Abstract
Background:
Venous access is a vital component of medical care for hospitalized patients, especially patients who present with difficult intravenous access and require long-term hospitalization. A promising solution for these challenges is the use of long peripheral catheters. This report highlights our experience with an innovative, easily inserted, over-the-needle-long peripheral catheter.
Methods:
A retrospective, observational study included patients admitted to internal medicine departments and underwent a new long peripheral catheter insertion. We provide data on patient characteristics, indications for long peripheral catheter insertion, insertion success rates, dwell times, and complications rates.
Results:
The study included 109 patients with a total of 128 long peripheral catheters. The leading indications for long peripheral catheter insertion were IV administration of antibiotics and vasopressors. A first-time user inserted 65% of long peripheral catheters on the first attempt, with an average insertion time of 131 ± 11 s. Success rates increased in parallel with experience gained (p < 0.05). The average dwell time was 8.8 ± 0.63 days. The long peripheral catheter was removed as the therapy was completed in 33.5% of the patients. The complication rate was 13/1000 catheter days, including dislodgement, occlusion, phlebitis and infiltration. In addition, 23 individuals were successfully discharged with the long peripheral catheter to complete therapy at home. The long peripheral catheters was used to draw blood in 28 patients and for vasopressor administration in 22 patients.
Conclusions:
The new long peripheral catheter provides a safe, rapid, and effective means of venous access. Its straightforward insertion technique, convenience, and suitability for “point of care” use make it a valuable option for patients requiring continuous intravenous therapy.
Introduction
Venous access is an essential component in the medical care of a hospitalized patient. More than 60% of acute care patients worldwide require vascular access. 1
Initially short peripheral catheters (SPCs) are used since they are simple and allow for short-term access. They are commonly replaced at regular intervals (72–96 h) to reduce the risk of infection and phlebitis, although the impact of this approach is debatable.2,3 When venous access is required for longer periods, a different intravenous catheter is often used. 2
Venous access devices can be divided into peripheral versus centrally inserted devices and short-, medium-, and long-term venous access.2,4 –7 When choosing which device to use one must strike a balance between maximizing functionality and usefulness and minimizing the risk of catheter-related complications. Clinical aspects (e.g., duration of treatment, type of infuscate), anatomical factors (e.g., difficult intravenous access (DIVA)), and other secondary factors such as local expertise may also affect the choice of catheter.
A major difference between central and peripheral catheters is their termination site. Central catheters terminate in the central circulation—the thoracic central veins or iliocaval venous system. In essence, they terminate at either the superior vena cava, inferior vena cava, or the right atrium. They can cope with agents that are vesicant, irritant, or hyperosmolar unsuitable for peripheral veins (e.g., antineoplastic, parenteral nutrition).
On the other hand, peripheral catheters that terminate distal to the subclavian vein are suitable for peripherally compatible agents and are subdivided into three categories: SPCs, long peripheral catheters (LPCs) and midline catheters (MCs).4,6,8 –10
Peripheral catheters differ one from the other in length, insertion area, expected duration, and materials: Short peripheral venous catheters (PVCs) vary between 3 and 6 cm in length; they are placed at the upper limb at or below the antecubital fossa, are reliable for 3–5 days, and are usually not recommended for prolonged use. MCs are 15–25 cm in length and are inserted into peripheral veins at or above the antecubital fossa. The distal tip generally extends into the axilla (some enter the infra/supraclavicular region). Viable MCs can be used for months. In the last decade LPC (or mini-midline) have been introduced into use. These catheters are longer than SPC but shorter than MC. LPCs are 6–15 cm in length (usually 8–10 cm). They are inserted in the forearm, antecubital fossa, or upper arm. Insertion to the deep veins of the upper arm requires ultrasound guidance. The distal tip terminates before the axilla, typically up to the mid-upper arm. They can remain in situ for up to 4 weeks.
