Abstract
Objectives:
To compare the mini-midline catheters (Mini-MCs) and peripheral intravenous catheters (PIVCs) in hospitalized children with digestive system diseases.
Methods:
In this study, children received either an 8-cm Mini-MC or a 24-gauge PIVC based on patient or guardian preference. Catheter outcomes, including dwell time, complications, and unplanned removal, were monitored during the indwelling period.
Results:
Sixty-four children received Mini-MCs and 61 received PIVCs. Median dwell time was significantly longer in the Mini-MC group (94 hours; Z = −4.37, P < .01). Kaplan-Meier analysis showed longer catheter survival for Mini-MCs (χ2 = 29.58, P < .001). The PIVC group had higher overall complication rates (χ2 = 8.23, P < .01) and more unplanned removals (χ2 = 9.30, P < .01).
Conclusion:
Mini-MCs were associated with longer dwell time, fewer complications, and reduced unplanned removal compared with PIVCs, and may be a suitable option for short-term intravenous therapy in this population.
Keywords
Introduction
Intravenous therapy is one of the most commonly used procedures in pediatric care and offers several advantages, including ease of administration, rapid drug delivery, and prompt therapeutic effects, making it essential in the management of pediatric diseases. 1 Children hospitalized with digestive system diseases frequently present with symptoms such as vomiting and diarrhea, which can lead to dehydration and electrolyte imbalances. In clinical practice, these conditions often require timely intravenous therapy to restore fluid balance and correct electrolyte disturbances. 2 Peripheral intravenous access is currently the most widely used method for short-term intravenous therapy in this population, and peripheral intravenous catheters (PIVCs) have long been the standard choice in clinical practice. 3 However, previous studies have reported a mean dwell time of only 50.1 hours for PIVCs in hospitalized children, 4 often necessitating the placement of 2 or more catheters during a single course of intravenous therapy. 5 Repeated venipunctures not only increase pain and anxiety in children but also contribute to higher healthcare costs and reduced family satisfaction with nursing care.6,7 In recent years, mini-midline catheters (Mini-MCs) have attracted increasing attention as an alternative form of peripheral venous access. Characterized by longer dwell times and lower complication rates, Mini-MCs have been gradually introduced into pediatric intravenous therapy.8,9
Mini-MCs also referred to long peripheral catheters (LPC), are a type of peripheral intravenous access device. They are intermediate in length between PIVCs (~3.5-5.2 cm) and standard midline catheters (15-25 cm). Typically measuring 6 to 15 cm in length, Mini-MCs are made of polyurethane and are usually inserted into superficial veins of the forearm or deeper veins of the mid-upper arm, with the catheter tip not extending beyond the axillary vein. 10 Some studies have recommended the use of Mini-MCs for patients requiring intravenous therapy for 1 to 4 weeks. 7 These catheters are widely used in adult patients, particularly those who are critically ill2,10,11 or have difficult peripheral venous access12 -14 and have demonstrated favorable clinical outcomes.
Qin et al 15 conducted a randomized controlled trial evaluating the use of Mini-MCs for intravenous therapy in hospitalized children and neonates. Compared with PIVCs, Mini-MCs were associated with a higher first-attempt puncture success rate (66.7% vs 19.4%), longer mean dwell time (4.7 vs 3.5 days), and a higher rate of treatment completion with a single catheter (80.6% vs 38.9%) in postoperative pediatric patients. Similarly, Romitti et al 16 reported that Mini-MCs were more suitable than PIVCs for neonates requiring intravenous therapy lasting longer than 3 days. A systematic review examining the use of Mini-MCs in hospitalized children reported median dwell times ranging from 5 to 14 days and treatment completion rates between 20% and 86%, with dislodgement, occlusion, and infiltration identified as the most common complications. 17 However, most existing evidence originates from studies conducted in the United States and Australia,3,6,17,18 with limited data available from Asian populations.
