Abstract
Background:
The INPULSIS trials revealed that nintedanib reduced the decline in lung function in patients with idiopathic pulmonary fibrosis. We aimed to evaluate the efficacy and safety of nintedanib in Japanese idiopathic pulmonary fibrosis patients in real-world settings.
Method:
Medical records of idiopathic pulmonary fibrosis patients, who received treatment with nintedanib in five institutions between July 2015 and June 2017, were reviewed. Patients with % forced vital capacity ⩾50% and % predicted diffusing capacity of the lung carbon monoxide ⩾30% were classified as the moderate group and those with more impaired lung functions as the severe group.
Result:
Among 158 patients analyzed, 132 (84.6%) were classified as the moderate group and 26 (15.4%) as the severe group. In the moderate group, changes in forced vital capacity in 12 months were significantly different between before and after nintedanib administration (−253 ± 163 vs −125 ± 235 mL;
Conclusion:
In real-world practice, nintedanib showed comparable efficacy to those observed in previous trials. In the severe group, the efficacy of nintedanib might be limited.
Keywords
Background
Idiopathic pulmonary fibrosis (IPF) is a debilitating, progressive, fibrosing lung disease caused by unknown etiology. 1 The prognosis of IPF is poor, with median overall survival ranging reported 2–4 years after diagnosis.2,3 Over the past 5 years, the introduction of novel antifibrotic agents in the form of pirfenidone and nintedanib has changed the management of IPF and provided improvement of clinical outcome. Several fibrotic mediators, including fibroblast growth factors (FGFs), platelet-derived growth factors (PDGFs), and transforming growth factor beta, are associated with the pathogenesis of IPF.4,5 Nintedanib is a potent, oral, small-molecule tyrosine kinase inhibitor that blocks the kinase activity of a variety of receptors, such as vascular endothelial growth factor (VEGF) receptor, 6 PDGF receptor, and FGF receptor. 7 It inhibits the intracellular signaling pathways that are crucial for the proliferation, migration, and transformation of lung fibroblasts.7,8 A phase II TOMORROW trial and two phase III trials (INPULSIS-1 and -2) have shown significant reductions in the rate of decline in forced vital capacity (FVC) and the incidence of acute exacerbation over a 52-week treatment period in IPF patients treated with nintedanib.9,10 In Japan, nintedanib was launched in 2015. In these clinical trials, however, patients, who had %FVC < 50% and % predicted diffusing capacity of the lungs for carbon monoxide (DLCO) < 30%, were excluded, which makes the efficacy of nintedanib in those with severe IPF unclear. Moreover, the number of IPF patients who received long-term oxygen therapy (LTOT) was less than 10% of the study subjects, making the efficacy and safety of nintedanib in such patients unclear. In this study, we aimed to evaluate the efficacy and safety of nintedanib for Japanese IPF patients in real-world practice.
Patients and methods
Study design
Our study was performed in five institutions in Aomori, Japan. This study was conducted between July 2015 and June 2017. This study is a retrospective study, and we did not set a clear number of cases. IPF patients who have received nintedanib were included. We reviewed the medical records of all the patients who were treated with nintedanib at the participating hospitals. We made a diagnosis of IPF based on the ATS/ERS/JRS/ALAT IPF guideline of 2011. 1 Patients with %FVC ⩾ 50% and %DLCO ⩾ 30% were classified as the moderate group and those with %FVC < 50% or %DLCO < 30% as the severe group. The study was performed according to the protocol approved by the ethics committee of each participating hospital. The ethics committee of Hirosaki University approved this study (approval number: 2018-1203). Because this was a retrospective cohort study, informed consent was waived. Opt out was posted on our website.
Evaluation and statistical analysis
The clinical data were expressed as mean ± standard deviation (SD). We compared the baseline characteristics, lung function data including the rate of decline in FVC, frequency of adverse events, and survival rate between the two groups. Acute exacerbations of IPF are defined as episodes of respiratory worsening that occur within 1 month and cannot be explained by other reasons such as pneumonia, pulmonary embolism, pneumothorax, or cardiac failure. We used two severity criteria of IPF, the gender, age, physiology (GAP) staging.