In the literature, there is considerable mislabeling and confusion between midlines and LPCs, including in clinical studies, guidelines, and evidence-based recommendations. They are, however, two different devices that vary in size, materials, and insertion technique.1 –8
Many patients admitted to internal medicine departments are elderly and present with challenging intravenous access, often requiring multiple insertion attempts. They frequently have comorbidities such as diabetes, obesity, and other chronic conditions, which can prolong their hospital stay and complicate venous access. Additionally, in Israel, most internal medicine departments include intermediate care units that admit ventilated or critically ill patients. These patients often require a continuous, stable venous access due to the need for multiple medications and ongoing treatments.
One option for treating these patients is to insert a central line; however, insertion of such a line requires considerable skill, consumes staff time, requires maximal sterile barrier precautions, and advanced imaging to verify location. It inevitably also involves high costs (direct and indirect). Another option is an LPC. LPCs have a relatively long dwell time (up to 28 days), lower infiltration/extravasation rates and lower rates of phlebitis compared to SPCs, as well as lower infection rates compared to central lines. 4 LPCs do not reach the central circulation so that the risk for pneumothorax or hemothorax is lower when compared to central venous catheter (CVC), as is the need to verify correct placement via chest radiography.1,11
These features explain the increasing use of LPCs in clinical practice—they are less expensive than MCs, and have a clinical performance superior to that of SPCs, lower complication risks and a higher duration. LPCs are appropriate in DIVA patients, or when expected duration of treatment is 1–4 weeks. 4
Typically, MCs are inserted by specialized vascular teams, such as anesthetic nurses or physicians skilled in ultrasound-guided vascular access, using the Seldinger technique.1,12 However, with the increasing use of ultrasound, including point-of-care ultrasound (POCUS), peripheral venous cannulation can now be performed at the bedside by less experienced physicians after a brief instructional session.
In this context, we present the clinical performance of a new LPC. This type of catheter, made by Bullpup Scientific is inserted over-the-needle, like a standard peripheral IV. It features a unique forward-grip introducer that facilitates easy insertion and minimizes direct contact with the catheter. This over-the-needle approach simplifies the learning process for practitioners, making it more accessible for those with limited experience.
Methods
This is a retrospective, observational study was conducted in internal medicine wards between July 2021 and March 2023. Adult patients (⩾18 years) who were admitted in these wards and underwent LPC insertion were included. Exclusion criteria were patients who already had a central line in place and were assessed by the treating physician as needing ongoing treatment with the latter. Patients were admitted either in regular internal medicine wards, or in an intermediate intensive care unit situated within an internal medicine department. In Israel, due to shortage of ICU beds, patients who are mechanically ventilated can be admitted to such an intermediate unit under the care of internal medicine physicians. These patients are usually older, sicker, and have a worse prognosis 13 than patients admitted to a regular intensive care unit. Data collection regarding LPC insertion was based on a retrospective review of medical charts. The study was performed at the Rambam Health Care Campus (RHCC) in Haifa, Israel. RHCC is a 1000-bed tertiary, academic hospital serving over 2 million residents in northern Israel with 80,000–90,000 inpatient admissions every year.
Routine patient care at RHCC includes comprehensive documentation of all complications associated with various types of catheters, including peripheral intravenous catheters, midlines, and CVCs. Recorded data also included reasons for LPC insertion, number of attempts per insertion, insertion time, catheter dwell time, and causes for catheter removal. Reasons for the removal of catheters included completion of intravenous therapy, catheter-related bloodstream infection (defined as clinical infection with a blood culture positive for a typical bacterial or fungal pathogen not related to an infection at another site), venous thrombosis (U.S. visualization of an echogenic intravascular structure),4,14 accidental catheter dislodgement, catheter occlusion, phlebitis, infiltration or extravasation of intravenous solution out of the vein, and pain during infusion. In addition, we documented demographic and clinical data that included sex, age, BMI, chronic comorbidities, hospitalization duration, medications administered via the LPC, and blood culture results whenever line infection occurred.
