Abstract
Crohn disease is an incurable inflammatory bowel disease that can involve the large or small bowel but typically is located at the terminal ileum. The disease ranges from mild inflammation to bowel obstruction, fistula, and abscess formation and is typically diagnosed during adolescence or the early 20s. Patients with Crohn disease undergo multiple follow-up imaging examinations for the disease during the course of their lives. Sonographic findings in an adolescent patient diagnosed with Crohn disease are discussed. Sonography is emerging not only as an effective imaging modality for long-term disease follow-up but also, with the addition of hydrosonography and improved transducer technology, as a primary diagnostic tool.
Crohn disease (CD) is an immune-induced inflammation of all layers in the gastrointestinal (GI) tract. Although it can potentially affect any area of the GI tract from the esophagus to the anus, it predominantly affects the small bowel at the terminal ileum and the colon. It typically occurs in younger patients in their teens and 20s, although diagnosis has been seen as young as age 5 and in patients older than 40.1,2 There is evidence that the incidence of CD is rising worldwide, particularly in the pediatric popuation.3,4 Crohn disease is incurable and requires frequent follow-up with diagnostic imaging because it features unpredictable periods of active disease and, when active, has serious complications. Sonography has become an important aid in the diagnosis of CD as well as monitoring the disease process.
Case Report
A teenage boy experienced prolonged vomiting and nausea along with extreme weight loss. He presented to his family physician with pelvic pain in the right lower quadrant. A flat and upright abdominal x-ray and a pelvic sonogram for appendicitis were ordered. The radiograph demonstrated normal bowel gas patterns and did not show any obstruction or free air. The sonogram demonstrated peristalsis of the small bowel and normal-appearing bowel loops (Figure 1). The appendix was not identified. Three weeks later, the patient returned for another pelvic sonogram for persisting abdominal pain. This sonogram demonstrated enlarged mesenteric lymph nodes and a possible mass superior to the bladder that was thought to be an intussusception or an abscess (Figures 2 and 3). A computed tomography (CT) examination of the abdomen and pelvis was ordered. The CT confirmed the sonographic finding of mesenteric lymph nodes and demonstrated a long segment of abnormal small bowel at the level of the terminal ileum that corresponded to the location of the mass seen on the sonogram. Proximal to the terminal ileum was dilation of the small bowel and fecalization, consistent with longstanding, partial bowel obstruction (Figure 4). The appendix was not identified on CT, and free fluid in the abdomen was noted.
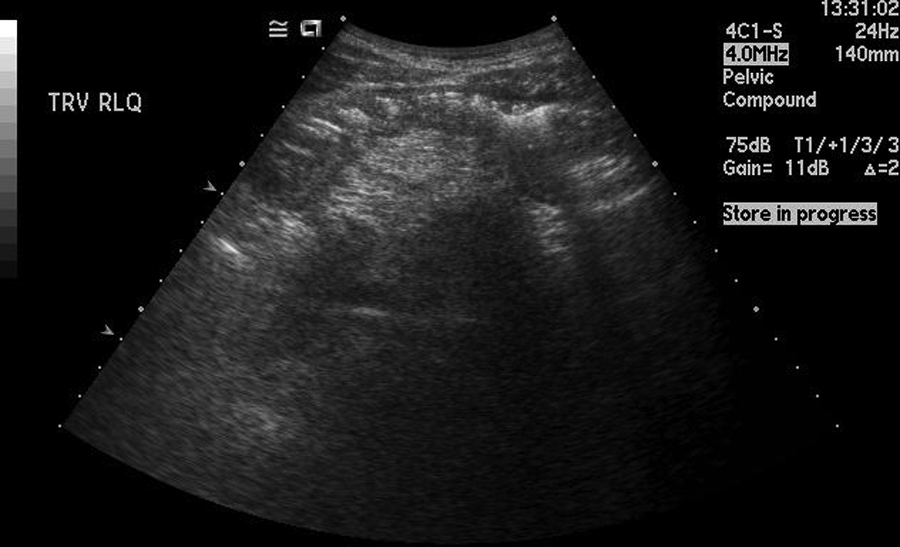
This sonographic image is from the initial sonogram that demonstrated peristalsis and normal-appearing bowel loops.