Therefore, this study conducted in China compares the clinical application of Mini-MCs and PIVCs in hospitalized children with digestive system diseases, thereby expanding the evidence base for Mini-MCs use in pediatric populations and clinical contexts. The findings may provide practical and scientific guidance for pediatric healthcare professionals when selecting appropriate peripheral intravenous access devices.
Materials and Methods
Study Design and Setting
This prospective observational cohort study was conducted between September and October 2024 in the pediatric ward of a tertiary grade A general hospital in China. The ward has 36 beds and admits more than 2000 pediatric patients annually, primarily children with digestive system diseases (~1200 cases/year). Participants were allocated to either the Mini-MCs group or the PIVCs group based on the voluntary choice of the child and/or their guardian.
Participant Recruitment and Selection
This study employed a prospective, consecutive enrollment design. From September to October 2024, all children admitted to the pediatric ward with a primary diagnosis of digestive system disease and an expected need for continuous intravenous therapy for more than 48 hours were screened for eligibility at admission.
Eligible children and their legal guardians were provided with informed consent by trained research nurses. The consent process included information on the study objectives, the characteristics of the 2 vascular access devices (Mini-MCs and PIVCs), their potential benefits and risks, the patient/guardian-based group allocation approach, and the right to withdraw from the study at any time. Written informed consent was obtained from the guardians prior to enrollment.
As this preliminary observational cohort study was designed to describe and compare the use of 2 catheter types in a real-world pediatric setting, no formal sample size calculation was performed. All eligible and consenting children were consecutively enrolled during the 2-month study period to obtain preliminary and representative clinical data. Participants were allocated to either the Mini-MCs or PIVCs group according to the preference of the child and/or their guardian, reflecting routine clinical practice. Inclusion criteria were: hospitalized children aged 4 to 16 years with digestive system diseases; an expected duration of intravenous therapy ≥48 hours and <14 days; and written informed consent from both the child and their legal guardian. Exclusion criteria included: pre-existing intravenous access (eg, peripherally inserted central catheters); known allergy to catheter materials; scars, prior vascular surgery, or a history of thrombosis at the intended insertion site; and communication disorders. Discontinuation criteria included withdrawal from the study at the request of the child or guardian due to discomfort during catheter insertion or the indwelling period.
Data Collection
Before catheter insertion, the research team recorded the basic information of hospitalized children, such as age, gender, BMI, diagnosis, infusion duration, and the type of fluids to be infused. During catheter insertion, the team documented the catheter device used, insertion site, punctured vein, operation time, and pain assessment during puncture. Operation time was defined as the time from the beginning of preparation until the catheter was secured after puncture, measured in seconds. Pain assessment was conducted using the internationally recognized Wong-Baker FACES Pain Rating Scale (FACES), 19 assessments were performed once at the time of catheter insertion and daily during the indwelling period. Catheter dwell time defined as the duration from the end of the puncture until catheter removal, was measured in hours. Complications were recorded, including catheter occlusion, phlebitis, infiltration, and catheter-related infection. Catheter occlusion was defined as the inability to infuse medication or flush the catheter. 19 Phlebitis was assessed using the Visual Infusion Phlebitis (VIP) scale from the 2024 Infusion Therapy Standards of Practice, 20 with scores ≥1 recorded as positive and absence recorded as negative. Infiltration was defined as medication leakage due to insecure dressing, prolonged infusion time, or catheter movement within the vein. 1 Infection was identified by pain at the puncture site, local redness, swelling, discharge, fever, elevated white blood cell count, and increased neutrophil count. 21 Unplanned catheter removal was due to the child’s actions (eg, struggling) or improper handling by medical staff, leading to catheter dislodgement or self-removal without consent. 1
Insertion Technique for Mini-Midline Catheters
The Mini-MCs used in this study are a type of peripheral venous access device, typically inserted into superficial veins of the forearm or deep veins of the mid-upper arm, with the catheter tip not extending beyond the axillary region. The Mini-MCs had a size of 3 F, an effective length of 8 cm, an internal lumen volume of 0.14 mL, and a flow rate of 27 mL/min, and were made of polyurethane. Mini-MCs insertion and maintenance were performed in accordance with the standardized puncture techniques of the Shandong Nursing Association training base for midline catheters specialty practice. A blind venipuncture technique was used above or below the elbow. During insertion, the patient’s arm was abducted, and the puncture site was selected to avoid the elbow joint and venous valves. After successful venipuncture with blood return, the needle angle was lowered and advanced ~0.5 cm along the vein, after which the catheter was fully advanced into the vessel using the stylet. The needle core and peel-away sheath were removed, a pre-flushed extension set and connector were attached, and the catheter was secured with a sterile transparent dressing. In the PIVCs group, 24-gauge peripheral intravenous catheters (0.7 × 19 mm; flow rate 19 mL/min) were used in accordance with the 2024 Infusion Nurses Society (INS) standards and the Nursing Practice Standard for Intravenous Therapy (WS/T 433-2023), 22 based on clinical infusion requirements.
