Abstract
Introduction:
Dengue fever is a major pediatric health challenge, with thrombocytopenia as a key severity marker.
Objectives:
To assess the predictive value of mean platelet volume, MPV-to-platelet ratio, and other complete blood count (CBC) indices for severe thrombocytopenia in pediatric dengue.
Methods:
This prospective observational study enrolled 127 children (1 month–15 years) with confirmed dengue at Hue Central Hospital, Vietnam (January 2023-July 2025). CBC parameters were measured on illness days 3 and 6. Severe thrombocytopenia was defined as platelet count <50 000/µL.
Results:
Severe thrombocytopenia occurred in 15.7% of patients. Day 3 MPV/PLT ratio was the strongest predictor (median 0.10 vs 0.05, P < .001; AUC 0.818; sensitivity 75.0%; specificity 88.8%). Elevated MPV, low platelet and lymphocyte counts were also significant. NLR showed moderate value, while PLR and SII were not predictive.
Conclusion:
MPV/PLT is a simple, early marker for identifying high-risk children with dengue.
Introduction
Dengue is a rapidly emerging mosquito-borne viral disease with a substantial burden among children, particularly in tropical and subtropical regions. Globally, it is estimated that 390 million dengue infections occur annually, with about 96 million manifesting clinically, and children under 15 years accounting for a significant proportion of severe cases and hospitalizations, especially in endemic areas of Southeast Asia and Latin America.1,2 In Vietnam, dengue is hyperendemic with frequent serotype shifts. A recent study in Quảng Nam Province found that DENV-1 replaced DENV-2 as the dominant strain and that about 50% of PCR-confirmed pediatric cases showed IgG positivity, indicating high rates of secondary infection—a risk factor for severe dengue. 3 These findings align with nationwide trends showing dynamic serotype circulation and highlight the ongoing risk to children, emphasizing the need for sustained surveillance and pediatric-focused interventions.
Early detection of predictors of dengue progression is vital, especially for anticipating thrombocytopenia, a hallmark of severe disease. Mean platelet volume (MPV) reflects platelet activation and consumption, with low values suggesting destruction and high values indicating marrow compensation. The MPV-to-platelet ratio (MPV/PLT) may provide stronger predictive value by integrating both measures.4 -6 These indices offer practical, cost-effective tools for early risk stratification in pediatric dengue cases, particularly in resource-limited settings.
CBC-derived markers such as NLR, PLR, and SII are low-cost tools for assessing infection severity and prognosis in both adults and children.7,8 However, evidence on MPV and related indices for predicting thrombocytopenia in pediatric dengue—a key complication tied to severity and bleeding risk—remains limited. Most studies focus on adults, highlighting the need for pediatric-specific research to identify early hematologic markers of clinical deterioration.
Despite increasing interest in these markers, studies focusing specifically on their predictive role in pediatric dengue—especially for forecasting thrombocytopenia—are limited. Most existing evidence comes from adult cohorts or cross-sectional studies with inconsistent results. Furthermore, few studies have assessed these indices at early phases of illness (eg, day 3) to anticipate subsequent platelet nadir. Therefore, this study aimed to evaluate the predictive value of MPV, MPV/PLT, and other CBC-derived indices (NLR, PLR, and SII) measured on day 3 of illness for the development of thrombocytopenia on day 6 in children with dengue fever.
Materials and Methods
Study Design and Population
This prospective observational study was conducted at the Pediatric Center of Hue Central Hospital, Vietnam, between January 2023 and July 2025. All eligible children diagnosed with dengue fever during this period were included in the study to maximize the sample size and ensure representation of the pediatric dengue population presenting to the hospital. No a priori sample size calculation was performed; instead, the sample size was determined by enrolling all eligible patients meeting the inclusion criteria during the study period to capture the broadest possible representation of pediatric dengue cases.
Children aged 1 month to 15 years with confirmed dengue fever were eligible for inclusion. Dengue fever was diagnosed according to the World Health Organization (WHO) 2009 guidelines, based on compatible clinical features and laboratory confirmation by a positive NS1 antigen test and/or dengue-specific IgM serology. 9
Patients were enrolled within the first 4 days of illness to enable early CBC follow-up.
