Abstract
Background/objectives:
This systematic review and meta-analysis quantified global and regional birth prevalence of hyperphenylalaninemia (HPA) caused by phenylalanine (Phe) hydroxylase deficiency.
Data sources and methods:
Searches of PubMed, Embase, the Cochrane Library and PROSPERO yielded 4297 records; 210 met inclusion and 97 contributed to a rasyndom-effects model stratified by blood Phe cut-off values.
Results:
The regionally weighted global prevalence of HPA was 0.84 per 10 000 live births (95% CI 0.44-1.24), ranging from 0.60 in South America to 1.30 in the Middle East and North Africa (MENA). Classic PKU (Phe: 1200 ± 200 μmol/l) occurred in 0.29 per 10 000 births overall, reaching 0.58 (0.34-0.87) in North America and 0.67 (0.28-1.23) in Europe.
Conclusion:
Prevalence patterns mirrored consanguinity rates, explaining the high burden in MENA and the low figures in Southeast Asia. The meta-analysis, applying rare-disease-specific minimum sample-size threshold, confirmed HPA, incl. PKU as a rare disease and validated prevalence estimates.
Keywords
Introduction
Phenylketonuria (PKU) was first reported in 1934 by Asbjørn Følling, who identified the disorder by detecting phenylpyruvic acid in the urine of siblings with intellectual disability, linking it to dietary phenylalanine (Phe). 1 PKU results from a genetic defect in the phenylalanine hydroxylase (PAH) enzyme, leading to Phe accumulation and associated disorders. Elevated Phe levels, termed hyperphenylalaninemia (HPA), are classified by severity, blood Phe levels, and enzyme impairment type. HPA can also result from tetrahydrobiopterin (BH4) deficiency, which impairs PAH activity. This study focuses on the HPA caused by PAH deficiency.
There are minor variations in the classification and management of PKU, based on plasma Phe levels, across different guidelines. The American College of Medical Genetics (ACMG) 2 advises initiating treatment when plasma Phe exceeds 360 µmol/l, whereas the European Society for Phenylketonuria (ESPKU) allows continued monitoring without treatment in mildly elevated cases if neurocognitive development remains normal.3-5 Figure 1 illustrates classification of HPA by serum Phe levels. 6 The most severe phenotype, classic PKU with the Phe level above 1200 µmol/l, is characterized by near-complete PAH deficiency and, without treatment, can cause intellectual disabilities, physical symptoms (eg, fair skin, eczema), and behavioral issues.7,8

Classification of hyperphenylalaninemia.
Early diagnosis via neonatal screening, typically within 24 to 48 hours of birth, identifies elevated Phe levels, 9 confirmed with genetic testing in some cases. 10 Treatment involves dietary Phe restriction, tyrosine supplementation, and pharmacological therapies, with intervention levels stratified by severity.2,5 For pregnant women with PKU, strict Phe reduction targets are applied to prevent fetal complications such as microcephaly.5,11
A few systematic literature reviews (SLRs) have been published on PKU epidemiology in the past.12-14 Updated data have the potential to shape healthcare policies, enhance newborn screening initiatives, and refine treatment practices for individuals living with PKU. Therefore, this SLR aimed to have an up-to-date understanding of the global epidemiology of HPA caused by PAH deficiency, with further stratification into 4 diagnostic-cutoff groups by Phe level. The HPA classification by ACMG, 2 ESPKU, 5 and prior literature11,12 were integrated to define diagnostic cutoff groups based on the blood Phe levels, including 120 ± 40 (mild HPA not requiring treatment), 360 ± 100 (mild HPA requiring treatment), 600 ± 100 (mild PKU), and 1200 ± 200 μmol/l (classic PKU). This review also involved the meta-analysis of epidemiology data by regional distribution and using the minimum sample size criteria, where an adequate sample size requirement was defined for each region based on expected birth prevalence.
Materials and Methods
Databases and Literature Search Strategy
The SLR was conducted following Centre for Reviews and Dissemination (CRD) guidance for conducting systematic reviews, 15 and reported in compliance with the PRISMA 2020 Statement. 16 A research protocol was prepared prior to the start of this SLR; it was not registered in any publicly accessible protocol database. The literature search was conducted in 4 major medical research databases, including Medline (via PubMed), Embase, Cochrane Library, and PROSPERO, on. January 12, 2024, with no prior date restrictions but limited to human studies published in English. Search strings were tailored for each database, with results summarized in Supplemental 1 Table 1.
