Abstract
Natural breast augmentation, or fat transfer, involves taking fat cells from one area of the body and injecting them into another area, such as the breast. This can be done for cosmetic reasons after a lumpectomy or other surgical procedures that affect the shape of the breast. Breast augmentation is also being used electively by some women to enhance the appearance of their breasts. Defects such as asymmetry, sagging, or indentations have been improved with fat transfers. The changes of the breast tissue from the reaction to the new fat can also change the outcome of mammograms and sonograms. The interpreting radiologist might recommend a biopsy to exclude the possibility of missing a malignant lesion. These two case reports give sonographers an idea of what additional questions they may need to ask patients to clarify their history.
Under general anesthesia, fat transfer is accomplished with a type of liposuction where fat is harvested from the patient’s own natural fat deposits, such as the thigh, buttock, or abdomen. The harvested fat is centrifuged to purify the fat. Using special syringes, the fat is injected meticulously into the fatty tissue of the breast through a cannula. The plastic surgeon may use a number of cannulas at different vantage points to evenly distribute the fat for a more natural look. It must be injected very slowly to decrease the amount of damage to the donor fat.1,2
Normal Breast Tissue
Breast sonography is a very useful tool that supplies information about the mass or lesion found on a mammogram. The breast tissue is imaged differently in each modality. The layers of tissue can be distinguished by comparing the shades of gray.
Mammographically, breast tissue is described as either radiolucent or radiopaque. Fatty tissue in the breast is a medium gray and more radiolucent. The parenchyma is a brighter white or radiopaque. Whether a mass is cystic or solid, it will appear on the mammogram as radiopaque and usually blends in with the parenchyma (Figure 1). Sonographically, breast tissue is described as hypoechoic or hyperechoic when compared to the gray scale. Fatty subcutaneous tissue should be medium gray. Tissue toward the darker end of the scale from the medium gray tissue is hypoechoic. Hyperechoic tissue is brighter when compared to the fatty tissue (Figure 2). 3
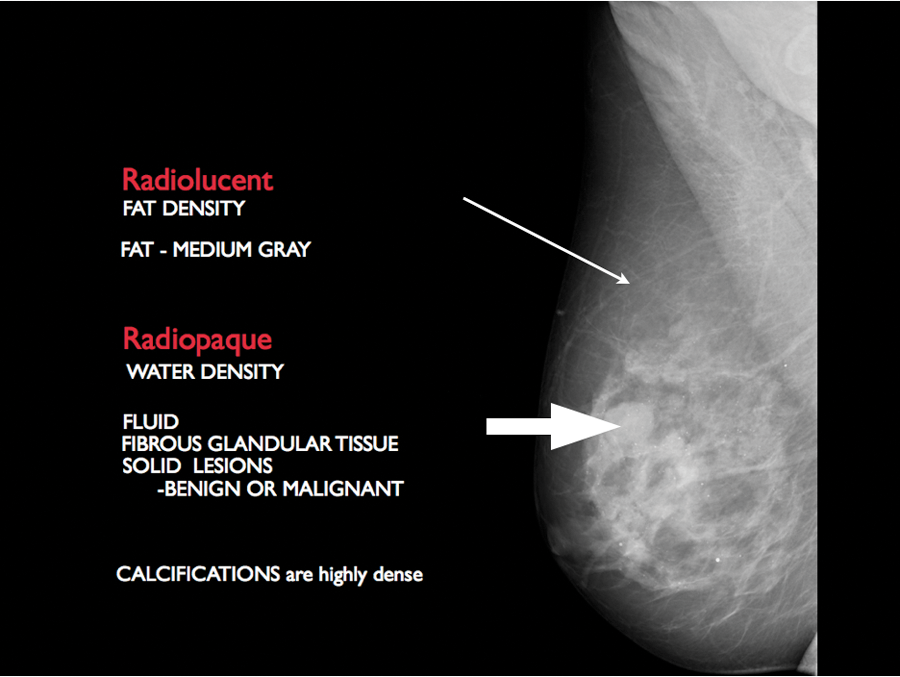
Radiolucent fatty tissue (thin arrow). Radiopaque fibrous glandular tissue (wide arrow).
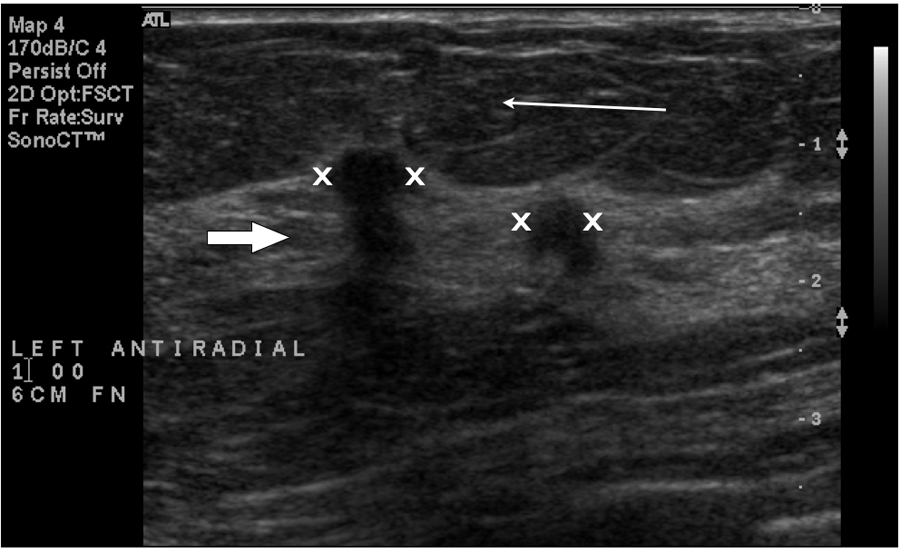
Fatty breast tissue (thin arrow). Hyperechoic fibroglandular tissue (wide arrow). Hypoechoic masses are indicated between the X marks.
