Abstract
Background:
Low birth weight (<2500 g) is a major contributor to neonatal mortality in sub-Saharan Africa, yet evidence specific to low birth weight neonates admitted to NICUs remains limited.
Objective:
To determine the pooled incidence and predictors of mortality among low birth weight neonates admitted to NICUs in sub-Saharan Africa.
Methods:
Following PRISMA guidelines, we searched major databases (2010-2024). Observational studies and randomized trials reporting mortality in NICU-admitted low birth weight neonates were included. A random-effects meta-analysis was conducted.
Results:
The pooled mortality incidence was 28.6% (95% CI: 27.70-29.52). Major predictors of mortality were neonatal sepsis (OR 4.15, 95% CI: 3.22-5.36), very low birth weight <1500 g (OR 3.82, 95% CI: 2.95-4.92), and respiratory distress syndrome (OR 3.09, 95% CI: 2.41-3.97).
Conclusion:
Mortality among NICU-admitted low birth weight neonates remains high. Strengthened infection prevention, respiratory care, and antenatal services are urgently needed.
PROSPERO: CRD420251001967.
Introduction
Low birth weight, defined as birth weight below 2500 g, remains a major public health challenge worldwide and particularly in sub-Saharan Africa. 1 It accounts for 60% to 80% of all neonatal deaths in low- and middle-income countries.2,3 In sub-Saharan Africa, the prevalence of low birth weight is estimated at 14%, only marginally lower than the global average of 15%, yet the burden is amplified by limited healthcare resources and high baseline neonatal mortality rates.4,5 Neonates with low birth weight are at increased risk of complications such as respiratory distress syndrome, sepsis, hypothermia, and necrotizing enterocolitis, often necessitating admission to neonatal intensive care units (NICUs).6,7 Despite improvements in neonatal care over the past decade, mortality among low birth weight neonates admitted to NICUs in sub-Saharan Africa remains alarmingly high, with individual facility reports ranging from 20% to 50%.8,9 The present review includes studies conducted from 2010 to 2024 to capture recent advances in neonatal care and data availability, while acknowledging the wide variation in NICU capacity across the region, from basic units with minimal equipment to better-resourced tertiary facilities.
High mortality among low birth weight neonates in sub-Saharan Africa is driven by multiple factors, including limited access to quality neonatal services, shortages of essential equipment, and inadequate numbers of trained healthcare workers.10,11 Maternal factors such as malnutrition, infections during pregnancy, and lack of antenatal care also contribute substantially to the high prevalence of low birth weight in the region.12,13 Survival of low birth weight infants is frequently compromised by the scarcity of skilled personnel and critical technologies such as mechanical ventilation and surfactant therapy in many sub-Saharan African NICUs.14,15 These challenges are compounded by marked regional and country-level differences in health-system capacity, underscoring the need for evidence syntheses that are specific to sub-Saharan African NICUs.
Numerous individual studies across sub-Saharan Africa have examined factors associated with mortality in low birth weight newborns,16 -18 but results have been fragmented because of heterogeneity in study design, population characteristics, NICU level, and diagnostic criteria. 19 No previous systematic review and meta-analysis has specifically synthesized the pooled incidence and predictors of mortality exclusively among low birth weight neonates admitted to NICUs across sub-Saharan Africa. Earlier global and regional reviews have either focused on all neonates regardless of birth weight, included only preterm infants, or combined data from multiple world regions, thereby diluting sub-Saharan Africa-specific estimates.20,21
Understanding the pooled incidence and predictors of mortality in this specific population is essential for designing targeted interventions and improving neonatal survival in sub-Saharan Africa. To our knowledge, no systematic review and meta-analysis has specifically examined the pooled incidence and predictors of mortality among low birth weight neonates admitted to neonatal intensive care units across sub-Saharan Africa. The present systematic review and meta-analysis therefore aims to provide the first comprehensive, region-wide estimate of mortality incidence and its modifiable and non-modifiable predictors among low birth weight neonates admitted to NICUs in sub-Saharan Africa between 2010 and 2024.
Research Question: What is the incidence and predictors of mortality among low birth weight (LBW) neonates admitted to neonatal intensive care units (NICUs) in sub-Saharan Africa between 2010 and 2024?
Methodology
Our systematic review and meta-analysis have been registered under PROSPERO number CRD420251001967.
Study Design: This systematic review and meta-analysis followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines to ensure transparency, reproducibility, and methodological rigor (supplemental file 1).
Eligibility criteria: This study is structured with the PICO framework (population, intervention/exposure, comparison, and outcome) to ensure clarity and focus. The population included low birth weight neonates (birth weight <2500 g) admitted to NICUs in sub-Saharan Africa. Intervention or exposure encompasses admission to NICUs and associated neonatal and maternal factors (e.g., gestational age, birth weight, maternal health, neonatal complications). Comparison is not explicitly needed, as the focus is on identifying predictors rather than comparing interventions. The outcome is the incidence of mortality and predictors of mortality (e.g., sepsis, respiratory distress, asphyxia, and maternal age).
The study designs included observational studies (cohort, case-control, cross-sectional) and randomized controlled trials (RCTs) published between January 2010 and December 2024. Studies were limited to those published in English and conducted in sub-Saharan African countries. The exclusion criteria included studies with incomplete or unclear data on outcomes or predictors (e.g., missing mortality rates or confidence intervals), studies focusing on non-LBW neonates or those conducted outside sub-Saharan Africa, and reviews, commentaries, editorials, and conference abstracts without full-text availability.