Exclusion criteria included: (1) known hematologic disorders (eg, leukemia, aplastic anemia), (2) co-infections (eg, malaria, typhoid), or (3) missing CBC data on either day 3 or day 6. Written informed consent was obtained from all participants’ parents or guardians. Additionally, an opt-out consent option was provided, allowing parents or guardians to withdraw their child’s participation at any point during the study.
Data Collection
Clinical and laboratory data were collected prospectively using a structured case report form.
Clinical variables included: age, sex, presenting symptoms, day of illness at admission, and the presence of dengue warning signs as per WHO 2009 guidelines (eg, abdominal pain, vomiting, mucosal bleeding). 9
Laboratory data included CBC parameters on illness day 3 and day 6: PLT, MPV, neutrophil count (NEU), lymphocyte count (LYM), and hematocrit (HCT). Day 3 was selected as the primary time point for CBC measurement because it corresponds to the typical day when pediatric dengue patients present to hospital with fever and other symptoms, ensuring uniformity in data collection. Patients with laboratory measurements taken on other days were excluded to maintain a standardized time point for analysis.
All CBC were analyzed using the Sysmex XN-1000 automated hematology analyzer (Sysmex Corporation, Kobe, Japan).
CBC-Derived Indices
The following indices were calculated from day 3 CBC:
These indices were chosen based on their emerging relevance in inflammatory and infectious conditions.
Outcome Definition
The primary outcome was severe thrombocytopenia, defined as a PLT <50 000/µL on day 6 of illness. This threshold is clinically significant due to its association with increased bleeding risk in dengue fever.
None of the enrolled patients progressed to clinically defined severe dengue (eg, shock, plasma leakage, organ dysfunction) during hospitalization, which is acknowledged as a study limitation.
Statistical Analysis
Data were analyzed with SPSS 20.0. Continuous variables were tested for normality (Shapiro-Wilk) and reported as medians (IQR); categorical variables as counts (%). Groups were compared using the Mann–Whitney U test for continuous data and chi-square or Fisher’s exact test for categorical data.
To identify independent predictors of severe thrombocytopenia, logistic regression analyses were conducted. Variables with a P-value < .1 in univariate analysis were included in the multivariate model. A P-value < .05 was considered statistically significant. To assess the predictive performance of CBC indices, receiver operating characteristic (ROC) curve analysis was performed. The area under the curve (AUC) was calculated for each index, and optimal cut-off values were identified using Youden’s Index. A P-value < .05 was considered statistically significant.
The relationship between MPV/PLT ratio on day 3 and platelet count on day 6 was analyzed using R version 4.3.3. Linear and quadratic (second-degree polynomial) regression models were fitted with the lm() function. Scatter plots and regression lines with annotated equations were generated using the ggplot2 package.
Ethical Approval and Informed Consent
The study was approved by the Ethics Committee of Hue University of Medicine and Pharmacy under approval number H2023/208. Written informed consent was obtained from the parents or guardians of all participants. An opt-out consent option was provided, allowing parents or guardians to withdraw their child’s participation at any point during the study.
Result
Patient Characteristics
A total of 127 pediatric patients with confirmed dengue fever were included in the study. Among them, 73 patients (57.5%) had dengue without warning signs, and 54 patients (42.5%) had dengue with warning signs based on the 2009 WHO classification. No patients progressed to severe dengue during the study period.
Overall, 20 patients (15.7%) developed severe thrombocytopenia on day 6 (PLT <50 000/µL), while 107 (84.3%) did not.
The median age of patients with severe thrombocytopenia was significantly higher compared to those without (14 vs 11 years, P = .001). Although the proportion of male patients and those with clinical symptoms such as lethargy, irritability, or bleeding signs did not differ significantly between groups, the presence of warning signs was notably more frequent in the severe thrombocytopenia group (70.0% vs 37.4%, P = .007; Table 1).
Baseline Demographic and Clinical Characteristics of Pediatric Dengue Patients (n = 127).

Baseline hematologic parameters showed that WBC and LYM were significantly lower, while MPV was significantly higher (P = .002) in the severe thrombocytopenia group. PLT were also significantly lower in this group (P < .001). Other variables, including NEU, HCT, and RDW, did not show significant differences.