Case Report 1
A woman in her late 40s had a negative screening mammogram one month prior to a breast fat transfer. The bilateral mammogram was imaged digitally using a Hologic LoRad Selenia (Siemens Medical Solutions USA, Malvern, Pennsylvania) unit. Routine cranial caudal (CC; Figure 3) and medial lateral oblique (MLO; Figure 4) views were obtained. Only the left breast is illustrated as a comparison for this case report.
Cranial caudal view of the screening mammogram.
Medial lateral oblique view of normal breast tissue on the screening mammogram.
The mammogram indicated extremely dense breasts with no suspicious findings present. She was to return in one year for a routine screening mammogram.
Nine months later, this patient returned with a new palpable lump felt in the upper central left breast. A unilateral left breast mammogram was ordered along with a sonogram.
Using a digital Mammomat Navation DR (Siemens Medical Solutions USA), CC (Figure 5) and MLO (Figure 6) views were performed, including compression views. A tiny marker (BB) was placed over the palpable nodule. The breast parenchyma was heterogeneously dense. A new finding was demonstrated in the upper central left area corresponding to the BB, measuring 4.6 × 4.1 cm in diameter. The mass was oval with completely fatty-appearing tissue, compatible with a benign lesion, likely a lipoma. This mass was not demonstrated in the screening mammogram done 9 months earlier.
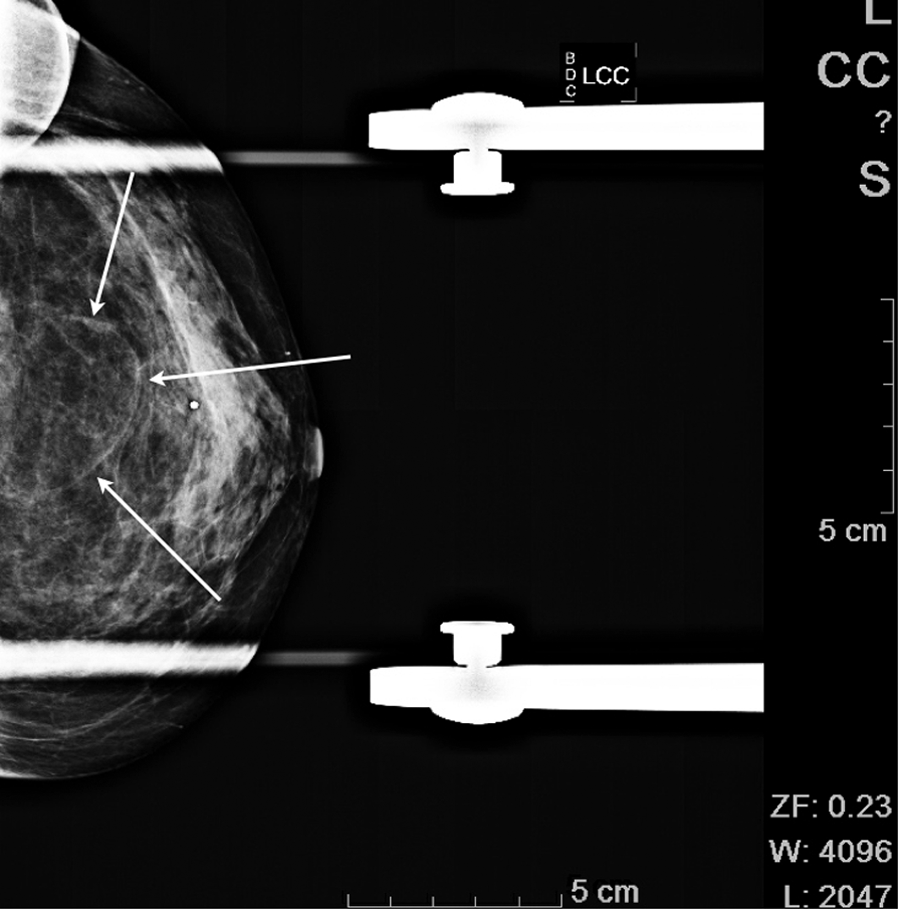
Cranial caudal view with the mass indicated by the arrows.
