Abstract
HIV remains a global public health issue; Uganda has nearly 7% of infants born to HIV positive mothers getting infected despite initiation on Nevirapine prophylaxis against HIV transmission among HIV-exposed infants. There is paucity of data regarding adherence to NVP in Western Uganda. This study aimed at assessing the predictors of adherence to nevirapine prophylaxis among HIV -exposed infants aged 0 to 6 months. A cross-sectional study was done among 200 HIV-exposed infants aged 0 to 6 months. Participants were consecutively recruited. Data was analyzed using SPSS version 26, logistic regression was used to infer predictors of Nevirapine adherence. Adherence to Nevirapine prophylaxis was at 95.0% (CI: 89-98). Factors associated with adherence included; disclosure of HIV status [aOR = 0.083; 95% CI: 0.007-0.92, P < .005] and having more than 1 child [aOR = 0.08; 95% CI: 0.007-0.82, P < .005]. Adherence to Nevirapine was suboptimal (95.0%) in comparison to the national set goals of 100% adherence by 2030.
Introduction
Globally, Human Immunodeficiency Virus (HIV) contributes to 40.4 million deaths. 1 In the Sub-Saharan Africa, HIV remains one of leading causes of death 2 and in 2020 around 1.5 million children were living with HIV/AIDS with approximately 130 000 new infections among children.3,4 Annually, over half a million new born are infected with HIV. 5 Majority of these new HIV infections in newborns occur in Eastern and Southern Africa. 6 Almost half of infants infected with HIV after birth and a quarter of infants infected through breastfeeding do not live to their second birthday which is higher than less than 5% infant mortality in HIV-exposed uninfected infants. 4 The risk reduction in vertical transmission of HIV/AIDS vary from 49% in developing countries to 100% in developed countries. 7 According to Ministry of Health, Uganda had an estimate of 5955 infants of 97 222 born to HIV-positive mothers, getting infected, hence Uganda ranked fourth due to high number of new infections in the year 2021. 8 Recent evidence indicates that the estimated mother to child transmission of HIV in Uganda stands at 1.8% during pregnancy and delivery, and 2.9% during breastfeeding. 9 The annual prevalence of pediatric HIV of 466 per 100 000 live births is still higher than the target of less than 50 per 100 000 live births despite the use of Nevirapine prophylaxis in the HIV Exposed Infants (HEI). 10 With access to effective HIV prevention, diagnosis, treatment and care, the rates of new infections can be lowered in all age groups. 11 Infant NVP prophylaxis has proven efficacy and effectiveness in prevention of vertical transmission of HIV, to eliminate mother-to-child transmission of HIV. 12 Nevirapine prophylaxis is key in reducing the MTCT of HIV in HEI mainly through vertical transmission to less than 5% with Current data shows that an HIV exposed infant is at risk of contracting HIV by 20% to 45% when no intervention has been done compared to 1 where interventions including Nevirapine prophylaxis have been. 13 Approximately 75% of HEI who miss Nevirapine prophylaxis acquire HIV in Uganda. 10 However, adherence to both maternal anti-retroviral therapy and infant prophylaxis would yield the prevention of mother-to-child transmission of HIV.9,14 optimal adherence.8,13
Elimination of mother to child transmission of EMTCT of HIV(EMTCT) is crucial if we are to achieve the Sustainable development goal of no new HIV infections among children by the year 2030. This study will provide a status update on the adherence to an infant’s NVP prophylaxis regimen and associated factors in Hoima City West, Western Uganda.
Methods
Study Design and Setting
A cross-sectional study design was employed. We used evidence from records drug refills and DNA/PCR results of dried blood samples for all infants. This study was conducted from 01st May to 31st June 2024 at Hoima Regional Referral Hospital (HRRH) in Western Uganda. 15 The study was conducted in ART clinic, the Young Child Clinic (YCC), Early Infant Diagnosis (EID) and Mother Baby care points of this hospital. HRRH has a bed capacity of 280, and mostly serves the neighboring rural and neighboring districts. It receives patients with limited financial resources.
Study Population
The study involved mothers of HIV exposed infants aged 0 to 6 months receiving their ART medications from HRRH.
Sample Size Determination
The Sample size was estimated using Yamane’s simplified formula (1967) for finite population size where the population size(N) was 430, which were the monthly projections of HEI at HRRH. We used a precision error (e) of 5% this resulted into 207 participants. Mothers of infants aged 0 to 6 months who were HIV exposed and had received Nevirapine prophylaxis at HRRR and were available during the days of data collection were included in the study. Mothers of children who required urgent medical care were not included in the study.