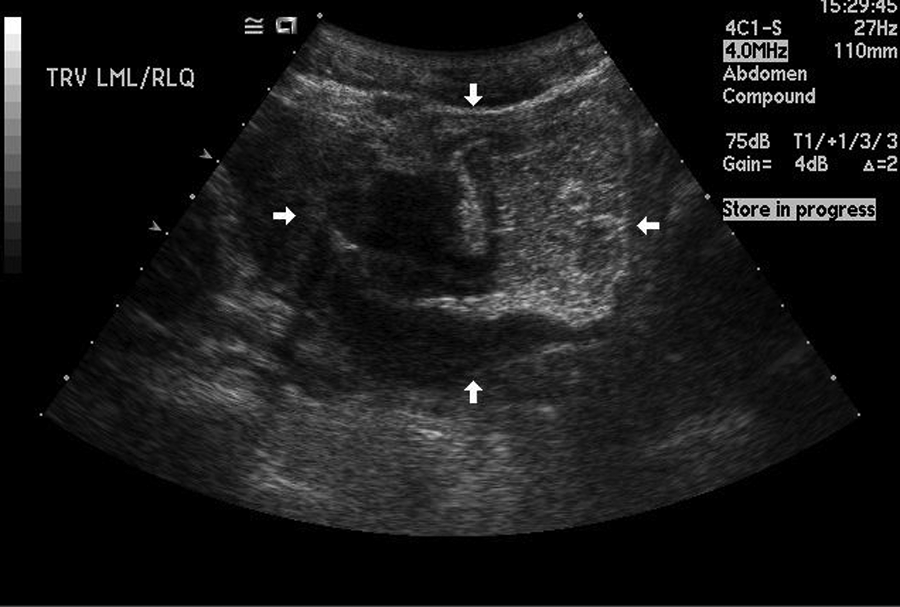
This transverse image is from the second sonogram, occurring three weeks after the initial sonogram. The arrowheads are pointing to the mass seen superior to the bladder.
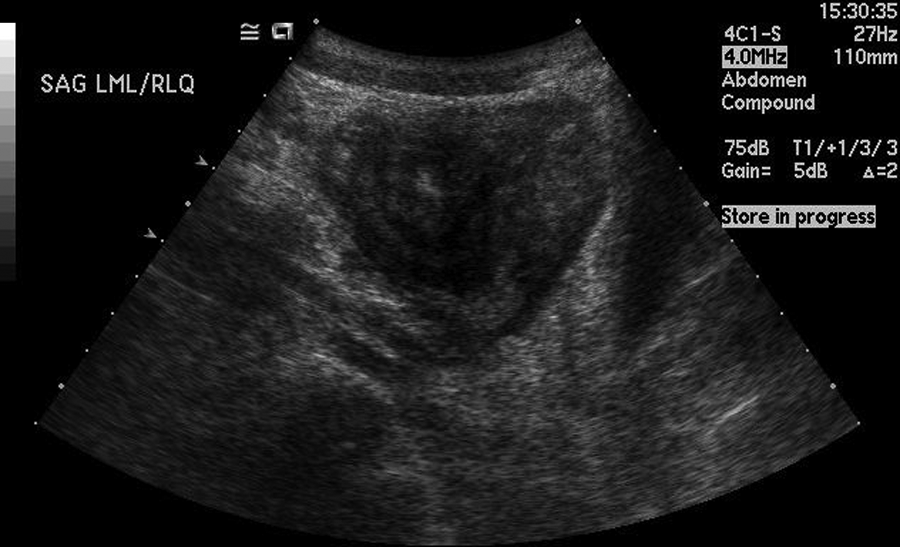
This longitudinal image is from the second sonogram, occurring three weeks after the initial examination. This is the same mass visualized superior to the bladder in Figure 2.