Comparison of CBC-Derived Indices
Table 2 presents the comparison of CBC-derived indices on day 3 between the 2 groups. The MPV/PLT ratio was significantly higher in patients who developed severe thrombocytopenia (0.10 vs 0.05, P < .001). The NLR was also significantly higher in this group (P = .027). However, no statistically significant differences were observed for the PLR or SII.
Comparison of Day 3 CBC Indices Between Patients With and Without Severe Thrombocytopenia on Day 6.
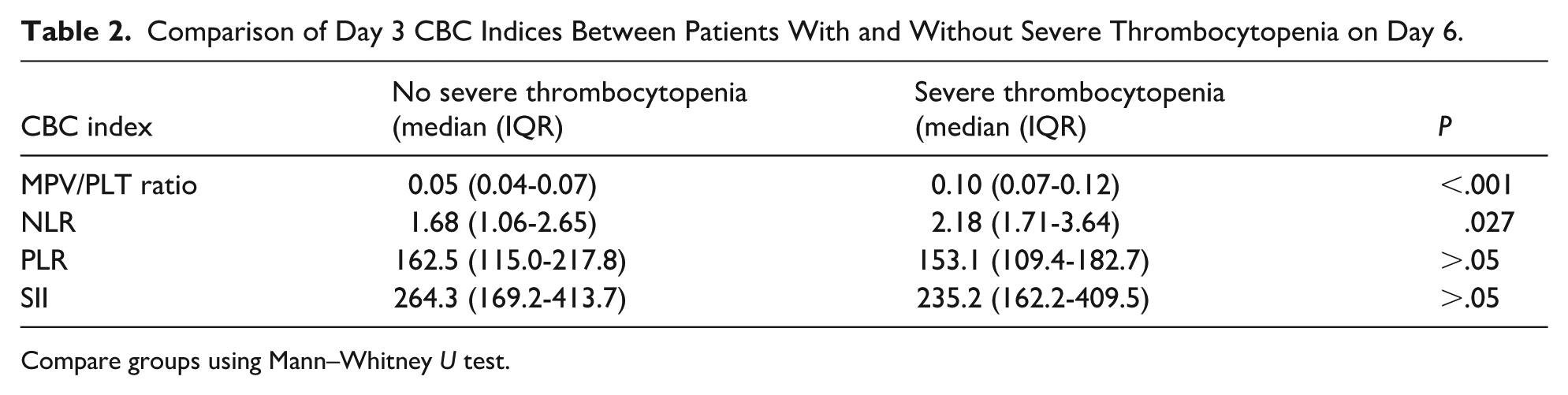
Compare groups using Mann–Whitney U test.
Predictors of Severe Thrombocytopenia
In univariate logistic regression analysis (Table 3), lower LYM (OR = 0.22; 95% CI: 0.06-0.76; P = .017), lower PLT (OR = 0.97; 95% CI: 0.96-0.99; P < .001), and higher MPV (OR = 1.65; 95% CI: 1.13-2.41; P = .009) were significantly associated with increased risk of severe thrombocytopenia. The MPV/PLT ratio (×10) also showed a significant predictive value (OR = 3.37; 95% CI: 1.32-8.55; P = .011). NLR was not a significant predictor in this model.
Logistic Regression Analysis of CBC Indices on Day 3 as Predictors of Severe Thrombocytopenia on Day 6.

In multivariate logistic regression analysis, the combination of lymphocyte count and MPV/PLT ratio remained significant, with ORs of 0.29 (95% CI: 0.09-0.96) and 3.10 (95% CI: 1.18-8.15), respectively, for predicting severe thrombocytopenia.
Discriminative Performance of CBC Indices
ROC curve analysis showed that the MPV/PLT ratio had the highest discriminative ability for severe thrombocytopenia with an AUC of 0.818, sensitivity of 75.0%, and specificity of 88.8% at an optimal cut-off of 0.0771 (P < .001; Figure 1). NLR demonstrated moderate predictive value (AUC = 0.656, P = .028), while PLR and SII did not show significant discriminative performance (AUC < 0.5, P > .05).