Data Collection Tools
Data were gathered using a researcher administered semi-structured questionnaire. The tool was developed by reviewing literature on similar studies on infant Nevirapine prophylaxis for HIV-exposed infants against HIV transmission. We pretested the questionnaire on 20 care givers of exposed children at Azur Health center IV, and made necessary corrections. The data collection tool was developed in English and translated to Runyoro, the major local language in the study area. All questions were written in English and translated in Runyoro by 2 different language experts to check for clarity and consistency
Study Variables
Adherence to NVP was calculated as total doses of NVP administered to the infant divided by age of infant in days expressed as a percentage (Doses of NVP/age of infant × 100]. An infant was also considered adherent if their DNA/PCR results from their respective dried blood samples (DBS) taken prior to or during the study were negative, the dependent variable was adherence to infant Nevirapine prophylaxis against HIV transmission. The independent variables included maternal age, education level, cultural beliefs, religious beliefs, place of delivery, influence of the media, family and friends, awareness campaigns, community leaders, reminders such as posters, awareness of the risks of not being adherence knowledge of HIV prevalence, attitude and norms toward adherence, information on HIV related mortality, knowledge of complications of HIV, cost and transportation to access the facility, shortages of NVP prophylaxis, misconceptions about NVP adherence, lack of access to the services, fear of NVP prophylaxis side effects, lack of time, knowledge gaps, cultural and religious beliefs, confidence in uptake of NVP syrup, adherence to the adherence schedule despite the barriers.
Data Quality Control
Data was collected through face-to-face interviews with the mother or caregiver of the infant using pretested interviewer administered questionnaire. Trained research assistants at a bachelor’s level were used to conduct these interviews in designated spaces at the ART clinic and Young Child Clinic (YCC). We used consecutive sampling method to select care givers who had come for immunization at the young child clinic, for ART refills and PCR testing who were willing to participate in the study. Participants were screened to select potential participants resulting into a sample size of 200 participants and thereafter, informed consent was sought from the study participants to conduct the study with interviews lasting between 15 and 30 minutes. We reviewed the questionnaire to ensure completeness and accuracy of responses given prior safe storage. Participants true names did not appear on the questionnaires.
Data Processing and Analysis
Data was entered using Epi Data version 3.0, cleaned using Microsoft excel prior final analysis using SPSS version 26 after it had been exported. Descriptive statistics were used to summarize demographic data. This was presented as frequencies and proportions for categorical data and as mean and standard deviation for discrete and continuous data. We used a logistic regression analysis at bivariate level to compare the predictors and the dependent variables. The model was assessed for goodness of fit using the Akaike Information Criteria (AIC) for logistic regression which fitted based on smaller values. A cut of P-value less than .2 at bivariate level was used, and the significant factors were taken to the multivariate level to adjust for confounding. A P-value of less than .05 was considered statistically significant at the multi variate level to determine the level of infant adherence and the predictors to adherence to infants NVP prophylaxis.
Ethical Considerations
Ethical approval of the study was obtained from the Lira University Research and ethics committee, Approval number (LUREC 2024-140) and thereafter, administrative clearance was obtained from the Hoima City. Principal Health Officer and the research ethics committee of Hoima Regional Referral Hospital where the study was conducted. Informed consent was sought from the study participants and were informed that they can withdraw from the study at any time.
Results
Sociodemographic, Obstetrics and Individual Characteristics of Participants
Only 200 respondents were recruited and participated giving a response rate of 97%. The mean age and SD of the infants were 3.96 ± 2.80 months, and almost a half of mothers who participated in the study were aged 21 to 29, 98 (49.0%). Almost all of the participants reported to have attended ANC whilst pregnant 190(95%) and delivered in a clinical setting 189 (94.5%). More than three quarters 155(77.5%) gave birth vaginally. Majority of the respondents had given to birth to more than 1 baby 157 (78.5%; Tables 1–3).
Socio-demographic Characteristics of the Study Participants (N = 200).

The bolded figures represent high frequencies and percentages.
Individual Characteristics of Participants.

Obstetric Characteristics of Participants.

Adherence to Nevirapine Prophylaxis
Majority of the participants were adherent to NVP prophylaxis were 189 (95%), CI (89-98).
Factors Associated with Adherence to Nevirapine Prophylaxis
The results from the binary logistic regression showed that employment status (aOR = 0.15; 95% CI: 0.02-1.16), P = .036, having given birth to more than 1 child (aOR = 0.21; 95% CI: 0.06-0.70) P = .006, ANC attendance during pregnancy (aOR = 0.11; 95% CI: 0.01-1.28) P = .033, place of delivery (aOR = 41.78; 95% CI: 3.46-504.8), P = .0001, mode of delivery (aOR = 0.22; 95% CI: 0.06-0.75), P = .009, babies born after HIV diagnosis (aOR = 0.21; 95% CI: 0.04-0.98), P = .030, disclosure of HIV status (aOR = 0.25; 95% CI: 0.07-0.84, P = .013 were significantly associated with adherence to NVP prophylaxis.
