Abstract
Objective
To evaluate the association between the uric acid-to-albumin ratio and adverse clinical outcomes in patients with hypertrophic cardiomyopathy.
Methods
This retrospective, observational single-center study included 124 patients with hypertrophic cardiomyopathy (58 with obstructive and 66 with nonobstructive hypertrophic cardiomyopathy) and 60 age- and sex-matched healthy controls. Baseline clinical, laboratory, and echocardiographic parameters were analyzed. A composite endpoint comprising cardiovascular death, heart failure hospitalization, and appropriate implantable cardioverter-defibrillator shocks was assessed. Logistic regression analyses were used to identify independent associations, and receiver operating characteristic analysis was performed to evaluate discriminative ability.
Results
The uric acid-to-albumin ratio was higher in patients with obstructive hypertrophic cardiomyopathy than in those with nonobstructive hypertrophic cardiomyopathy and controls (1.56 ± 0.15 vs 1.21 ± 0.08 vs 1.15 ± 0.11; p < 0.001). Adverse events occurred in 32 patients with hypertrophic cardiomyopathy (25.8%) and were more frequent in the obstructive group (34.5% vs 18.2%; p = 0.038). In multivariable analysis, higher uric acid-to-albumin ratio (per 0.1 increase; OR, 1.50; 95% confidence interval: 1.18–1.90; p = 0.001) and septal thickness (OR, 1.65; 95% confidence interval: 1.30–2.10; p < 0.001) were independently associated with adverse outcomes. Receiver operating characteristic analysis yielded an area under the curve of 0.717 (p < 0.001).
Conclusions
Elevated uric acid-to-albumin ratio was independently associated with adverse outcomes in hypertrophic cardiomyopathy and may serve as a simple and inexpensive biomarker for risk stratification.
Introduction
Hypertrophic cardiomyopathy (HCM) is one of the most common inherited cardiovascular diseases, with a prevalence of approximately 1 in 500 in the general population. It is characterized by unexplained left ventricular hypertrophy and a heterogeneous clinical course.1,2 Despite substantial advances in diagnosis and management, risk stratification in HCM remains challenging due to substantial variability in phenotypic expression and clinical outcomes.3,4
Recent studies have highlighted the genetic diversity underlying HCM, with sarcomeric and nonsarcomeric mutations contributing to disease development and progression. 5 This heterogeneity underscores the need for additional biomarkers to refine risk assessment beyond established clinical and imaging parameters.
Among circulating biomarkers, uric acid has been associated with oxidative stress, endothelial dysfunction, and adverse cardiovascular outcomes in various populations. 6 Serum albumin reflects nutritional and inflammatory status, with lower levels associated with poor prognosis in heart failure and ischemic heart disease. 7 The uric acid-to-albumin ratio (UAR), which integrates these two parameters, has been proposed as a better indicator of cardiovascular risk than either marker alone. 8
However, the clinical relevance of UAR in patients with HCM has not been investigated. Therefore, this study aimed to evaluate the association between UAR and adverse clinical outcomes in patients with HCM.
Materials and methods
Study design and population
This retrospective, single-center observational study was conducted in accordance with the Strengthening of Reporting of Observational Studies in Epidemiology (STROBE) guidelines 9 and the Declaration of Helsinki 1975, as revised in 2024.
A total of 124 consecutive patients diagnosed with HCM between 2017 and 2025 were included in the analysis. The diagnosis of HCM was established according to current guideline criteria. 1 Patients were categorized as having obstructive HCM (left ventricular outflow tract (LVOT) gradient ≥30 mmHg at rest or with provocation; n = 58) or nonobstructive HCM (n = 66). A control group of 60 age- and sex-matched apparently healthy individuals with no structural heart disease, chronic kidney disease, inflammatory or autoimmune disorders, or malignancy was also included.
