Abstract
Atrial septal defects (ASDs) are one of the most common heart defects diagnosed in the adult population. If left unrepaired, they may lead to significant complications including right heart volume overload, pulmonary complications, and heart failure. Percutaneous transcatheter closure by use of an occluder device is clinically indicated for most patients with a secundum ASD and who are symptomatic. Although this procedure is safe, in rare instances, complications may be encountered, including atrial wall perforation, arrhythmias, and, in very rare instances, device migration. Echocardiography has become a widely used and reliable form of patient imaging that relies on the skillset of the sonographer as well as the interpreting physician. This case study illustrates the specific role of the cardiac sonographer in taking a careful clinical history, imaging of an ASD occluder device, and its location as it migrated into the aortic arch. This case also demonstrates the importance of imaging beyond the standard echocardiographic protocol, given this clinical manifestation.
Case Study
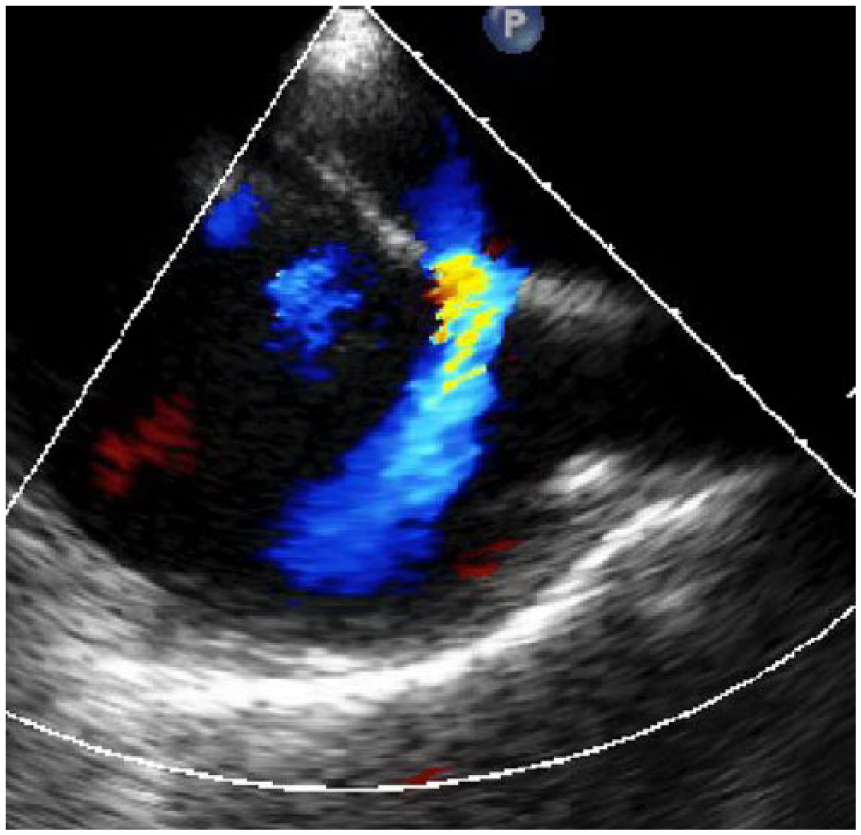
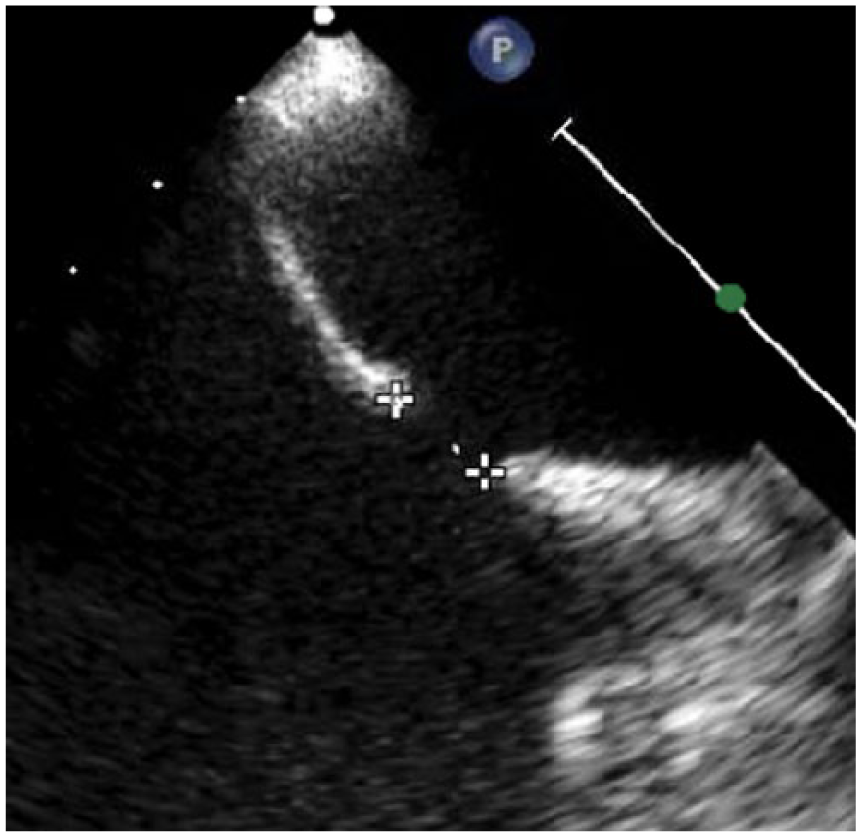
A 46-year-old patient was referred to the outpatient heart clinic with a history of a left posterior circulation cerebrovascular accident. The patient underwent a workup including a transesophageal echocardiogram, which discovered a 19-mm secundum atrial septal defect (ASD) (Figures 1 and 2) with an associated atrial septal aneurysm (Figure 3). Due to the septal defect, the patient was consulted for a possible atrial septal