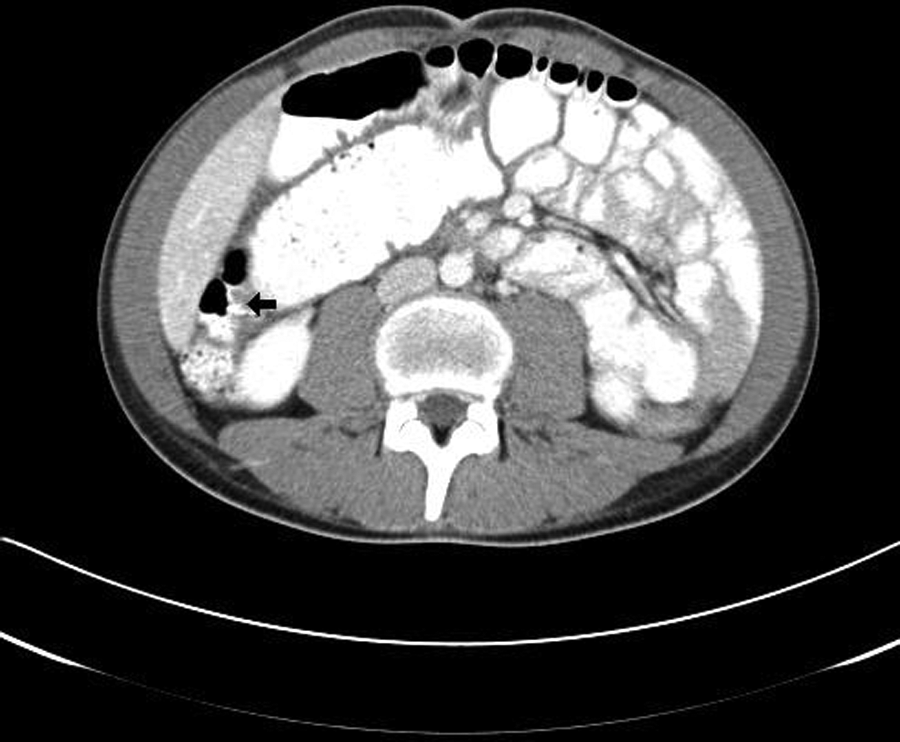
This image is from the computed tomography examination following second sonogram. The small arrow indicates the area of partial obstruction at the level of the terminal ileum.
The patient was immediately sent to a large metropolitan children’s hospital where he was admitted through the emergency department and remained for approximately one week. During this inpatient stay, the patient was diagnosed with Crohn disease, treated medically with steroid therapy, and discharged. He was instructed to have a follow-up sonogram of the small bowel at the hospital in his hometown three weeks later. The transabdominal pelvic sonogram performed at that time examined the level of distention and thickness of the bowel. The sonologist’s report concluded that the bowel remained thickened and still demonstrated a mass effect just superior to the bladder. The bladder had no abnormalities, and the bowel appeared less pronounced compared with prior sonograms (Figures 5 and 6). The sonographic findings were believed to be consistent with the recent CD diagnosis. The patient was instructed to follow up with his primary care physician and is currently undergoing long-term follow-up care with his physician.
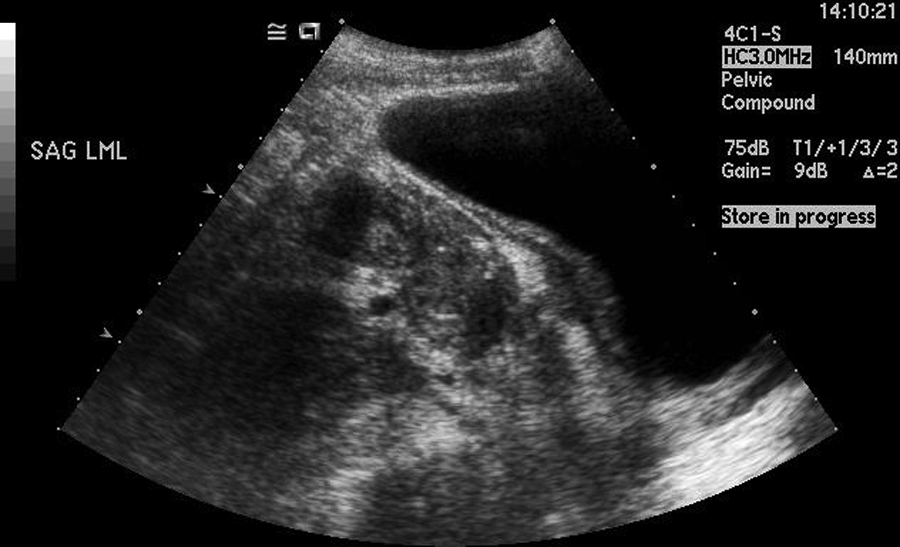
This sonographic image is from the third (follow-up) sonogram three weeks after discharge from the hospital. The bowel is visibly less thickened as compared with the second sonogram.