Title and Abstract Screening
Screening and extraction were performed by 2 reviewers working independently (ie, each reviewer screened/extracted separately, then compared results and resolved disagreements by consensus with a third reviewer). Records from database searches were imported into Covidence software (Veritas Health Innovation, Melbourne, Australia; www.covidence.org) and de-duplicated. Subsequently, two researchers conducted title and abstract screening, resolving disagreements through consultation with the project leader. Using the Population, Intervention, Comparison, Outcome, and Study design (PICOS) hierarchical approach, records lacking primary epidemiological data on HPA were excluded, with detailed exclusion criteria in Supplemental 1 Table 2. The key inclusion criterion was unselected population-level screening, regardless of participant age group. Studies based on pre-selected or high-risk populations (eg, family history of HPA/PKU, clinical suspicion) were excluded.
Full-Text Screening
Full-text articles passing the title and abstract screening were evaluated for inclusion by 2 researchers. Articles not meeting exclusion criteria were included, with reasons documented. The project leader resolved disagreements on inclusion. Hierarchical criteria were applied to exclude articles lacking primary epidemiological data on HPA, detailed in Supplemental 1 Table 3. A snowball sampling approach was employed to enhance review sensitivity, which involved screening references from relevant systematic reviews and meta-analyses.
Data Extraction
Two researchers conducted data extraction, with a third researcher resolving any discrepancies. Covidence or a tailored Microsoft Excel data extraction sheet was used depending on study complexity. Specifically, single-center studies with uniform screening methods and regional data were extracted in Covidence, while multi-center studies with varied methodologies or regional data were extracted in Excel. Birth prevalence was calculated for regional and global determinations across 4 Phe concentration cutoff levels: 120 ± 40, 360 ± 100, 600 ± 100, and 1200 ± 200 μmol/l. Cutoff values were selected based on the clinical classification of HPA (Figure 1) and recent publications to allow for benchmarking of the results.12-14 Threshold ranges of ±40 μmol/l or ±100 μmol/l or ±200 μmol/l were applied to the respective Phe level categories to account for significant variance in the reported diagnostic cutoff values. If a study provided birth prevalence for ranges between different Phe intervals, we added all the values above the respective cutoff level. When a publication presented multiple birth prevalence estimates for multiple single cutoff values, we only considered one estimate to avoid double counting. A detailed explanation of the methodology with examples is provided in Supplemental 1 Table 4. We also conducted an unstratified meta-analysis that included estimates from studies where Phe cutoff values were not reported or did not fit into the 4 cutoff groups to determine the overall birth prevalence. A comprehensive list of collected data items is presented in Supplemental 1 Table 5.
Quality Assessment
To enhance the robustness of the systematic review, the methodological quality of included studies was assessed by 1 reviewer and cross-checked by another. A quality assessment matrix with 9 domains, based on the Joanna Briggs Institute (JBI) critical appraisal checklist for prevalence studies, 17 was applied (Supplemental 2 Section 1). Each study was evaluated against JBI criteria for methodological rigor. Given the low prevalence of rare diseases, a sufficiently large sample size is critical to ensure accuracy, reliability, and statistical validity. A specific formula for calculating sample size in prevalence studies, adjusted for rare diseases, was used. 18 This formula accounts for expected prevalence, the z-score for a 95% confidence level, and the desired precision. Expected regional prevalence data were used from the SLR by Foreman et al. 12 Accordingly, a minimum required screening population was calculated for each region, and only studies meeting or exceeding this threshold were included in the respective subgroup analyses. The regional thresholds and calculation methodology are described in detail in Supplemental 2 Section 2. Diagnostic methods for HPA, including tandem mass spectrometry, chromatographic techniques (ion-exchange, HPLC, column chromatography), genetic diagnosis, and enzymatic or fluorometric methods, were considered adequate for study inclusion in the review. Supplemental 2 Section 2 presents a detailed introduction to the methodology used to assess quality.
Meta-analysis
Studies failing domain no. 7 (“Test method”) of the quality assessment matrix were classified high risk and excluded from the meta-analysis. Potential overlap among patient populations was carefully evaluated by reviewing countries, study periods, data sources, and cohorts to ensure no duplication exists. The publication with the largest sample size was selected for inclusion in geographic and temporal overlap studies. Two statisticians performed a random-effects meta-analysis, including studies with no temporal or geographic overlaps. For each study we computed birth prevalence as the number of confirmed cases divided by the total screened population. One statistician used STATA SE 15.0 with the Metaprop module, which pooled proportions using the Freeman-Tukey double arcsine transformation. Confidence intervals for individual and pooled estimates were calculated using the exact binomial (Clopper-Pearson) procedure. Parallel analyses in R (version 4.4.1) utilized the “meta” package with a random-effects model and the same transformation and weighting methods. Between-study heterogeneity was assessed with the Cochrane Q, I2, and τ2 statistics.