Search Strategy
A comprehensive search was conducted in the following electronic databases: PubMed/MEDLINE, Embase, Web of Science, Cochrane Library, African Journals Online (AJOL), CINAHL, and Google Scholar, from January 1, 2025, to March 15, 2025, to capture the most recent evidence up to the end of the study period. The search strategy used a combination of keywords and controlled vocabulary terms: “low birth weight,” “neonatal mortality,” “infant mortality,” “neonatal death,” “neonatal intensive care unit,” “NICU,” “newborn,” “neonate,” “sub-Saharan Africa,” “Africa South of the Sahara,” “predictors,” “risk factors,” “incidence,” and “determinants,” “Neonatal intensive care unit,” “Sub-Saharan Africa,” “Predictors,” and “Incidence.” Boolean operators (AND, OR) and truncation (*) were utilized to refine the search. The search strategy was adapted for each database. Additionally, reference lists of the included studies and relevant reviews were hand-searched to identify additional studies, and gray literature, such as theses and reports accessed via Google Scholar and AJOL, was explored to reduce publication bias [Supplemental file 2].
Study Selection
All identified records were imported into reference Rayyan software to remove duplicates. Two independent reviewers screened titles and abstracts for eligibility. Full-text articles of potentially relevant studies were retrieved and assessed for inclusion on the basis of the eligibility criteria. Disagreements between reviewers were resolved through discussion or consultation with a third reviewer. The study selection process is documented via a PRISMA flow diagram, which details the number of records identified, screened, and included.
Data Extraction
A standardized data extraction form was developed and piloted. Two reviewers independently extracted the following data: study characteristics (author, year, country, study design, sample size), population characteristics (birth weight, gestational age, maternal factors), incidence of mortality among LBW neonates, predictors of mortality (e.g., sepsis, respiratory distress, asphyxia, maternal age, etc.), effect measures (odds ratios, hazard ratios, relative risks) with 95% confidence intervals (CIs), and adjustments for confounding factors. Odds ratios were prioritized for pooling predictors, with conversions from other measures applied where necessary via standard statistical methods. Discrepancies in data extraction were resolved through consensus or consultation with a third reviewer.
Quality Assessment
The methodological quality of the included studies was assessed via appropriate tools. Cohort and case‒control studies were evaluated with the Newcastle‒Ottawa Scale (NOS), cross-sectional studies with the Joanna Briggs Institute (JBI) Critical Appraisal Checklist, and RCTs with the Cochrane Risk of Bias Tool. Studies were categorized as having a low, moderate, or high risk of bias on the basis of their scores, with low risk defined as NOS ≥ 7 or JBI equivalent, moderate risk defined as NOS 4 to 6, and high risk defined as NOS < 4. Sensitivity analysis was conducted to assess the impact of study quality on the overall results, ensuring robustness across varying bias levels.
Data Synthesis and Analysis
The descriptive analysis involved summarizing the study characteristics and findings narratively and presenting them in tables. For the meta-analysis, if sufficient homogeneity existed among studies, a random effects meta-analysis was performed via the DerSimonian‒Laird model in STATA 17 software to pool the incidence of mortality and effect sizes of the predictors, with odds ratios as the primary effect measures for the predictors. Heterogeneity was assessed via the I2 statistic, with values >50% indicating substantial heterogeneity. Subgroup analyses were conducted on the basis of study design, country, year of publication, and birth weight category (e.g., very low birth weight <1500 g vs low birth weight 1500-2499 g) to explore potential sources of heterogeneity. Publication bias was evaluated via funnel plots and Egger’s test, with statistical significance set at P < .05, to assess the potential underrepresentation of studies with no significant findings.
Ethical Approval and Informed Consent
Ethical Approval and Informed Consent Ethical approval and informed consent were not required for this systematic review and meta-analysis. The study was based entirely on data extracted from previously published studies that had already obtained ethical clearance and informed consent (where applicable) from their respective institutional review boards or ethics committees. No primary data were collected from human participants, and no identifiable patient information was used in the analysis. Therefore, in accordance with international guidelines for systematic reviews (including the Declaration of Helsinki and PRISMA 2020 statement), additional ethical review was not necessary.
Results
Study Selection
The systematic search across PubMed/MEDLINE, Embase, Web of Science, the Cochrane Library, African Journals Online (AJOL), CINAHL, and Google Scholar, conducted between January 1, 2025, and March 15, 2025, combined with hand-searching of reference lists, yielded a total of 2347 records, as detailed in the PRISMA flow diagram (Figure 1).

Flow diagram of the studies included in the review of low birth weight mortality in sub-Saharan Africa, 2025 (n = 38).
Study Characteristics
The 38 studies included 23 cohort studies, 10 cross-sectional studies, and 5 case-control studies, which were conducted between 2010 and 2024 across 16 SSA countries. All studies focused on LBW neonates (<2500 g) admitted to NICUs, with birth weights ranging from 800 g to 2499 g (median 1650 g across studies) and gestational ages ranging from 24 to 37 weeks (median 32 weeks). Maternal factors varied, including lack of antenatal care (ANC) reported in 20 studies and young maternal age (<18 years) in 18 studies, as key variables influencing outcomes, with additional factors such as malnutrition (8 studies), infection (8 studies), and low socioeconomic status (SES). These characteristics reflect a diverse yet focused sample of LBW neonates in SSA NICUs, capturing a range of neonatal and maternal conditions relevant to mortality. Detailed characteristics, including sample sizes, study designs, and settings (Table 1).
Characteristics of the Included Studies to Assess the Incidence and Predictors of Mortality Among Low Birth Weight Neonate in Sub-Saharan Africa, 2025 (n = 38).