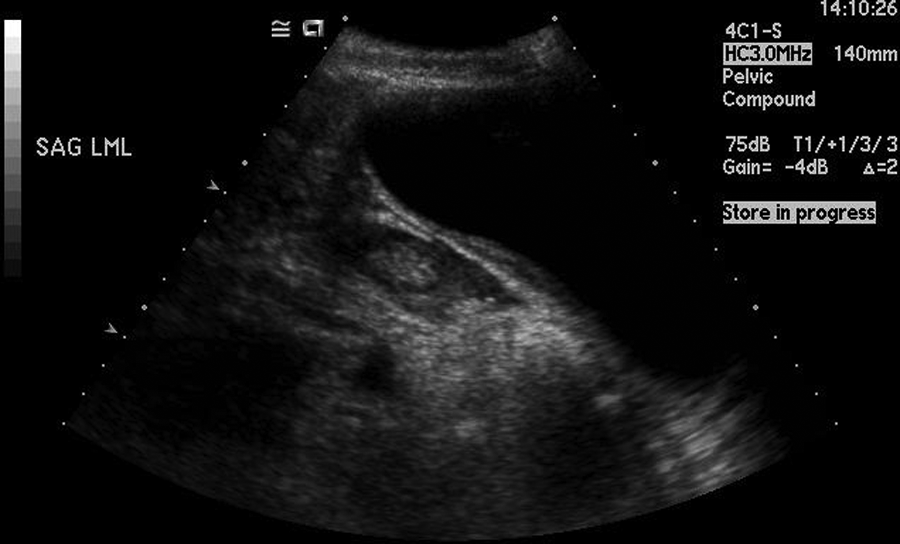
This image is from the third (follow-up) sonogram occurring three weeks following discharge from the hospital. This longitudinal image shows a reduction in the “mass” seen on the second sonogram.
Discussion
Anatomy
The GI tract consists of the esophagus, stomach, small intestine, colon, rectum, and the anus. The small intestine measures about 6 to 7 meters long and comprises three sections beginning at the pylorus: the duodenum, jejunum, and ileum. 5 The ileum terminates at the cecum, where it joins the ascending colon. Where the cecum and ileum join, there is muscular valve, called the ileocecal valve (also called the ileocecal fold), that is made up of folds of the muscular ileum. The ileocecal valve functions as a sphincter, regulating the passage of waste materials into the large intestine and preventing reflux. The ascending colon begins at the cecum and ascends through the right upper quadrant. At the level of the liver, it turns leftward, becoming the transverse colon. The large intestine is approximately 1.5 meters long and is responsible for absorption of fluids and forming feces but does not aid in digestion. 5 The colon has multiple sacs called haustra and has a larger diameter as compared with the small intestine.
The bowel has four anatomic wall layers. The outermost layer, the serosa, keeps the bowel anchored in the peritoneum. The next layer, moving inward, is the muscularis propria, which contains both circular and longitudinal muscles that are responsible for moving waste products through the bowel. Inward to the muscularis layer is the submucosal layer, followed by the mucosal layer. The mucosal layer of the small intestine has villi that project into the lumen and secretes a mucoid substance that aids digestion, the absorption of nutrients, and the movement of waste products.