11
The GAP score was calculated by the following status: gender (0: female; 1: male), age (0: ⩽60; 1: 61–65; 2: >65), %FVC (0: >75%; 1: 50%–75%; 2: <50%), and %DLCO (0: >55%; 1: 36%–55%; 2: <35%; 3: cannot perform). The total point score is used to classify patients as stage I: 0–3 points, stage II: 4–5 points, or stage III: 6–8 points. All statistical analyses were performed using JMP 13 (SAS Institute, Cary, NC). Overall survival (OS) and the ratio of acute exacerbation were estimated using the Kaplan–Meier method. The toxicity profiles were graded by Common Terminology Criteria for Adverse Events v5.0 (CTCAE). A statistical significance was considered when
Results
Patient characteristics
A total of 177 patients with IPF were administrated nintedanib at the participating hospitals between July 2015 and June 2017. Nineteen patients were excluded from analysis due to the lack of initial spirometry data. Finally, a total of 158 patients were subjected to analysis (Figure 1). In 45 patients, the rate of FVC decline over 12 months (ΔFVC) was evaluated both before and after the introduction of nintedanib. One hundred thirty-two patients (84.6%) were classified as the moderate group and 26 (15.4%) as the severe group. Patient demographics and baseline characteristics were summarized in Table 1. There were no significant