As both catheter types were classified as peripheral venous access devices, the same standardized maintenance protocol was applied to all participants. This protocol included: (1) covering the catheter securement device with a transparent semipermeable membrane dressing; (2) replacing the dressing every 7 days or immediately if it became loosened, soiled, or compromised; (3) flushing the catheter lumen with 2 to 3 mL of 0.9% normal saline using a pulsatile technique before and after each infusion and after intermittent infusion to prevent occlusion; and (4) daily assessment and documentation of the insertion site, dressing integrity, and local skin condition, as well as monitoring for symptoms such as extremity edema, pain, or signs of leakage or infection.
Quality Control
Prior to the study, all research team members involved in venipuncture and data collection received standardized training and competency assessments to ensure consistency. The 3 venipuncture practitioners were certified specialist nurses through the Shandong Nursing Association, each holding a bachelor’s degree and more than 15 years of pediatric nursing experience. They practiced extensively on simulation models according to operational standards and passed formal assessments before performing procedures on participants. Data were collected by 2 trained researchers using standardized observation and recording forms. Daily assessments included the condition of puncture sites, dressing integrity, and any complications. Criteria for evaluating complications, such as catheter occlusion, phlebitis, infiltration, and catheter-related infections were standardized through uniform training.
In this study, catheter removal was determined solely by the clinical medical team based on the patient’s actual clinical needs. The main indications for removal were as follows: (1) when complications related to the catheter (such as occlusion, phlebitis, infiltration/extravasation, or infection) occurred, and the clinical team assessed that the catheter could no longer be used safely and (2) when intravenous therapy was completed and vascular access was no longer required. Once the clinical team decided to remove a catheter, it was withdrawn from the study. This process ensured clinical decision-making independence, prioritized patient safety, and maintained the standardization and objectivity of data collection.
Statistical Analysis
Statistical analyses were performed using IBM SPSS Statistics for Windows, version 26.0 (IBM Corp, 2019, Armonk, NY, USA). Categorical variables, such as sex and the incidence of catheter-related complications, were summarized as frequencies and percentages. Continuous variables, including puncture time and pain scores, were described using median values. The chi-square test was used to compare complication rates between the Mini-MCs and PIVCs groups. As the continuous data were not normally distributed, the Mann-Whitney U test was used to compare pain scores and puncture times. Kaplan-Meier survival analysis was applied to evaluate catheter dwell time, and the log-rank test was used to compare survival curves between groups. A 2-sided P < .05 was considered statistically significant.
Ethical Approval
The study was approved by the Ethics Committee of Shandong Provincial Hospital Affiliated to Shandong First Medical University (approval no SWYX: 2023-479). In accordance with the Declaration of Helsinki, the privacy and personal information of all participants were protected, and written informed consent was obtained from the children and/or their legal guardians prior to enrollment.