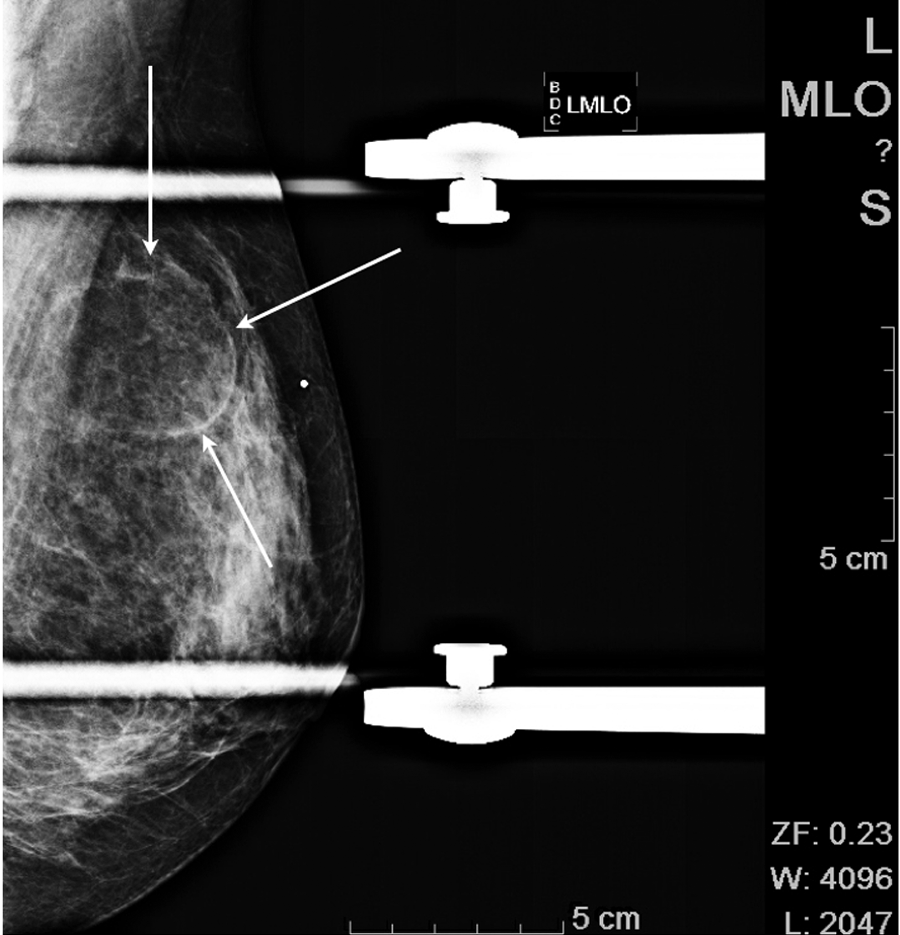
Medial lateral oblique view with arrows indicating the mass. The BB corresponds to the palpable lump.
Once the mass was visualized on the mammogram, the patient was sent for a sonogram, which was performed using a HDI 5000 (Philips Medical, Bothell, Washington) with a 12.5 MHz–50-mm linear probe. At the 12 o’clock position, 7 cm from the nipple, an oval-shaped, circumscribed mass with mixed echogenicity was found. It was imaged in the radial (Figure 7) and antiradial (Figure 8) planes and measured approximately 3.4 × 1.6 × 4.2 cm in diameter. Color Doppler (Figure 9) was applied to check for vascularity. No color flow was detected.
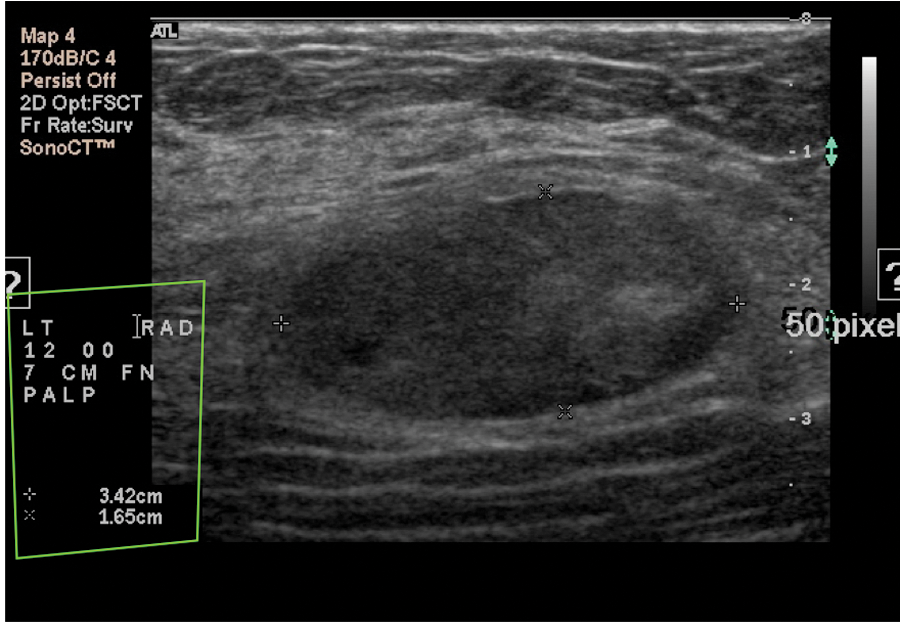
Radial plane of the palpable mass. The location corresponds to the palpable area. The internal echo pattern is heterogeneous with hyperechoic and hypoechoic features. Measurements were taken to assess the approximate volume.