differences between the two groups in age, height, smoking status, and serum levels of lactate dehydrogenase (LDH) and sialylated carbohydrate antigen KL-6 (KL-6). On the other hand, there were significant differences in body weight (59.3 ± 11.6 kg in the moderate group vs 52.2 ± 11.8 kg in the severe group;
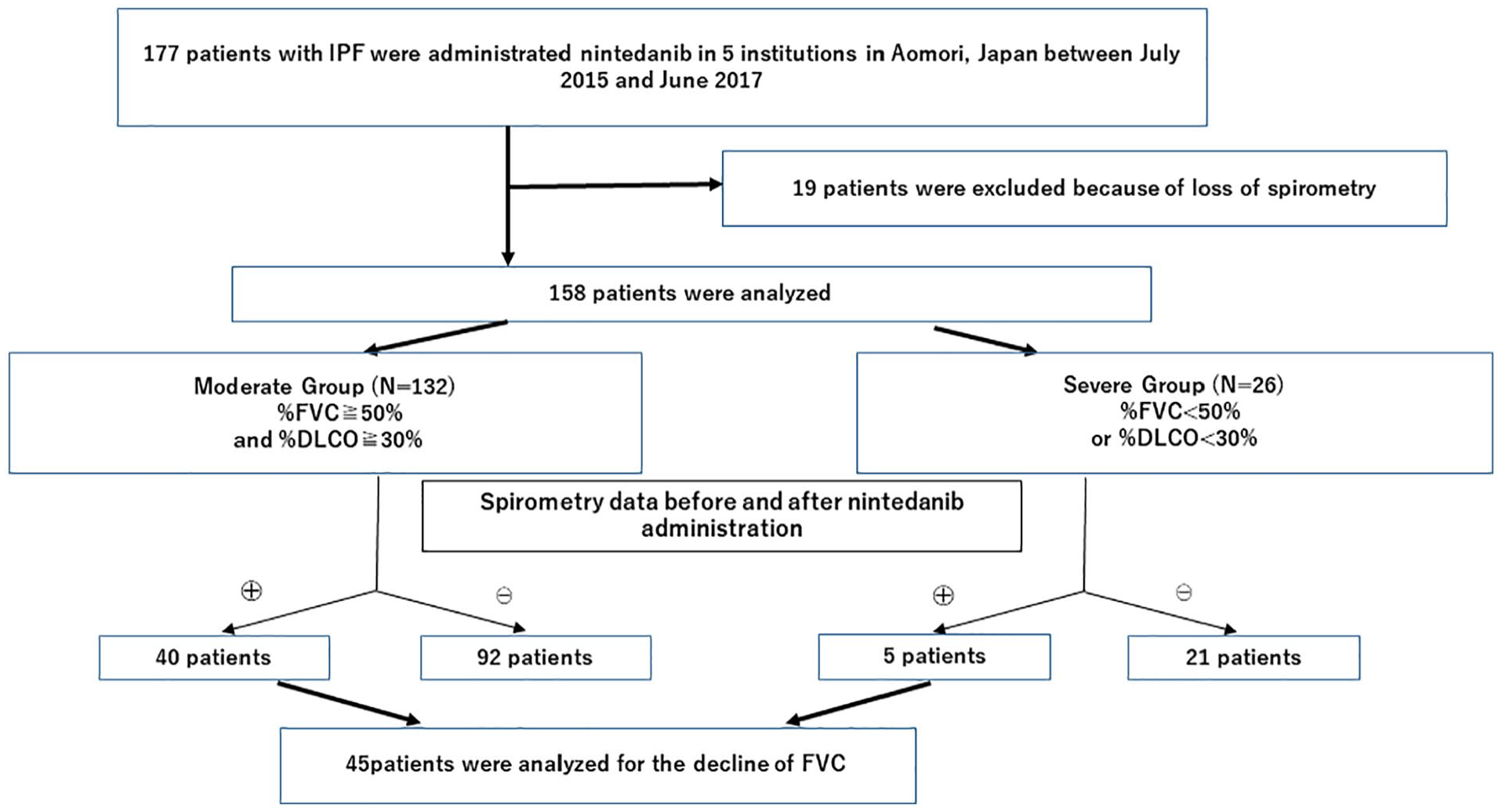
Enrollment of the study subjects.
Baseline Characteristics of Total 158 Patients.

COPD: chronic obstructive pulmonary disease; BMI: body mass index; DLCO: diffusing capacity of the lung carbon monoxide; FVC: forced vital capacity; GAP: gender, age, physiology; KL-6: sialylated carbohydrate antigen KL-6; LDH: lactate dehydrogenase; LTOT: long-term oxygen therapy.
Rate of decline in FVC
ΔFVC was calculated from spirometry data conducted before and 12 months after the introduction of nintedanib. In the moderate group, ΔFVC was significantly different between before and after the introduction of nintedanib (−253.2 ± 163.2 vs −124.5 ± 235.3 mL;
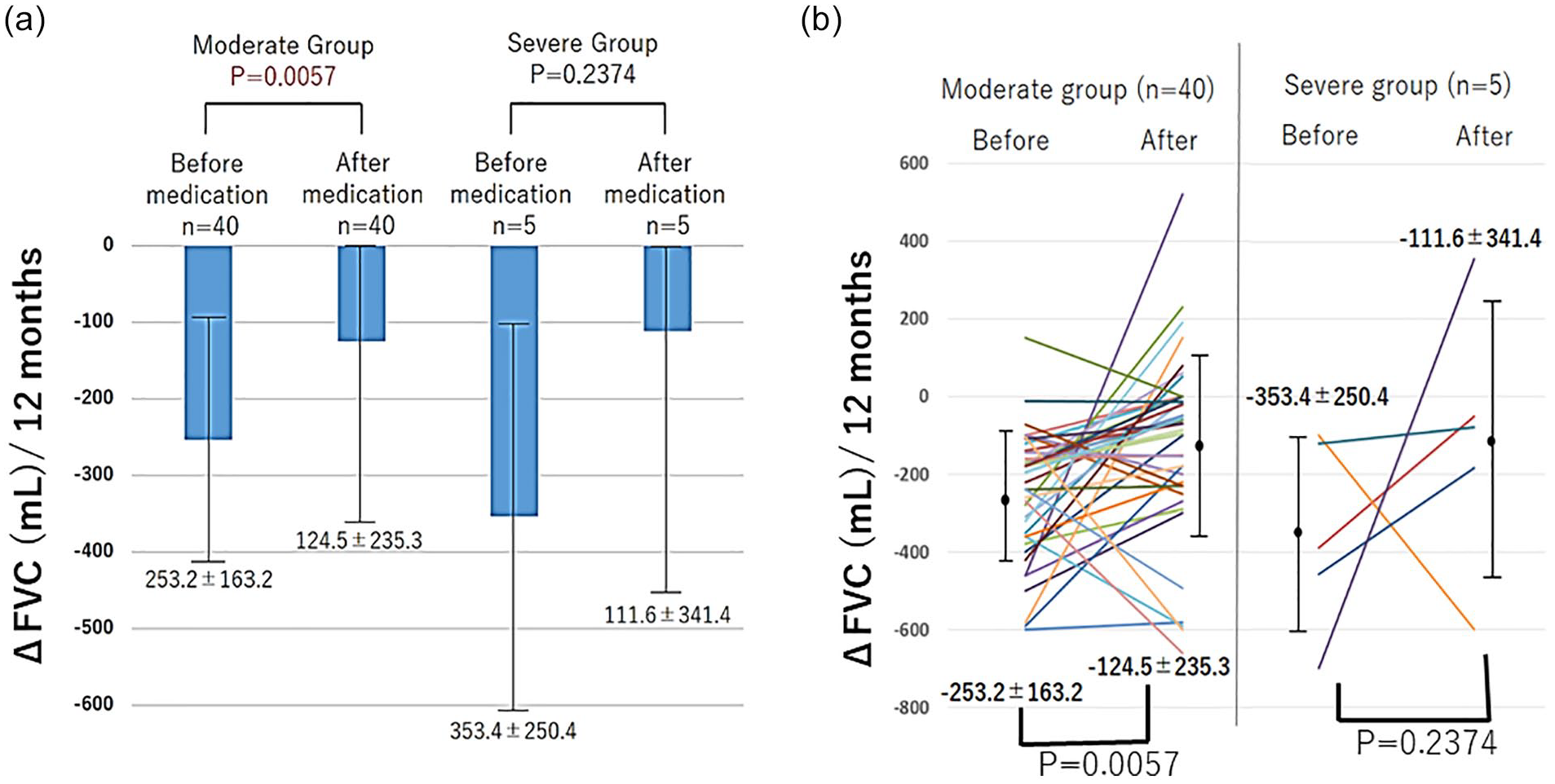
Changes in FVC in 12 months (ΔFVC) before and after treatment. In the moderate group, ΔFVC was significantly different between before and after the introduction of nintedanib (−253.2 ± 163.2 vs −124.5 ± 235.3;
Survival outcome
The OS was 17.2 months in the moderate group and 10.1 months in the severe group, which was significantly different (

Kaplan–Meier analysis of overall survival and acute exacerbation. The overall survival in the moderate and severe groups were 17.2 months (95% CI: 15.8–21.3) and 10.1 months (95% CI: 7.0– 18.1), respectively. There was a significant difference in overall survival between the two groups (
Acute exacerbation
The incidence of acute exacerbation in the severe group was significantly higher than in the moderate group (Figure 3(b)). During 52 weeks after the introduction of nintedanib, acute exacerbation occurred in 12.3% of the moderate group and 27.3% of the severe group (

Incidence rate of acute exacerbation. Overall incidence rate of acute exacerbation in both the moderate and the severe groups. The incidence rate of acute exacerbation was 18.9% in the moderate group (mean follow-up time: 563 days) and 30.8% in the severe group (mean follow-up time: 550 days) (a). The rate of acute exacerbation in patients who received LTOT and severe group was 44.4% (b).
Association of Baseline Patient Parameters With Acute Exacerbations.

BMI: body mass index; DLCO: diffusing capacity of the lung carbon monoxide; FVC: forced vital capacity; GAP: gender, age, physiology; KL-6: sialylated carbohydrate antigen KL-6; LDH: lactate dehydrogenase; LTOT: long-term oxygen therapy.