Analyses were stratified by region, including Global, Asia, North America, South America, Europe, Middle East and North Africa (MENA), Sub-Saharan Africa, and Australia & Oceania. North America included the Caribbean, and Turkey was classified under MENA, following UNSD guidelines. 19
Regional populations and the list of countries included in each region are presented in Supplemental 1 Table 6; regional populations were considered as per the latest UN report and were used to calculate a regionally weighted global prevalence for the disease. 20 The regionally weighted global prevalence was calculated by weighting each region’s results according to its relative share of the global population: North America (0.0882), South America (0.0642), Asia (0.6505), Europe (0.1092), MENA (0.0813), and Australia & Oceania (0.0066).
Meta-analyses included all qualifying studies (unstratified) and data stratified into 4 Phe cutoff groups (120 ± 40, 360 ± 100, 600 ± 100, and 1200 ± 200 μmol/l). Regional meta-analysis for each cutoff group required a minimum of 2 qualifying studies, while global meta-analysis allowed a region to be represented by at least 1. An additional minimum sample-size criterion, defined based on the expected regional prevalence, was applied; only studies with a screened population exceeding the calculated thresholds described in Section 2.5 were included in the subgroup analyses.
Epidemiological Terminology
Studies on HPA exhibit inconsistent use of epidemiological terms such as incidence, prevalence, rate, and frequency. Incidence refers to new cases within a specified time, measured as cumulative incidence (proportion developing the outcome over time) or incidence rate (new cases per person-time “at risk”). Prevalence reflects the proportion of the population affected, expressed as a percentage or cases per 10 000 or 100 000 individuals, measured at a specific time (point prevalence) or over a period (period prevalence). Rates typically include a time component, while frequencies often use total sample size as the denominator. Inconsistent terminology in reviewed studies complicates comparisons. However, most studies adopted birth prevalence, focusing on HPA cases detected through newborn screening. While this term emphasizes prevalence, one should know that it refers to incident (new) cases identified during screenings.21,22
Despite this limitation, we have used the term “birth prevalence” to present our findings on the frequency of HPA cases detected among screened newborns. The epidemiological terminology used in both the screened and the finally included studies varied significantly (eg, incidence, prevalence, frequency, rate). However, the calculation methodology used for newborn screening was largely consistent. Studies with unclear calculation methodologies were excluded from the analysis.
Ethical Approval and Informed Consent
This study is a systematic review and meta-analysis of previously published, aggregate, de-identified data and did not involve collection of new data from human participants, prospective recruitment, or access to identifiable personal information. Accordingly, ethical approval and individual informed consent were not required.
Results
Literature Review
The search identified 4272 records across 4 databases: Embase (n = 2305), PubMed (n = 1920), PROSPERO (n = 42), and Cochrane (n = 5). After de-duplication, 2903 database records and 25 from snowball sampling were screened, with 434 advancing to full-text review. Of these, 210 studies were included to progress to data extraction. Included studies were published between 1963 and 2024. All of the included studies (Supplemental 2 section 4) focused on newborn screening, except 2 studies focusing on screening of healthy pregnant women.23,24 The PRISMA chart of the systematic review process is presented in Figure 2.