We used the M/29 Bullpup mini-MC. The catheter is 8.5–10 cm polyurethane over-the-needle catheter. It inserts similarly to a standard IV cannula so that any trained IV inserter can insert an extended dwell catheter system without touching the catheter itself. We inserted 18 or 20 G (22 G is also available but was not used in our study). All catheters were inserted under ultrasound guidance.
Catheters were inserted mainly by a novice physician who had completed a POCUS course, including a short session regarding vascular access, and underwent a didactic training session for the specific catheter. A specific standardized insertion protocol was used.15 –17
First, after both arms were examined, the most appropriate vein was selected in terms of position, diameter (above 4 mm) and depth (no more than 20 mm deep as measured vertically from the skin to the vein). Upper arm veins were chosen over forearm veins. The potential sites were the cephalic, basilic and brachial veins. If the brachial vein was selected a planned search was performed in order to identify the brachial artery and brachial nerve, in order to avoid puncturing them.
Then, the puncture site was scrubbed with 0.5% chlorhexidine with alcohol 70% before placing a protective sterile drape around the designated site. Portable ultrasound devices, including sterile gel, ultrasound probe cover and sterile gloves, were used. Local anesthesia was administered before venipuncture under U.S. guidance if the patient’s condition required it.
Using the dynamic needle tip positioning technique, the vein was punctured under direct ultrasound guidance in a short-axis view. Once the needle was confirmed to be within the vein, the catheter was advanced over the needle into the vein, and the needle was withdrawn. Successful cannulation was verified by the return of blood. The end was then closed with a short extension tubing connected to a needless connector.
The entry site and the extension tubing were dressed with a suture-less device (3M™ Tegaderm™ I.V. Advanced Securement Dressing), and the line was then flushed with saline under ultrasound visualization to confirm correct placement.
Afterward documentation was added in the medical records, and a sign was placed above the patients’ bed indicating that a LPC was in place.
This study was approved by the Institutional Review Board (IRB) of our institute (Helsinki Committee of the Rambam Medical Center), with approval number RMB-D-0299-22. Written informed consent was waived by the IRB for this retrospective study.
Statistical analysis
We used a convenience sample of 109 patients to estimate success and complication rates. Patients’ characteristics were summarized using descriptive statistics. For normally distributed continuous variables, we used means and standard errors. The student’s t-test was used to compare normally distributed continuous variables. To analyze the differences between categorial variables, Chi-square test was used. Statistical significance was defined as a p-value of <0.05. Data analysis was conducted with R studio software 1.4 (Posit, Boston, MA, USA) and Microsoft Excel version 14.0 (Microsoft Corporation, Redmond, WA, USA).
Results
One hundred nine patients were included in the study; they underwent the insertion of 128 LPCs. The patients’ characteristics including age, gender, Charlson Comorbidity Index (CCI) and the primary diagnosis at discharge are presented in Table 1. 18
Characteristics of patients.

Data presented as average ± standard error.
The indications for LPC insertion are presented in Figure 1. 49/109 (44.9%) patients had DIVA, while 99/109 (90.8%) patients had a need for IV access of more than 6 days. Out of the patients who required prolonged IV access, the indications for line insertion included antibiotic therapy in 77 (77.7%) patients, prolonged IV fluid administration in 13 (13.1%) patients, long-term sedative and analgesic treatment in 21 (21.2%) patients, administration of vasopressors to 22 (22.2%) patients, and peripheral total parenteral nutrition in 4 (0.04%) patients. Some patients had more than one indication.

The indications for LPC insertions.
Figure 2(a) illustrates the number of attempts required to successfully insert a functional LPC. In 84 insertions (65.6%), the catheter was successfully inserted on the first attempt. A second attempt was necessary in 33 insertions (25.7%), while 10 insertions (8%) required a third attempt, and 1 patient (0.7%) needed a fifth attempt. As the physician gained experience with the LPC insertion technique, the number of attempts significantly decreased, as demonstrated by the Pearson’s correlation test (Figure 2(b)). The average time for insertion was 131.54 ± 11.7 s.