ROC Curves for CBC-Derived Indices in Predicting Severe Thrombocytopenia on Day 6.
Predictive Relationship Between MPV/PLT Ratio on Day 3 and PLT on Day 6
We investigated the predictive value of the MPV/PLT ratio measured on day 3 for the PLT on day 6 using regression analysis. Both linear and quadratic models were fitted to the data to characterize the relationship (Figure 2).
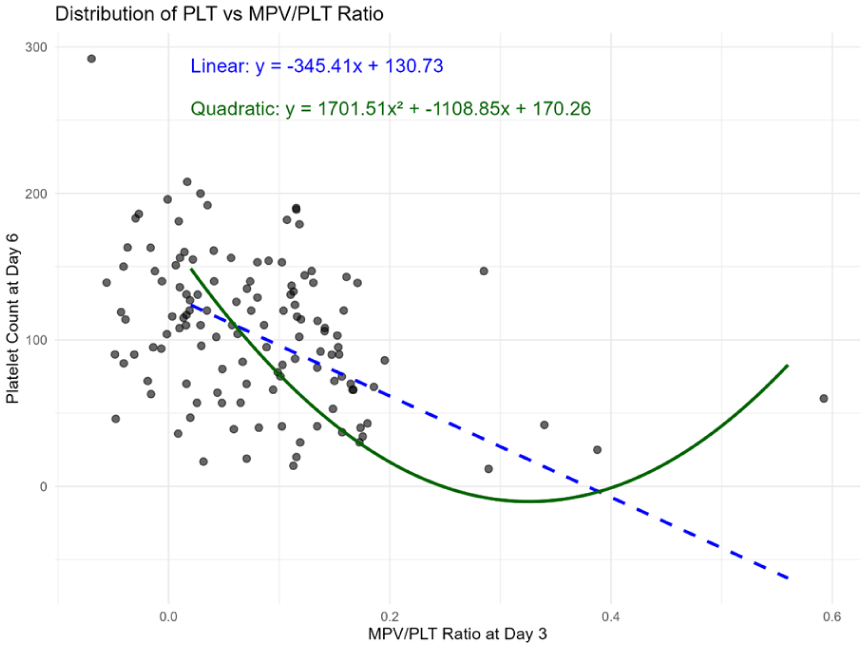
Relationship Between MPV/PLT Ratio and Platelet Count With Linear and Quadratic Fits. The quadratic regression formula is y = 1701.51x² − 1108.85x + 170.26. The quadratic model indicates that the inverse relationship between MPV/PLT on day 3 and platelet count on day 6 strengthens markedly beyond a threshold MPV/PLT value, reflecting increased risk of severe thrombocytopenia.
The linear regression model indicated a significant negative association between MPV/PLT on day 3 and PLT on day 6, described by the equation:

This suggests that higher MPV/PLT ratios on day 3 are associated with lower PLT on day 6.
However, the quadratic regression model provided a better fit, capturing a nonlinear relationship between these variables:

This curvilinear pattern implies that the association between MPV/PLT and subsequent PLT is more complex than a simple linear trend, possibly reflecting biological thresholds or compensatory mechanisms.
Discussion
In our study, severe thrombocytopenia was observed in 15.7% of the pediatric cohort, which is consistent with the range reported in previous studies of dengue fever in pediatric and mixed populations. For instance, Sontakke et al reported severe thrombocytopenia in 26.38% of dengue fever (DF) cases and 26.31% of dengue hemorrhagic fever/dengue shock syndrome (DHF/DSS) cases in a pediatric cohort, higher than our findings, possibly due to the inclusion of more severe cases. 6 In contrast, a study in Nepal during the 2022 dengue outbreak reported severe thrombocytopenia in 1.98% of 252 dengue patients (not exclusively pediatric), suggesting lower rates in some settings. 10 Similarly, a study during the 2016-2017 dengue outbreak in Burkina Faso reported severe thrombocytopenia in 2.5% of 296 patients, with 66.6% having thrombocytopenia (<150 000/µL). 11 Our observed prevalence of 15.7% falls within the reported range (1.98%-26.38%), with variations likely attributable to differences in study population, disease severity, serotype distribution, and timing of platelet measurement. The absence of severe dengue cases in our cohort may explain the relatively moderate prevalence compared to studies including DHF/DSS cases.