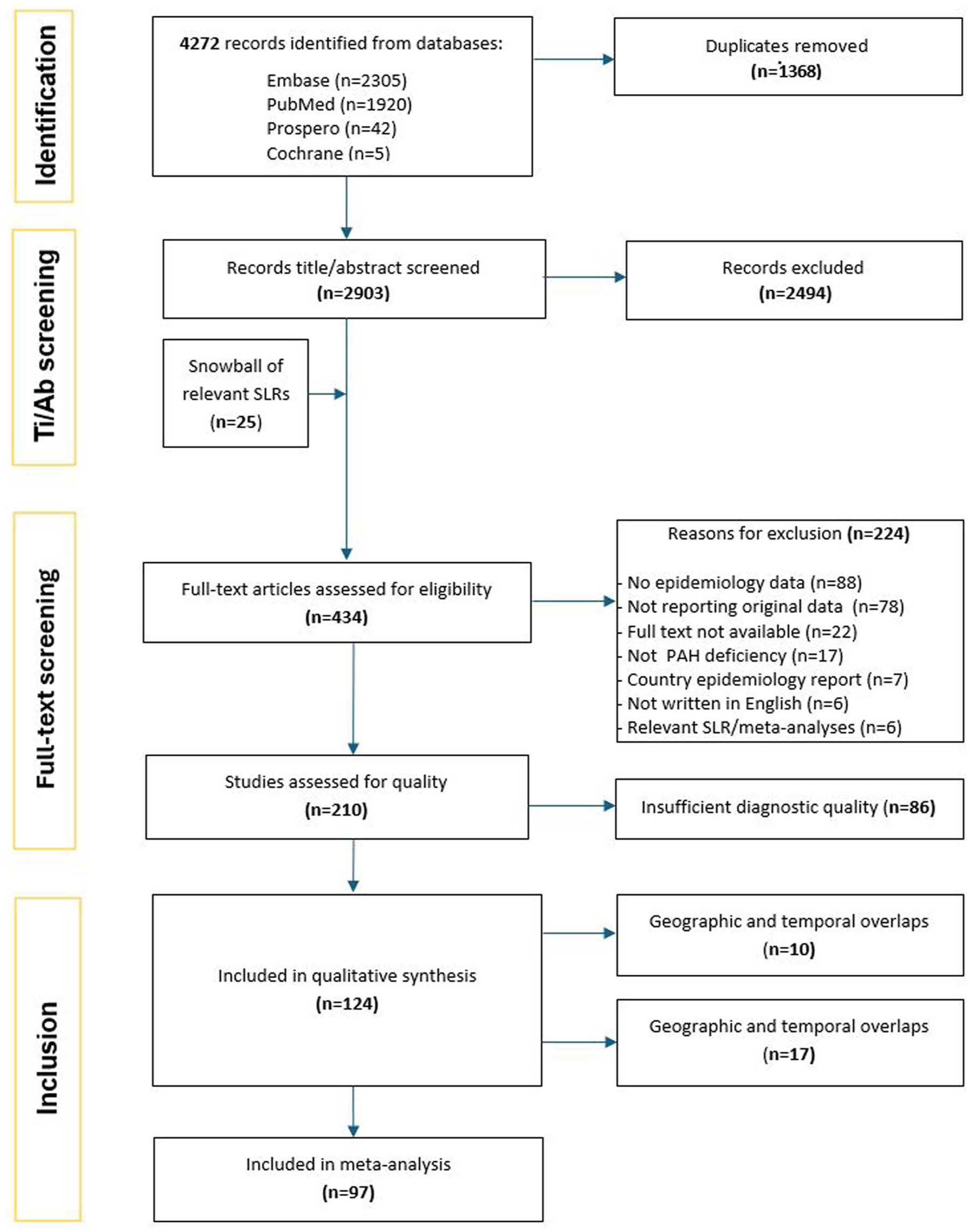
PRISMA chart of the systematic review process.
Data Synthesis
An overview of the quality assessment results is provided in Supplemental 2 Sections 3 and 4. Individual birth prevalence estimates were taken as the numerator (confirmed cases) and denominator (number of screened newborns) as reported by the original study; no individual-level patient data were accessed. A total of 208 birth prevalence estimates (from 86 studies) were excluded due to insufficient or poorly documented diagnostic methods. Two studies included mixed birth prevalence estimates, of which not all passed the diagnostic criteria; of these only those meeting the diagnostic criteria were used in the quantitative synthesis.25,26 Studies performed particularly well in detailing subjects and settings, with 386 birth prevalence estimates meeting this criterion. Sample size adequacy was achieved in 207 birth prevalence estimates, and 199 (from 124 studies) met the criteria for diagnostic methods. Birth prevalence per 10 000 newborns was calculated for various Phe cutoff groups based on population and confirmed cases, with results presented by region and country in Supplemental 3 Tables 1 to 8. Data for a 900 ± 100 μmol/l cutoff group were unavailable due to insufficient reporting. Recent publications on HPA epidemiology from North America and Europe are limited, while China has contributed robust studies with solid sample sizes. In contrast, recent studies from MENA, though numerous, often feature smaller sample sizes.
Meta-analysis
The removal of 35 birth prevalence estimates from (27 studies) due to geographic and temporal overlaps finally left 163 birth prevalence estimates (from 97 studies) to form the basis of birth prevalence calculations. A subgroup analysis was conducted for 92 birth prevalence estimates (from 47 studies) reporting data from adequately large cohorts.