The multivariate analyses were conducted by entering all the plausible variables in the bivariate analyses as independent variables into a logistic regression model. After controlling forconfounding factors, those who had disclosed their HIV status (aOR = 0.08; 95% CI: 0.007-0.92) P < 0.005 and mothers who had more than 1 child (aOR = 0.076; 95% CI: 0.007-0.82) P < .005 were more likely to adhere to Nevirapine prophylaxis (Table 4).
Bivariable and Multivariable Analyses of Factors Associated with NVP Adherence.
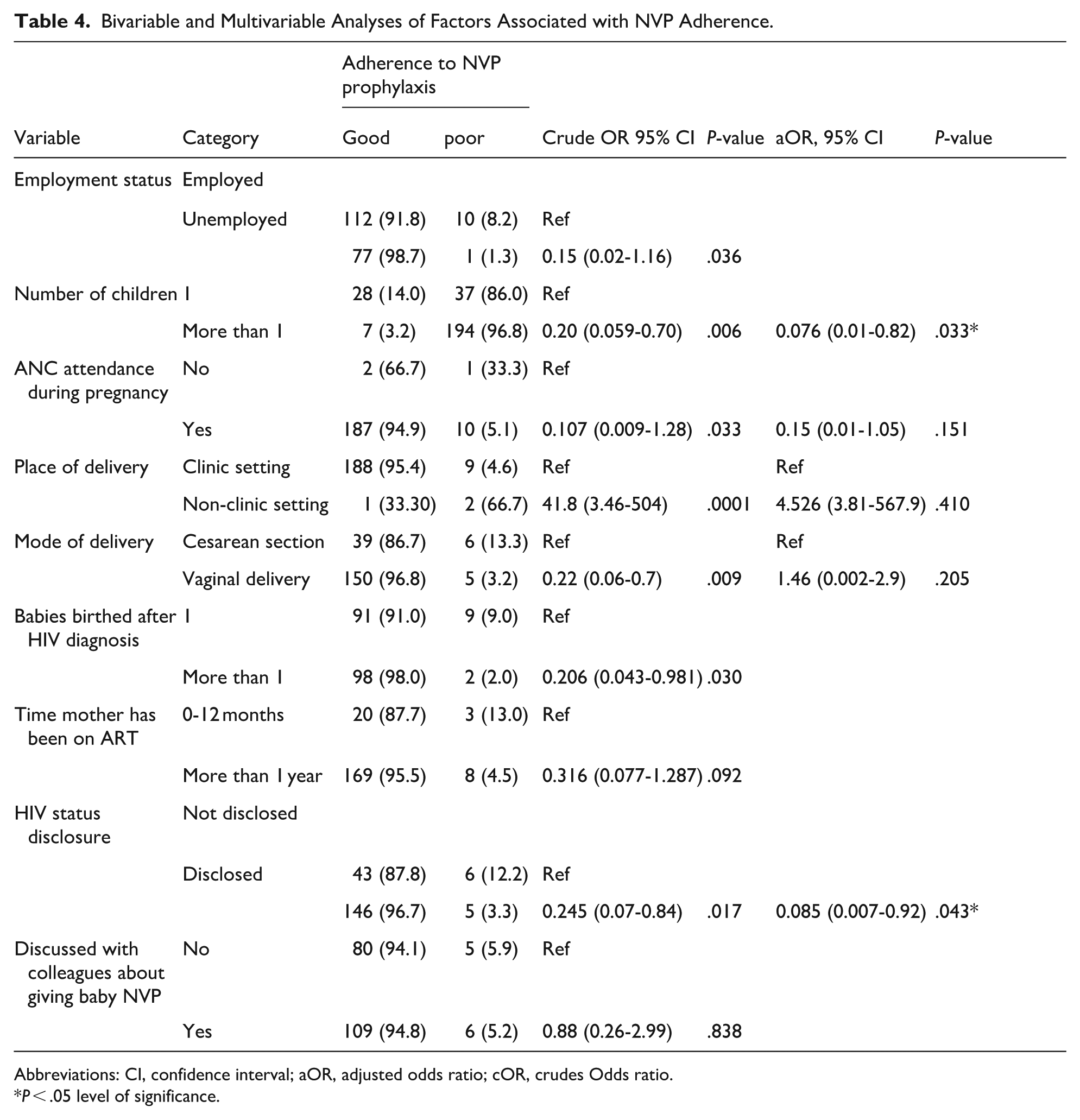
Abbreviations: CI, confidence interval; aOR, adjusted odds ratio; cOR, crudes Odds ratio.