This prospective observational study evaluated the predictive value of mean MPV, MPV/PLT, and other CBC-derived indices in forecasting severe thrombocytopenia in children with dengue fever. Our findings demonstrate that elevated MPV and MPV/PLT ratio measured on day 3 of illness were significantly associated with the development of severe thrombocytopenia on day 6. Among the markers assessed, the MPV/PLT ratio showed the highest diagnostic accuracy, with an AUC of 0.818, a sensitivity of 75.0%, and a specificity of 88.8%. Additionally, decreased LYM and increased NLR were associated with thrombocytopenia, although NLR had a lower predictive power. From a clinical perspective, the ROC analysis identified an optimal cut-off value of 0.0771 for the MPV/PLT ratio on illness day 3. In practical terms, a ratio above this threshold may help clinicians recognize children at higher risk of developing severe thrombocytopenia. These patients could benefit from closer monitoring, such as daily CBC follow-up, and extended observation before hospital discharge, even if their platelet counts have not yet reached critical levels. Such an approach may facilitate earlier detection of platelet nadir and timely supportive interventions.
These findings support the potential clinical utility of MPV and MPV/PLT as early, low-cost biomarkers to identify children at risk for hematologic complications during dengue infection. Our results align with and extend previous observations in pediatric and adult dengue populations. For instance, Nandwani et al demonstrated that early hematological parameters, including lower platelet counts, were predictive of adverse outcomes in children with dengue in northern India, highlighting the importance of early CBC monitoring in pediatric settings. 5 Similarly, Islam et al found that altered hematological profiles, including reduced platelet counts, were associated with disease severity in children with dengue in a non-endemic region of Bangladesh, reinforcing the relevance of platelet indices in pediatric populations. 4 However, neither of these studies evaluated the MPV/PLT ratio, which our study identifies as a robust early predictor, thus adding novel insights to the pediatric dengue literature. In comparison, Sontakke et al reported a significant correlation between higher MPV and thrombocytopenia severity, highlighting MPV as a reflection of increased platelet activation and consumption, though MPV/PLT, NLR, PLR, and SII were not evaluated. 6 In adults, Shahila and Jothilingam observed elevated MPV with lower platelet counts, consistent with our findings, but did not assess MPV/PLT. 12 Similarly, Ojha et al noted higher MPV linked to thrombocytopenia in adults, reflecting platelet turnover. 13 Navya et al found NLR and PLR predictive of dengue severity in adults, suggesting stronger roles for these inflammatory indices compared to our pediatric cohort, where PLR was non-significant and NLR had moderate value. 14 SII is rarely studied in adult dengue, limiting comparisons, but its lack of predictive power in our study may reflect milder inflammation in non-severe pediatric cases. These differences likely stem from immunological factors, with pediatric patients often having primary infections and milder inflammatory responses, while adults in hyperendemic settings experience secondary infections with more pronounced inflammation, enhancing NLR and PLR’s predictive roles. 3
The MPV/PLT ratio, by combining 2 interrelated variables, has been increasingly explored as a robust indicator of platelet kinetics and inflammatory burden. Studies in sepsis and critical illness suggest that MPV/PLT may outperform MPV alone in predicting poor outcomes. 15 However, its application in dengue fever—particularly in children—remains underexplored. Our study adds to the literature by showing that the MPV/PLT ratio on day 3 is a strong early predictor of severe thrombocytopenia on day 6. This may provide clinicians with an accessible tool for identifying high-risk patients before their PLT reaches critical levels.
While NLR was significantly higher in the thrombocytopenia group, its AUC was moderate (0.656), indicating only fair discriminative ability. This aligns with previous studies which suggest that while NLR reflects systemic inflammation, it lacks disease-specific predictive power. 16 PLR and SII did not differ significantly between groups in our cohort. This contrasts with findings from a cross-sectional study in Southern India, which reported a mild association between PLR and dengue severity in adults. 14 This discrepancy may be due to differences in study population, disease stage at measurement, or age-related immune responses.