The regional and global birth prevalence estimates, with and without stratification by Phe levels, are presented in Table 1, Figure 3, and Supplemental 3 Tables 9 and 10. The estimated global birth prevalence of HPA per 10 000 newborns was 0.84 (0.44-1.24) with regional weighting and 0.96 (0.81-1.13) without weighting. When applying sample size criteria, the regionally weighted global prevalence was 0.85 (0.45-1.25), while the unweighted global prevalence was 1.04 (0.82-1.29). Calculated birth prevalences by country are presented in Supplemental 3 Tables 1 to 8.
Global and Regional Birth Prevalences of Hyperphenylalaninemia Calculated with Meta-Analysis Per 10 000.


(A and B) Global and regional birth prevalences of HPA and classic PKU calculated with meta-analysis per 10 000.
Based on the Phe stratification levels, for classic PKU (1200 ± 200 μmol/l group), regional prevalences were reported highest in Europe [0.67 (0.28-1.23)], followed by North America [0.58 (0.34-0.87)]. Whereas the MENA region reported the highest rates for mild HPA not requiring treatment [360 ± 100 μmol/l group: 1.47 (0.97-2.06)].
In Asia, the birth prevalence of HPA was estimated at 0.78 (0.48-1.14) based on 23 studies,27-49 and 0.77 (0.41-1.26) when considering studies meeting sample size requirements (12 studies). For specific Phe cutoff groups, the prevalence was 1.03 (0.70-1.43) for the 120 ± 40 μmol/l group, 0.61 (0.34-0.96) for the 360 ± 100 μmol/l group, 0.17 (0.12-0.24) for the 600 ± 100 μmol/l group, and 0.16 (0.04-0.36) for the 1200 ± 200 μmol/l group.
In North America, the overall birth prevalence was 0.61 (0.47-0.78) based on 14 studies,50-63 but increased to 0.68 (0.41-1.02) when considering only studies that met sample size criteria (3 studies). For the 360 ± 100 μmol/l cutoff group, the prevalence was 0.52 (0.18-1.03), and for the 1200 ± 200 μmol/l cutoff group, it was 0.58 (0.34-0.87). Insufficient data were available to estimate prevalences for the 120 ± 40 and 600 ± 100 μmol/l groups.
In South America, the overall birth prevalence was estimated at 0.60 (0.31-0.96) based on 6 studies23,64-68 and 0.64 (0.33-1.05) when considering only studies met the sample size criteria (3 studies). The prevalence in the 600 ± 100 μmol/l group was 0.47 (0.30-0.68), and 0.48 (0.39-0.58) in the 1200 ± 200 μmol/l group. Data for the 120 ± 40 and 360 ± 100 μmol/l groups were insufficient.
In Europe, the overall birth prevalence of HPA was 1.20 (1.04-1.39) based on 32 studies24,26,54,69-99 and 1.29 (1.08-1.50) when accounting for studies that met the sample size criteria (19 studies). Within Phe cutoff groups, the prevalence was 1.21 (0.78-1.74) for the 360 ± 100 μmol/l group, 1.04 (0.83-1.29) for the 600 ± 100 μmol/l group, and 0.67 (0.28-1.23) for the 1200 ± 200 μmol/l group. There were insufficient data for the 120 ± 40 μmol/l group.
In the MENA region, the overall birth prevalence was 1.30 (0.90-1.76) based on 19 studies100-118 and 1.21 (0.74-1.79) based on studies that met the sample size criteria (9 studies). By Phe cutoff groups, the prevalence was 1.09 (0.56-1.76) for the 120 ± 40 μmol/l group, 1.47 (0.97-2.06) for the 360 ± 100 μmol/l group, 0.26 (0.10-0.46) for the 600 ± 100 μmol/l group, and 0.40 (0.13-0.76) for the 1200 ± 200 μmol/l group.
For Australia and Oceania, the birth prevalence was 0.93 (0.63-1.30) based on 2 studies.119,120 Data were insufficient to perform analyses by sample size criteria or Phe cutoff groups.