Sonography of the Bowel
By sonography, the normal bowel has five distinct, identifiable layers, four of which correspond to the anatomical layers. When imaged in transverse, from outer to inner, the sonographic appearance is as follows: the serosa is hyperechoic, the muscularis propria is hypoechoic, the submucosa is hyperechoic, the muscosa is hypoechoic, and the fifth sonographic layer is not a functional anatomical layer but represents the bowel lumen. It appears hyperechoic and is commonly filled with air. Peristalsis should be present with normal small bowel activity, and the haustra can be visualized as part of the normal large intestine. Normal stratifications exist if all layers are intact and visualized. Normal wall thickness is generally 3 mm or less and less than 2.5 mm at the terminal ileum.6,7
When scanning the bowel transabdominally, the patient should fast for more than six hours because this reduces the amount of motility and air in the small intestines. The sonographic examination should begin with the patient in the supine position. The sonographer should survey the entire abdomen using a 2.5- to 5-MHz frequency curve-linear transducer. To image the layers of the bowel wall and to demonstrate wall stratification, a higher frequency (7.0–15 MHz) transducer is recommended. When air and bowel gas obscure the image, the sonographer can administer oral contrast agents specifically designed for sonographic imaging that will displace the contents in the bowel. 6 The electrolyte solutions used for bowel cleansing during colonoscopy are the most preferred oral contrast agent to image the small bowel, but drinking water may also accomplish this task. 8 An alternate approach to visualize the small bowel is to administer either water or an oral contrast agent into the small bowel through a nasogastric tube. 9 The addition of a clear liquid into the gastrointestinal system distends the intestines and allows for visualization of the bowel wall.
Another effective technique to view the bowel is called hydrosonography.10–13 In this procedure, water is injected through the rectum in conjunction with intravenous administration of a smooth muscle relaxant, which causes the intestines to distend. This technique is effective when imaging the length of the colon up to the ileocecal valve because the water removes the lumen obscuring gas as well as providing effective through transmission. Echogenicity, wall thickness, intraluminal changes, and stratification are examined with hydrosonography. 14 Hydrosonography or oral contrast agents should not be used if there is a known bowel obstruction, and side effects may include nausea, vomiting, and abdominal distention. Furthermore, if examination of the rectum is of importance, hydrosonography is not effective, and other imaging modalities such as CT or barium x-ray should be considered.11–13
The sonographic examination begins in the right lower quadrant by identifying the terminal ileum as it passes over the right psoas muscle. If hydrosonography is used, the scan should begin while water is injected.6,8 If oral contrast agents are used, then the sonographer should wait approximately 10 minutes after ingestion to begin the scan. 8 The small bowel can be differentiated from the colon by the presence of peristalsis and mucous villi that project into the lumen. The terminal ileum can be traced to the cecum and ascending colon and toward the periumbilical region where the jejunum is typically located. 6 The duodenum can be visualized by placing the transducer in the subxiphoid region in a longitudinal plane and locating the pylorus. By moving the transducer to the right side of the body, the duodenal bulb can be identified, traced in a “C” shape downward, and then moved toward the head of the pancreas. 6 In general, patients with CD tend to be thin, and visualization of the bowel is not hindered by the body wall. However, large and small bowel can appear similar, and the sonographer must have a good working knowledge of bowel anatomy to successfully locate and discern the large and small intestines.
Superior mesenteric Doppler examinations may enhance the sonographic evaluation of the small bowel. 14 By following a standard fasting superior mesenteric artery (SMA) study, sonography can obtain protocol peak systolic velocity measurements, peak end-diastolic velocity measurements, flow volume, resistive indices, and diameter measurement.6,15 If a meal is given to test postprandial values, the same values as the fasting scan should be assessed about 30 minutes after ingestion of a meal. Normal resistive index (RI) values for the fasting SMA are .80 or higher, and the Doppler signal should represent that of a high-resistance vascular bed. 15 Approximately 20 to 30 minutes following a meal, the normal SMA should experience a doubling of the fasting end-diastolic velocity and will transform to a low-resistance waveform.