We also observed that lower LYM on day 3 were associated with increased risk of thrombocytopenia on day 6. This likely reflects early immune suppression or bone marrow involvement in dengue. Similar hematologic patterns have been described in pediatric tuberculosis, where lymphopenia reflects immune exhaustion during acute infection. 7 Dengue virus may cause direct marrow suppression and cytokine-induced lymphocyte apoptosis, explaining this association.
Importantly, the relationship between MPV/PLT and subsequent PLT appeared non-linear, with a potential threshold effect. This suggests that elevated MPV/PLT may be most predictive once a certain inflammatory or consumptive burden is reached. Such behavior has been proposed in other infectious diseases, 17 and highlights the potential of MPV/PLT as a dynamic early biomarker rather than a linear predictor.
This study has several strengths. It used a prospective design, employed standardized WHO 2009 criteria for diagnosis, and analyzed data from a relatively homogeneous pediatric population. Automated hematology analyzers minimized inter-observer variability in laboratory values. Nevertheless, the study has limitations. First, the modest sample size (n = 127) limited multivariable modeling and subgroup analysis. Second, the absence of severe dengue cases (eg, shock, plasma leakage, severe hemorrhage, or organ dysfunction) in our cohort means our findings are primarily applicable to non-severe dengue cases. This limits generalizability to severe dengue, where the predictive performance of MPV/PLT and other indices could differ due to massive bleeding, fluid shifts, or systemic inflammation. Third, we did not assess the impact of comorbidities such as sepsis, rheumatoid arthritis, inflammatory bowel disease, autoimmune diseases, renal failure, cirrhosis, B12 deficiency, or aplastic anemia, which could potentially alter the MPV/PLT ratio and mask its predictive value in dengue. While patients with known hematologic disorders (eg, leukemia, aplastic anemia) were excluded, other unrecorded comorbidities may have influenced our findings. Fourth, the correlation between MPV/PLT ratio on day 3 and platelet count on day 6, as shown in Figure 2, may be sensitive to outliers. The robustness of this correlation after excluding cases with elevated platelet counts or high MPV/PLT ratios remains uncertain and requires further validation. Fifth, only 2 time points (day 3 and day 6) were assessed, possibly missing temporal trends. Finally, this was a single-center study, which may reduce external validity. Future multicenter studies should evaluate MPV/PLT and MPV in larger cohorts, consider longitudinal trends, integrate additional biomarkers, and assess utility in severe dengue to expand clinical applicability.
Conclusion
In pediatric dengue, elevated MPV and MPV/PLT ratio on day 3 were linked to severe thrombocytopenia by day 6, with MPV/PLT showing the highest predictive accuracy. An MPV/PLT ratio above 0.0771 may help identify high-risk children for closer monitoring, guide daily CBC tracking, and support discharge decisions. Larger studies are needed to validate these findings and their clinical utility.
Footnotes
Acknowledgements
We would like to acknowledge all individuals and the hospital involved in the implementation of the survey.
Ethical Considerations
This study was conducted in accordance with the ethical standards outlined in the 1964 Helsinki Declaration and its subsequent amendments. Ethical approval was obtained from the Ethics Committee of Hue University of Medicine and Pharmacy, Hue City, Vietnam (Approval Number: H2023/208, Approval Date: May 24, 2023). Written informed consent was obtained from the parents or legal guardians of all pediatric participants prior to enrollment. Personal information, including names, phone numbers, and addresses, was not collected, and all data were anonymized and maintained with strict confidentiality to protect participant privacy.
Author Contributions
Chau-Duc Nguyen-Huu, as the Principal Investigator, conceptualized and designed the study, oversaw the overall research process, contributed to data interpretation, and drafted and revised the manuscript. Thi Lan Nguyen was primarily responsible for data collection, ensuring accurate and timely acquisition of clinical and laboratory data. Nhu-Huy Pham and Van-Tuy Nguyen developed the methodology, including the study protocol and statistical analysis plan, and contributed to data interpretation. Quoc Bao Vo, Hoang Le, Tuan Tai Manh, and Thi Tam Dang contributed equally to study design, data collection, data analysis, and manuscript preparation, ensuring comprehensive involvement in all stages of the research process. All authors reviewed and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