Safety
Table 3 shows the adverse events (AEs) observed during the treatment with nintedanib. Any AEs were observed in 136 patients (86.1%). AEs of CTCAE grade 3 and 4 were observed in 17 patients (10.8%). The most common AEs were diarrhea, which was observed in 76 patients (57.8%) in the moderate group and 14 (53.8%) in the severe group, followed by liver disorder (39.4% in the moderate group and 46.2% in the severe group), anorexia (34.8% in the moderate group and 26.9% in the severe group), and nausea (12.9% in the moderate group and 3.8% in the severe group). Malaise was significantly more frequent in the severe group than in the moderate group (8.3% in the moderate group and 23.1% in the severe group;
Toxicity in Patients Treated With Nintedanib.
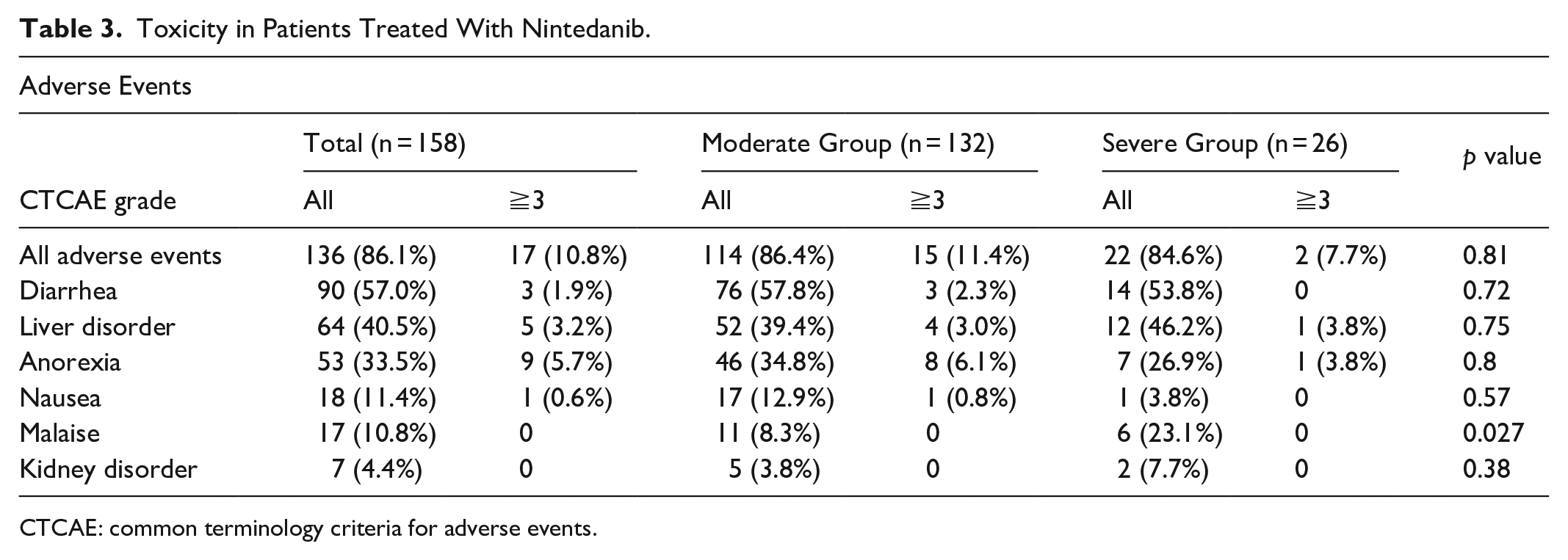
CTCAE: common terminology criteria for adverse events.
Twenty-eight patients needed to reduce the dose of nintedanib. Liver disorder was the most common reason for dose reduction of nintedanib (40.5%).