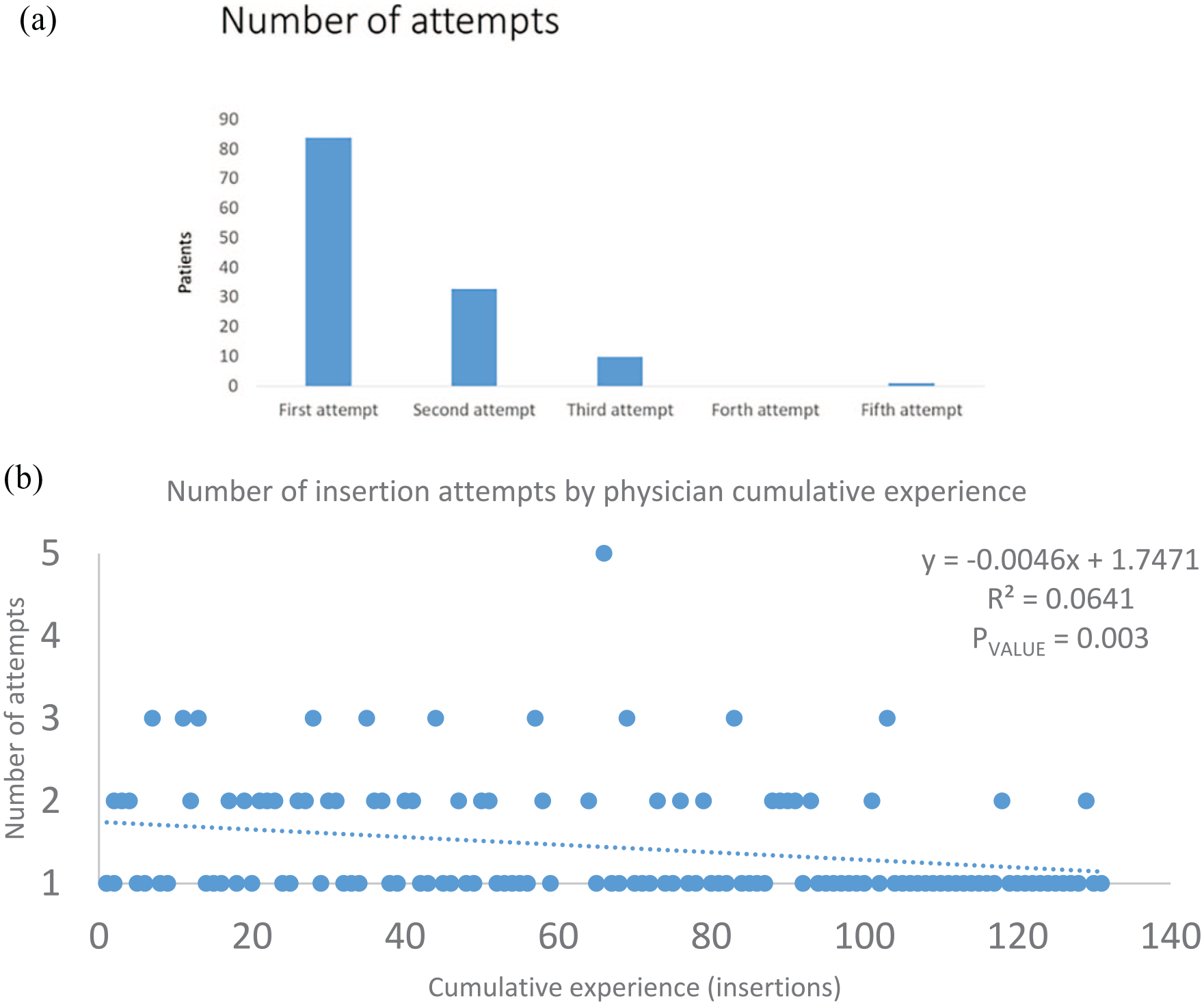
Number of attempts and success rate. (a) Number of insertions attempts and (b) Number of attempts to achieve a functional LPC.
