Abstract
Background:
Pain is a common symptom at cancer diagnosis and often worsens with treatment and disease progression. Comprehensive evidence on its prevalence and severity in Africa remains limited.
Objectives:
This systematic review and meta-analysis aimed to estimate the prevalence and severity of cancer-related pain among adult cancer patients in Africa using available evidence.
Methods:
Google Scholar, PubMed, and the Cochrane Library were searched for articles that reported the prevalence of pain in cancer patients in African countries. The methodological quality of included studies was assessed using a quality assessment tool for prevalence studies, and the certainty of the evidence was assessed using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) framework. Pooled estimates of the prevalence and severity of cancer-related pain were calculated. Study heterogeneity was assessed using I², while sensitivity analyses were performed to test the robustness of the findings.
Results:
This meta-analysis included 23 studies conducted in African nations. In all included studies, 5378 patients with cancer at various stages of anticancer treatment were included. The pooled prevalence of pain was 76% (95% CI: 71%−81%,
Conclusion:
More than three-quarters of patients suffer from cancer-related pain, with a substantial proportion experiencing moderate-to-severe levels with moderate certainty. Considerable variability exists in pain prevalence and severity across studies and regions of Africa. As a result, findings should be interpreted with caution. Improving access to analgesics may help alleviate pain and enhance quality of life for cancer patients.
Introduction
Pain is the most common, feared, untreated, and debilitating symptom in patients with a diagnosis of cancer. 1 Cancer-related pain is either persistent or intermittent and is associated with tumor progression. 2 This pain can result from tissue compression and may also arise from diagnostic procedures or treatments such as surgery, chemotherapy, or radiotherapy.3 –5 Additionally, cancer-related pain may be caused by tumor metastasis, which can inflame or erode bones, viscera, or nerves. 6
Among patients with a diagnosis of cancer, pain has been reported in 55%−59% in patients undergoing anticancer treatment, 64%−70% in those with advanced metastatic or terminal illness, 53% across all disease stages, and 33%−39.3% in patients who have completed curative treatment. 7 Categorized by region, a median of 51.9% in Asia, 42.1% in Latin America, 1 more than 50% in Europe and North America 8 and 50%−92.2% in Africa.9 –11 Categorized by severity, 33%−58% report moderate-to-severe pain.8,12 These highlight the significant burden of cancer-related pain across various stages of the disease, treatment phases, and geographical areas.
Poorly treated pain remains a significant issue for many cancer patients. 13 Some studies suggest that inadequately managed pain may decrease survival rates in cancer patients. 14 Despite the development of various pain treatment modalities and guidelines for cancer-related pain management by the World Health Organization and other pain societies, reported levels of cancer-related pain remain high, particularly in low-resource settings. Several contextual factors may contribute to challenges in effective pain management in these settings, including patients’ reluctance to report pain, fear of the side effects of opioids, and inadequate knowledge and practices regarding pain management among health professionals. 15 A small number of African nations have national palliative care policies or incorporated palliative care services into their healthcare systems.16,17 In this context, opioid availability and consumption remain limited,18,19 highlighting systemic barriers to comprehensive pain management. In addition, the long distance to facilities and inadequately trained health workforce, particularly in rural centers, limit access to analgesic medications and palliative care services. 20 Furthermore, high transport costs, a weak referral system, and out-of-pocket payments restrict patients from seeking and receiving adequate pain management services.21,22
Previous meta-analyses have largely focused on high- and middle-income countries, including a limited number of studies from Africa. Existing African studies are heterogeneous, small in sample size, and geographically limited. To the authors’ knowledge, no pooled analysis has reported the prevalence and severity of cancer-related pain across Africa. This meta-analysis addresses this gap by including studies from West Africa, East Africa, and Southern Africa, estimating both prevalence and severity, and performing regional subgroup analyses, thereby providing evidence to inform cancer-related pain management and health system planning in low-resource settings.
Methods
Registration
This systematic review and meta-analysis was registered in the Research Registry with the identifying code of reviewregistry1977.