The individual meta-analysis for the United States (Supplemental 3 Table 7) showed an overall birth prevalence of 0.68 (0.53-0.84) based on 12 studies50,51,53,55-63,69 and 0.68 (0.41-1.02) when considering only studies that met the sample size criteria (3 studies). For the 360 ± 100 μmol/l group, the prevalence was 0.67 (0.24-1.30), and for the 1200 ± 200 μmol/l group, it was 0.58 (0.34-0.87). Data were insufficient for the 120 ± 40 and 600 ± 100 μmol/l groups. In Saudi Arabia (Supplemental 3 Table 8), the unstratified birth prevalence was 0.68 (0.52-0.86) overall based on 3 studies102,103,115 and 0.72 (0.56-0.90) based on those that met the sample size criteria. No data were available to perform analyses for individual Phe cutoff groups in Saudi Arabia.
Discussion
To the best of our knowledge, this is the most recent and comprehensive systematic review of the global epidemiology of HPA caused by PAH deficiency. A total of 124 articles were included in the qualitative phase, with 97 high-quality articles progressing to the quantitative phase, primarily based on data from newborn screening programs. Our study offers a novel contribution to HPA epidemiology by employing a rigorous meta-analysis framework incorporating minimum sample size calculations tailored for rare diseases. This practice aligns with recognized guidelines for epidemiological research on rare conditions and improves the reliability of regional and global prevalence estimates. 121 Additionally, it stratifies prevalence by specific Phe cutoff groups (120 ± 40, 360 ± 100, 600 ± 100, and 1200 ± 200 μmol/l), providing a detailed understanding of disease severity across regions.
Despite the rarity of the disease, robust newborn screening programs adopted in various countries enable precise data collection; hence, the epidemiology of HPA is well-documented in scientific literature. The global birth prevalence of HPA was 0.84 (0.44-1.24) per 10 000 births (regionally weighted) and 0.96 (0.81-1.13) per 10 000 births (non-weighted). Studies meeting sample size requirements showed a slightly higher prevalence of 0.85 (0.45-1.25) per 10 000 births (regionally weighted) and 1.04 (0.82-1.29) per 10 000 births (non-weighted). The sample-size restriction reduced imprecision (narrower CIs) for several region/cutoff strata; detailed comparisons are presented in Supplemental 3. North America exhibits moderate to high prevalence of classic PKU (Phe: 1200 μmol/l) with 0.58 (0.34-0.87) per 10 000 births, indicating that the region’s screening programs effectively identify more severe cases. This aligns with the National Organization for Rare Disorders (NORD) report of PKU occurring in approximately 1 in 13 500 to 19 000 newborns in the United States. 122 Europe consistently reports higher prevalence rates across several Phe cutoff groups, particularly at the higher Phe levels (600 and 1200 μmol/l). Similar to North America, this pattern likely reflects comprehensive and rigorous screening programs, which may be more effective at identifying both mild and severe cases of HPA.
The MENA region showed the highest prevalence in the unstratified group and the 360 μmol/l cutoff, with 1.30 (0.90-1.76) and 1.47 (0.97-2.06) per 10 000 births, respectively. This aligns with the higher consanguinity rates in the region, 123 though prevalence drops significantly at higher cutoffs, suggesting most cases detected are less severe. In contrast, Asia recorded the lowest HPA birth prevalence at 0.16 (0.04-0.36) per 10 000 newborns for the 1200 ± 200 μmol/l cutoff group. Asia exhibited a high prevalence at lower cutoffs but a sharp decline at higher cutoffs, highlighting potential regional variations in genetic factors, healthcare practices, and screening program implementation.
How this review extends prior work: Compared with the systematic reviews published in 202013,14 and 2021, 12 this review (1) includes studies identified up to 12 January 2024, capturing multiple new population-level screening reports (notably from China) published after those reviews; (2) applies pre-specified minimum sample-size criteria to reduce imprecision from small screened cohorts in pooled estimates; and (3) presents prevalence estimates stratified using a clinically conventional set of Phe cutoffs (≥360, ≥600, and ≥1200 µmol/l) plus an additional intermediate cutoff (≥120 µmol/l) to describe the broader distribution of elevated Phe at birth. These elements provide complementary information to earlier SLRs: the conventional cutoffs retain direct clinical relevance for treated disease burden, while the 120 µmol/l category helps describe the epidemiologic continuum and identify populations with high numbers of borderline elevations that may warrant monitoring or targeted research.