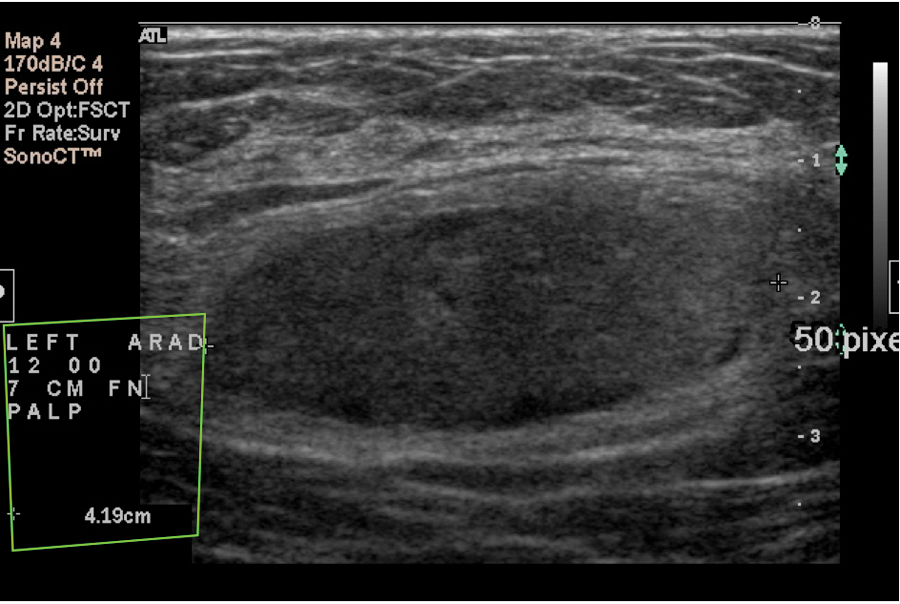
Antiradial plane of this smooth oval lesion.
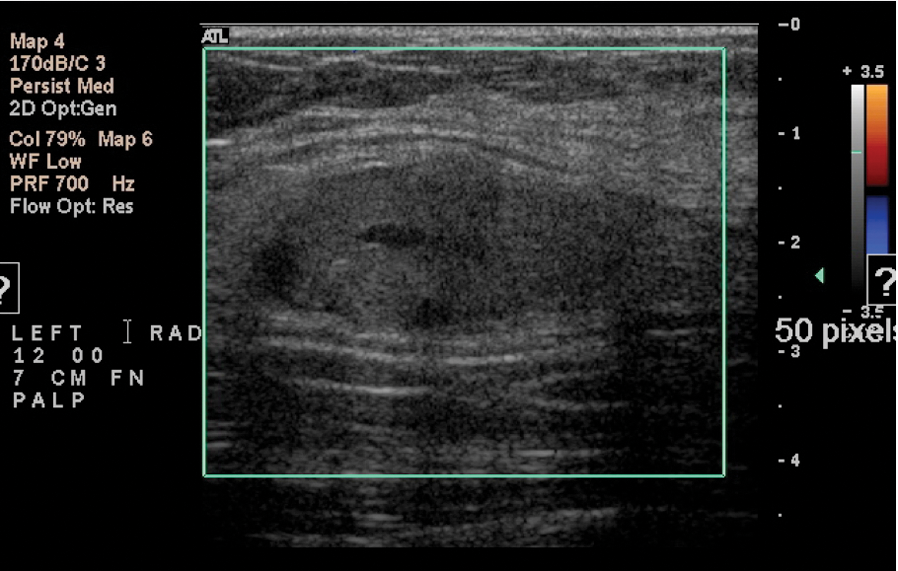
There was no blood flow detected in this lesion.
The radiologist discussed these findings with the patient and suggested sonographic-guided core biopsy. The patient informed the radiologist that approximately six to seven months ago, she underwent augmentation of both breasts. This was accomplished using fatty tissue from her buttocks using liposuction. This additional clinical history added to the differential diagnosis that the palpable mass could represent the transferred fatty tissue. The atypical sonographic features likely reflected postoperative changes. She was advised to proceed with the core biopsy for confirmation. The histology from the biopsy revealed benign breast tissue with fat necrosis. No further follow-up was determined to be necessary. Routine screening was suggested.
Later, the patient returned for a screening mammogram. Routine bilateral films were done. The right breast (Figures 10 and 11) did not demonstrate any sign of the augmentation. The left breast (Figures 12 and 13) views demonstrated the biopsied mass with the clip in place.
