Abstract
An implantable cardioverter defibrillator (ICD) is placed in the chest wall of patients who have reduced cardiac function and recurrent episodes of ventricular tachycardia or fibrillation. The purpose of the ICD is to detect life-threatening arrhythmias with subsequent cardioversion to a normal rhythm. Standard positioning of ICD leads are isolated to the right atrium and right ventricle, respectively. This case report documents an incidental but rare finding of an ICD lead traversing a suspected atrial septal defect in a patient with worsening dyspnea and a history of pulmonary embolism. Transthoracic echocardiography revealed what appeared to be a looped right atrial lead within the left atrium traversing the atrial septum, demonstrating an unusual ring appearance artifact. Subsequent imaging by contrast-enhanced computed tomography of the chest and transesophageal echocardiography confirmed the initial transthoracic echocardiographic findings.
Keywords
Implantable cardioverter defibrillators (ICDs) are placed in the chest wall of patients who have reduced cardiac function and recurrent episodes of ventricular tachycardia or fibrillation to detect life-threatening arrhythmias and provide cardioversion. This case describes an incidental but rare finding of an ICD lead traversing a suspected atrial septal defect (ASD) in a patient with worsening dyspnea and a history of pulmonary embolism.
Case Report
A man in his late 70s presented to the emergency room with worsening dyspnea and hypotension. Past medical history included coronary artery disease consistent with ischemic cardiomyopathy, atrial fibrillation, pulmonary fibrosis, recurrent deep vein thrombosis, and pulmonary embolism. The patient was intolerant to anticoagulation therapy. An echocardiogram performed eight months earlier