Abstract

Keywords
Introduction
In health, exercise capacity is limited by reaching cardiovascular limits, which occurs prior to exhausting ventilatory reserve. Normal lung growth affords healthy adults more lung capacity than is needed during exercise. An excess of ventilatory capacity allows many patients to maintain reasonable exercise capacity despite partial lung resection. However, surgical removal of a complete lung usually results in a substantial loss of ventilatory reserve and exercise capacity.1,2 This case describes a patient who maintained normal spirometry and no exercise limitation following total pneumonectomy of the left lung.
Case summary
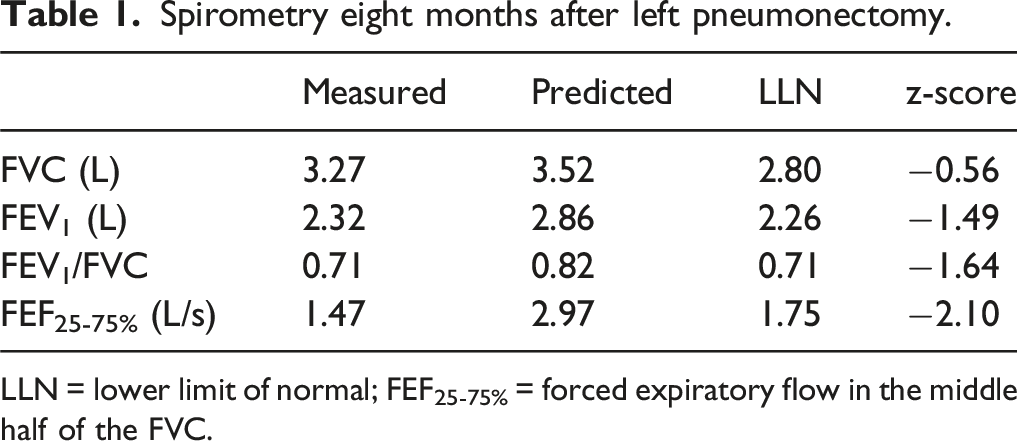
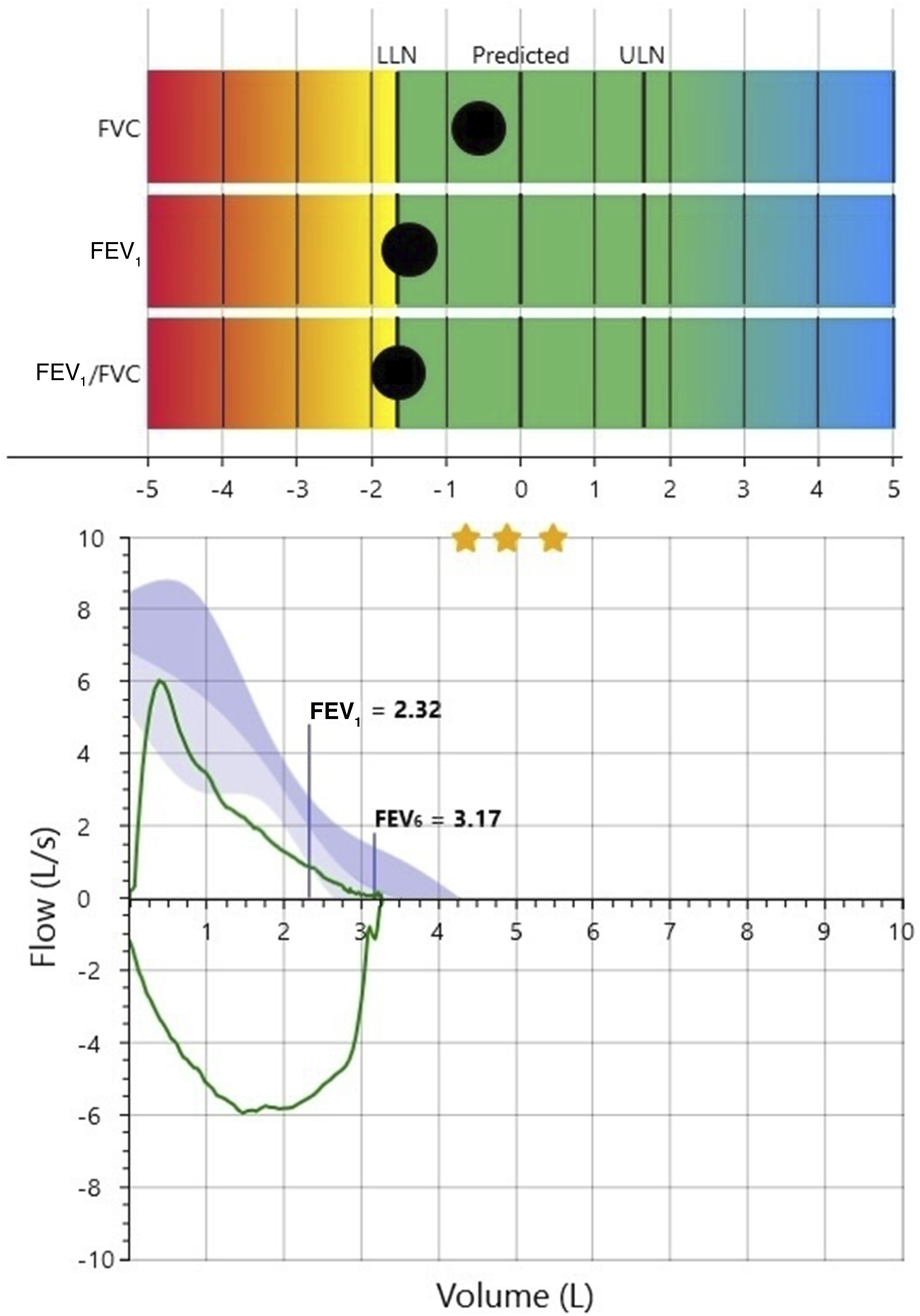
A 43-year-old woman presented with a chronic cough and unintentional weight loss. The patient had a history of asthma controlled with a long-acting beta agonist and inhaled corticosteroid. She was a former smoker of ½ pack-per-day for 16 years until quitting at age 32. At age 35, the patient underwent left upper lobe wedge resection for chondroid hamartomas. At age 43, the work-up for chronic cough and unintentional weight loss included a chest radiograph which revealed a large left perihilar mass and post-obstructive bullae formation (Figure 1). A ventilation/perfusion scan demonstrated marked asymmetry with only 15% of ventilation and 13% of perfusion to the left lung. Subsequently, the patient underwent pneumonectomy of the remaining left lung. A chest radiograph taken 4 months after pneumonectomy showed compensatory hyperinflation that had markedly shifted the trachea and right lung into the left chest cavity, and repositioned the heart laterally (Figure 2). Eight months following pneumonectomy, the patient underwent spirometry testing (Table 1, Figure 3). Remarkably, despite the loss of one lung, the FVC and FEV1 were within the normal range according to the Global Lung Function Initiative reference equations.
3
The FEV1/FVC was at the lower limit of normal. Chest radiograph showing a large left perihilar mass and post-obstructive bullae formation. Chest radiograph taken 4 months after left pneumonectomy showing compensatory hyperinflation and shifting of the trachea and right lung into the left chest cavity, and repositioning the heart laterally. Spirometry eight months after left pneumonectomy. LLN = lower limit of normal; FEF25-75% = forced expiratory flow in the middle half of the FVC. Spirometry z-score graph and flow-volume loop 8 months after pneumonectomy. LLN = lower limit of normal; ULN = upper limit of normal; FEV1 = forved expiratory volume at 1 second; FEV6 = forced expiratory volume at 6 seconds.