Results
Between September and October 2024, a total of 245 children were admitted to the pediatric ward, of whom 145 were diagnosed with digestive system diseases. Among these, 126 eligible children were enrolled in the study, including 64 in Mini-MCs group and 62 in the PIVCs group. One participant withdrew during the study period, resulting in a final sample of 125 children included in the analysis (Figure 1). The median age of the participants was 9 years (range, 4-15 years), and 67 were male (54.0%). Gastritis was the most common diagnosis, accounting for 45 cases (36.3%). The first-attempt success rate for venipuncture was 100% in both groups. Baseline characteristics and comparisons between the 2 groups are summarized in Table 1.

Inclusion flow chart.
Baseline Patient Characteristics.
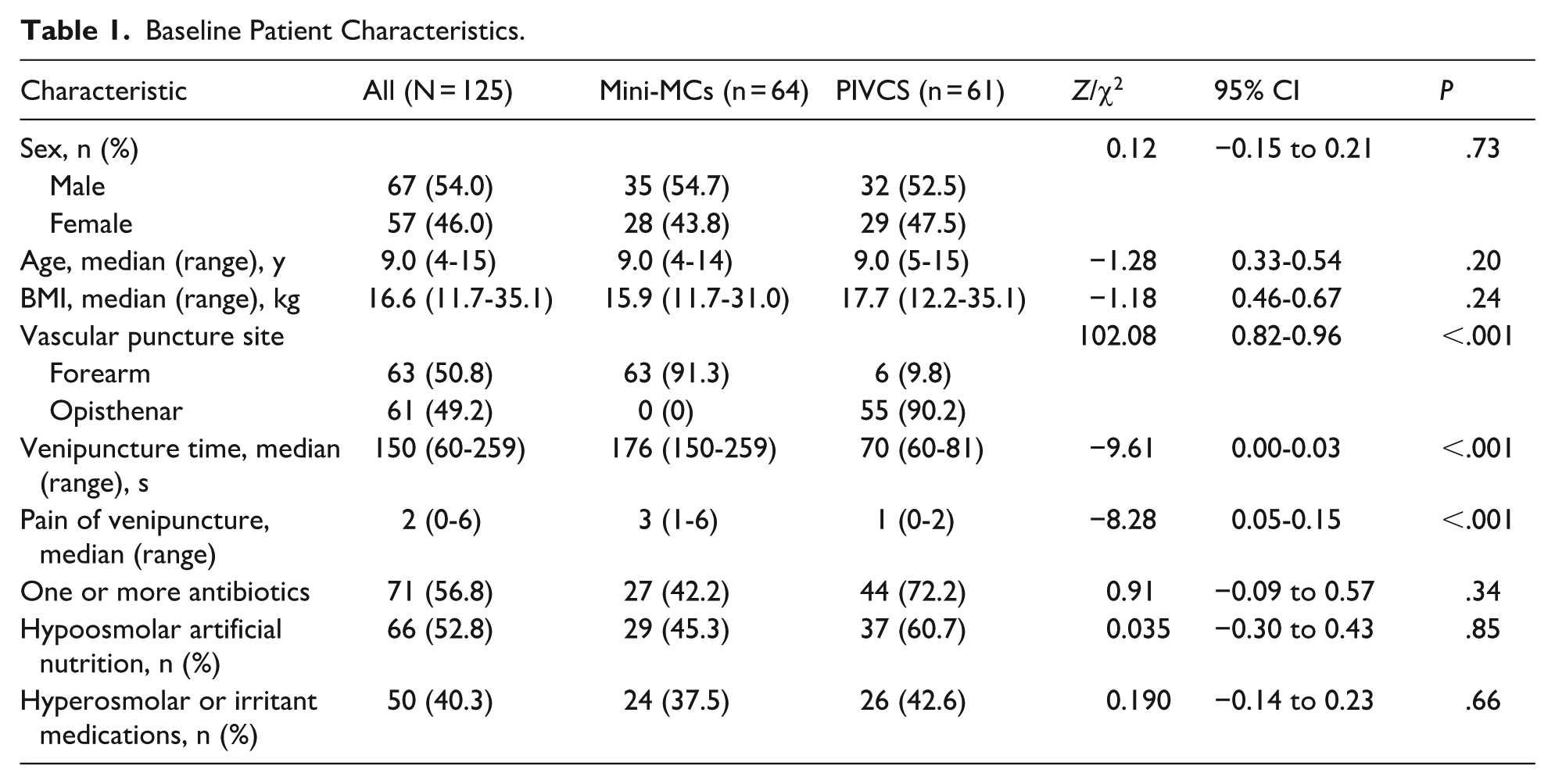
The median catheter dwell time in the Mini-MCs group was 94 hours (range, 21-304 hours). Compared with the PIVCs group, the Mini-MCs group had a significantly longer catheter dwell time (Z = −4.37, P < .01). Kaplan-Meier survival analysis further showed a significant advantage in dwell time for the Mini-MCs group (χ2 = 29.578, P < .001; Figure 2). Pain scores during the catheter dwell period were low in both groups, with no statistically significant difference observed (Z = −1.97, P = .05). Regarding complications, the overall incidence was significantly higher in the PIVCs group than in the Mini-MCs group (χ2 = 8.23, P < .01). Specifically, catheter occlusion occurred more frequently in the PIVCs group (χ2 = 7.25, P < .01), whereas no significant differences were observed between the groups for other complications. In addition, the proportion of unplanned catheter removal was significantly higher in the PIVCs group compared with the Mini-MCs group (χ2 = 9.3, P < .01). Detailed comparisons are presented in Table 2.

Kaplan-Meier curve revealing the percentage survival of Mini-MCs and PIVCs over time. The P value was calculated by using the log-rank test.
Outcomes of the Mini-MCs and PIVCS.

Discussion