Search strategy
A literature review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. 23 Two major electronic databases—PubMed and the Cochrane Library—were systematically searched to identify relevant studies on the prevalence of pain among cancer patients in Africa. Additionally, Google Scholar was explored to capture further pertinent literature. The Population (P), Exposure (E), and Outcome (O), (PEO) framework for observational and cross-sectional studies was used to guide the formulation of the research question and to structure the literature search strategy. Population (P) comprises cancer patients who have been diagnosed with any type of malignancy and receive treatment in different healthcare settings in African nations. Acute or chronic cancer-related pain and cancer-related conditions (diagnosis, treatment, stage) constitutes the exposure (E). The main outcome (O) is the prevalence of cancer-related pain, including the percentage of patients who experience pain and the percentage severity of pain classifications (mild pain, moderate pain, and severe pain). The initial literature search conducted on June 10, 2025 was restricted to studies published in English. However, a subsequent comprehensive search conducted on January 25, 2026 without language restrictions. A sensitivity check confirmed that no additional eligible studies were identified between the initial and updated searches.
Medical Subject Headings (MeSH) and free-text terms were combined to create the search strategy, using Boolean operators (AND, OR) to maximize sensitivity and specificity. The search terms included “cancer,” “tumor,” “neoplasm,” “pain,” “prevalence,” and “epidemiology” were used to access eligible studies. The detailed search strategy for databases and Google Scholar is provided in (Supplemental Table 1). To ensure inclusive regional representation, the names of specific African nations were included in the search, such as “prevalence of pain in cancer patients.” The reference lists of all included studies were manually reviewed to find more relevant studies that were not identified through electronic searching.
Eligibility criteria
Inclusion criteria
This meta-analysis included studies conducted in African countries that reported the prevalence or severity of cancer-related pain among patients of all genders, across all cancer types and stages, and at any phase of anticancer treatment.
Exclusion criteria
Nonpeer-reviewed studies, editorials, case reports, studies with overlapping populations, scoping reviews and narrative reviews that did not report the prevalence or severity of pain quantitatively were excluded from the meta-analysis.
Study selection
Based on the inclusion and exclusion criteria, two investigators (GDG and AGA) screened the title and abstract. Full texts of potentially eligible studies were independently assessed by the same investigators. Any disagreements during screening were resolved through discussion with the principal investigator (MAT).
Data extraction
Two researchers (GDG and AGA) developed and piloted the data extraction form in accordance with the study objectives. Data extraction was performed independently by the same two investigators, and discrepancies were resolved by discussion with the principal investigator (MAT). The extracted data included methodological characteristics, participant demographics (gender distribution, mean or median age), and bibliographic information (first author’s name and year of publication). The extraction form also included additional fields for the geographic area of the African continent where the study was conducted (West Africa, East Africa, and Southern Africa). Furthermore, each included study’s quality appraisal score, the presence of cancer-related pain, and the key findings or conclusions of the study were methodically documented.
Measurement of outcomes
The primary outcome of this meta-analysis was the prevalence of pain in cancer patients in Africa, particularly in West Africa, East Africa, and Southern Africa and the secondary outcome was the severity of pain in these regions. Different studies used various pain assessment tools, including a tool for the diagnosis of neuropathic pain, 24 the Memorial Symptom Assessment Schedule Short Form (MSAS-SF), 25 Numeric Rating Scale,9,26 –32 Verbal Rating Scale,33,34 Brief Pain Inventory (BPI),35 –41 Universal Pain Assessment Tool, 42 Shoulder Pain and Disability Index, 43 Quality of Life and Functional Scale, 44 and unspecified pain assessment tool. 45 Despite differences in pain assessment tools, all were developed to measure the presence or absence of pain. These tools have been widely validated and applied in clinical and epidemiological research. For this meta-analysis, pain prevalence was extracted as yes/no to allow comparability across studies, while pain severity was extracted using each study’s original definitions, reflecting differences in severity classifications and cut-off points.