closure. A 20-mm AMPLATZER closure device (St. Jude Medical, St. Paul, MN) (Figure 4) was used to repair the defect. A resting echocardiogram with a bubble study was ordered several months postimplantation of the atrial septal occluder device. Echocardiographic views of the parasternal short axis demonstrated an absence of an echogenic structure consistent with the known occluder device. The apical windows also demonstrated similar findings of the septum and absence of an apparent ASD closure device. At the end of the examination, the suprasternal notch view was investigated to inspect the distal ascending aorta, transverse aortic arch, and proximal descending aorta. The suprasternal notch view at the level of the aortic arch revealed a bright echogenic structure that was visualized close to the origin of the head vessels (Figure 5). Color flow Doppler imaging confirmed that the echogenic structure was not an artifact by demonstrating mosaic color flow distal to the structure in the descending aorta, leading us to believe that there was a true obstruction in the aorta producing the mosaic flow pattern (Figure 6). This led to the strong suspicion that the atrial septal occluder device lay butted against the arterial walls.
Transesophageal echocardiography demonstrating the secundum atrial septal defect with color Doppler before closure device deployment.
Measurement of the secundum atrial septal defect with transesophageal echocardiogram.
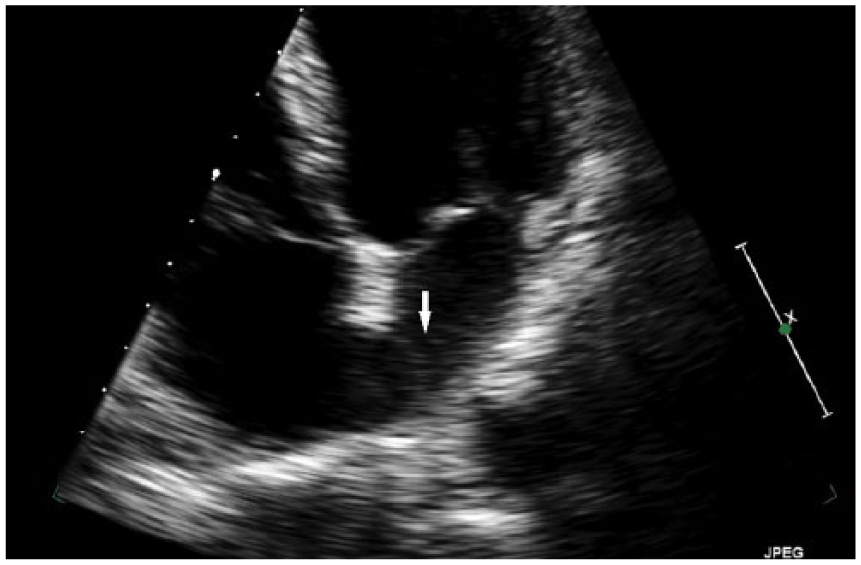
Atrial septal aneurysm (white arrow) bulging into the left atrium eluding to a hypermobile atrial septum.