In this study, the Mini-MCs associated a longer dwell time compared to PIVCs, with a median dwell time of ~4 days. This finding is consistent with the results reported by Qin et al 15 and may be related to catheter length and the caliber of the accessed vessels. The Mini-MCs used in this study had an effective length of 8 cm, which is longer than that of 24-gauge peripheral intravenous catheters. In addition, more than 90% of Mini-MCs were inserted into relatively larger veins of the upper arm, whereas PIVCs were predominantly placed in forearm veins. In pediatric patients with limited tolerance for infusion therapy, greater arm movement during the indwelling period may contribute to shorter catheter dwell time, which could partly explain the prolonged dwell time observed with Mini-MCs. Recent findings by Kleidon et al 18 demonstrated that in children with respiratory diseases, the use of Mini-MCs reduced the need for general anesthesia for peripherally inserted central catheter placement, further supporting the potential benefits of Mini-MCs in pediatric care. For children hospitalized with digestive system diseases, gastrointestinal endoscopy is frequently required for diagnosis and treatment. Periprocedural administration of sedatives, analgesics, and large volumes of fluids and electrolytes is common, and these infusions often have high osmolarity, which can irritate peripheral veins and negatively affect PIVCs dwell time. This may explain the shorter dwell time observed for PIVCs in the Kaplan-Meier survival analysis.
Pain, recognized as the fifth vital sign, has received increasing attention in recent years. 23 In hospitalized children, pain related to venipuncture and catheter indwelling, in addition to venous access difficulty, represents a major concern for clinical nursing care. 24 Therefore, this study evaluated pain during both catheter insertion and the indwelling period, an aspect often underreported in previous studies, providing a more comprehensive assessment of Mini-MCs use. As shown in Table 1, pain scores during insertion were higher in the Mini-MCs group than in the PIVCs group, with a median score of 3. This difference is likely attributable to the larger needle size required for Mini-MCs placement. However, insertion-related pain was transient and generally well tolerated by pediatric patients. During the indwelling period, no significant difference in pain scores was observed between the 2 groups, indicating acceptable comfort with Mini-MCs. This may partially explain the longer dwell time observed with Mini-MCs in this study.