Discussion
The physiologic impact of pneumonectomy is much more complex than simply a ∼50% loss of total lung capacity. In the immediate post-operative period, the chest cavity of the removed lung is filled with air. However, within days the post-pneumonectomy space begins to fill with fluid, the ipsilateral hemidiaphragm shifts cephalad, and the mediastinum migrates into the post-pneumonectomy space. Complete opacification of the post-pneumonectomy space often occurs several months after surgery. 4
The impact of pneumonectomy on lung capacity and spirometry can be profound, so much so that formulas have been developed to predict spirometry values following lung resection. Predicting post-operative lung capacity can be based on perfusion studies or using preoperative spirometry values and the number of segments expected to be removed. Two commonly used spirometry-based formulas to predict post-pneumonectomy FVC and FEV1 are shown below:
Kristersson and Olsen:
5
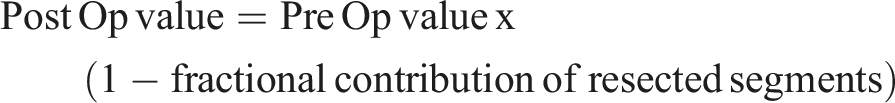
Juhl and Frost:
6

Smulders and colleagues 1 assessed the accuracy of the Kristersson/Olsen and Juhl/Frost equations to predict post-pneumonectomy lung function in 32 patients >2 years after undergoing pneumonectomy. While they found that both equations demonstrated good correlation between predicted and actual post-pneumonectomy FVC and FEV1, the Kristersson/Olsen equations were judged to be superior with higher correlation coefficients than the Juhl/Frost equations for both FVC (0.75 vs 0.62) and FEV1 (0.79 vs 0.69). The Juhl/Frost equations predicted a post-pneumonectomy FEV1 ∼300 mL smaller than the Kristersson/Olsen equations. Examination of longitudinal data showed an average yearly decline in FEV1 of 44 mL. Six subjects showed longitudinal improvements in FEV1 which the authors attributed to COPD treatment and hyperinflation of remaining alveoli.
An important compensatory adaptation following pneumonectomy is hyperinflation. Deslauriers et al 2 measured lung function in 100 subjects who had undergone pneumonectomy ≥5 years prior to entering the study. 68% of subjects had undergone an extended pneumonectomy (removal following previous ipsilateral partial resection), similar to the patient in this case. Also similar to the patient in this case, subjects who had undergone left pneumonectomy had smaller post-operative reductions in FVC (−25%) and FEV1 (−34%) compared to those who had undergone right pneumonectomy (−38% and −44%, respectively). In this cohort radiographic evidence of hyperinflation in the remaining lung was associated with a larger FEV1. As shown in Figure 2, the patient’s right lung extends well into the left side of the thoracic cavity and is noticeably larger than it appeared preoperatively (Figure 1).
A more intriguing possibility regarding compensatory adaptation to pneumonectomy is lung regeneration. Indeed, while mammals are not capable of growing new airways and major pulmonary blood vessels, in some species (e.g., rodents) there is an ability to generate new gas exchange tissue. 7 Theoretically, post-pneumonectomy hyperinflation resulting in lung stress and strain could trigger parenchymal lung growth since similar mechanical forces from a growing and expanding rib cage during the maturation process stimulates lung growth. 7 Takeda and colleagues 8 studied the effect of pneumonectomy on the remaining lung in foxhounds who underwent lung removal at 2 months of age. Exercise capacity, lung function, and histological examination was performed at the age of 18 months. Despite removal of an entire lung, exercise capacity and diffusion capacity (measured via the rebreathing method) had achieved normal status. Morphometric examination of the remaining lung demonstrated a greater than two-fold increase in endothelium, interstitial, and endothelial volume. Moreover, the parenchymal proliferation exceeded typical growth rates. A similar experiment in adult foxhounds following left pneumonectomy found that compensatory hyperinflation, alveolar enlargement, and thinning of the alveolar capillary membrane were solely responsible for increases in diffusing capacity; there was no morphometric evidence of lung regeneration. 9 However, the same group of researchers did find evidence of lung regeneration in adult foxhounds following right pneumonectomy. 10 Morphometric analysis 16 months after pneumonectomy showed growth of the endothelium, epithelium, interstitium, and an increase in capillary blood volume. With regard to the discordant findings in adult foxhounds, the researchers speculated that the removal of the larger right lung may have resulted in a greater adaptive response.