Methodological quality assessment of included studies
The methodological quality of included studies was assessed using standardized criteria for prevalence studies across four domains: representativeness of the population, quality of data, reporting of methods and results, and definition of pain prevalence. The assessment was based on 11-item checklist incorporating sample representativeness, response rates, use of validated data collection tools, completeness of reporting (patient characteristics, disease stage, sample size), and whether prevalence recall periods were stated. 7 Each item was assigned a weighted score according to predefined criteria, yielding a maximum possible score of 20 points. Based on total scores, the included studies were classified as low quality (⩽10 points), moderate quality (11–15 points), and high quality (⩾16 points) 46 (Supplemental Table 1). Methodological quality assessment was independently conducted by two investigators (ATA and DGD). Disagreements were resolved through discussion with the last author (TBA) (Supplemental Table 2).
Assessment of certainty of evidence (GRADE)
The certainty of evidence was assessed using the GRADE framework across five domains: risk of bias, inconsistency, indirectness, imprecision, and publication bias. Because the outcomes were pooled prevalence and severity, evidence was initially rated as high and was subsequently downgraded to serious or very serious limitations were identified in any domain. In GRADE, a serious limitation resulted in downgrading by one level, whereas a very serious limitation resulted in downgrading by two levels, and the cumulative downgrades across domains determined the overall certainty of evidence. Risk of bias was assessed based on the methodological quality of the included studies, and inconsistency was evaluated using the
Statistical analyses
Statistical analyses were performed using STATA version 17 to ensure accurate computations. Within each defined patient group, the pooled prevalence and percentage severity of pain were calculated. Heterogeneity among studies was evaluated using the I² statistic, with values <25% indicating low heterogeneity, 25 to 50% indicating moderate heterogeneity, and >50% indicating substantial heterogeneity. 48 Due to substantial heterogeneity, pooled prevalence and prevalence estimates were reported using a random-effects model.
Subgroup analyses were conducted to explore variation in cancer-related pain prevalence by study setting (single-center vs. multicenter), sample size (less than 100 versus 100 or more), study quality (moderate vs. high), and regional distribution (West Africa, East Africa, Southern Africa). Sensitivity analysis using a leave-one-out approach assessed the robustness of the findings. Meta-regression was performed to identify factors contributing to differences in prevalence across studies. Publication bias was evaluated visually with funnel plots and statistically using Egger’s regression test. Findings are presented as point estimates with 95% confidence intervals (CIs), and statistical significance was defined as
Results
Literature search
Through database and citation searches, 41,945 records were identified, of which 23 studies were ultimately included in the statistical analysis (Figure 1).

Selection process for included studies in the meta-analysis.
Characteristics of included studies
This systematic review incorporated 23 studies conducted at different hospital settings in various parts of Africa. In the included studies, 5,378 patients with histopathologically confirmed cancer and at different phases of treatment. The majority of patients, 3,674, were females. The meta-analysis incorporated studies conducted on different types of cancer, including breast, gynecologic, head and neck, lung and bronchi, Kaposi’s sarcoma, genitourinary, gastrointestinal, hematologic, bone, cancer of unknown primary origin, skin, other cancers, and mixed unspecified cancers. The studies were primarily conducted to determine prevalence of cancer-related pain,24,26 –28,30,32, 35 –38,43 to determine the pain management strategies29,34 to identify the adequacy of pain treatment,11,41 to measure the impact of cancer-related pain on health-related quality of life,31,33,40,44 to identify the barriers to pain management 37 to determine the pain treatment outcomes,9,39 and to identify the clinical presentations of patients with a diagnosis of cancer.25,45 The methodological quality assessment score for the majority of the included studies was greater than 15. Most of the studies included more than three types of cancer, with some specifically focusing on patients with breast cancer and cervical cancer. The majority of included studies were conducted in East Africa (Table 1).
Characteristics of included studies.

NR: Not reported, Age (mean/median).
In pain severity and overall prevalence categories, minor difference was due to approximation.