Pathology
Inflammatory bowel disease refers to two chronic diseases that cause inflammation of the intestines: ulcerative colitis and Crohn disease. Crohn disease affects mainly the bowel lumen, includes inflammation that extends deep into the layers of the intestinal wall, and generally involves all layers of the bowel wall. The inflammation is typically localized, is granulomatous, and may affect several areas of the bowel in a discontinuous fashion, sometimes referred to as skip lesions. Fistulizing CD is the other most commonly appearing form and may be present in up to 40% of cases.16–18 The area of the small bowel that is most commonly affected by CD is the terminal ileum, followed by the ascending colon. Crohn disease may be isolated to either the small intestine or the colon, or it may appear in either location at different times or simultaneously. When CD appears in the childhood, it is more likely to occur in the ileum and the colon rather than isolated to one system. 2 Ulcerative colitis is specific to the colon and rectum and does not affect the small bowel. It affects only the lining of the bowel and is a continuous lesion of the entire length of the colon. 16 When CD is isolated to the colon, differentiation between the two diseases is difficult. Perianal involvement is exclusively related to CD, and indeterminate colitis is diagnosed when a case is unable to be diagnosed as either ulcerative colitis or CD.16,17
The etiology of CD is unknown, but it is believed to be an abnormal immune response that is triggered by an environmental stressor. 19 There is also evidence of a genetic component to the disease, with a first-degree relative being the strongest indicator for the risk of developing the disease.20,21 Use of the tetracycline class of antibiotics, particularly doxycycline, which is commonly prescribed for acne in the teenage years, appears to increase the risk of developing CD, with the most risk occurring in patients who have more than seven courses of antibiotics.22,23 Smoking increases the risk of developing CD, and other proposed risk factors include urban living, high socioeconomic status, increased use of nonsteroidal anti-inflammatory drugs, and high-fat diet.24–28
Crohn disease typically develops in adolescence and young adulthood, occurring mainly between the ages of 15 and 35, but it can develop at any age.1,2,19 It is more common in women than in men, and recent epidemiologic studies suggest that the incidence of CD in children has doubled over a 10-year period in Europe, whereas the incidence of ulcerative colitis has remained stable.3,29 The United Kingdom reports an incidence rate of 5.2/100,000 children younger than age 16.30,31 The overall incidence in the United States is 5 per 100,000 persons and is 50/100,000 in Westernized countries worldwide.16,26,29
Diagnosis
The clinical symptoms of CD are abdominal pain, nocturnal or prolonged diarrhea, nausea, vomiting, fever, severe weight loss, anemia, hypoalbuminemia, and malnutrition. Blood may be present in the stool when CD involves the colon. 16 Abdominal pain can be described as intermittent cramping, and the abdomen may be tender to the touch. As the condition progresses, abdominal pain may persist to a continuous aching, and the patient may experience malnutrition due to failure to absorb nutrients through the affected bowel. In children, the initial symptoms may be vague and present as cryptic as growth failure. 30 Associated external conditions may include arthritis, inflammation of the eye, ankylosing spondylitis, rheumatoid arthritis, and sclerosing cholangitis.16,19
In addition to being confused with other inflammatory bowel diseases, CD may be misdiagnosed as irritable bowel syndrome (IBS). This syndrome presents with similar clinical symptoms but does not include inflammation of the GI tract. Celiac disease, Bechet syndrome (inflammatory ulcerative disease of the oral and genital tract), appendicitis, food intolerance, and diverticulitis are also possible differential diagnoses that do not include focal inflammation of the bowel but have similar presenting symptoms. Medical history, clinical symptoms, physical examinations, imaging modalities, and laboratory tests are the primary tools for diagnosing CD and distinguishing it from other similar presenting pathologies.