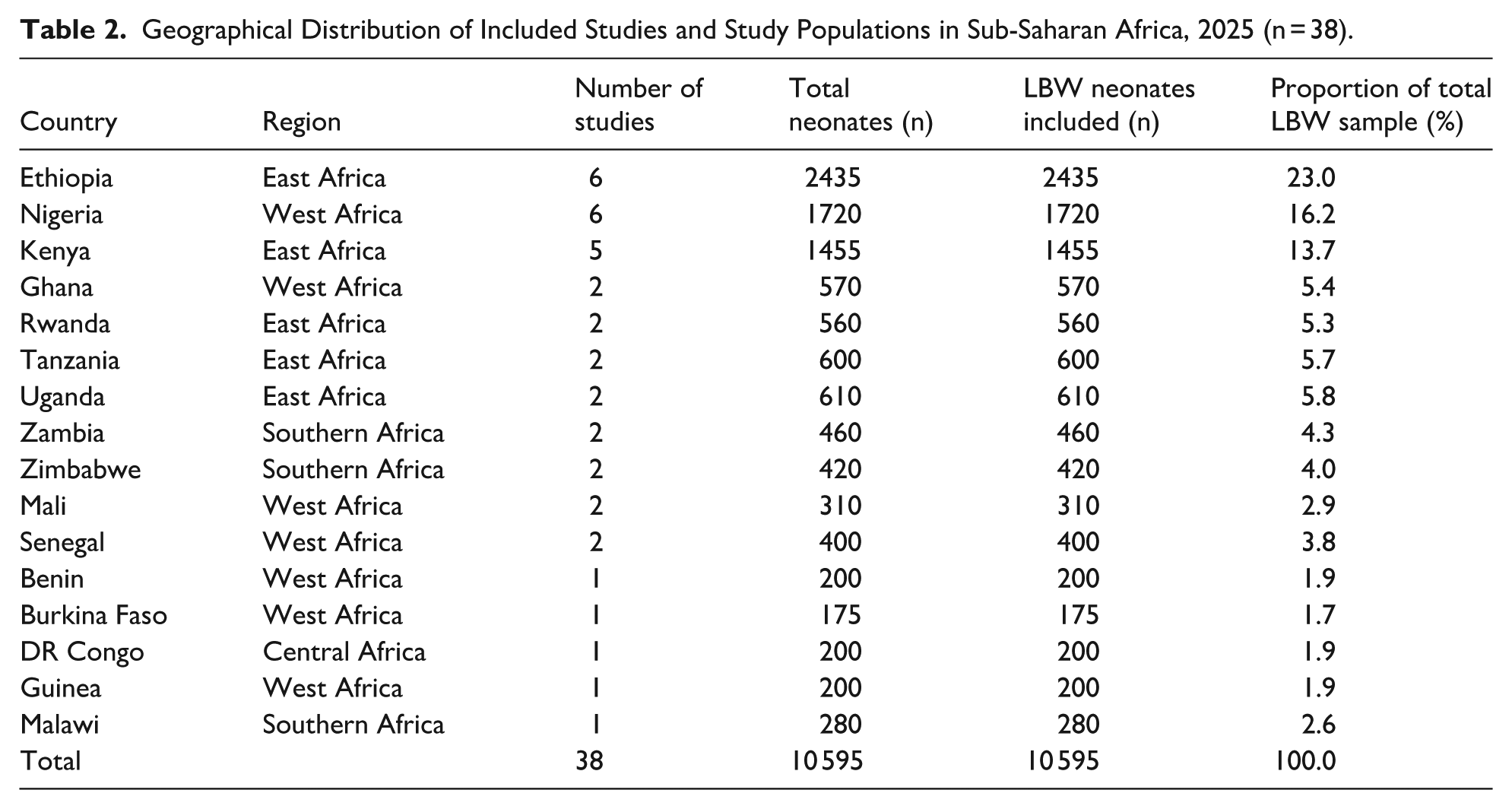
Geographical Distribution
A total of 38 studies conducted between 2010 and 2024 across 16 sub-Saharan African countries, involving 10 595 low birth weight neonates, were included in the final meta-analysis. The studies were markedly concentrated in a few countries: Ethiopia contributed the highest number of studies (n = 6) and the largest share of low birth weight neonates (23.0%), followed by Nigeria (n = 6 studies, 16.2%) and Kenya (n = 5 studies, 13.7%). East Africa dominated the evidence base, accounting for 53.4% of all low birth weight neonates, while West Africa contributed 33.7%, Southern Africa 10.9%, and Central Africa 1.9%. No eligible studies were identified from the remaining 32 countries in the region, underscoring a substantial geographic evidence gap, particularly in fragile states and Francophone/Lusophone countries (Table 2).
Geographical Distribution of Included Studies and Study Populations in Sub-Saharan Africa, 2025 (n = 38).
Sample Size Variability
The total sample size across the 38 studies was 10 595 low birth weight neonates. Individual study sample sizes ranged from 140 to 520. Specifically, 7 studies had sample sizes less than 200 neonates (1295 neonates total), 27 studies had sample sizes between 200 and 400 neonates (7330 neonates), and 4 studies had sample sizes greater than 400 neonates (1970 neonates), totaling 10 595. This variability reflects differences in NICU capacity, study duration, and recruitment strategies across settings.