Certainty of evidence (GRADE)
For all pain outcomes, including the prevalence of mild, moderate, and severe pain, as well as the pooled prevalence of cancer-related pain, this review initially rated the certainty of evidence as high, in accordance with WHO GRADE guidance for prevalence studies, which suggests a starting at high certainty for population-based representative studies. However, the certainty of evidence for each outcome was downgraded by two levels, resulting in moderate certainty due to significant inconsistency (high heterogeneity, with I² values ranging from 94.5%−98.1%) and a significant risk of bias arising from consistently variable methodological quality concerns across the included studies in the meta-analysis (Table 2).
Certainty of evidence.

Prevalence of cancer-related pain
Across the included studies from West Africa, East Africa, and Southern, the reported prevalence of cancer-related pain varied widely, ranging from approximately 52%−93%, indicating substantial between-study variability. Consistent with this, heterogeneity was extremely high (

Forest plot for prevalence of cancer-related pain in Africa.
Severity of cancer-related pain
Among the included studies, 14 assessed pain severity using three categories: mild, moderate, and severe. The results showed that the pooled prevalence of mild pain was 27% (95% CI: 20%−34%), moderate pain was 44% (95% CI: 34%–54%), and severe pain was 25% (95% CI: 15%−35%).
Subgroup analyses
Subgroup analyses were conducted by study setting, sample size, methodological quality, and geographic region to explore potential sources of heterogeneity in cancer-related pain prevalence across Africa. The prevalence estimates were similar between multicenter (76%, 95% CI: 64%–89%,
Sensitivity analysis
Leave-one-out sensitivity analysis indicated that excluding any single study did not materially affect the pooled prevalence. The pooled prevalence remained stable, ranging from 0.75 (95% CI: 0.70–0.81) to 0.77 (95% CI: 0.71–0.82), with all estimates remaining statistically significant (

Leave-one-out sensitivity analysis for prevalence of cancer-related pain in Africa.
Meta-regression
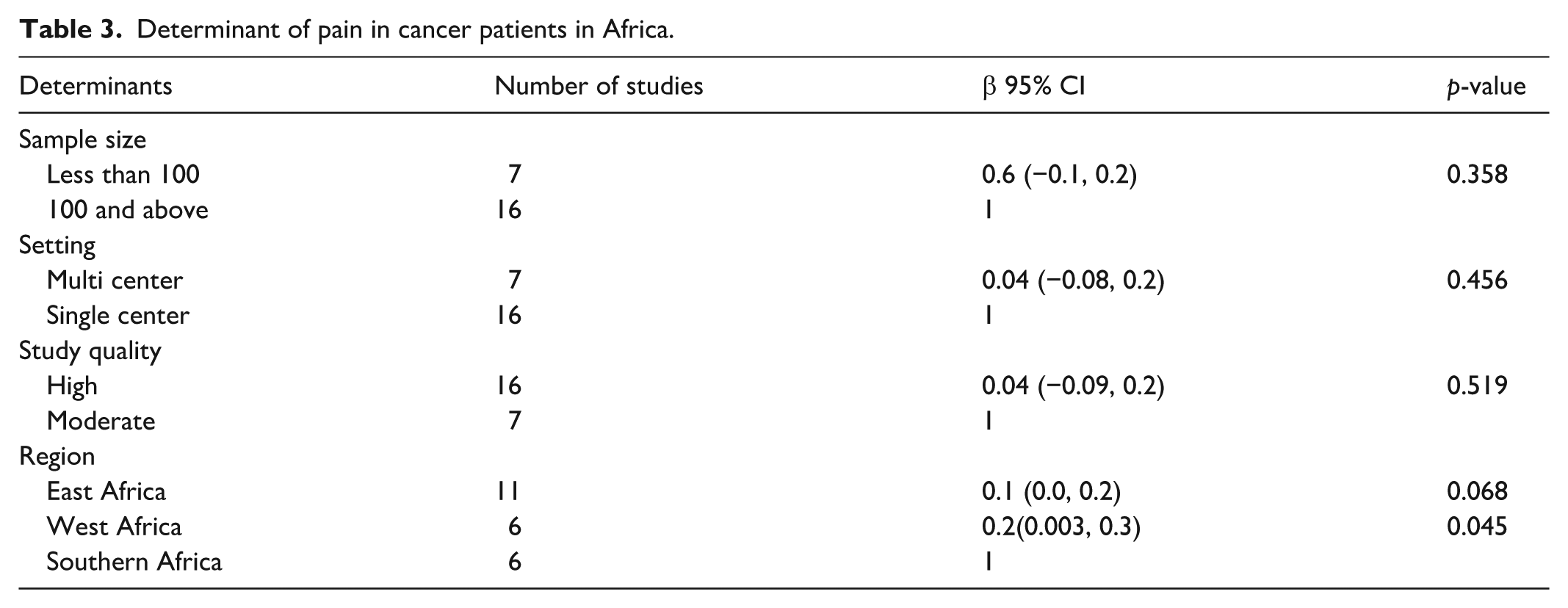
Exploratory meta-regression analyses were performed to assess whether study-level characteristics explained variability in the prevalence of cancer-related pain. Sample size, study quality, and study setting were not significantly associated with pooled prevalence. Subgroup analyses indicated a trend toward higher pooled prevalence estimates in West Africa relative to East and Southern Africa, with substantial between study heterogeneity (Table 3).