Sonography was once considered an unreliable modality to diagnose CD, but with the advent of high-frequency imaging, sonography is emerging as a primary diagnostic tool as well as part of an effective long-term disease management strategy. When CD is present, the bowel wall thickness (BWT) via sonography measures greater than 3 mm, and thickening will be localized. 6 The thickened bowel in transverse sections will look like the target or “bull’s-eye” sign similar to what is seen sonographically in acute appendicitis. Another 2-D, gray-scale sonographic finding is the pseudo-kidney sign, in which the thickened bowel wall mimics the appearance of kidney cortex and the echogenic lumen appears like renal sinus. In addition to the target or pseudo-kidney sign, the sonographer may notice reduced peristalsis, stiffened intestine, and flattened appearance of the haustra if the disease is in the colon. 6
The loss of stratification of the bowel wall layers is an indicator of CD and can be used as a distinguishing feature to diagnose CD over ulcerative colitis.10,11 The loss of one or more layers, which is attributed to both edema and fibrosis, may be imaged by both standard bowel wall sonography and by hydrosonography.32,33 Patients with more active disease are more likely to show loss of stratification as well as patients with increased erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) in their laboratory blood tests. 33 Additional sonographic findings associated with CD include abdominal fluid, the presence of mesenteric lymph nodes, bowel stenosis, mesenteric inflammation, fistulas, and abscesses.34,35
A stenotic intestine is a complication of more severe disease and sonographically has a thickened wall and narrowed lumen and may lack peristalsis.6,35 Proximal to the narrowing, the examiner may note dilation of the bowel that exceeds 3 cm. 35 Fistulas are seen as echo-poor, duct-like structures with fluid or air content communicating between two intestinal loops or skin. 35 The most common types of fistulas include those that connect bowel to bowel, followed by bowel to skin, and bowel to bladder. 16 Abscesses typically appear as anechoic peri-intestinal lesions larger than 2 cm. 6 In the presence of CD, there may be decreased resistance and increased flow volume in the fasting SMA. 15 The RI may drop to .75 or lower, and the diameter of the SMA may increase to accommodate increased volume of flow. 15
The sensitivity and specificity for detecting CD by sonography are varied and depend on the differences in patient habitus, examiners’ skill, and equipment quality. Sensitivity and specificity may also vary based on the cutoff values for the BWT. Bremner and associates 7 report in a prospective, blinded cohort study of 44 pediatric patients, a sensitivity of 48%, a specificity of 93%, and a positive predictive value (PPV) of 83% of sonography compared with endoscopy when a value greater than 2.9 mm for bowel wall thickness is used, but they report a sensitivity of 75%, specificity of 92%, and a PPV of 88% when greater than 2.5 mm is used. In a meta-analysis of five cohort and two case control studies, Fraquelli and associates 34 demonstrated that the overall range of sensitivity of sonography for the detection of CD lies between 75% and 93%. When 3.0 mm is used as the cutoff for BWT, the reported sensitivity is 88% and specificity is 93%. 34 When 4.0 mm is used as the cutoff, the sensitivity and specificity are 75% and 97%, respectively. 34 For ruling out disease, any bowel wall measurement less than 3 mm is unlikely to be diseased.6,34 Using a cutoff BWT measurement of 4.0 mm reduces the number of false positives and increases specificity, but to screen for inflammatory processes, using a BWT cutoff value of 3.0 mm or greater is appropriate. 34
Most of the validity statistics for sonography are based on diagnostic follow-up examinations on patients with known disease. Not much is known about sonography as a primary diagnostic tool, but Parente and associates, 36 in evaluating 336 patients, demonstrated that sonography is an effective primary diagnostic tool when compared with endoscopy and pathological specimens. Parente et al. found the sensitivity and specificity of sonography as a primary diagnostic tool for all bowel disorders to be 85% and 95%, respectively, and when CD was isolated to the terminal ileum, the sensitivity and specificity of sonography were 96% and 75%, respectively. 36 When barium x-ray was used as the gold standard, sonography had a sensitivity and specificity of 92% and 87%, respectively. 36 As compared with barium x-rays, sonography was more likely to miss disease that was located in the rectum, jejunum, and duodenum. 36
Hydrosonography is successful at imaging lesions of the colon such as colon cancer, polyps, and diverticulitis. 13 Reported sensitivities range from 70% to 100% for detecting colon cancers and from 6% to 100% for detecting colon polyps. 8 Hydrosonography also shows promise in the diagnosis of CD, especially when it is located in the colon.10–13 Bru and associates, 12 in examining 68 patients with active inflammatory bowel disease, reported that the sensitivity of hydrosonography for detecting CD was 83.3% compared with 66.1% for scintigraphy. Limberg and Osswald, 11 in a prospective study of 440 patients who had clinical symptoms for inflammatory bowel disease, found that hydrosonography had a sensitivity and specificity of 86% and 100% in diagnosing active CD. Both sonography and hydrosonography demonstrate high detection rates for CD as compared with other imaging modalities, and thus sonography can be used as a reliable tool in the diagnosis and management of CD.