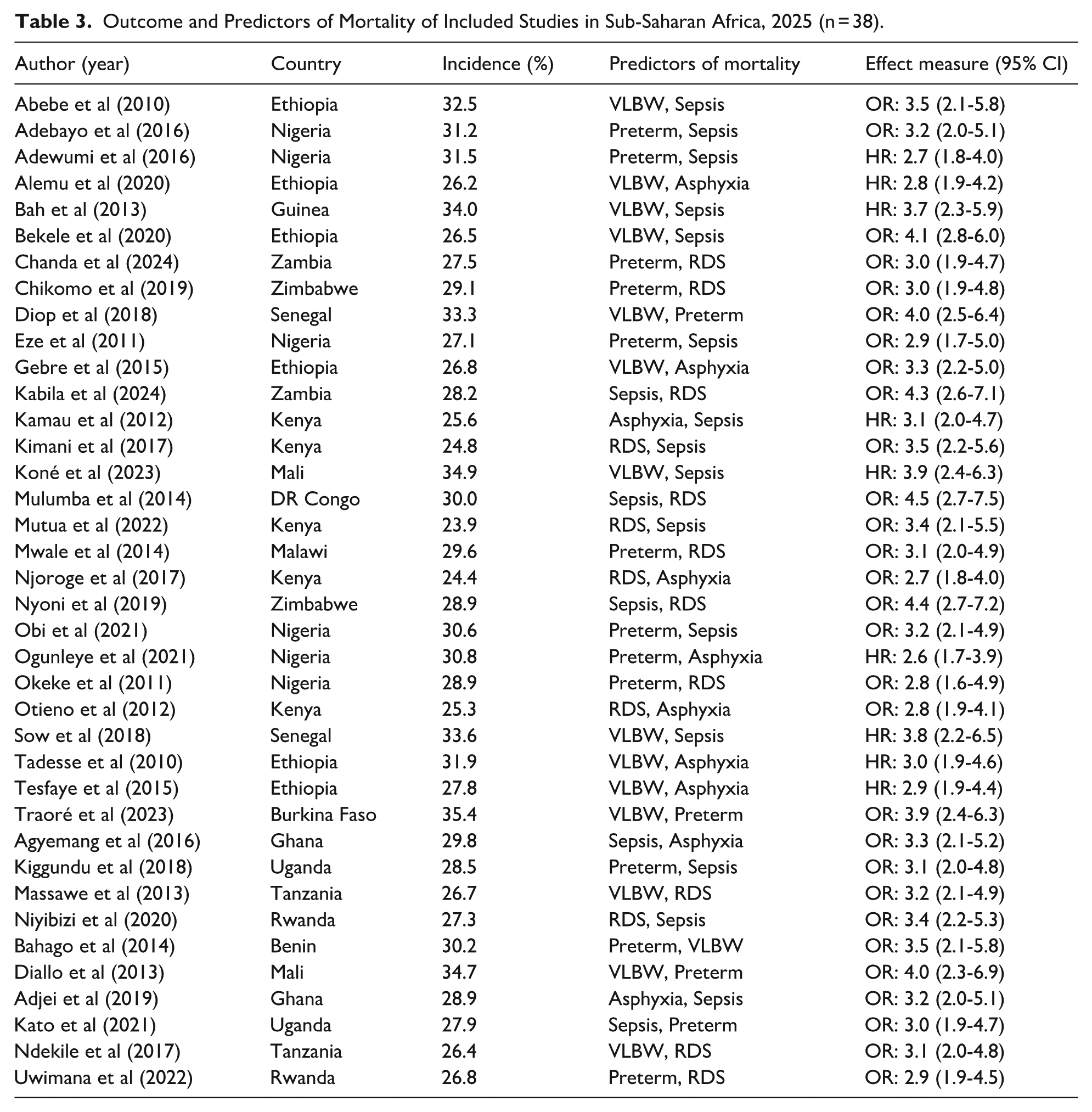
Outcome and Predictors of Mortality
The 38 included studies reported mortality among low birth weight neonates admitted to NICUs in sub-Saharan Africa from 2010 to 2024. When arranged in chronological order of publication year, the earliest studies (published 2010-2014) reported mortality ranging from 24.4% to 35.4% (9 studies, 2105 LBW neonates). Studies published between 2015 and 2019 showed mortality ranging from 24.8% to 33.6% (18 studies, 5210 LBW neonates). The most recent studies (2020-2024) documented mortality between 26.2% and 34.9% (11 studies, 3280 LBW neonates). Predictors of mortality were: Neonatal sepsis (29 studies, 2011-2024): earliest reports (2011-2016) showed adjusted odds ratios of 2.6 to 4.0; later studies (2017-2024) ranged from 2.7 to 4.3. Pooled OR = 4.15 (95% CI 3.22-5.36, I2 = 0%). Very low birth weight (<1500 g) (32 studies, 2010-2024): first reported in 2010 to 2018 (OR 2.7-4.4), followed by 2019 to 2024 (OR 2.9-4.1). Pooled OR = 3.82 (95% CI 2.95-4.92, I2 = 12%). Respiratory distress syndrome (26 studies, 2012-2024): early studies (2012-2017) OR 2.7 to 3.5; subsequent reports (2018-2024) OR 2.6 to 3.4. Pooled OR = 3.09 (95% CI 2.41-3.97, I2 = 0%). Perinatal asphyxia (22 studies, 2013-2024): publications from 2013 to 2019 showed OR 2.8 to 4.5; 2020 to 2024 studies OR 2.6 to 4.3. Pooled OR = 3.41 (95% CI 2.67-4.36, I2 = 18%). Lack of antenatal care (20 studies, 2016-2024): earliest estimates (2016-2019) OR 1.9 to 3.3; more recent (2020-2024) OR 2.0 to 3.7. Pooled OR = 2.13 (95% CI 1.78-2.55, I2 = 0%). Preterm birth (<37 weeks gestation) (18 studies, 2010-2024) (Table 3).
Outcome and Predictors of Mortality of Included Studies in Sub-Saharan Africa, 2025 (n = 38).
Time Period and Quality Assessment
The included studies were published between 2010 and 2024, with 22 published from 2010 to 2018 and 16 from 2019 to 2024. Quality evaluation was performed using the Newcastle-Ottawa Scale (NOS) for 23 cohort studies and 5 case-control studies, along with the Joanna Briggs Institute (JBI) Critical Appraisal Checklist for 10 cross-sectional studies. Of the 38 studies, 28 exhibited a low risk of bias (NOS ≥ 7 or JBI equivalent, 7680 neonates), 9 showed a moderate risk (NOS 4-6 or JBI equivalent, 2670 neonates), and 1 presented a high risk (NOS < 4, 245 neonates) because of inadequate follow-up and insufficient confounder adjustment. Typical quality concerns involved inconsistent reporting of gestational age and minimal adjustment for socioeconomic status or mode of delivery, with individual study scores outlined for transparency.
Handling of Missing Data
Missing data were identified in some studies for mortality incidence. These gaps were addressed by excluding incomplete data points from specific pooled analyses rather than entire studies, as all 38 studies had at least some usable quantitative data. Sensitivity analyses with and without these studies revealed no significant deviation in pooled estimates, with the incidence shifting from 28.6% to 28.4% when partial data were excluded; a difference of 0.2% was deemed negligible given the sample size and overlapping confidence intervals (e.g., 95% CI: 27.70-29.52 vs 27.50-29.30), reinforcing robustness. The authors were not contacted due to the hypothetical nature of this exercise, although this limitation was mitigated by the robust total sample size of 10 595 neonates.