Illustration from St. Jude Medical of an AMPLATZER™ Septal Occluder device. AMPLATZER and St. Jude Medical are trademarks of St. Jude Medical, Inc. or its related companies. Reproduced with permission of St. Jude Medical, ©2016. All rights reserved.
Two-dimensional echocardiography at the level of the suprasternal notch demonstrating the dislodged occluder device resting in the aortic arch postimplantation.
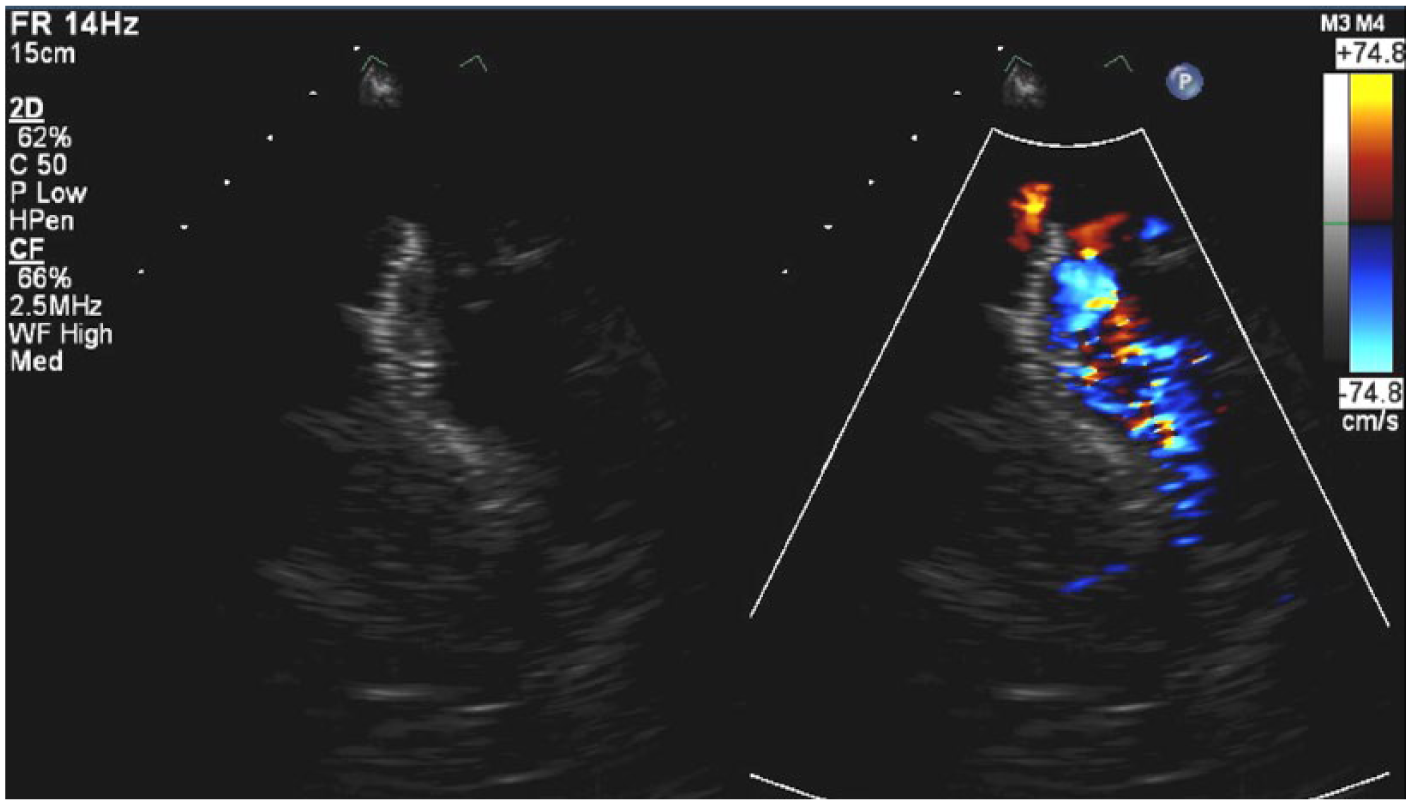
Color Doppler demonstrating the turbulent flow distal to the occluder device in the descending aorta via the suprasternal notch window that is not characteristic of typical blood flow in a normal aorta.