revealed moderate to severe left ventricular dysfunction with an ejection fraction of 30% to 35%, right heart dilation, severe pulmonary hypertension, severe tricuspid regurgitation, and moderate mitral regurgitation. Five months prior, the patient had ICD replacement with original leads remaining in place.
Following admission, a transthoracic echocardiogram was performed. There were no significant changes in the results of the echocardiogram compared with the prior study. However, the right atrial ICD lead appeared to traverse the atrial septum, forming a loop within the left atrium, as demonstrated by several echocardiographic views (Figures 1–4). Doppler color flow imaging demonstrated significant left-to-right shunting at the midlevel of the interatrial septum coinciding with the position of the right atrial lead (Figure 5). The lead in the right ventricle appeared to be in the appropriate location. Findings were most consistent with displacement of an ICD lead and a small ostium secundum ASD.
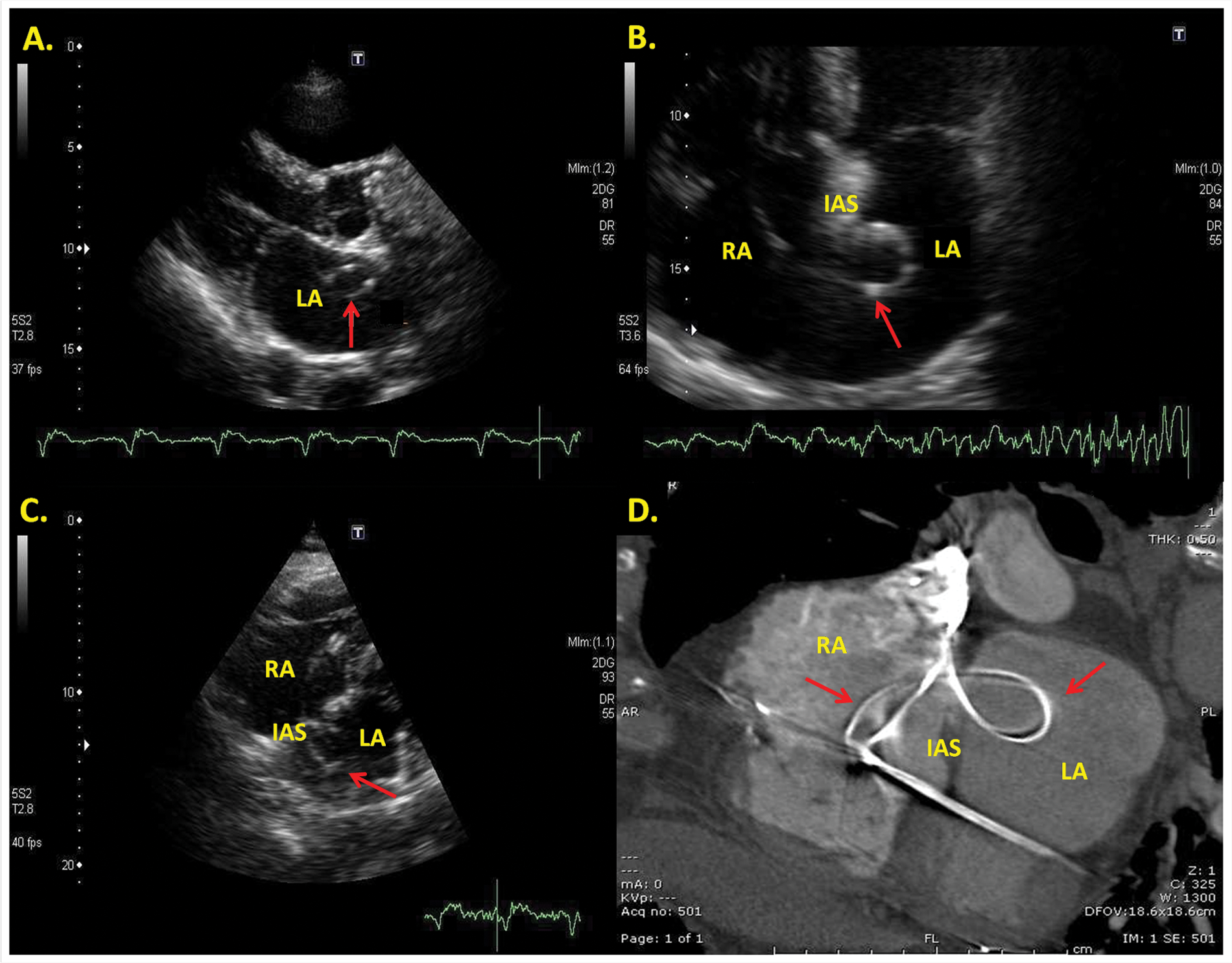
Parasternal long-axis view demonstrating the looped ICD lead (arrow) within the left atrium (A); apical four-chamber view demonstrating the lead crossing the IAS and looping in the LA (arrow), before crossing back across the IAS (B); subcostal view of the atria displaying the lead (arrow) within the LA and RA (C); cross-sectional view of contrast-enhanced CT of the heart demonstrating the lead traversing the IAS, creating a “bowtie” appearance (arrows) (D). LA, left atrium; RA, right atrium; IAS, interatrial septum; ICD, implantable cardioverter defibrillator; CT, computed tomography.
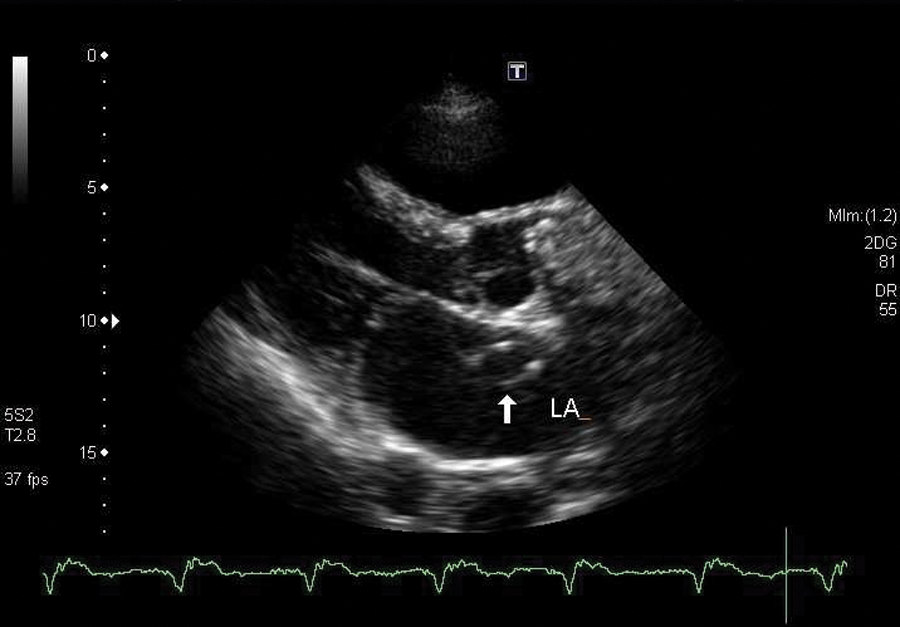
Parasternal long-axis view demonstrating the looped implantable cardioverter defibrillator lead wire within the left atrium (LA).