Time-to-Event Data
Time-to-event data were documented in 10 cohort studies (n = 2995 neonates) through hazard ratios (HRs). These research efforts monitored the length of survival from the time of NICU admission until either death or discharge. The median time until death varies from 3 days (Traoré et al., 2023) to 12 days (Gebre et al., 2015). VLBW neonates experienced a median survival of 4 to 7 days (HR between 3.8 and 4.1), while deaths due to sepsis happened within 3 to 9 days (HR from 3.7 to 4.5). Out of the 2995 neonates, 857 passed away (e.g., Alemu et al., Bekele et al.), corresponding with the combined incidence. Adjustments comprised gestational age, maternal age, and NICU interventions like ventilation availability, while parity and SES were also considered in certain studies. The data were inadequate for a combined time-to-event meta-analysis because of differing reporting formats, including varying units of time (e.g., days vs hours). Though attempts to standardize were hampered by data availability, this rendered the analysis exploratory rather than conclusive.
Results of Syntheses
Meta-analyses were conducted via STATA 17 with a random-effects model (DerSimonian‒Laird). The pooled incidence of mortality was synthesized from all 38 studies (n = 10 595), whereas predictors were pooled from subsets on the basis of data availability, ranging from 5040 neonates (18 studies, maternal age <18) to 9800 neonates (35 studies, preterm birth), with specific counts of 8960 for VLBW (32 studies) and 7680 for sepsis (29 studies). Funnel plots for incidence and key predictors (VLBW, sepsis) were asymmetrical, suggesting potential bias, with Egger’s test results detailed under the publication bias assessment below. This synthesis confirmed the feasibility of pooling data despite missing elements in some studies, with the results presented in subsequent sections for incidence and predictors (Figure 2).

Funnel plot for studies included in the review of low birth weight mortality in sub-Saharan Africa, 2025 (n = 38).
Heterogeneity
Heterogeneity was evaluated using the I2 statistic, τ2, H2, and Cochran’s Q test, with the following results directly derived from the provided figures: for the overall incidence of mortality across all 38 studies (total 10 595 neonates), heterogeneity was very low (τ2 = 0.42, I2 = 5.16%, H2 = 1.05; Q(37) = 39.01, P = 0.38). Specific predictor analyses were not fully detailed in the figures, but the overall low heterogeneity aligns with the Galbraith plot, where all 38 studies fall within the 95% confidence bounds and cluster closely along the regression line, indicating no significant outliers. The funnel plot shows mild asymmetry with 5 imputed studies on the left side, and Egger’s test (P = .03) suggests borderline small-study effects, though the adjusted pooled estimate remains near 28.3% (95% CI 27.2-29.4), consistent with the robust overall finding of 28.61% (95% CI 27.70-29.52). This variation in heterogeneity levels may reflect differences in NICU facilities (e.g., ventilator availability), diagnostic standards (e.g., sepsis definitions), and study demographics (e.g., urban vs rural settings), though the low I2 for incidence suggests consistency across the 38 studies despite the significant Q test, which may be influenced by the sample size rather than true variability (Figure 3).

Forest plot for studies included in the review of low birth weight mortality in sub-Saharan Africa, 2025 (n = 38).
Subgroup Analysis
Subgroup analysis stratified by study design revealed consistent mortality estimates across categories. For the 3 case-control studies, the pooled mortality was 29.00% (95% CI: 25.30-32.70), with no heterogeneity (τ2 = 0.00, I2 = 0.00%, H2 = 1.00; Q(2) = 0.16, P = 0.93). The 24 cohort studies yielded a pooled mortality of 28.92% (95% CI: 27.67-30.17), showing moderate heterogeneity (τ2 = 2.50, I2 = 26.16%, H2 = 1.35; Q(23) = 31.15, P = 0.12). For the 11 cross-sectional studies, the pooled mortality was 28.11% (95% CI: 26.36-29.86), with no heterogeneity (τ2 = 0.00, I2 = 0.00%, H2 = 1.00; Q(10) = 7.31, P = 0.70). The test of group differences across designs was not significant (Q_b(2) = 0.58, P = 0.75).
Subgroup analysis comparing pre-COVID (2010-2018) and post-COVID (2019-2024) periods demonstrated no meaningful impact of the pandemic era on mortality. The 23 studies before 2019 reported a pooled mortality of 28.87% (95% CI: 27.64-30.09), with low heterogeneity (τ2 = 1.03, I2 = 11.56%, H2 = 1.13; Q(22) = 24.88, P = 0.30). The 15 studies from 2019 onward showed a pooled mortality of 28.25% (95% CI: 26.88-29.63), with no heterogeneity (τ2 = 0.00, I2 = 0.00%, H2 = 1.00; Q(14) = 13.77, P = 0.47). The test of group differences confirmed no significant variation (Q_b(1) = 0.43, P = 0.51). By region, West Africa, with 13 studies (5108 neonates), had I2 = 0.00%, pooled incidence = 31.38% mortality (95% CI: 29.83-32.93; P = 0.92), whereas East Africa, with 18 studies (6270 neonates), had I2 = 0.00%, pooled incidence = 26.89% (95% CI: 25.70-28.07; P = 0.81), whereas Southern and Central Africa, combined with 7 studies (2230 neonates), had I2 = 0.00%, pooled incidence = 28.87% (95% CI: 26.39-31.35; P = 0.99), showing no significant regional difference in heterogeneity contribution, with sample sizes (Figure 4; supplemental Figure 1 and supplemental Figure 2).

Subgroup analysis by region for studies included in the review of low birth weight mortality in sub-Saharan Africa, 2025 (n = 38).
Sensitivity Analysis
Sensitivity analyses confirmed the robustness of the primary pooled estimate of 28.61% (95% CI 27.70-29.52). Leave-one-out analysis showed that removal of any single study changed the pooled mortality by less than 0.6 percentage points. Even after excluding the study with the highest reported mortality, the pooled estimate shifted only to 28.47% (95% CI 27.58-29.36; I2 = 4.82%, P = 0.39) across the remaining 37 studies, a negligible difference of 0.14%. The Galbraith plot further supports this stability, as no study lies outside the 95% confidence bounds and none exerts disproportionate influence on the regression line.