The average dwell time for the LPC was 8.8 ± 0.63 days. It was successfully used to administer medications such as fluids, midazolam, fentanyl, antibiotics, and antivirals. Some of the medications administered are typically reserved for central venous access like norepinephrine and propofol. The overall complication rate for LPCs used for these medications was comparable to that of LPCs used for other medications. Additionally, the LPC was successfully employed for drawing blood samples in 28 patients (25.7%).
Of the patients, 41 (37.6%) required an additional IV access: 30 needed PVCs, 9 needed CVCs, 1 had a PERM-CATH, and 1 patient had a dialysis line. Twenty-three patients (21.1%) were discharged with the LPC for home IV treatment, and all lines were easily removed upon completion of their planned therapy.
The reasons for extraction are presented in Figure 3; 43 (33.5%) of the catheters were used until completion of therapy. In 53 (41.4%) catheters (inserted to 49 patients) a dislodgement of the LPC was noted and necessitated its removal. Eight (6.2%) catheters (7 patients) were removed due to phlebitis, and 5 catheters (3.9%) were occluded. In a single patient, the LPC was removed because of extravasation, and in a single patient, it was removed due to a suspected catheter-associated blood stream infection. The only predictor of a lower rate of therapy completion was the CCI; as the average CCI of patients who completed the planned therapy was 2.25 and those who did not was 2.73 (p = 0.034). Reinsertion of LPC was performed in 14 patients (12.8%).
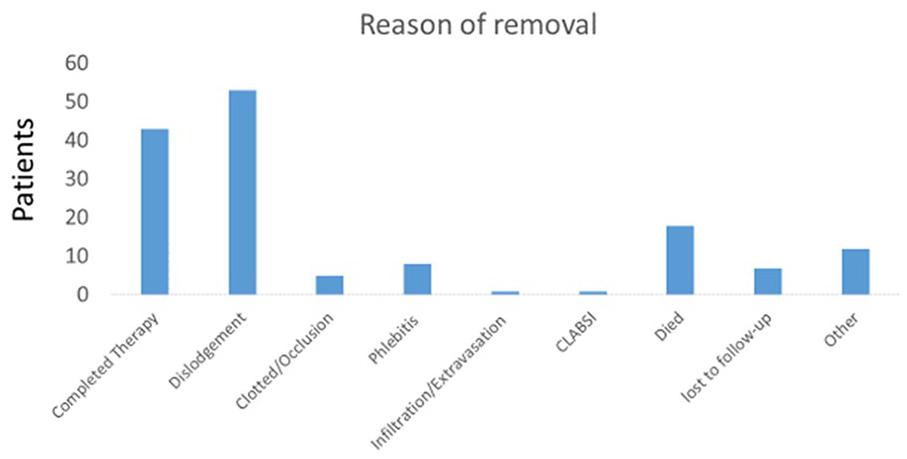
Reasons for LPC removal.
Discussion
In this study, we described our experience with a unique and new over-the-needle LPC catheter in patients requiring prolonged intravenous treatment or having difficult peripheral venous access.
The Bullpup M/29 is designed to address the challenges associated with longer over-the-needle catheters, which can become harder to control as needle length increases. With traditional catheters, the further the hand is from the needle tip, the more flexible and less precise the device becomes, making deep vein access particularly challenging in patients with thicker tissue. The Bullpup M/29 introduces a different approach by allowing clinicians to grip the needle closer to the insertion point, which may help improve control and make longer catheters easier to handle. Additionally, its design reduces exposure to contaminants by keeping most of the catheter enclosed during insertion, which could contribute to better infection control compared to conventional long catheters.
These unique features—a forward-grip introducer, no direct contact between the physician’s hands and the catheter, and an “over-the-needle” insertion technique—contribute to a relatively high success rate on the first attempt and a shorter insertion time, as demonstrated by our outcomes (Figure 4).