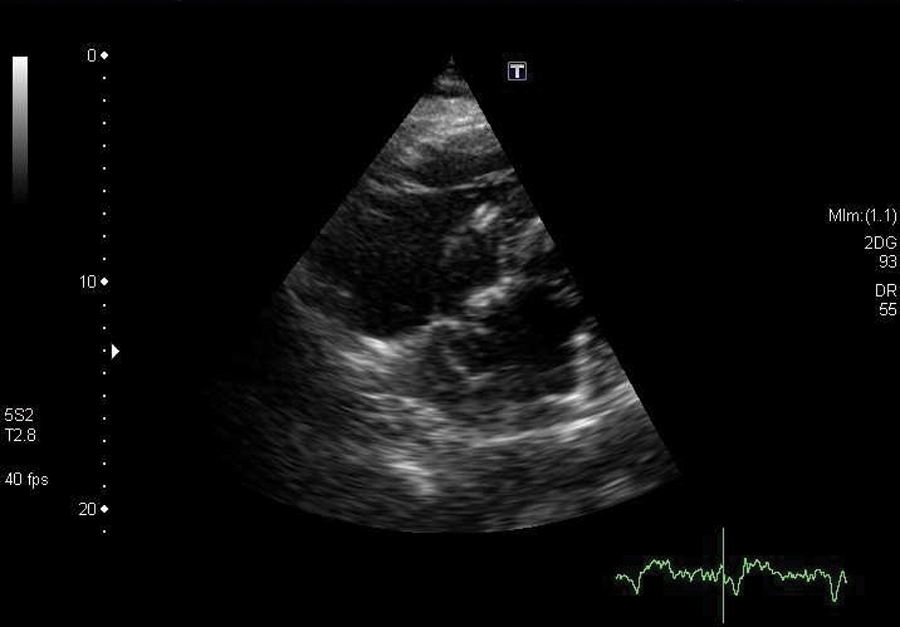
Subcostal view of the atria displaying the lead wire within the left atrium.
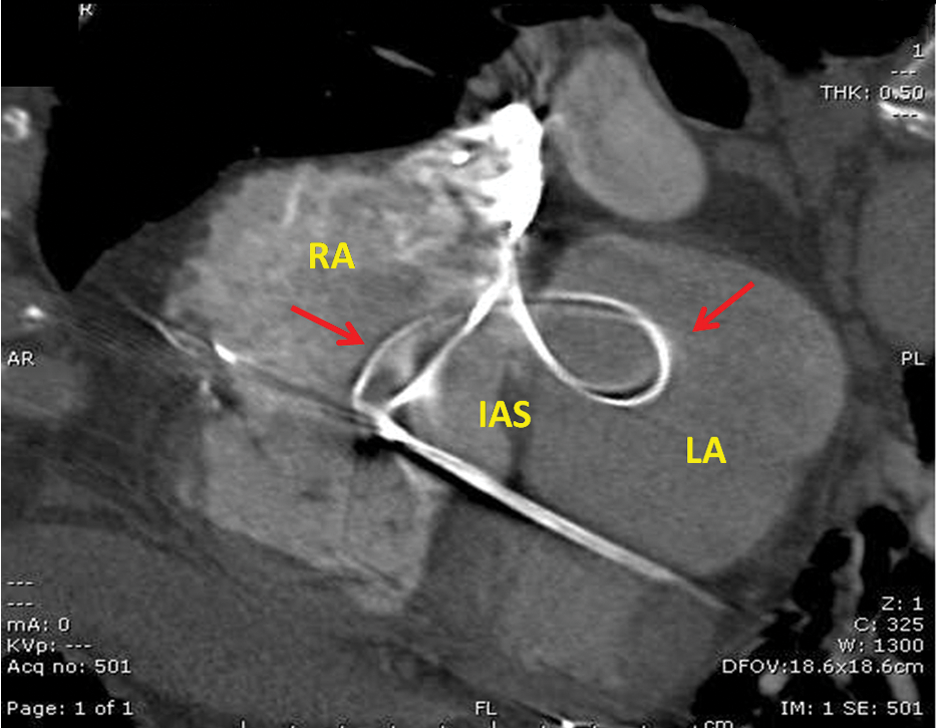
Cross-sectional computed tomography image of the heart demonstrating the lead wires traversing the septal defect.
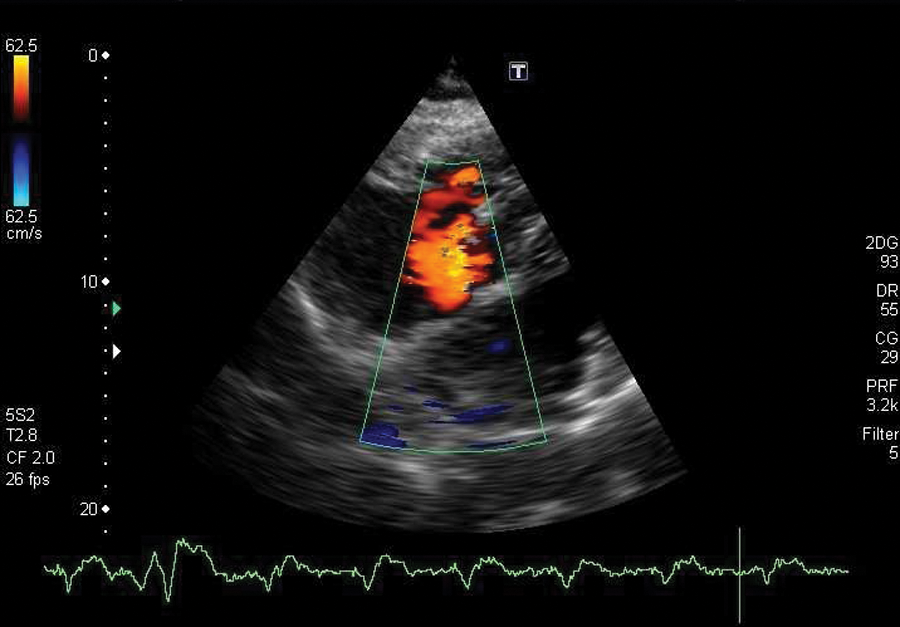
Subcostal view with color Doppler revealing the presence of left-to-right shunting of blood across the atrial septum.