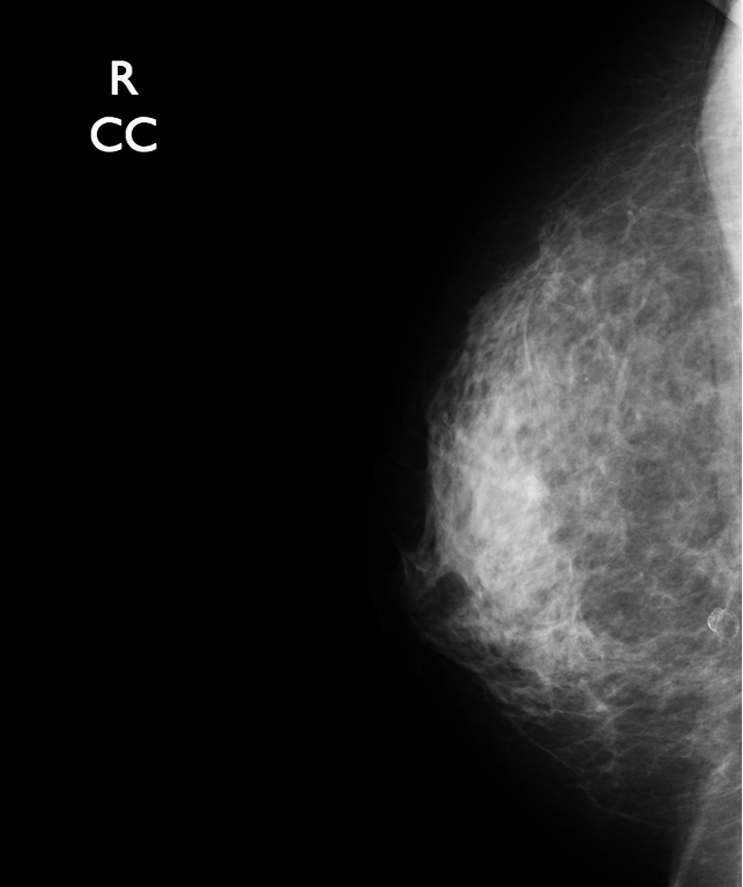
Cranial caudal (CC) view of normal breast tissue in the right breast. The transferred fat blends in with the fat tissue of the breast.
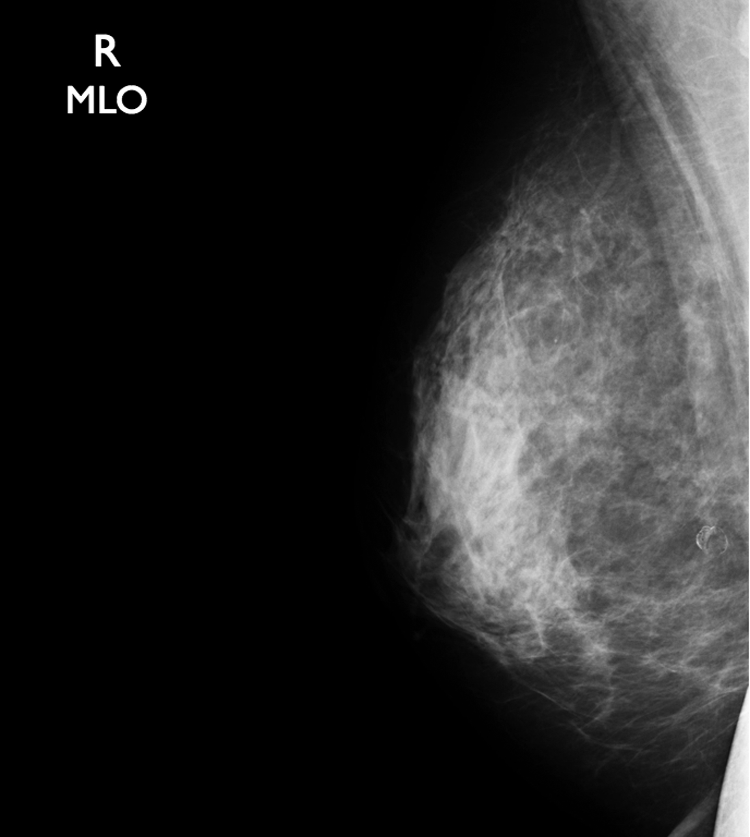
Medial lateral oblique view of the right breast. No sign of the augmentation was detected.
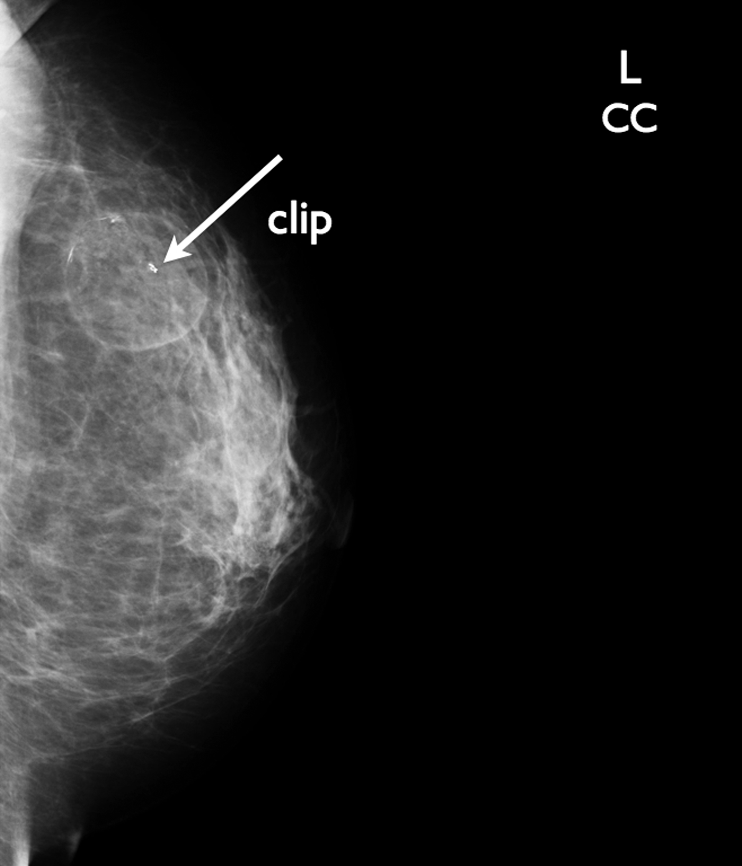
Follow-up cranial caudal (CC) view of the left breast with the clip (arrow) noted in the mass to designate the biopsied area.