Evidence of lung growth following pneumonectomy in adult humans is limited. Butler et al 11 reported a case of apparent lung regeneration in a 33-year-old female patient who underwent right pneumonectomy for hilar adenocarcinoma. Three months after pneumonectomy, the patient’s FVC and FEV1 were reduced by 52% and 65% from the preoperative values, respectively. However, 15 years after pneumonectomy the patient’s FVC and FEV1 had increased 35% and 51%, respectively, defying expected age-related declines. Over the 15-year follow-up period, serial CT imaging demonstrated a near doubling of lung tissue volume. At year 15, after pneumonectomy MRI performed during a breath hold of hyperpolarized helium-3 suggested a 64% increase in the number of alveoli with reduced alveolar depth that the authors speculate could be additional evidence of new alveoli formation.
The patient whose case we describe demonstrated impressive compensatory adaptation to left pneumonectomy which is most likely due to right lung hyperinflation. Unfortunately, we cannot compare pre- and post-pneumonectomy data because spirometry was not performed prior to surgery. Lung volume testing before and after surgery was also not performed. Following pneumonectomy, this patient attained a high level of physical fitness, regularly running, biking, and hiking, and denied any dyspnea-related limitations. While the patient’s regular and vigorous physical activities could theoretically provide enough mechanical force in her lung to stimulate some lung regeneration, such an assertion would only be conjecture. Moreover, the spirometry test and chest x-ray we report were performed only 8 months after surgery. The impressive increases in lung size and function partially attributed to lung regeneration by Butler et al 11 were performed 15 years after pneumonectomy. Another factor that may have contributed to the patient’s normal vital capacity after pneumonectomy is having above average lung capacity ante morbum. The patient did have PFTs 3 months prior to the left upper lobe wedge resection for chondroid hamartomas at age 35. At that time, the total lung capacity was slightly above the mean (z-score 0.24) and the FVC was at the mean (z-score −0.01).3,12
As noted above, the FEV1/FVC was at the lower limit of normal which could be attributed to her diagnosis of asthma. However, at the time of testing the patient’s asthma was well controlled, she regularly exercised without respiratory symptom limitation, and she showed no response to bronchodilator. The most likely reason for the FEV1/FVC being at the lower limit of normal is a consequence of the 3.27 L vital capacity having to be exhaled forcefully through a bronchial tree with reduced cross sectional area, which could be described as acquired dysanapsis. 13
Dysanapsis is present when airway size and lung size are not proportional. There are many forms of dysanapsis including low dysanapsis (large lungs-normal airways; large lungs-small airways; normal sized lungs-small airways) and high dysanapsis (small lungs-normal airways).13,14 While some degree of mild dysanapsis is expected among a population of healthy subjects, significant dysanapsis during the period of lung growth and adulthood is associated with a higher risk of asthma and COPD. 13
Spirometry data has been used to identify the presence of dysanapsis. Shimada and colleagues used CT imaging to identify and quantify dysanapsis using the airway-to-lung ratio. 15 Subjects were separated into quartiles based on their airway-to-lung ratio, and spirometric indices from each group were compared. Women in the quartile with the most dysanapsis had a mean FEF25-75%/FVC of 0.78. FEF25-75%/FVC has been used as a measure of small airways disease and to quantify dysanapsis. Eight months after pneumonectomy, this patient had an FEF25-75%/FVC of 0.45, even lower than the mean value of female subjects with the most dysanapsis in the study by Shimada et al.16 We would characterize this type of altered airway-to-lung ratio as a form of acquired dysanapsis, a consequence of pneumonectomy where a preserved vital capacity due to hyperinflation is exhaled forcefully through a bronchial tree with reduced cross-sectional area. 13 Additional studies such as oscillometry and specific airway conductance might be helpful in determining if the FEV1/FVC at the lower limit of normal was due to dysanapsis. This case illustrates the remarkable adaptability of the respiratory systems to pneumonectomy.
Footnotes
Author contributions
Manuscript development and review: JH, DK, and GR.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclosures
JH: Consultant, Morgan Scientific, Inc.; DK: MGC Diagnostics, Inc., UpToDate, Inc., and Elsevier, Inc.; and GR: None.