Given the patient’s history, worsening dyspnea, and unusual appearance of ICD leads, contrast-enhanced computed tomography (CT) of the chest was performed the following day to evaluate for recurring pulmonary embolism. The CT revealed a small pulmonary embolus at the bifurcation of the right and main pulmonary artery. In addition, the CT confirmed the presence of a lead traversing the atrial septum, creating an unusual “bowtie” appearance (Figure 1D). A transesophageal echocardiogram (images not shown) confirmed the findings observed on the CT and transthoracic echocardiogram.
Discussion
Placement of ICD and pacemaker leads is the most common surgery involving the heart. 1 These small devices are surgically placed in the chest wall to help detect and control potentially dangerous and life-threatening arrhythmias. A pacemaker uses low-energy electrical pulses to maintain normal sinus rhythm. In contrast, an ICD uses both low-energy electrical pulses and high-energy electrical shocks for cardioversion. 2 Conventional placement of these leads courses through the subclavian vein and superior vena cava to gain right heart access.
The majority of patients with ICDs have two leads implanted, with one placed in the right ventricle and one in the right atrium. The ICD leads can perform two functions: (1) continually monitor the patient’s cardiac rhythm and (2) if abnormal rhythms are detected, the device is designed to use electrical pulses to cardiovert or shock the heart into normal rhythm. If the device needs to be replaced, it is common practice to leave the original leads in place to prevent damage to the heart wall. 3
An ASD is an absence of tissue in the atrial septum, which permits blood to predominantly shunt from the left atrium to the right atrium. ASDs comprise approximately 10% of all congenital heart disease, with ostium secundum defects accounting for 60% to 70% of all ASDs. 4 Patients can be asymptomatic, and often the initial finding of an ASD may not occur until adulthood. A patent foramen ovale (PFO), by contrast, is a valvular-type opening at the ostium secundum region in the atrial septum that does not close spontaneously after birth. 4 Transient left-to-right, right-to-left shunting may occur in patients with PFOs, without significant chamber dilation. An ASD, unlike PFO, demonstrates more continuous shunting, resulting in significant volume overload and right heart dilation. 4
The determination was made in this case that the defect was a small secundum ASD rather than a PFO, based on the size, direction, location, and duration of the shunt. Multiple echocardiographic views are required to fully assess the presence, location, and size of an ASD. The view of choice is the subcostal four-chamber view.5,6 Imaging of the interatrial septum from a standard apical four-chamber view is less desirable because of potential shadowing and “echo dropout” of the mid-atrial septum. This echo-free area can create the potential for false-positive results. 6 The subcostal four-chamber view places the atrial septum more perpendicular to the sonographic beam, permitting better delineation of atrial structure by 2D and color Doppler imaging. The subcostal approach is also used to differentiate between different types of defects such as primum, secundum, and sinus venosus ASDs. 6
ICD leads visualized within the left side of the heart, although rare, can occur by accidental passing of a wire through an ASD, PFO, or by perforation of the atrial septum. 7
There have been reports of a pacing lead being inadvertently placed within the left side of the heart.1,7 To our knowledge, this is the first report of an ICD lead traversing a suspected ASD, as demonstrated by contrast-enhanced CT, transthoracic echocardiography, and transesophageal echocardiography.
Conclusion
Echocardiography is often the first cardiac imaging modality used for symptomatic patients with a known history of cardiac disease. In this case, we were able to use multiple imaging modalities to confirm the presence of incidental but rare findings within the heart. An understanding of normal placement and location of intracardiac leads by the sonographer is necessary.
Footnotes
Acknowledgements
Special thanks to the entire Mount Desert Island Hospital radiology staff, David McQuade, MD, and Sherri B. Zeisloft, RDMS, RVT, RRT, for their support and help with the manuscript.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