The Bullpup long peripheral catheter. Top: closed form demonstrating the forward grip introducer, Bottom: opened form demonstrating the entire length of the cannula.
Major complication rates (e.g., DVT, CRBSI) were low while minor complication rates (e.g., phlebitis, clotted, infiltration) were similar to that published in the literature 19 for conventional midlines—except for a higher dislodgement rate. The LPC was also successfully used after discharge in patients’ homes.
For intravenous access of more than 6 days current recommendations support the use of LPC 12 and also suggested by the Michigan appropriateness guide for intravascular catheters, MAGIC. 1 In our study slightly more than half of the catheters (60 patients, 55.0%) were inserted for this reason alone.
DIVA was also a common reason for LPC use and was defined as a clinician having two or more failed attempts at IV access using a traditional technique, or lack of physical examination findings (no visible or palpable vessel) or a patient’s stated or documented history of DIVA. 20 Many factors can interfere with routine peripheral IV placement, such as obesity, intravenous drug abuse, or hypovolemia. 21 Our experience suggested that the LPC could be an affective IV access device in these patients by preventing unnecessary needle sticks, and potentially shortening the time to administration of IV medications. Some of these medications may be urgently indicated for conditions such as severe sepsis or shock.
The majority of the LPCs were inserted in the first attempt 84 (65.6%), with an average number of 1.44 attempts per line. To note, though the majority of catheters were inserted in the first attempt, our first attempt success rate was lower than anticipated. This could easily be explained by the fact that the catheters were inserted by a novice physician. Furthermore, a previous study reported similar average success rates per line, with traditional midlines inserted over a wire. 8 Our data also indicated that constant improvement in the insertion success rate occurred throughout the study. All LPCs were inserted by a physician who had completed a formal POCUS course and practiced LPC insertion on a model, but had had little experience in U.S. guided IV access. The success rate is therefore encouraging, and the learning curve reflects real-life scenarios.
As, all in all, LPC insertion is similar to that of a standard peripheral IV, the insertion time from skin puncture to connection of the extension set is relatively short (131 s), and probably offers an advantage over traditional over-the-wire midlines. A study by Nielsen et al. 22 provides an indirect measurement of time spent to catheterization using traditional MCs. In this study midline insertion time was 14 min measured from entering the room to completion of the procedure. We believe that the relatively short insertion time of the LPC, and the relatively high success rate result from the simpler insertion technique of the LPC.
The average dwell time was 8.8 days. This is longer than dwell times reported in studies performed in hospitals8,22 but naturally shorter than dwell times of midlines used mainly in the community. 19 Roughly 30% of the LPCs were extracted because therapy was completed, so the potential dwell time could have been much longer if additional therapy had been required.
A significant number of catheters (53, 41.4%) were removed prematurely due to dislodgement. Most of these were accidentally pulled by staff or patients (n = 40, 31.2%) or found to be incorrectly placed in the vein by attending staff (n = 13, 10.1%). Since the LPCs were introduced for the first time, the staff required an adjustment period. The high rate of accidental removals may be attributed to the inexperience of the nursing staff with the new device or inadequate securement of the lines. Additionally, the complexity of care for patients with multiple comorbidities, especially in intermediate care units, likely contributed to the accidental catheter dislodgement. Self-removal by patients also played a significant role in premature catheter extraction. 23 Consequently, these issues with premature catheter removal affected the average dwell time.
Some known complications of venous catheter insertion, such as bleeding or accidental artery cannulation, were not observed at all. In general, complications were relatively uncommon, with the exception of a relatively high incidence of catheter dislodgement, (see Figure 3). Significantly, no case of VTE occurred, although systematic U.S. assessments were not performed. Catheter related blood stream infection (CRBSI) was suspected only once, and blood cultures were consistently negative in this case too. This data confirm the low incidence of major complications caused by MCs. 16
Rates of catheter occlusion (3.9%) and phlebitis (6.2%) were low and are similar to those reported previously for regular MCs. 19 On the whole, this emphasizes the need for daily catheter inspection for signs of phlebitis or occlusion, and highlights the fact that catheter ultrasound insertion is only the initial stage in the use of a catheter, and that a systematic inspection protocol is required to detect complications.