To confirm the position of the ASD occluder device, a chest computed tomography (CT) angiogram was performed. The CT angiogram confirmed the suspicion of an occluder device that had migrated to the aortic arch (Figure 7). The device was resting obliquely, extending from the innominate artery to the left subclavian artery. The patient was placed under conscience sedation with full anesthesia and transesophageal echocardiography guidance. Surgery demonstrated that the device was significantly adhered to the aortic wall during a limited aortotomy. This was a significant finding due to the potential risk of perforation through the aorta.
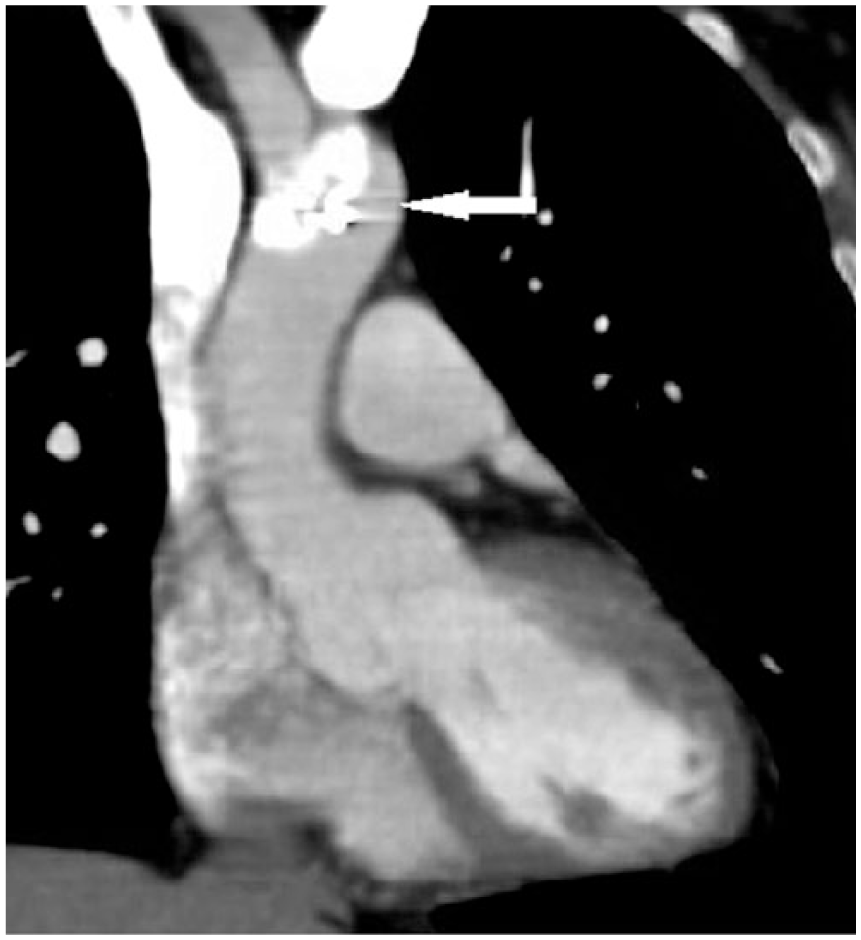
Computed tomography confirming the position of the septal occluder device that migrated to the aortic arch.