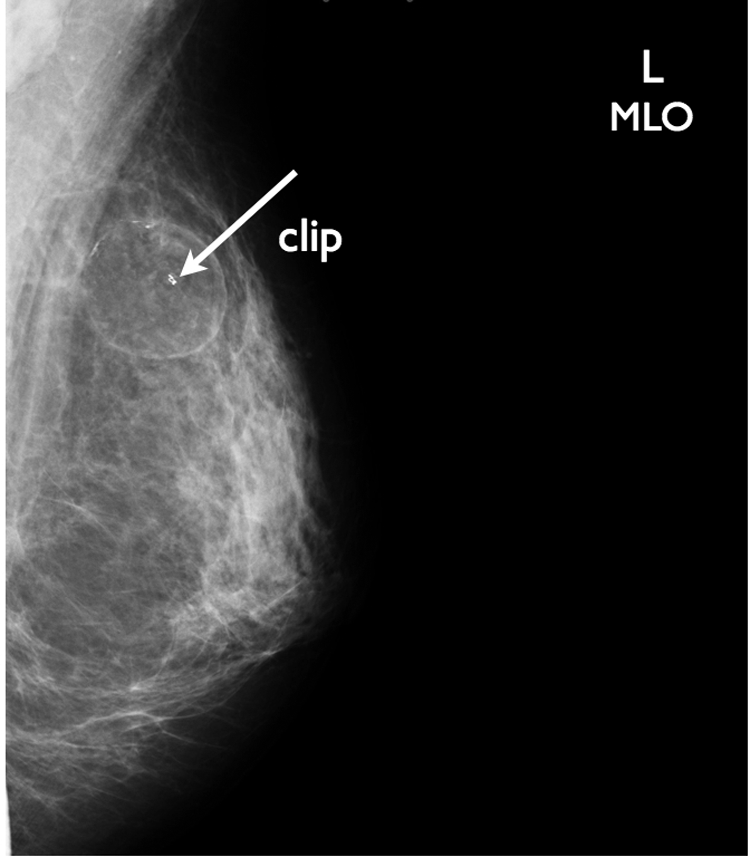
Medial lateral oblique view with clip in place (arrow).
Case Study 2
A woman in her early 40s presented with a palpable lump in her left breast. The patient reported a recent fat injection into her breast in the area of the new lump. A BB was placed over the palpable abnormality, before the CC (Figure 14) and MLO (Figure 15) views were performed.
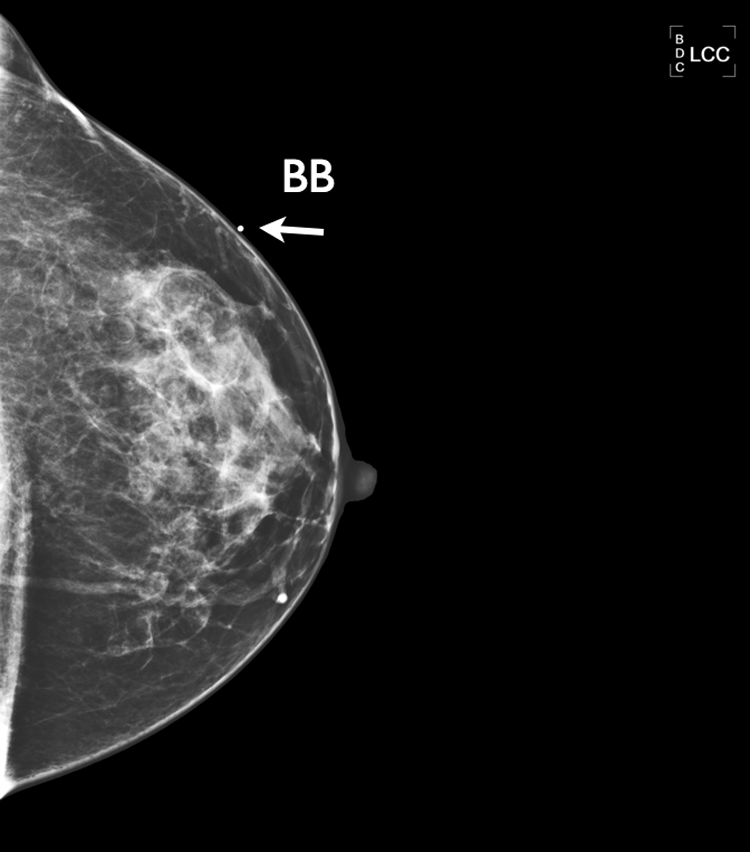
Cranial caudal (CC) view of the left breast with a BB (arrow) placed on the breast where the lump was felt by the patient. Heterogeneously dense breast with no appreciable difference from the earlier mammogram.