Determinant of pain in cancer patients in Africa.
Publication bias
Funnel plot and Egger’s test
The funnel plot indicated that the included studies are mostly outside the funnel, and there is some asymmetry, with most studies deviating to the right of the estimated effect size (Supplemental Figure 5). Egger’s test indicates no statistically significant evidence of small study effects or publication bias, with a beta coefficient of −3.37, a standard error of 2.14, and a
Discussion
This meta-analysis was conducted to provide comprehensive evidence on the observed pooled prevalence and severity of cancer-related pain across the included studies. It examined studies involving patients with a cancer diagnosis at various stages of anticancer treatment, using studies from West Africa, East Africa, and Southern Africa. The meta-analysis comprised 23 studies, all of which were assessed as having moderate to high methodological quality. The results demonstrate that more than 75% of patients reported cancer-related pain, with approximately 70% of those experiencing moderate-to-severe intensity. However, these pooled estimates were accompanied by substantial heterogeneity.
The significant heterogeneity within, and among subgroups suggests that study-level characteristics are insufficient to explain the observed variability, even though high prevalence estimates are consistent. This heterogeneity may be caused by variations in cancer types, disease stages, pain assessment tools, and healthcare facilities across countries and settings. Furthermore, the significant heterogeneity could be explained by variable definitions of pain without specifying the duration and etiology. Pain related with diagnostic or therapeutic interventions may contribute to inconsistent prevalence of pain. In addition, the significant heterogeneity could be explained by inability to differentiate tumor related pain and treatment related pain and the type of pain (somatic pain and neuropathic pain). Therefore, the pooled prevalence estimates should be interpreted as average values not universally values.
Regardless of study setting, sample size, or methodological quality, the subgroup analyses show that the prevalence of cancer-related pain in Africa is consistently high. This indicates that pain is still a major issue among African cancer patients. Sensitivity analyses showed that no single study had a disproportionate impact on the overall prevalence and supporting the strength of pooled estimates. Although Egger’s test did not indicate statistically significant publication bias, funnel plot asymmetry may suggest differences across study populations rather than selective reporting.