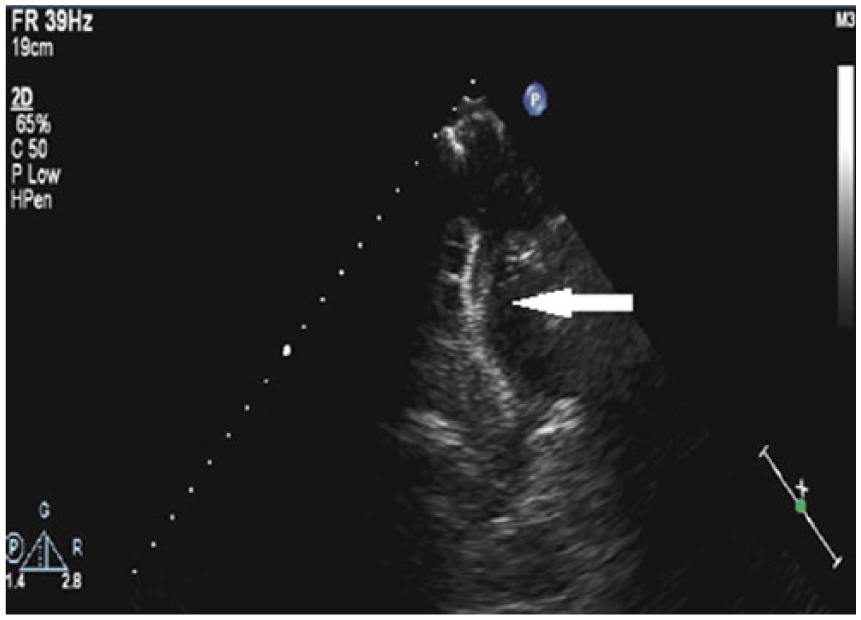
Furthermore, the edges of the device were beginning to erode through the intima and media. The device also contained substantial amounts of fibrin deposit and thrombus, putting the patient at an even greater risk for a cerebrovascular event. Along with the removal of the foreign body from the aortic arch, the patient’s residual secundum ASD was visualized again with intraoperative transesophageal echocardiography (Figure 8) and was repaired with a primary closure, which included a dissected portion of the patient’s own pericardium implanted over the defect. 1 Because the patient’s native tissue was used to repair the defect, there is no discernible echogenicity of the new primary occluder on echocardiogram as seen in Figure 9. In addition, surgical repair of the left common carotid origin and aortic arch was necessary. A postoperative transesophageal echocardiogram demonstrated a normal functioning left ventricle with no residual flow crossing through the atrial septum. The patient also underwent a follow-up CT angiogram to evaluate the status of his aortic repair, which was unremarkable. Serial echocardiograms with bubble studies were also done to confirm the successful closure of the ASD.
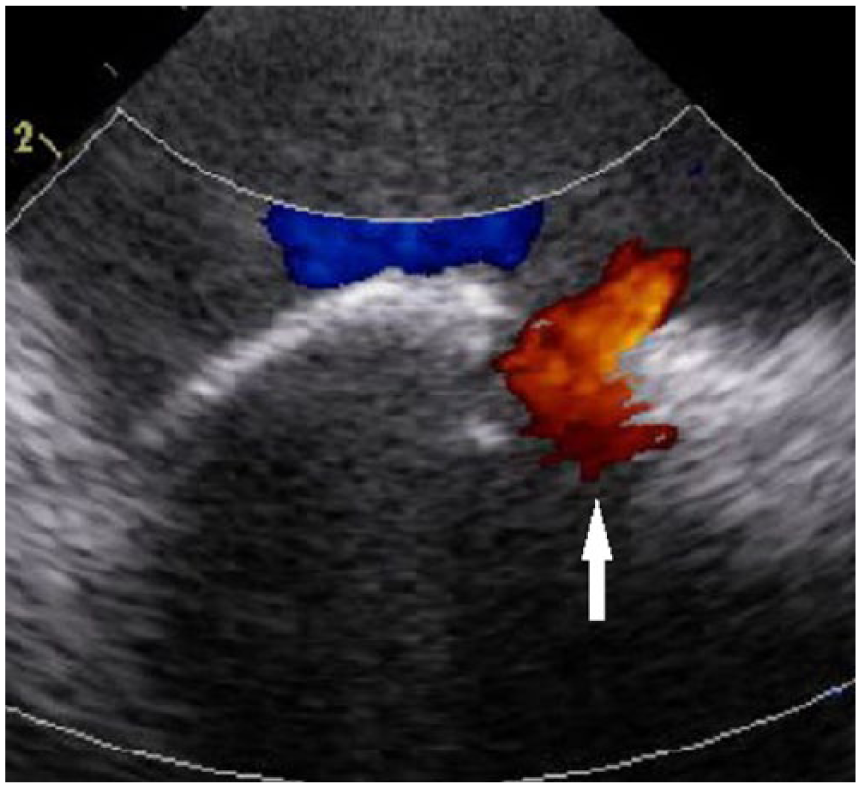
Intracardiac echocardiogram demonstrating the residual atrial septal defect with an absence of the atrial septal occluder device.