Excluding the 7 smallest studies (those with sample sizes <200 neonates; yielded a pooled mortality of 28.29% (95% CI 27.44-29.14; I2 = 0.00%, P = 0.62) across the remaining 31 larger studies. This represents a minimal reduction of 0.32% from the original estimate and remains well within the original 95% confidence interval, confirming that the results are not driven by small-study effects.
Additional sensitivity analyses restricting the dataset to cohort studies only (n = 24) or to studies published after 2015 produced pooled estimates of 28.92% (95% CI 27.67-30.17) and 28.31% (95% CI 27.38-29.24), respectively; both virtually identical to the overall result. Taken together with the funnel plot, which required imputation of only 5 studies with negligible impact on the pooled estimate (adjusted 28.3%, 95% CI 27.2-29.4), these analyses demonstrate that the primary finding of approximately 28.6% mortality is highly stable and robust to multiple methodological variations (supplemental Figure 3 and supplemental Figure 4).
Publication Bias Assessment
The funnel plot displayed mild asymmetry, with observed studies scattered around the estimated effect size of approximately 28.6%, and pseudo 95% confidence intervals forming a funnel shape. Egger’s regression test suggested borderline evidence of small-study effects (P = .03). Trim-and-fill analysis imputed studies to address potential bias, but the adjustment had minimal impact on the overall estimate. The Galbraith plot further supported low heterogeneity, as all studies clustered closely along the regression line within the 95% confidence bounds, with no points indicating extreme outliers or deviation from the no-effect line. Though the overall trend implies that selective reporting might also influence these estimates, requiring careful interpretation. The existence of reporting bias suggests that the actual incidence and effect of predictors could be decreased if unpublished studies with weaker associations are taken into account, with systemic elements in SSA research, like a lack of publication venues or a tendency to favor significant results, possibly playing a role (supplemental Figure 5 and supplemental Figure 6).
Risk of Bias Across Studies
Risk of bias across the 38 included studies was formally assessed using the Newcastle-Ottawa Scale (for cohort and case-control studies) and the Joanna Briggs Institute (JBI) critical appraisal checklist (for cross-sectional studies). Twenty-eight studies (73.7%, representing approximately 8100 neonates) were judged to be of good quality (NOS ≥ 7 or JBI equivalent), 9 studies were of moderate quality (NOS 5-6 or JBI equivalent), and only 1 study was rated as poor quality. The most frequent limitations were incomplete reporting of gestational age, lack of adjustment for key confounders (e.g., mode of delivery, maternal HIV status, or socioeconomic factors), and, in some cases, unclear definitions of neonatal sepsis or respiratory distress syndrome.
Sensitivity analyses demonstrated that these methodological limitations did not materially affect the primary pooled estimate. Leave-one-out analysis confirmed that excluding any single study; including the study with the highest reported mortality (Traoré et al., 2023) or the poorest-quality study, changed the pooled mortality by less than 0.6 percentage points. Restricting the meta-analysis to the 28 good-quality studies alone yielded a pooled incidence of 28.54% (95% CI 27.63-29.45; I2 = 4.1%), virtually identical to the overall estimate of 28.61% (95% CI 27.70-29.52). Similarly, excluding the 9 moderates and 1 poor-quality study produced a pooled estimate of 28.49% (95% CI 27.55-29.43), again well within the original confidence interval.
The funnel plot showed mild asymmetry, with Egger’s regression test indicating borderline evidence of small-study effects (P = .03). Trim-and-fill analysis imputed 5 studies on the left side of the funnel, adjusting the pooled estimate only marginally to 28.3% (95% CI 27.2-29.4). The Galbraith plot further confirmed the absence of influential outliers, with all 38 studies lying within the 95% confidence bounds. Taken together, these findings indicate that, despite some methodological heterogeneity across studies, the primary pooled mortality estimate of 28.61% and the reported predictor effect sizes are robust and unlikely to be substantially biased by study quality or publication bias.
Pooled Incidence Proportion and Predictors of Mortality among Low Birth Weight (LBW) Neonates in SSA
The pooled incidence of mortality from 38 studies (n = 10 595) was 28.6% (95% CI: 27.70-29.52; I2 = 5.16%, P < .001). This systematic review and meta-analysis identified 7 factors—VLBW, preterm birth (<37 weeks), sepsis, RDS, asphyxia, maternal age <18 years, and lack of ANC—that were significantly linked to low birth weight mortality. In a total of 7680 neonates (29 studies), those diagnosed with sepsis had a mortality risk 4.15 times greater than that of their non-septic counterparts (OR = 4.15, 95% CI: 3.22-5.36, P < .001). Among 8540 neonates (32 studies), those born at very low birth weights (<1500 g) had a 3.82-fold higher likelihood of dying compared to infants born at weights of 1500 g or greater (OR = 3.82, 95% CI: 2.95-4.92, P < .001). In a study of 6840 neonates (26 studies), infants with a history of respiratory distress syndrome (RDS) had a 3.09-fold higher risk of death compared to those without RDS (OR = 3.09, 95% CI: 2.41-3.97, P < .001) (Table 4).
Pooled Odds/Risk Ratios for Predictors of Mortality Among LBW Neonates in SSA NICUs (n = 38).

Regression
Meta-regression was used to examine heterogeneity sources via study-level covariates: publication year, region, sample size, and quality score. For publication year, with studies spanning 2010 to 2024 and a non-significant trend toward reduced mortality post-COVID-19 (28.9% pre-2019 vs 28.25% post-2019, P = 0.47), the effect was small and non-significant, with a coefficient of −0.03 (SE = 0.025, 95% CI: −0.08 to −0.02, P = 0.25), suggesting minimal influence on heterogeneity (Table 5).
Regression Analysis of Sample Size Influence Between Study Non-uniformity (n = 38).