Regional differences in pain prevalence, particularly the higher pooled estimate observed in West Africa, should be interpreted cautiously. These regional differences may reflect unmeasured contextual factors, such as variation in palliative care development and resource availability, which could not be examined directly in this meta-analysis.49,50
The pooled prevalence of pain in this meta-analysis is higher than that reported in previous meta-analysis; 53% across all disease stages, 55% during anticancer treatment, to 66% in advanced or metastatic diseases. 51 Furthermore, the pain prevalence identified in the present study exceeds that reported in a meta-analysis of patients with solid tumors, where the prevalence was estimated at 47%. 52 Similarly, the finding of this meta-analysis is higher than those of an earlier meta-analysis of observational studies examining persistent postsurgical pain following breast cancer surgery, which reported a prevalence of 35% and an average pain intensity of 3.9 cm on a 10-cm visual analogue scale. 53 The discrepancy between the findings of the current meta-analysis and those of previous meta-analyses may be attributed to several factors. One important explanation is the inclusion of a larger number of studies in earlier systematic reviews. 54 Moreover, earlier studies largely included researches particularly from middle-income and high-income countries with comparatively greater resources for cancer-related pain management. 55 On the other hand, the current meta-analysis is carried out in Africa where there may be less access to institutional guidelines, and pain management services.55,56 The higher prevalence of pain in Africa compared to other regions may be due to variations in healthcare infrastructure, access to pain management specialists, knowledge, and attitude of cancer-related pain.20,57,58
According to a more thorough analysis of 14 studies that reported the severity classification of cancer-related pain, 27% of cancer patients have mild pain that can be controlled with common analgesic medications, including nonsteroidal anti-inflammatory drugs (NSAIDs) or acetaminophen. 59 This meta-analysis found that 44% of cancer patients in Africa experienced moderate pain and 25% experienced severe pain, both of which can negatively affect health-related quality of life, daily functioning, and contribute to anxiety and depression. These findings emphasize the significant burden of cancer-related pain in Africa and the pressing need for efficient pain management techniques.60,61 These findings indicate that the burden of cancer-related pain in Africa is higher than reported in previous studies conducted in other regions and in global focus. For instance, substantial proportion of patients reported moderate-to-severe pain during all stages of anticancer treatment, according to a global meta-analysis of cancer-related pain prevalence. 62
The significant burden of cancer-related pain identified in Africa, particularly in West African, East Africa, and Southern Africa indicate a substantial unmet need for effective cancer-related pain management. These findings demonstrate the importance of strengthening pain assessment, expanding access to palliative care, and improving the availability and appropriate use of analgesic therapies. However, rather than being based only on these prevalence estimates, policy and practice recommendations in Africa should be supported by implementation research and context-specific data.
Strength and limitations of the study
The meta-analysis possesses several key strengths. First, it addresses the critical issue of poorly managed pain in West Africa, East Africa, and Southern Africa by offering a thorough pooled estimate of the frequency and intensity of pain in cancer patients. Second, the inclusion of a large patient cohort from diverse African nations enhances the precision of these estimates, supported by the results of Egger’s test, which show nonsignificant publication bias and small study effects. Third, the meta-analysis may help prioritize pain management resources in low-income settings by classifying pain according to severity. Fourth, it identifies regional variations in the prevalence of cancer-related pain which could help put regional variations in the burden of cancer-related pain into perspective. However, certain limitations exist. First, the substantial heterogeneity may suggest significant variability of cancer related pain between population and setting. Second, underrepresentation of Central and Northern Africa may limit the generalizability to the whole African continent. Third, differences in reporting procedures, recall periods, severity classifications, and pain measurement tools among studies may have impacted comparability and increased measurement bias. These variables could lead to an overestimation or underestimation of the frequency and intensity of pain. Fourth, meta-regression did not take into account certain possible factors that could influence pain related to cancer. These include the type of cancer, the analgesics used, and the stage of the cancer. Fifth, the initial search was limited to studies published in English; however, a subsequent comprehensive search without language restrictions identified no additional eligible studies. Nevertheless, the potential for language bias cannot be entirely exclude. Lastly, the inability to differentiate between pain types (acute vs. chronic, neuropathic vs. somatic, treatment- vs. tumor-related) may contributed to the observed heterogeneity.
Conclusion
Cancer-related pain is a significant and often inadequately treated public health issue across African regions, with prevalence generally comparable between West, and East Africa but lower in Southern Africa. The pooled estimates represent averages across highly heterogeneous contexts and should not be interpreted as a single definitive value. Overall, these findings underscore the substantial burden of cancer-related pain while emphasizing cautious interpretation.