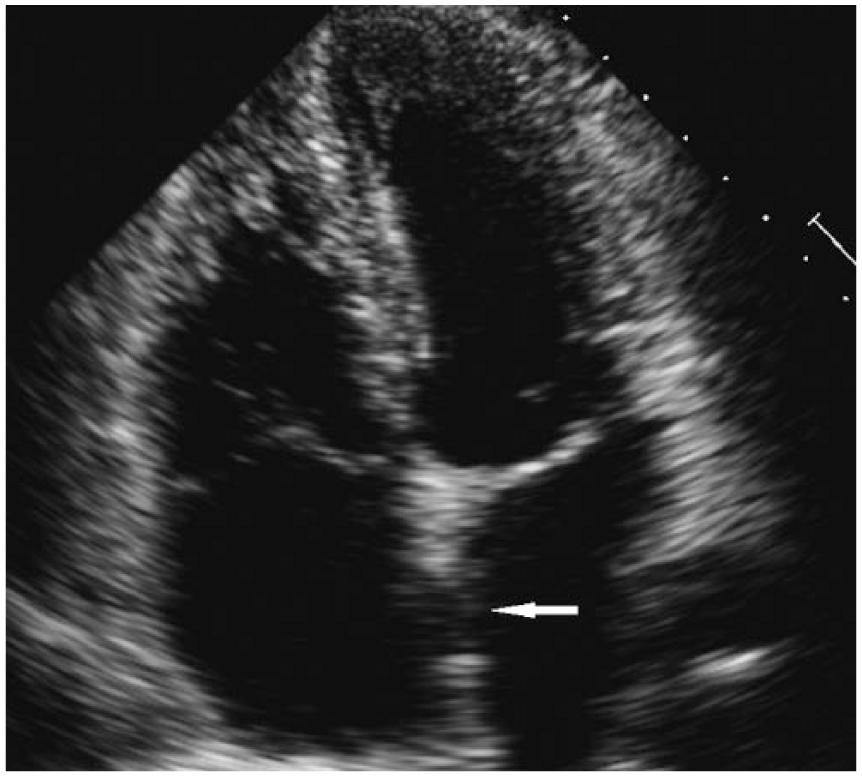
Apical four-chamber view demonstrating the primary closure used to close the secundum atrial septal defect.
Discussion
Atrial septal communications account for approximately 6% to 10% of congenital heart defects, with an incidence rate of 1 in 1500 live births. 2 There are four different types of ASDs: secundum, primum, sinus venosus, and coronary sinus (in order of frequency). 3 A patent foramen ovale (PFO) is not considered a true ASD because no structural deficiency of the atrial septal tissue is present. 4 Patent foramen ovales are more common and can be present in up to 20% to 25% of the adult population. 2 Smaller septal defects and PFOs may spontaneously close on their own during infancy or early childhood. 2 Patent foramen ovales in asymptomatic patients are not recommended for closure and may be treated medically with aspirin; however, patients who experience recurrent strokes or transient ischemic attacks may have their PFO fixed percutaneously. 5 Larger defects or defects that go undetected into adulthood may damage the heart and lungs over long periods of time and will need surgical closure, as in this case presentation.
As blood flows through the heart in the presence of an ASD, the oxygenated blood travels through the defect back into the right side of the heart, mixing with the deoxygenated blood. Over time, this can cause the right side of the heart to enlarge and weaken and cause pulmonary hypertension. 4 There is a potential for Eisenmenger’s syndrome to develop with longstanding pulmonary hypertension, which is a left-to-right shunt that has reversed direction. 6 Right heart pressures can be evaluated by measuring the peak pulmonary artery systolic pressure from tricuspid regurgitation. 3 Mild pulmonary hypertension is assessed at 30 to 40 mmHg and severe pulmonary hypertension is greater than 70 mmHg. 7 Another measurement to calculate the severity of atrial shunting is the Qp/Qs equation. 6 By calculating right ventricular stroke volume and left ventricular stroke volume through the respective outflow cross-sectional areas, the cardiac sonographer can supply the cardiologist with a pulmonary-to-systemic flow ratio. A normal ratio, indicating no shunt, is 1:1. A significant shunt has a Qp/Qs ratio of 1.5:1 or greater. 6 For further measurements and quantification, cardiac sonographers should use electronic calipers and measure the size of the defect, if possible. A large defect is considered to be 1.0 cm or greater. 6
Transthoracic and transesophageal echocardiograms are effective tools to determine the presence, type, and hemodynamic effect of an ASD. These shunts are best visualized and interrogated using the subcostal four chambers (due to blood flow being parallel with the Doppler signal), apical four chamber, and the parasternal short axis at the level of the aortic valve. An associated atrial septal aneurysm may be present in patients with an ASD. 6 A true aneurysm is described as a bulging of the atrial septum greater than 1.5 cm. 6 Not only can ASDs be found in the area of the atrial septal aneurysm, but the aneurysm can also be a cause of embolism. 6 Other echocardiogram findings associated with an ASD are right ventricle dilatation, right atrium dilatation, a dilated pulmonary artery, and a flattening of the right ventricle in both systole and/or diastole, indicating a pressure overload and volume overload, respectively. Electrocardiography may show the presence of a right bundle branch block and other arrhythmias in patients with an ASD due to conduction interruption. 6