The global prevalence of HPA by Phe cutoff groups was reported recently by Foreman et al 12 in a comprehensive study. Additionally, 2 SLRs published within the past 5 years have reported on the global prevalence of PKU. Shoraka et al 14 identified studies reporting the birth prevalence of classical PKU in newborns and applied meta-analysis. Hillert et al 13 utilized unpublished information from local screening programs through a literature search to estimate the global prevalence of PKU in newborns. This paper applied the same calculation method for regionally weighted global prevalence as Foreman et al 12 by weighting each region by its relative contribution to the total population. Geographical regions applied by this paper, however, are somewhat different; instead of West-Pacific (China, Taiwan, Japan, Korea) and Southeast Asia (Thailand), as reported by Foreman et al, 12 we used a single Asia geographical region, while North America for the purposes of this paper includes the Caribbean. Our estimated birth prevalence figures are lower for North America and South America than those reported by Foreman et al 12 in the unstratified category. The difference may be attributed to the number of studies included in each review. The birth prevalence for North America was calculated based on 10 birth prevalence estimates (from 6 studies) in the Foreman et al 12 study, as opposed to 21 birth prevalence estimates (from 13 studies) in the present review. Similarly, the prevalence for South America was calculated based on 7 birth prevalence estimates (from 3 studies) in the Foreman et al 12 study, while the present study was based on 10 birth prevalence estimates (from 6 studies). To our knowledge, only Foreman et al 12 has previously estimated birth prevalence figures in multiple Phe cutoff groups. A comparison of these results is presented in Supplemental 3 Tables 11 and 12. Both global and regional comparisons of birth prevalence data in the unstratified and the 360 ± 100, 600 ± 100, and 1200 ± 200 μmol/l Phe cutoff groups are aligned with those of Foreman et al. 12 Birth prevalence in the 120 ± 40 μmol/l Phe cutoff group, which was not previously covered in other reviews, would potentially also include patients with no dietary restrictions required.
This study has several limitations. Our search was limited to English-language databases, which could potentially omit studies published only in regional languages. In practice, studies with large newborn-screening cohorts are typically published in English or include bilingual abstracts to reach the international community, hence any residual language bias is expected to be small. Geographically, significant gaps in data exist for regions such as Sub-Saharan Africa and Australia & Oceania, limiting global representativeness. No eligible studies werefound for African countries. During 1979 to 1986, a pilot newborn screening program for amino acidopathies, particularly PKU, was conducted by the Department of National Health and Population Development in South Africa. 124 From a cohort of 59 600 newborns screened over a period of 8 consecutive years, only 1 case of PKU was detected. The screening program was discontinued in 1986, concluding that the newborn screening for PKU and other amino acidopathies was not cost-effective and justifiable. Comprehensive data on PKU prevalence in other African countries are scarce, reflecting limited newborn screening programs covering PKU in the region. Data imbalances, with robust birth prevalence estimates from Europe and Asia but sparse data from low- and middle-income regions, hinder global extrapolation. The reliance on newborn screening data excludes late-diagnosed cases, potentially underestimating prevalence and missing point prevalence data in the general population. Additionally, the use of Phe cutoff groups introduces variability, as reported diagnostic thresholds differ across studies, potentially affecting consistency in prevalence estimates. Future research should address these limitations by expanding regional data collection, refining cutoff group standardization, and capturing undiagnosed cases.
Conclusions
The observed variability in HPA prevalence across regions and Phe cutoff levels underscores the need for standardized diagnostic criteria and screening practices globally. Standardization would facilitate more accurate comparisons and improve understanding of HPA’s global burden. Meta-analysis using adequately sized subgroups confirmed HPA’s rarity while reinforcing the reliability of reported prevalence estimates.
With a birth prevalence of 1.06 (0.78-1.37) per 10 000 births in the 120 ± 40 μmol/l cutoff group, HPA qualifies as a rare disease under definitions in both the U.S. (fewer than 200 000 people affected, translating to approx. 1 in 1700 individuals)20,125 and the European Union (fewer than 1 in 2000 individuals). 126 Even within this mild phenotype, HPA consistently meets the criteria for rare disease classification across all studied cutoff groups.
While substantial data exist on HPA’s epidemiology, gaps remain, particularly in Sub-Saharan Africa, where no epidemiological studies were identified. Moreover, while precise birth prevalence data are available, point prevalence in the general population remains unclear, potentially leading to underestimation and a high rate of cases lost to follow-up.
Our findings highlight the importance of accurately determining the global birth prevalence and severity distribution of HPA, particularly classic PKU and other forms requiring treatment, to guide efficient screening protocols and resource planning for disease management. By ensuring early diagnosis and prompt therapeutic intervention, and by dedicating efforts to continued research and monitoring, healthcare systems can prevent severe complications, including irreversible neurocognitive damage, and significantly enhance the quality of life for individuals affected by this rare disorder.