Recommendations
The results of this meta-analysis suggest that oncology care in West Africa, East Africa, and Southern Africa should place more emphasis on cancer-related pain management. At all levels of care, protocols should incorporate routine pain assessment and management. Healthcare providers should routinely assess pain using validated and standardized tools and modify treatment based on reported pain levels. Governments and health systems should examine supply chain and regulatory barriers that restrict access to necessary analgesics, balancing responsible opioid use and effective pain relief. Furthermore, it should be a top priority to expand and incorporate palliative care services into national cancer control plans, particularly in underserved and rural areas where access to care is restricted. Training healthcare professionals in pain assessment and management techniques, as well as initiating pain management early in the course of the disease, is necessary. These recommendations should be interpreted in light of the observational nature of the evidence and the absence of data from North and Central African regions. Further research is needed to clarify regional differences in cancer-related pain, barriers to pain management, intervention effectiveness, and pain patterns across cancer types.
Supplemental Material
sj-docx-1-smo-10.1177_20503121261434145 – Supplemental material for Epidemiology of cancer-related pain in Africa: A systematic review and meta-analysis of studies from 2001 to 2024
Supplemental material, sj-docx-1-smo-10.1177_20503121261434145 for Epidemiology of cancer-related pain in Africa: A systematic review and meta-analysis of studies from 2001 to 2024 by Molla Amsalu Tadesse, Getachew Mekete Deress, Gezahagn Demsu Gedefaw, Eniyew Assimie Alemu, Abere Gebru Abuhay, Asnake Tadesse Abate, Tsehayu Melak Siyoum, Daniel Getaneh Damtie, Agernesh Dereje Misker, Habtie Bantider Wubet, Walelign Asmie Afework, Amanu Addis Melesew, Negesse Zurbachew Gobezie, Biruk Demissie, Begizew Yimenu Mekuriaw and Temesgen Birlie Asmare in SAGE Open Medicine
Supplemental Material
sj-docx-2-smo-10.1177_20503121261434145 – Supplemental material for Epidemiology of cancer-related pain in Africa: A systematic review and meta-analysis of studies from 2001 to 2024
Supplemental material, sj-docx-2-smo-10.1177_20503121261434145 for Epidemiology of cancer-related pain in Africa: A systematic review and meta-analysis of studies from 2001 to 2024 by Molla Amsalu Tadesse, Getachew Mekete Deress, Gezahagn Demsu Gedefaw, Eniyew Assimie Alemu, Abere Gebru Abuhay, Asnake Tadesse Abate, Tsehayu Melak Siyoum, Daniel Getaneh Damtie, Agernesh Dereje Misker, Habtie Bantider Wubet, Walelign Asmie Afework, Amanu Addis Melesew, Negesse Zurbachew Gobezie, Biruk Demissie, Begizew Yimenu Mekuriaw and Temesgen Birlie Asmare in SAGE Open Medicine
Supplemental Material
sj-docx-3-smo-10.1177_20503121261434145 – Supplemental material for Epidemiology of cancer-related pain in Africa: A systematic review and meta-analysis of studies from 2001 to 2024
Supplemental material, sj-docx-3-smo-10.1177_20503121261434145 for Epidemiology of cancer-related pain in Africa: A systematic review and meta-analysis of studies from 2001 to 2024 by Molla Amsalu Tadesse, Getachew Mekete Deress, Gezahagn Demsu Gedefaw, Eniyew Assimie Alemu, Abere Gebru Abuhay, Asnake Tadesse Abate, Tsehayu Melak Siyoum, Daniel Getaneh Damtie, Agernesh Dereje Misker, Habtie Bantider Wubet, Walelign Asmie Afework, Amanu Addis Melesew, Negesse Zurbachew Gobezie, Biruk Demissie, Begizew Yimenu Mekuriaw and Temesgen Birlie Asmare in SAGE Open Medicine
Supplemental Material
sj-docx-4-smo-10.1177_20503121261434145 – Supplemental material for Epidemiology of cancer-related pain in Africa: A systematic review and meta-analysis of studies from 2001 to 2024