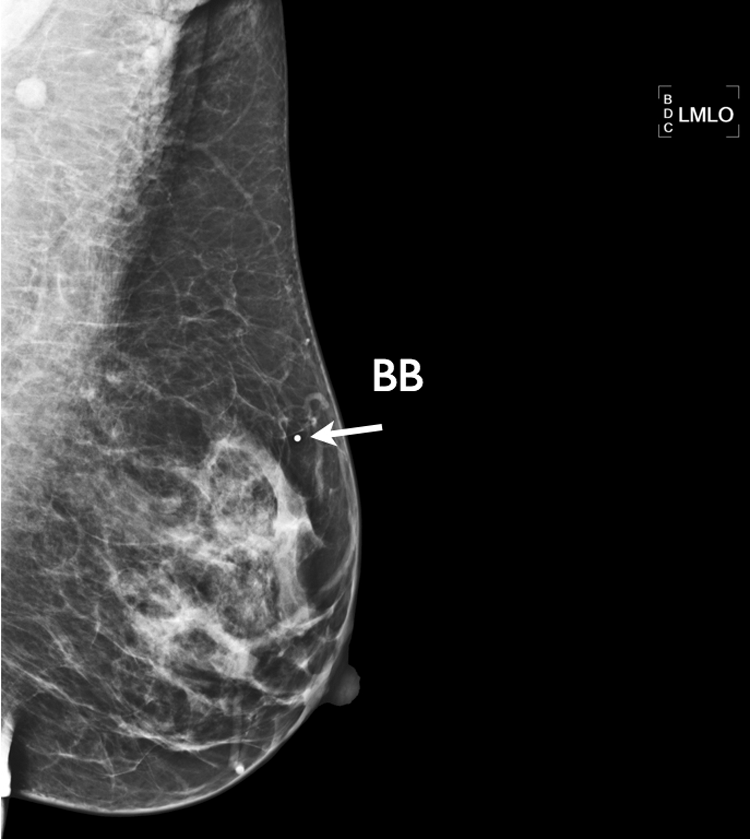
Medial lateral oblique view of BB (arrow) indicating the palpable area.
Mammographic findings were interpreted as having heterogeneously dense breast tissue bilaterally. There was no real appreciable difference from her earlier screening mammogram. All views were digitally done on Siemens Hologic LoRad Selenia. Since the patient had a new palpable lump, she was sent for a sonogram, which was performed with a Philips HDI 5000 unit using a 12.5 MHz–to 50-mm linear probe. The left breast was scanned in the region of the palpable abnormality as identified by the patient. Radial and antiradial images and measurements were taken in the 1 o’clock to 2 o’clock position. Different angles and planes were used to characterize the internal echoes and surrounding tissue (Figures 16 and 17). Several mixed echogenic and hypoechoic masses were identified in this region. Given the history of fatty tissue injection into the breast, it was determined that the areas seen by sonography likely represented areas of fat necrosis. A six-month follow-up was suggested to evaluate for stability.
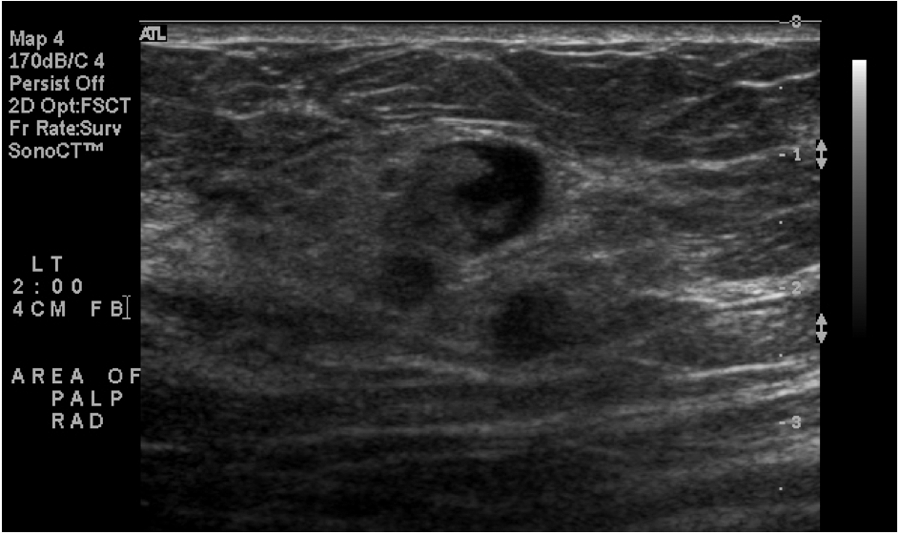
Radial plane of the palpable area with mixed echogenicity.