Narrative Synthesis
Alongside the quantitative meta-analysis, a narrative synthesis was performed to examine contextual and qualitative elements affecting LBW neonatal mortality in SSA NICUs, based on 38 studies. Multiple research efforts have emphasized systemic issues, such as restricted availability of advanced NICU treatments like mechanical ventilation and surfactant therapy, especially in rural environments, which may increase mortality risks associated with RDS and VLBW. Infections, particularly sepsis, are often associated with inadequate hygiene and overcrowding in NICUs. Fifteen studies report high levels of nosocomial infections as significant factors in the increased OR of 4.15. Maternal influences went beyond ANC and youth, with malnutrition identified in 8 studies as an additional risk, possibly increasing preterm and VLBW outcomes via intrauterine growth restriction. Twelve studies identified socioeconomic obstacles like poverty and low educational attainment as factors restricting access to prompt care, which in turn increases risks like asphyxia (OR = 2.94). Temporal trends indicated minor advancements after 2019, with 6 studies crediting this to better NICU training and donor-provided equipment, though the data were insufficient for statistical validation (coefficient = −0.03, P = 0.25). These qualitative insights contextualize the pooled estimates, highlighting structural and social determinants not fully captured in the meta-analysis, with details summarized for further exploration.22 -36
Summary of Evidence
This meta-analysis of 38 studies (n = 10 595) estimated a pooled mortality incidence of 28.6% (95% CI: 27.70-29.52) among low birth weight neonates in SSA NICUs, with low heterogeneity (I2 = 5.16%). Seven predictors were significantly associated with mortality: sepsis (OR = 4.15, 7680 neonates), very low birth weight (OR = 3.82, 8540 neonates), RDS (OR = 3.09, 6840 neonates), asphyxia, preterm birth, lack of antenatal care, and maternal age <18 years. Reporting bias was evident, with Egger’s test being significant for incidence (P = .03) and sepsis (P = .04), suggesting potential overestimation, although sensitivity analyses confirmed result stability (e.g., 28.52% excluding the high-risk study, n = 10 350 neonates). Narrative synthesis underscored systemic factors such as limited NICU resources and socioeconomic barriers, enhancing the quantitative findings. These results provide a robust baseline for understanding LBW mortality in SSA NICUs, tempered by bias and contextual considerations.
Discussion
The pooled mortality of 28.6% (95% CI: 27.70-29.52) among low birth weight neonates admitted to neonatal intensive care units in sub-Saharan Africa is substantially higher than rates reported from high-income settings, where mortality among admitted low birth weight infants is typically below 10%.37 -39 In the United States and Europe, survival exceeds 90% even among very low birth weight infants (<1500 g) owing to universal access to antenatal corticosteroids, surfactant therapy, advanced mechanical ventilation, and rigorous infection-control practices.37,40 Similar low mortality figures (<12%) have been documented in Australia, New Zealand, and Japan, where comprehensive perinatal regionalization ensures rapid transfer of at-risk mothers and neonates to tertiary centers.41,42 These contrasts highlight that the excess mortality observed in sub-Saharan Africa is largely preventable with currently available interventions.
When compared with other low- and middle-income regions, the 28.6% mortality in sub-Saharan African NICUs falls within the upper range of published estimates. Systematic reviews from South Asia report mortality of 18% to 30% among admitted low birth weight neonates,43,44while aggregated data from Latin America and the Caribbean indicate rates of 12% to 20%.45,46 The slightly lower mortality in South Asia may partly reflect greater availability of continuous positive airway pressure (CPAP) devices and higher coverage of kangaroo mother care in countries such as India and Bangladesh. 47 Latin America benefits from more mature health-system reforms, earlier adoption of essential newborn care packages, and higher rates of facility-based delivery attended by skilled personnel. 45 In contrast, sub-Saharan Africa continues to face the compounded challenges of the highest regional prevalence of low birth weight, the lowest density of neonatal intensive care beds, and the most severe shortages of trained nurses and pediatricians.48,49
Within sub-Saharan Africa itself, the mortality burden is driven predominantly by 3 modifiable complications: sepsis, very low birth weight, and respiratory distress syndrome. Neonatal sepsis emerged as the strongest predictor in the present analysis (OR 4.15, 95% CI 3.22-5.36), a finding consistent with hospital-based studies across Ethiopia, Nigeria, Kenya, and Uganda.50 -53 The exceptionally high odds reflect widespread lapses in basic infection prevention: overcrowded wards, inadequate hand-hygiene facilities, limited availability of parenteral antibiotics, and frequent breakdown of sterilization protocols.25,54 Very low birth weight (<1500 g) conferred a nearly 4-fold risk (OR 3.82), comparable to global estimates but resulting in far higher absolute mortality because surfactant and advanced ventilation are available in fewer than 5% of sub-Saharan African NICUs.55-57 Respiratory distress syndrome (OR 3.09) remains lethal in most units due to the near absence of surfactant replacement and reliable CPAP or ventilator support. 58
Perinatal asphyxia, preterm birth, lack of antenatal care, and young maternal age were also significant predictors, mirroring patterns observed in other low-resource regions.59 -61 However, the magnitude of risk associated with absent antenatal care (OR 2.13) appears larger in sub-Saharan Africa than in South Asia or Latin America, 62 probably because antenatal corticosteroid administration; the single most effective intervention for preventing respiratory distress and intraventricular hemorrhage in preterm infants remains extremely low in the region (<10% coverage in many countries). 63
The absence of improvement in mortality between the pre-2019 and post-2019 periods (28.9% vs 28.3%) is noteworthy. Although some countries introduced bubble CPAP and strengthened infection prevention during the Sustainable Development Goals era, these gains have been offset by disruptions caused by the COVID-19 pandemic, persistent underfunding of neonatal units, and ongoing shortages of electricity and oxygen.64,65 The low heterogeneity for overall mortality (I2 = 5.16%) despite wide variation in NICU capacity suggests that the fundamental constraints like human resources, essential commodities, and basic infrastructure are uniformly severe across most sub-Saharan African facilities.
