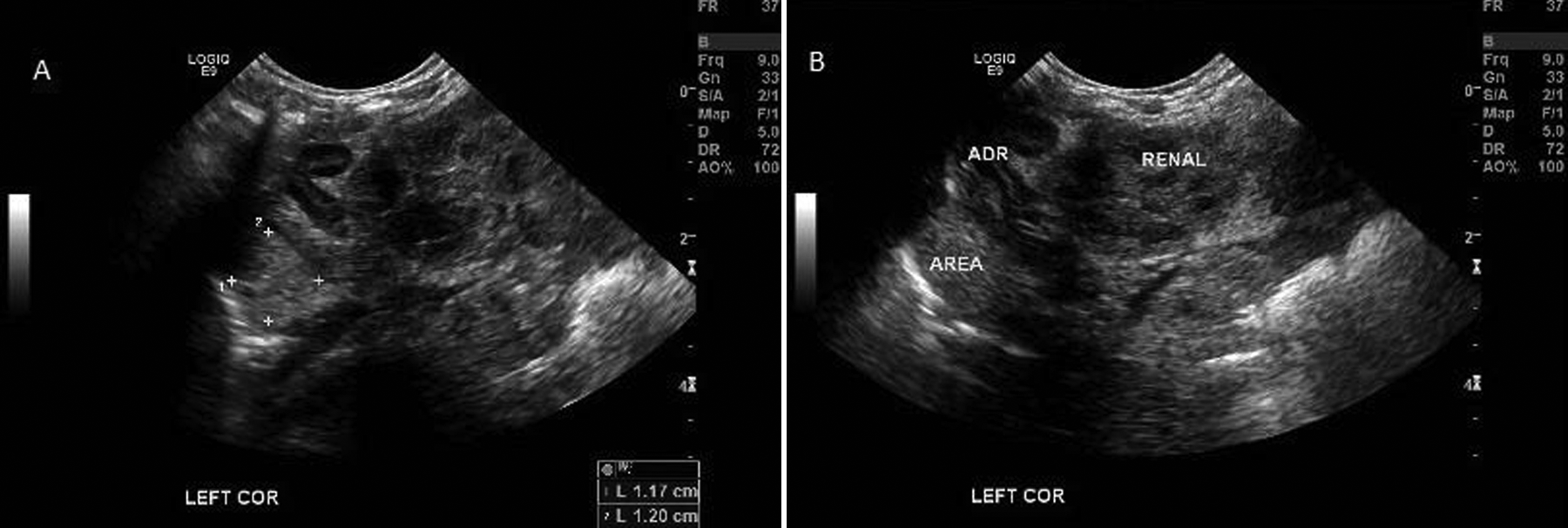
Postnatal scan of a one-day-old neonate illustrating the left upper quadrant in a sagittal view from the coronal plane. (A) Measurement of a mildly echogenic intra-abdominal pulmonary sequestration. (B) An image similar to A. The intra-abdominal pulmonary sequestration (AREA) and the relational anatomy of the left adrenal gland (ADR) and left kidney (RENAL).
Treatment and Prognosis
The prognosis is determined by the patient’s age and stage of disease at diagnosis. Early detection of neuroblastoma allows for a better prognosis because the disease can be treated at a younger age and at a lower stage. Patients with neuroblastoma detected before age one year have an exponentially better prognosis to be disease free than older children, regardless of the stage (Table 1). 2 If undetected during the first year of life, the neoplasm is usually at a more advanced stage and results in a poor prognosis. Radiation or chemotherapy does not have a significant impact at this point in development of the tumor. The cure rate drops dramatically to less than 10% compared with more than 75% in infants who are diagnosed at one year of age or younger. 8
Surgery alone is used to manage low-stage (stages I and II) neuroblastomas. 2 Multiple-agent chemotherapy is the conventional therapy for patients in the more advanced stages of neuroblastoma to shrink the neoplasm. Moderate-dose chemotherapy can be used alone or in conjunction with surgery for intermediate-risk neuroblastoma. High-risk neoplasms require aggressive therapy, such as myeloablative treatment with hematopoietic stem cell transplantation or high-dose chemotherapy to shrink the tumor before surgical resection.5,12 Radiation therapy will be used if the tumor does not respond to chemotherapy.