The discontinuation rate was 47.5% (75/158) in all patients. The reasons for discontinuation were toxicity (37.3%), death due to disease progression (24.0%), acute exacerbation (16.0%), insufficient efficacy (5.3%), and others (17.3%). Discontinuation rates were not significantly different between the moderate group and severe group (47.4% vs 50%;
Discussion
The present study is one of the largest real-world studies that evaluated the efficacy and safety of nintedanib for Japanese IPF patients including those who did not meet the inclusion criteria of the clinical trials. Moreover, this is the first report that showed the rate of acute exacerbation in IPF patients receiving LTOT during treatment with nintedanib.
In this study, ΔFVC in 12 months was significantly reduced after nintedanib administration in patients with moderate IPF, which was comparable with the data shown in phase III trials. In INPULSIS-1 and -2 trials, nintedanib reduced the rate of decline in FVC by 125.3 and 93.7 mL/year, respectively. 10 In addition, ΔFVC was reduced after the introduction of nintedanib in four of five patients with severe IPF although the difference was not statistically significant. In the recent INPULSIS-ON study, patients were classified according to the baseline FVC % predicted (⩽50% or >50%). It was revealed that there were no significant differences in the decline of FVC between the two subgroups. 12 However, the number of patients with FVC ⩽ 50% predicted was only 24, which made the efficacy of nintedanib in patients with more advanced IPF unclear. Recently, Yoon et al. reported a prospective study that evaluated the efficacy and safety of nintedanib in patients with advanced and nonadvanced IPF. 13 A significant reduction of FVC decline rate was observed even in those with advanced disease (n = 45). In another retrospective study, Abe et al. reported the effectiveness of nintedanib in patients with severe IPF. 14 A total of 51 patients received nintedanib (34 with mild-to-moderate IPF and 17 with severe IPF). Because ∆FVC was significantly reduced after the introduction of nintedanib, they concluded that the efficacy of nintedanib might be regardless of the disease severity. However, the frequency of side effects tended to be higher and the prognosis was significantly worse in the severe group. 13 Moreover, Tzouvelekis et al. reported that nintedanib was as effective in patients with %FVC < 50% as in those with %FVC > 50%. 15
In the present study, the annual rate of acute exacerbation during 12 months in patients with moderate IPF was 12.3%, which was higher than the numbers shown in phase III trials. We considered the following reasons for the higher incidence rate of acute exacerbation. First, in our study, the number of patients who had been receiving medical treatment including pirfenidone was as high as 37.1% in the moderate group. Second, the number of patients who received LTOT was also high in the moderate group. These factors may have affected the frequency of acute exacerbations. In fact, the annual incidence of acute exacerbation was 27.3% in the severe group and 44.4% in those receiving LTOT, which was considerably high. Acute exacerbation is the leading cause of death of IPF patients in Japan, and its incidence has been reported to be 5%–19%.3,16,17 The pooled analysis of the data from the INPULSIS trials showed that a lower percentage of the subjects treated with nintedanib experienced acute exacerbations in a 52-week period compared with those in the placebo group. 17 Therefore, we considered that there might be some effective cases even in those with severe IPF, although the patients on LTOT might receive little benefit from the treatment.
The incidence of AEs in our study was similar to that reported in the INPULSIS studies. 9 Incident rates for diarrhea, nausea, and malaise were comparable with those observed in the trials. Yoon et al. reported that the frequencies of diarrhea and anorexia were 50.0% and 45.4%, respectively, which were similar to those in our study. 13 On the other hand, we observed liver disorder in 40.5% of the patients, which was high than that shown in the INPULSIS trials. Azuma et al. reported the Japanese subset analysis of the INPULSIS trial, showing that abnormal hepatic function and increased liver enzymes were more frequent in the Japanese subjects compared with overall population.18,19 Ikeda et al. 20 reported an association between BMI and liver dysfunction due to nintedanib, which indicated that the dose of nintedanib should be adjusted according to BMI of patients. In our study, patients with severe IPF had significantly lower body weight and BMI than those with moderate disease. In addition, they were more likely to have received previous treatment, such as corticosteroid and immunosuppressants, which might contribute to the high incidence of liver disorders. In comparison to the moderate group, the severe group tended to experience more side effects although the difference was not statistically significant except for malaise.