P < .05 level of significance.
Discussion
There was suboptimal adherence to Nevirapine prophylaxis. Our study revealed 95% adherence to Infant prophylaxis, Adherence to which is lower that the target required by UNAIDS before 2030 16 The suboptimal levels of adherence to NVP prophylaxis in this study suggests possible negative impacts aimed at elimination of mother-to-child transmission without ultimate success in HRRH as recommended by UNAIDS 2030. The results of this study are related to those in studies previously conducted in rural settings of Uganda, 95.8%, 17 Ethiopia 18 adherence, Kenya 90.7%. 19 Contrary, studies done elsewhere in Lira regional Referral Hospital, Addis Ababa Ethiopia, and South Africa where adherence levels were at 85.2%, 83.1% and 74.5% respectively.14,20 The possible reason for this variation could be due to differences in the socio-demographics, the countries’ interventional and promotional policies towards MTCT polices. This implies that despite fair adherence, optimal adherence to NVP prophylaxis has not yet been achieved thus a need to strengthen linkage and care of HEI in the fight against MTCT of HIV to achieve the goal of UNAIDS that no new HIV infections among children by the year 2030.21,22
Infants of mothers who had disclosed their HIV status were more likely to adhere to NVP prophylaxis compared to their counterparts who had not disclosed their status, this could be due to support from the immediate partners, relatives and friends by reminding them to take their medications. The study result aligns with studies carried out by Napyo et al, 14 Menbere et al 3 that showed non-disclosure was a risk factor to non-adherence to NVP prophylaxis. This finding underscores the need to disclose the HIV status in order to avoid stigma and discrimination in order to improve self-esteem consequently contributing to adherence. Mothers who had more than 1 child were more likely to adhere to infant NVP prophylaxis compared to mothers with only 1 child, this could be attributed to the experience of the previous pregnancy. This contradicts with a study by Nwaiwu et al. 23 There is need for heightened strategies to help increase social support for mothers with HIE infants by the community.
Study Limitations and Strengths
The study evaluated adherence using the previously done DNA/PCR test, so mothers who were reporting self-adherence (verbal) was cross checked with results from the last DNA/PCR test. Results are limited by the cross-sectional design in that causal-effect inferences of the findings cannot be made.
Conclusion
The levels of adherence to infant Nevirapine prophylaxis in Hoima City West, Western Uganda were below the recommended UNAIDS and the Ugandan Ministry of health. The number of births and the ability to disclose the HIV status appear to have an impact on the caregiver’s ability to facilitate infant adherence to NVP prophylaxis. Ultimately, non-adherence to infant’s NVP prophylaxis could contribute to a rising burden of pediatric HIV/AIDS. Future strategies should aim at improving disclosure and measures for adherence to NVP prophylaxis.
Recommendations
We recommend Intensive adherence counseling on Nevirapine adherence among mothers, and concerted efforts of government, health workers to promote disclosure among HIV pregnant women to work toward to achieving the UNAIDS goal of no new pediatric HIV infections by 2030. Further research on evidence-based interventions to increase demand for adherence to NVP prophylaxis among HIV exposed infants aged 0 to 6 months should be implemented, through strategies such as client reminder and recall systems, outreach clinics and addressing concerns about NVP effectiveness and safety.
Footnotes
Acknowledgements
We want to thank the participants for their precious time, the administration of Hoima Regional Referral Hospital and the research assistants who collected the data.
Ethical Considerations
Ethical approval of the study was obtained from the Lira University Research and Ethics Committee, Approval number LUREC 2024-140 and thereafter, administrative clearance was obtained from the Hoima City Principal Health Officer and the research ethics committee of Hoima Regional Referral Hospital where the study was conducted.
Consent to Participate
Informed Consent was sought from the parents or caregivers. All the participants voluntarily participated in the study and also were able to withdraw at any time if they felt uncomfortable.
Consent for Publication
Not applicable.
Author Contributions
PA conceptualized the study, designed the study and wrote the manuscript, MT conceptualized the study, designed the study and collected data and wrote the first version of the manuscript, SU conceptualized the study, designed the study, analyzed the data and revised the manuscript, MA analyzed the data and wrote the manuscript, DFA analyzed the data and wrote the manuscript, PA collected the data and revised the manuscript, GW analyzed data and revised the manuscript, and BN supervised the study, designed the study wrote and reviewed the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during this study are available from the corresponding author upon reasonable request.