Supplemental material, sj-docx-4-smo-10.1177_20503121261434145 for Epidemiology of cancer-related pain in Africa: A systematic review and meta-analysis of studies from 2001 to 2024 by Molla Amsalu Tadesse, Getachew Mekete Deress, Gezahagn Demsu Gedefaw, Eniyew Assimie Alemu, Abere Gebru Abuhay, Asnake Tadesse Abate, Tsehayu Melak Siyoum, Daniel Getaneh Damtie, Agernesh Dereje Misker, Habtie Bantider Wubet, Walelign Asmie Afework, Amanu Addis Melesew, Negesse Zurbachew Gobezie, Biruk Demissie, Begizew Yimenu Mekuriaw and Temesgen Birlie Asmare in SAGE Open Medicine
Supplemental Material
sj-docx-5-smo-10.1177_20503121261434145 – Supplemental material for Epidemiology of cancer-related pain in Africa: A systematic review and meta-analysis of studies from 2001 to 2024
Supplemental material, sj-docx-5-smo-10.1177_20503121261434145 for Epidemiology of cancer-related pain in Africa: A systematic review and meta-analysis of studies from 2001 to 2024 by Molla Amsalu Tadesse, Getachew Mekete Deress, Gezahagn Demsu Gedefaw, Eniyew Assimie Alemu, Abere Gebru Abuhay, Asnake Tadesse Abate, Tsehayu Melak Siyoum, Daniel Getaneh Damtie, Agernesh Dereje Misker, Habtie Bantider Wubet, Walelign Asmie Afework, Amanu Addis Melesew, Negesse Zurbachew Gobezie, Biruk Demissie, Begizew Yimenu Mekuriaw and Temesgen Birlie Asmare in SAGE Open Medicine
Supplemental Material
sj-docx-6-smo-10.1177_20503121261434145 – Supplemental material for Epidemiology of cancer-related pain in Africa: A systematic review and meta-analysis of studies from 2001 to 2024
Supplemental material, sj-docx-6-smo-10.1177_20503121261434145 for Epidemiology of cancer-related pain in Africa: A systematic review and meta-analysis of studies from 2001 to 2024 by Molla Amsalu Tadesse, Getachew Mekete Deress, Gezahagn Demsu Gedefaw, Eniyew Assimie Alemu, Abere Gebru Abuhay, Asnake Tadesse Abate, Tsehayu Melak Siyoum, Daniel Getaneh Damtie, Agernesh Dereje Misker, Habtie Bantider Wubet, Walelign Asmie Afework, Amanu Addis Melesew, Negesse Zurbachew Gobezie, Biruk Demissie, Begizew Yimenu Mekuriaw and Temesgen Birlie Asmare in SAGE Open Medicine
Supplemental Material
sj-docx-7-smo-10.1177_20503121261434145 – Supplemental material for Epidemiology of cancer-related pain in Africa: A systematic review and meta-analysis of studies from 2001 to 2024
Supplemental material, sj-docx-7-smo-10.1177_20503121261434145 for Epidemiology of cancer-related pain in Africa: A systematic review and meta-analysis of studies from 2001 to 2024 by Molla Amsalu Tadesse, Getachew Mekete Deress, Gezahagn Demsu Gedefaw, Eniyew Assimie Alemu, Abere Gebru Abuhay, Asnake Tadesse Abate, Tsehayu Melak Siyoum, Daniel Getaneh Damtie, Agernesh Dereje Misker, Habtie Bantider Wubet, Walelign Asmie Afework, Amanu Addis Melesew, Negesse Zurbachew Gobezie, Biruk Demissie, Begizew Yimenu Mekuriaw and Temesgen Birlie Asmare in SAGE Open Medicine
Footnotes
ORCID iDs
Author contributions
MAT conceived the study, developed the protocol, performed meta-analysis, and drafted the manuscript. MAT, GDG, and AGA designed the search strategy, managed reference databases, participated in study selection and data extraction. ATA, TBA, and DGD participated in the quality assessment of included studies. BD, HBW, WAA, and AAM revised the first draft manuscript. EAA, TMS, ADM, NZ, BYM, and GMD supported statistical analysis and interpreted the findings, TBA supervised the meta-analysis. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data generated and analyzed for this study can be retrieved in the databases were individual articles were searched and can be submitted by the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
Research registration
It was registered in the Research Registry with the unique identifier “reviewregistry1977” to improve reproducibility and transparency.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