Disease Course and Treatment
In the early stages, CD causes the inner surface of the bowel to have erosions called aphthous ulcers. Over time, the erosions become large and deep, causing true ulcers that will create scarring and stiffness of the bowel. As the inflammation progresses, the bowel may narrow to the point of obstruction, or the ulcers may perforate, forming an abdominal abscess or possibly fistulas. Patients with CD in the colon have an increased risk of colorectal cancer, and in rare cases, CD may progress to small bowel cancer.16,17,37
Crohn disease is incurable and typically requires lifelong treatment and continuous follow-up care, and patients may experience periods of active and nonactive disease. The course of CD varies in each patient and may be classified as mild, moderate, severe, or even in remission. One of the more common ways to evaluate CD is through the Crohn’s Disease Activity Index (CDAI), which is a questionnaire that gathers primarily subjective information from the patient about quality and consistency of stool, general well-being, weight, symptoms of other inflammatory processes, and other various clinical presentations. In general, higher scores on the CDAI indicate more active or severe cases of the disease. 38
Mild CD typically corresponds to a CDAI score of 150 to 220, and patients in this stage typically experience abdominal pain and dehydration but have not lost more than 10% of their weight. 16 Moderate disease corresponds to a CDAI score of 220 to 450. Patients are considered to have moderate disease when they have failed to respond to treatment for mild disease, present with abdominal pain, have significant weight loss, and may experience nausea and vomiting but do not have a bowel obstruction. 16 Severe disease correlates to a CDAI greater than 450. Severe disease does not respond to treatment and may present with evidence of bowel obstruction, high fevers, and intractable vomiting. 16
The location and amount of the intestine affected are factors in the selection of treatment. The most common treatment approach of CD is to prescribe anti-inflammatory medications and/or corticosteroids such as prednisone.17,19 Mild disease also may be treated with diet and lifestyle modifications. Most patients can expect to be treated with steroids at some point during the course of the disease. However, approximately 50% of patients will become steroid dependent or steroid resistant and will have to discontinue that line of therapy.16,39 Biologic therapy is a treatment option recently approved by the US Food and Drug Administration. This therapy delivers agents via oral medications, injections, or through intravenous administration that targets and suppresses the immune system, thus reducing the inflammatory response. Patients with moderate or severe disease who have failed to respond to steroid treatments may benefit from biologic therapy.16,17 Patients who are at risk for steroid dependence or for whom steroids are contraindicated are also good candidates for biologic therapy.
Approximately 75% of patients will require, at some point during the course of the disease, surgical resection of the bowel. 17 Surgery is reserved for patients who do not respond to traditional steroid or biologic treatment, when an obstruction or fistula is present, or when neoplastic lesions are either present or suspected. 16 Surgical resection may include the placement of a colostomy or ileostomy. Although surgery does not cure CD, patients may be able to live for extended periods without relapse. Recurrence of the disease is common, as only about 13% of patients will be successfully treated and experience no relapse. 16 For patients who experience remission for at least one year, there is an 80% chance that the disease will remain in remission for an additional year. 16 However, if the patient has had active disease in the past year, he or she has a 70% chance of continuing with active disease in the next year and only a 50% chance of achieving remission in the following three years. 16 Overall, about 20% of patients relapse each year, and 67% have a combination of relapse and remission in the first eight years following diagnosis. 16 Less than 5% of patients will have a continuous course of the disease with no remission. 16 Patients living with the disease for more than eight years are at increased risk for bowel cancers, particularly colon cancer. 17
Conclusion
Patients with CD participate in regular and frequent follow-up imaging examinations to monitor disease activity, assess treatment effectiveness, and assess any other related complications. Barium x-ray and CT are common imaging modalities for following CD; however, the radiation dose is cumulative and can be worrisome, especially when repeated examinations begin in childhood. Because bowel wall thickness measurements and visual assessment of bowel wall stratification by sonography are accurate, reproducible, noninvasive, and do not expose the patient to ionizing radiation, it is a logical choice for long-term disease follow-up once the diagnosis of CD has been established. With the addition of hydrosonography and advances in high-frequency transducer technology, sonography shows promise in becoming a primary tool for screening and diagnosing CD.
Footnotes
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