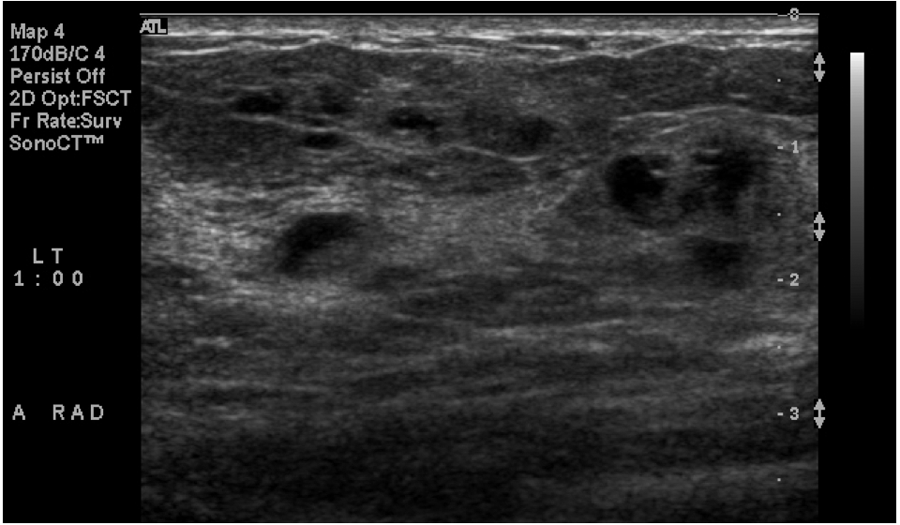
Antiradial plane demonstrating several areas that are not well circumscribed, likely fat necrosis.
Discussion
Complications
Using fat obtained from the patient’s own body should lower the rate of rejection, but there is a problem of reabsorption. To compensate for the absorption, the surgeon may overinject the fat. 4 This could cause the patient to have multiple injections over time at a continuing expense. The total cost can be much higher than having an implant placed in the breast. Dead cells can dissolve or form chronic inflammatory reactions. The tissue can cause a granulomatous reaction or form fatty cysts. Lumpiness can also occur. Other complications that could result are fat necrosis, scarring, and perhaps infection.4–6 One of the biggest challenges is differentiating between calcifications associated with fat transfer and those associated with breast cancer. The procedure can be painful and can last anywhere between six and eight hours, increasing the risk of complications with anesthesia. 4 There have been no long-term clinical trials. 6
Illouz and Sterodimas, 5 with 25 years of fat transplantation experience, evaluated 820 women operated on between 1983 and 2007. The authors reported 163 patients with some type of complication. Six hundred seventy patients underwent mammography at six months and one year. The majority of complications were noted in the first six months after each session. “Breast lesions, including calcifications, cysts, and cancer, that are not apparent in the first year after the final procedure of lipofilling, we believe may not be directly associated with the autologous fat grafting to the breast.” 5 Patients with a susceptible breast lesion less than one year after the final intervention should be evaluated further.
Similar tissue types should blend together, but changes can occur to the donor fat after it is redistributed into the breast. A patient who has a routine screening mammogram without any breast complaints might have findings warranting a call back for more mammographic images and/or a sonogram. In the two cases in this study, the patients were referred for a mammogram and a sonogram because of a new palpable lump. In the first case, the fat looked like a lipoma, which is a noncancerous lump of fatty tissue. The results of the sonogram showed a heterogeneous mass of mixed echogenicity. Characterizing this mass by sonography gave additional information to the radiologist. A biopsy was necessary to get the correct diagnosis and make sure that management of the mass was in the best interest of the patient’s breast health. In the second case, the mammogram did not demonstrate any appreciable abnormalities. Again, the sonogram demonstrated areas of mixed echogenicity in the region, representing possible areas of fat necrosis. Because of the history of a fat transfer to the breast, a six-month follow-up was recommended. The sonogram was the key element in both cases. It helped determine the final outcome of the palpable lumps to complete the breast imaging.
According to the American Society of Plastic Surgeons, 7 “lipofilling [after] breast cancer treatment leads to low complication rate and does not affect the radiologic follow-up after breast conserving surgery.” Three French and Italian hospitals also evaluated 513 women who underwent lipofilling after breast cancer surgery from 2000 to 2010. Follow-up showed a recurrence of breast cancer that developed in 5.6% of patients. There was no evidence that lipofilling interfered with the detection, but a biopsy of the area should be obtained if there is any doubt. Researchers have emphasized long-term follow-up studies to confirm the safety of the procedure. 7
In another study, the authors compared breast tissue density in 20 patients with preoperative and postoperative imaging between 2000 and 2008. They concluded that breast tissue treated with fat grafting was not problematic and should not be a hindrance to the procedure. 6
The American Society of Aesthetic Plastic Surgery and the American Society of Plastic Surgeons set guidelines in 2009 stating that they do not recommend breast augmentation by fat transfer, except in a clinical trial. Fat transfer is a safe procedure in select cases, and the results depend on the expertise of the surgeon. The short-term effects are starting to be seen more and more in diagnostic imaging, and this can complicate the patient’s breast health.7–9
Conclusion
An accurate patient history should be obtained from all patients. Additional or more detailed information may be necessary to assist in summarizing overall breast health. Clinical histories could include a question about previous surgeries or injections that might complicate the imaging of the natural breast tissue. Some patients do not consider fat transfer as a type of augmentation or the procedure as surgery. Therefore, this question could help to educate patients that any change in their breast is important information. This may decrease additional imaging. Fat transfer is not a common procedure for most women, but breast health care providers need to be aware of the possibility.
Mammograms are the first defense in detecting cancer before it becomes invasive. The complications following the fat transfer can increase the radiation and/or procedures needed to assess these changes. As more and more women have these elective surgeries, radiologists interpreting the mammograms and sonograms need to be aware of natural breast augmentations and what effects they can have on the breast.
Footnotes
The author declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author received no financial support for the research and/or authorship of this article.