Determining the correct size of the ASD is imperative to assure proper deployment of the occluder device and to eliminate the potential of device migration. Recently, 3D transesophageal echocardiography (TEE) and intracardiac echocardiography have been helpful in accurately measuring the ASD and properly positioning the closure device. 3 Three-dimensional TEE may also play a major role in finding the appropriate rims to securely fasten the closure device to the heart. 3 Rims are edges surrounding the defect that are ideal locations for anchoring the occluder device. 3 Treatment varies depending on the size and location of the defect. Currently, simple secundum ASDs are the most common type of defect closed percutaneously with an atrial occluder device. 3 This type of secundum defect involves one single hole and is not associated with other forms of congenital defects. It is paramount that cardiac sonographers obtain accurate information prior to implantation of these types of devices with the use of transthoracic echocardiography and TEE to accurately define the heart’s anatomy.
During a postimplantation echocardiogram, the typical appearance of an occluder device has very high echogenicity when seen with sonography. The occluder device is noticeably thicker than a normal atrial septum and typically has a “clamshell” appearance. Due to the risk of residual shunting, which is not a rarity in the early stages of endothelialization, it is common for cardiologists to order serial echocardiograms with bubble studies every 3, 6, and 9 months from implantation of atrial septal occluder devices. To interrogate the full functionality of the septal occluder device, 5- to 10-mL injections of agitated saline should be performed with and without a Valsalva maneuver.4,6 Obtaining high-quality diagnostic information by investigating all the sonographic windows when assessing the heart and its related structures minimizes the risk of missing pathology.
As with any invasive procedure, adverse events can occur. Complete separation and migration of these devices from the atrial septum are a rarity. However, this does occur in 0.4% to 1.1% of patients.8–12 Device embolization and erosion are potential life-threatening complications requiring immediate removal by percutaneous or surgical intervention. 10 Reasons for embolization are inappropriate sizing of the device, inadequate rims of the ASD for anchoring, improper deployment of the device, “Minnesota wiggle,” extensive Valsalva by the patient, and unwarranted cable tension. 13 The Minnesota wiggle is a technique used to check the secure placement of the occluder device within the atrial septum by slightly tugging on the wire. 14 Regardless of the nature and risk of adverse events, as in this instance, routine echocardiograms investigating the device are quantitatively and qualitatively necessary. Sonographers remain the hands-on examiner for the cardiologist—the cardiologist’s “eyes and ears.” Sonographers act as detectives and careful clinical investigators in the echocardiography lab. This is coupled with the careful mindset that allows the physician to obtain as much information as possible.
Conclusion
As health care providers continue to balance providing quality care with lower reimbursement, echocardiography labs can feel pressure to increase the quantity of examinations performed. This case study presents 2D transthoracic, endoscopic, and intracardiac echocardiography images of an adult patient who received a 20-mm AMPLATZER atrial septal occluder device that migrated to the transverse aortic arch and lodged itself against the arterial walls, putting the patient at a high risk. This case also illustrates the importance of maintaining quality imaging, independent of the cardiac imaging windows, to ensure that pathology is not inadvertently missed. Along with all necessary imaging windows, the suprasternal notch window provided critical information that allowed the cardiologist to confidently provide swift decisions regarding this patient’s care and possibly saved the patient from further unnecessary imaging in looking for the migrated device. Furthermore, this case study highlights the importance of a complete, thorough echocardiogram examination, not only in complex patients. Although abnormalities can be rare, sonographers should feel confident in providing the ordering and referring physician with an exhaustive examination. This case study magnifies the importance of the duties that sonographers have in performing high-quality echocardiograms that increase diagnostic confidence.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