We have noted several advantages of the over-the-needle LPCs: the use of vasopressors through these lines, the use of LPCs for withdrawing blood tests, and their use in home care.
Twenty-two patients (20.1%) received norepinephrine through their LPCs, highlighting the catheter’s suitability for critical treatments in intensive care settings. Typically, vasopressors are administered via central lines due to concerns about tissue damage when delivered through small peripheral lines. 24 However, since LPCs are placed in larger veins, they offer a larger lumen for the vasopressor, potentially reducing the risk of tissue injury. No complications related to vasopressor administration were observed in this study, but a formal prospective trial is needed to confirm these findings. Future research should focus on factors such as vasopressor dosage, duration of administration through the LPC, and associated complication rates.
In 28 patients (25.7%), the LPC was successfully used to draw blood samples; therefore, preventing unnecessary venous punctures. Twenty-three (21.1%) patients were discharged with the LPC, most of whom (18/23, 78.2%) successfully completed therapy at home. The convenience of the use of LPC is therefore an appealing option for the growing field of home care.
The study has several limitations. As an initial single-center study without a control group and with a relatively small sample size (i.e., a convenient sample), definitive conclusions cannot be drawn. As a formal sample size calculation was not performed—the study may have been under powered to detect some uncommon complications of this venous catheter. The retrospective nature of the study may have led to incomplete documentation of some clinical aspects of LPC care, despite staff being instructed to examine the line daily, document their findings in the electronic chart, and record all events according to a prespecified checklist. Due to the small sample size, rare adverse events may have been missed. Since studies to assess Deep Vein Thrombosis (DVT) and catheter-associated blood-stream infections were carried out only when clinical suspicion arose, some subclinical events may have been missed. Additionally, the reason for catheter dislodgement was unknown in 12.8% of cases, and some patients were not available for follow-up.
Conclusion
In conclusion, this study is the first to evaluate a new over-the-needle LPC. We demonstrated its clinical utility in hospitalized patients, including many who were critically ill, with a quarter requiring mechanical ventilation.
Several key advantages of this new catheter were highlighted: it achieved relatively high insertion success rates and shorter insertion times compared to traditional midlines, with a notable learning curve associated with the technique. These positive outcomes are likely due to the relatively simple insertion method. Complications such as line infection or phlebitis were rare, and notably, vasopressors were administered through the LPCs without any observed complications. However, accidental dislodgement remained a recurring issue.
The LPC catheter presents a promising option for continuous IV access, offering simplicity, ease of use, and cost-effectiveness. A randomized controlled trial comparing this device with other LPCs is warranted to further validate these findings.
Footnotes
Acknowledgements
The authors thank AM and TH for their valuable assistance during the study, Dr. Belina Neuberger for language editing services, and would also like to thank Einav Blanche for the encouragement and support offered throughout all stages of manuscript preparation.
Ethical considerations
This study was approved by the Institutional Review Board (IRB) of our institute (Helsinki Committee of the Rambam Medical Center), with approval number RMB-D-0299-22.
Consent to participate
Written informed consent was waived by the Institutional Review Board (IRB) for this retrospective study.
Consent for publication
Not applicable.
Author contributions
YW: Conceptualization, Study design, Data collection, Data analysis, Manuscript drafting and preparation. DC-S and SK: Data collection, Data analysis, Data interpretation, Statistical analysis, manuscript preparation. AN and GB: Manuscript review and editing, Supervision, Critical revision of the manuscript. All authors have read and approved the final version of this manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability Statement
Data is available upon request from the corresponding author.
Trial registration
This is a retrospective descriptive study—hence not applicable (the approval number is mentioned in the ethical approval section).