Regarding complications, the overall incidence during the indwelling period was lower in the Mini-MCs group than in the PIVCs group, consistent with findings reported by Kleidon et al. 6 Occlusion and extravasation, which are major causes of peripheral intravenous infusion failure, 25 occurred at rates of only 1.6% and 1.8%, respectively, in the Mini-MCs group, both of which were significantly lower than those observed in the PIVCs group. These differences may be related to variations in catheter insertion sites and catheter characteristics. Unlike PIVCs, which are commonly placed in more mobile distal upper-limb veins, Mini-MCs are typically inserted in the upper arm or proximal forearm, where limb movement is relatively limited. In addition, the larger internal diameter of Mini-MCs may reduce the risk of occlusion when infusing solutions with similar molecular properties.
The primary goal of utilizing Mini-MCs was to achieve “1 needle until discharge” for hospitalized children. This means using only 1 peripheral intravenous catheter throughout the infusion period. The aim was to minimize pain from repeated venipunctures. In this study, ~65% of the hospitalized children were able to achieve a single puncture throughout the infusion period, effectively meeting the infusion needs of most hospitalized children with digestive system diseases and showing a significant advantage compared to PIVCs. The remaining instances of unplanned catheter removal were largely due to various complications occurring during the indwelling period. This highlights the importance of implementing scientific and effective maintenance practices when using Mini-MCs. Recommendations include assessing the catheter at least once every 4 hours 20 ; using a pulsatile flush with normal saline or heparinized saline equivalent to twice the internal volume of the catheter system immediately before and after each infusion to prevent occlusion and related complications20,26; and using sterile, transparent, breathable dressings, with transparent dressings replaced at least every 7 days.20,27
Limitations
This study has several limitations. First, as a single-center observational study with a small sample size, its statistical power and generalizability are limited. A non-randomized design was adopted due to the novelty of Mini-MCs in pediatric care in China, where randomization could pose ethical challenges and affect parental acceptance. Consequently, the study reflects real-world clinical practice. However, the observational design may introduce selection bias from unmeasured factors, such as vascular conditions and treatment complexity, which could influence the assessment of catheter efficacy. Future multi-center, large-scale randomized trials are needed to minimize bias and provide more robust evidence. Second, the study design did not allow full separation of the effects of catheter size (24 G) and insertion site (forearm vs upper arm) on dwell time. Thus, observed differences may partly reflect anatomical and hemodynamic variations related to insertion location rather than catheter type alone. Finally, although the study suggests potential advantages of Mini-MCs in specific clinical scenarios, causal inferences regarding their performance are limited by the observational nature of the study.
Conclusion
This study applied Mini-MCs in hospitalized children with digestive system diseases and demonstrated their advantages in this population, potentially expanding their clinical application. For children requiring intravenous therapy for 3 to 10 days, Mini-MCs were associated with fewer complications, greater comfort during the indwelling period, and facilitated a “1 needle until discharge” approach, reducing the pain of repeated venipunctures. Therefore, Mini-MCs may represent a suitable and effective option for pediatric peripheral infusion.
Footnotes
Ethical Considerations
The study was approved by the Ethics Committee of Shandong First Medical University Affiliated Provincial Hospital (approval no SWYX: 2023-479). In accordance with the ethical principles of the Declaration of Helsinki, the privacy and personal information of pediatric patients was kept confidential.
Consent to Participate
All participating or their guardians signed written informed consent forms.
Author Contributions
LJ contributed to conception and design, contributed to acquisition, drafted the manuscript, critically revised manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy. BC contributed to design, contributed to analysis, drafted the manuscript, critically revised manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy. ZY contributed to design, contributed to interpretation, drafted the manuscript, critically revised manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy. JY contributed to design, contributed to interpretation, drafted the manuscript, critically revised manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy. SW contributed to design, contributed to interpretation, drafted the manuscript, critically revised manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy. HJ contributed to conception and design, contributed to acquisition, drafted the manuscript, critically revised manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