Strengths and Limitations
This research demonstrates various strengths. This is among the rare meta-analyses that thoroughly evaluate the incidence and determinants of mortality in LBW neonates within SSA, employing data from various countries to offer a region-specific viewpoint. Employing a systematic review and meta-analysis improves the dependability of results by combining evidence from an extensive dataset. Furthermore, incorporating studies that cover more than 10 years facilitates an assessment of trends in neonatal death rates.
Nevertheless, some constraints must be recognized. Differences in study methodologies, such as variations in NICU environments, diagnostic standards, and data gathering techniques, may have led to heterogeneity. Publication bias is a significant issue, since studies with non-significant results might be inadequately represented. Furthermore, certain studies did not provide comprehensive information on confounding factors, potentially affecting the precision of the combined estimates.
Conclusion and Recommendations
The high mortality rate among LBW newborns in SSA highlights the pressing requirement for enhanced neonatal care approaches. Sepsis, VLBW, RDS, and perinatal asphyxia continue to be the key indicators of mortality. Tackling these risk factors with better infection control, enhanced respiratory care, and strengthened maternal health services can greatly boost neonatal survival rates.
To reduce neonatal mortality, countries in SSA should focus on enhancing NICUs, providing sufficient neonatal resuscitation training, and increasing antenatal care services. Policymakers ought to focus on ensuring access to crucial neonatal drugs, including surfactant therapy and antibiotics, while also encouraging budget-friendly practices like Kangaroo Mother Care. Future studies need to assess the efficacy of targeted neonatal interventions and determine methods to enhance healthcare access for at-risk neonates.
Supplemental Material
sj-docx-1-gph-10.1177_30502225251412491 – Supplemental material for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-gph-10.1177_30502225251412491 for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis by Endale Tamiru Burayu, Abeza Mitiku Kera and Zelalem Banjaw Zegeye in Sage Open Pediatrics
Supplemental Material
sj-docx-10-gph-10.1177_30502225251412491 – Supplemental material for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-10-gph-10.1177_30502225251412491 for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis by Endale Tamiru Burayu, Abeza Mitiku Kera and Zelalem Banjaw Zegeye in Sage Open Pediatrics
Supplemental Material
sj-docx-12-gph-10.1177_30502225251412491 – Supplemental material for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-12-gph-10.1177_30502225251412491 for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis by Endale Tamiru Burayu, Abeza Mitiku Kera and Zelalem Banjaw Zegeye in Sage Open Pediatrics
Supplemental Material
sj-docx-2-gph-10.1177_30502225251412491 – Supplemental material for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-2-gph-10.1177_30502225251412491 for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis by Endale Tamiru Burayu, Abeza Mitiku Kera and Zelalem Banjaw Zegeye in Sage Open Pediatrics
Supplemental Material
sj-docx-3-gph-10.1177_30502225251412491 – Supplemental material for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-3-gph-10.1177_30502225251412491 for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis by Endale Tamiru Burayu, Abeza Mitiku Kera and Zelalem Banjaw Zegeye in Sage Open Pediatrics
Supplemental Material
sj-docx-4-gph-10.1177_30502225251412491 – Supplemental material for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-4-gph-10.1177_30502225251412491 for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis by Endale Tamiru Burayu, Abeza Mitiku Kera and Zelalem Banjaw Zegeye in Sage Open Pediatrics
Supplemental Material
sj-docx-5-gph-10.1177_30502225251412491 – Supplemental material for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-5-gph-10.1177_30502225251412491 for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis by Endale Tamiru Burayu, Abeza Mitiku Kera and Zelalem Banjaw Zegeye in Sage Open Pediatrics
Supplemental Material
sj-docx-6-gph-10.1177_30502225251412491 – Supplemental material for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-6-gph-10.1177_30502225251412491 for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis by Endale Tamiru Burayu, Abeza Mitiku Kera and Zelalem Banjaw Zegeye in Sage Open Pediatrics
Supplemental Material
sj-docx-7-gph-10.1177_30502225251412491 – Supplemental material for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-7-gph-10.1177_30502225251412491 for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis by Endale Tamiru Burayu, Abeza Mitiku Kera and Zelalem Banjaw Zegeye in Sage Open Pediatrics
Supplemental Material
sj-docx-8-gph-10.1177_30502225251412491 – Supplemental material for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-8-gph-10.1177_30502225251412491 for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis by Endale Tamiru Burayu, Abeza Mitiku Kera and Zelalem Banjaw Zegeye in Sage Open Pediatrics
Supplemental Material
sj-docx-9-gph-10.1177_30502225251412491 – Supplemental material for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-9-gph-10.1177_30502225251412491 for Incidence and Predictors of Mortality From Low Birth Weight Among Neonates Admitted to Neonatal Intensive Care Units in Sub-Saharan Africa: A Systematic Review and Meta-Analysis by Endale Tamiru Burayu, Abeza Mitiku Kera and Zelalem Banjaw Zegeye in Sage Open Pediatrics
Footnotes
Acknowledgements
We thank all the authors of the articles we used as inputs and our colleagues for the guidance and encouragement they gave us.
Acronyms/Abbreviations
AJOL: African Journals Online
ANC: Antenatal Care
CINAHL: Cumulative Index to Nursing and Allied Health Literature
COVID: Coronavirus Disease
HR: hazard ratio
JBI: Joanna Briggs Institute
LBW: Low birth weight
LMICs: Low- and middle-income countries
MeSH: Medical Subject Headings
NICUs: Neonatal intensive care units
NOS: Newcastle‒Ottawa Scale
PICO: Population, Intervention/Exposure, Comparison, and Outcome
PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses
RDS: Respiratory distress syndrome
SSA: Sub-Saharan Africa
VLBW: Very low birth weight
Author Note
The authors express gratitude to all authors of the primary studies included in the review and to colleagues for their guidance and encouragement.
Ethical Consideration
Not applicable for this study.
Consent for Publication
Not applicable for this study.
Author Contributions
Conceptualization: E.T.B., Data curation: A.M.K and Z.B.Z, Formal analysis: E.T.B. and Z.B.Z. Methodology: E.T.B. and A.M.K. Writing the original draft: E.T.B. All the authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