Supplemental Material
sj-docx-1-gph-10.1177_30502225251414610 – Supplemental material for Epidemiology of Hyperphenylalaninemia: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-gph-10.1177_30502225251414610 for Epidemiology of Hyperphenylalaninemia: A Systematic Review and Meta-Analysis by Tamás István Dóczi, Shailja Vaghela, George Dennis Obeng, David Ameyaw, Tamás Zelei, Zoltán Vokó, Vanja Sikirica, Orsolya Varga, Gábor Kovács, Judit Józwiak-Hagymásy and Sue Perera in Sage Open Pediatrics
Supplemental Material
sj-docx-2-gph-10.1177_30502225251414610 – Supplemental material for Epidemiology of Hyperphenylalaninemia: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-2-gph-10.1177_30502225251414610 for Epidemiology of Hyperphenylalaninemia: A Systematic Review and Meta-Analysis by Tamás István Dóczi, Shailja Vaghela, George Dennis Obeng, David Ameyaw, Tamás Zelei, Zoltán Vokó, Vanja Sikirica, Orsolya Varga, Gábor Kovács, Judit Józwiak-Hagymásy and Sue Perera in Sage Open Pediatrics
Supplemental Material
sj-docx-3-gph-10.1177_30502225251414610 – Supplemental material for Epidemiology of Hyperphenylalaninemia: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-3-gph-10.1177_30502225251414610 for Epidemiology of Hyperphenylalaninemia: A Systematic Review and Meta-Analysis by Tamás István Dóczi, Shailja Vaghela, George Dennis Obeng, David Ameyaw, Tamás Zelei, Zoltán Vokó, Vanja Sikirica, Orsolya Varga, Gábor Kovács, Judit Józwiak-Hagymásy and Sue Perera in Sage Open Pediatrics
Supplemental Material
sj-docx-4-gph-10.1177_30502225251414610 – Supplemental material for Epidemiology of Hyperphenylalaninemia: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-4-gph-10.1177_30502225251414610 for Epidemiology of Hyperphenylalaninemia: A Systematic Review and Meta-Analysis by Tamás István Dóczi, Shailja Vaghela, George Dennis Obeng, David Ameyaw, Tamás Zelei, Zoltán Vokó, Vanja Sikirica, Orsolya Varga, Gábor Kovács, Judit Józwiak-Hagymásy and Sue Perera in Sage Open Pediatrics
Supplemental Material
sj-docx-5-gph-10.1177_30502225251414610 – Supplemental material for Epidemiology of Hyperphenylalaninemia: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-5-gph-10.1177_30502225251414610 for Epidemiology of Hyperphenylalaninemia: A Systematic Review and Meta-Analysis by Tamás István Dóczi, Shailja Vaghela, George Dennis Obeng, David Ameyaw, Tamás Zelei, Zoltán Vokó, Vanja Sikirica, Orsolya Varga, Gábor Kovács, Judit Józwiak-Hagymásy and Sue Perera in Sage Open Pediatrics
Footnotes
Acknowledgements
During the preparation of this manuscript, the authors used GPT 4o for the purposes of improving language, grammar and structure of this study. The authors declare that no AI tool was applied to produce content such as generating references, text, images or any other form of content. The authors have reviewed and edited the output and take full responsibility for the content of this publication.
Ethical Considerations
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Author Contributions
Conceptualization, S.V., T.Z., V.S., J.J., and S.P.; methodology, T.D., S.V., T.Z., J.J., and S.P.; formal analysis, Z.V. and G.K.; investigation, T.D., G.O., D.A., O.V., and G.K.; data curation, T.D., G.O., D.A., O.V., and G.K.; validation, T.D., G.O., D.A., O.V., and G.K.; writing—original draft preparation, T.D., G.O., and D.A.; writing—review and editing, S.V., T.Z., V.S., J.J., and S.P.; supervision, S.V., T.Z., V.S., J.J., and S.P.; project administration, S.V., T.Z., J.J., and T.D. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by MODERNA, INC.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:Moderna employee-authors (VS and SP) and a contracted consultant (SV) contributed to the conceptualization, methodology, supervision and critical review of the manuscript in their scientific capacities. The sponsor had no additional influence over the collection, analysis or interpretation of data. The content of this manuscript, as well as the views and opinions expressed therein are those of the Authors and not the organizations that employ them. All authors—including those affiliated with the sponsor—jointly decided to submit the manuscript.
Data Availability Statement
The original data supporting this study are provided in the Supplementary Materials; further inquiries can be directed to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