There were some big limitations in our study. First, as this study was retrospective, a selection bias might exist concerning the introduction of nintedanib for the patients with IPF. Sample size/power analysis was not performed. This effect may result in an inadequate analysis of the severe group. Second, the number of patients in the severe group was too small to conclude whether nintedanib is effective for these patients. Pulmonary function assessment is not inadequate due to more acute exacerbations and death events in the severe group. Third, ΔFVC could be compared before and after the introduction of nintedanib only in a limited number of patients. Fourth, the follow-up time was short in our study. The discontinuation rate of nintedanib was as high as 47.5% and almost 20% had the dose reduced which makes the assessment of the efficacy challenging in this study.
Conclusion
In this real-world study, we observed ∆FVC similar to those shown in the INPULSIS trials. On the contrary, the rate of acute exacerbation was higher, especially in the patients with severe IPF. Further clinical data are required to evaluate the clinical benefit of nintedanib, especially in patients with severe IPF and those receiving LTOT.
Supplemental Material
sj-tif-1-smo-10.1177_20503121211023357 – Supplemental material for The efficacy of nintedanib in 158 patients with idiopathic pulmonary fibrosis in real-world settings: A multicenter retrospective study
Supplemental material, sj-tif-1-smo-10.1177_20503121211023357 for The efficacy of nintedanib in 158 patients with idiopathic pulmonary fibrosis in real-world settings: A multicenter retrospective study by Masaki Dobashi, Hisashi Tanaka, Kageaki Taima, Masamichi Itoga, Yoshiko Ishioka, Toshihiro Shiratori, Fumihiko Okumura, Chiori Tabe, Yoshihito Tanaka, Takeshi Morimoto, Yukihiro Hasegawa, Hideo Yasugahira, Koichi Okudera, Shingo Takanashi and Sadatomo Tasaka in SAGE Open Medicine
Supplemental Material
sj-tif-2-smo-10.1177_20503121211023357 – Supplemental material for The efficacy of nintedanib in 158 patients with idiopathic pulmonary fibrosis in real-world settings: A multicenter retrospective study
Supplemental material, sj-tif-2-smo-10.1177_20503121211023357 for The efficacy of nintedanib in 158 patients with idiopathic pulmonary fibrosis in real-world settings: A multicenter retrospective study by Masaki Dobashi, Hisashi Tanaka, Kageaki Taima, Masamichi Itoga, Yoshiko Ishioka, Toshihiro Shiratori, Fumihiko Okumura, Chiori Tabe, Yoshihito Tanaka, Takeshi Morimoto, Yukihiro Hasegawa, Hideo Yasugahira, Koichi Okudera, Shingo Takanashi and Sadatomo Tasaka in SAGE Open Medicine
Footnotes
Acknowledgements
The authors are grateful to the patients and their family and medical staff who contributed to this study.
Author contributions
M.D. prepared the article and made contributions to acquisition of data. H.T. made a concept of this study; H.T. and K.T. conducted statistical analysis. M.I., Y.I., T.S., F.O., and C.T. treated and observed patients in Hirosaki University; Y.T. treated and observed patients in Hirosaki National Hospital; T.M. and Y.H. treated and observed patients in Aomori Prefectural Central Hospital; H.Y. treated and observed patients in Hachinohe City Hospital; K.O. treated and observed patients in Hirosaki Central Hospital; Sh.T. evaluated the CT image of the patients included this study. S.T. advised and revised statistical analysis critically for important intellectual content and reviewed the article. All authors read and approved the final article.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from Hirosaki University Research Ethics Review Committee prior to the study (approval number: 2018-1203).
Ethical approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study was approved by the ethics committee of Hirosaki University (approval number: 2018-1203). Because this was a retrospective cohort study, informed consent was waived. Opt out was done on the website of Hirosaki University.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was not sought for the present study because this was a retrospective cohort study, and informed consent was waived. Opt out was posted on the website of Hirosaki University.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