Children older than one year with high-risk neuroblastoma have very poor survival rates despite intensive multimodal therapy (Table 2). 8 Fifty percent of children with high-risk disease will relapse from drug-resistant residual disease. 2 A study of 3539 children with neuroblastoma revealed that African American children were diagnosed at an older age, 18 months or older, and at a higher risk. Also, progression or relapse of neuroblastoma two or more years from diagnosis occurred more often in this population, suggesting a patient population possibly more resistant to chemotherapy. 17 Adrenal neuroblastomas are reported to have an excellent prognosis if detected prenatally. A prenatally detected neuroblastoma has a better outcome with early postnatal surgical resection, even with distant metastases. 8 Because of the noninvasive nature of diagnostic sonography, it has played a major role in the early detection and treatment of neuroblastoma.

Age <1 year with localized or regional disease and Myc-N amplification are a rare subgroup, and it is not yet well established if the unfavorable implications of Myc-N amplification override the favorable characteristics of age <1 year. 7
Myc-N: a gene with an amplification that is associated with tumors, notably neuroblastoma.
The course of preoperative and surgical treatments depends to a large degree on whether an intravascular tumor thrombus is present. Intravascular tumor thrombus poses a risk of morbidity from intraoperative bleeding. 3 Furthermore, if the right atrium of the heart is involved in tumor thrombus, cardiopulmonary bypass may be necessary for complete tumor resection. Preoperative chemotherapy may be necessary to shrink the tumor and/or the thrombus if they are too large and pose a risk in surgery. In some instances, surgery can be avoided if the tumor starts to spontaneously regress. In a review of the literature by Lee and Cho, 8 97% of patients with prenatally diagnosed neuroblastoma were alive without recurrence.
Conclusion
The quality of patient care associated with neuroblastomas of all stages increases with technological and medical advances. Neuroblastoma is a common malignant neoplasm of infancy and early childhood, and if diagnosed and treated before age one year, the survival rate dramatically increases. Neuroblastoma must be staged accurately to provide the best treatment available. The ability to expedite the diagnostic process by early prenatal detection will allow adequate time for physicians to individually tailor treatments for each patient, in the hopes of limiting long-term side effects while maintaining a high rate of cure. Sonography, CT, MRI, and general radiology all have different strengths and abilities. A risk/benefit ratio must be carefully balanced when imaging studies are performed on children. Early detection and the postnatal management of neuroblastoma are greatly attributed to sonography. It has proved to be the optimal imaging modality of abdominal neoplasms because of its noninvasive nature and ability to provide extensive analysis of the mass and its surrounding structures both pre- and postnatally.
Neuroblastoma is a malignancy that requires a multitude of imaging and other tests to correctly diagnose and stage the tumor. Sonography is the starting point in the continuum of care for neuroblastoma diagnoses. The prognosis of these malignant suprarenal masses in infants has improved dramatically with early prenatal detection in conjunction with early postnatal surgical intervention. Early detection by sonography not only improves treatment management and survival but also improves the quality of patient care by allowing family members to prepare for a child who will need more postnatal care than normal. Not only has sonography improved the diagnostic evaluation of neuroblastoma by noninvasive prenatal detection, but it has also allowed physicians to study the natural progression of the disease to further evaluate postnatal treatment options.
Footnotes
Acknowledgements
The author thanks Terry Read, BA, BS, RDMS, RDCS, RVT, of Seattle University, Department of Diagnostic Ultrasound; Carolyn T. Coffin, MPH, RT, RDMS, RVT, RDCS, FSDMS, of Seattle University, Department of Diagnostic Ultrasound; and Michael S. Cronan, RT, RDMS, chief sonographer of UC Davis Health System Sacramento, Department of Radiology, for their support and guidance.
The author declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author received no financial support for the research and/or authorship of this article.