Eligible participants were ≥18 years old and had complete baseline laboratory and echocardiographic data. To minimize confounding, only individuals with preserved renal function (estimated glomerular filtration rate (eGFR) ≥60 mL/min/1.73 m2) were included. Exclusion criteria included acute infection, chronic inflammatory or autoimmune disease, malignancy, advanced chronic kidney disease (eGFR <60 mL/min/1.73 m2), incomplete data, or current use of uric acid–modifying agents (e.g. allopurinol, febuxostat, and probenecid) or long-term diuretic therapy.10,11
Data collection
Demographic and clinical data, including comorbidities (hypertension, diabetes mellitus, and hyperlipidemia) and New York Heart Association (NYHA) functional class, were extracted from electronic medical records and independently verified by two investigators. Laboratory tests included serum uric acid, albumin, creatinine, and C-reactive protein (CRP) measured using standardized automated assays. The UAR was calculated as serum uric acid (mg/dL) divided by albumin (g/dL). Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) formula was used to determine eGFR. 12
All patients underwent comprehensive transthoracic echocardiography in accordance with current recommendations. 13 Interventricular septal thickness, posterior wall thickness, left atrial diameter, and left ventricular ejection fraction (LVEF) were recorded. LVOT gradients were assessed at rest and with provocation using continuous wave Doppler.
Adverse clinical events were identified retrospectively from patient records and verified using discharge summaries and outpatient follow-up documentation. Appropriate implantable cardioverter-defibrillator (ICD) shocks were defined as therapies delivered for documented ventricular tachyarrhythmias, as confirmed through device interrogation and reviewed by an electrophysiologist.
The median follow-up duration was 32 months (interquartile range, 24–48 months). The composite endpoint comprised cardiovascular death, hospitalization for heart failure, and appropriate ICD shocks. All patient data were deidentified to ensure confidentiality.
Statistical analysis
Continuous variables were expressed as mean ± SD or median (interquartile range) and compared among groups using one-way analysis of variance (ANOVA) or Kruskal–Wallis test with post hoc pairwise analysis as appropriate. Categorical variables were presented as frequencies (%) and compared using the chi-square test or Fisher’s exact test with Bonferroni correction. Correlations between UAR and clinical or echocardiographic parameters were assessed using Pearson’s correlation coefficient.
Binary logistic regression analysis was performed to identify independent associations with the composite endpoint. Variables with p <0.10 in univariable analysis or those with known clinical relevance in HCM (septal thickness, LVOT gradient, UAR, and CRP) were included in the multivariable model. Due to potential collinearity, uric acid and albumin were not analyzed separately. Variables were retained in the final model if they remained statistically significant. Model calibration was assessed using the Hosmer–Lemeshow test, and explanatory power was reported as Nagelkerke R2.
Receiver operating characteristic (ROC) curve analysis was used to assess discriminative ability, and the optimal cutoff value for UAR was determined using the Youden index. A two-tailed p-value <0.05 was considered statistically significant. All statistical analyses were performed using Statistical Package for Social Sciences (SPSS) version 26 (IBM Corp.; Armonk, NY, USA).
As this was a retrospective observational study, the sample size was determined by the number of eligible patients who met the inclusion criteria during the study period (2017–2025). Although no formal sample size calculation was performed a priori, the number of variables included in multivariable models was limited to prevent overfitting, following the commonly recommended rule of at least 10 events per variable. This approach helps ensure reasonable model stability given the available number of adverse outcomes.
Results
A total of 184 individuals were included: 60 controls, 66 patients with nonobstructive HCM, and 58 with obstructive HCM. Baseline clinical, laboratory, and echocardiographic characteristics are summarized in Table 1. There were no significant differences in age or sex distribution across groups. Hypertension and diabetes were more prevalent in patients with HCM, particularly in the obstructive group (both p < 0.001). Patients with obstructive HCM also demonstrated higher rates of moderate-to-severe mitral regurgitation and had more advanced NYHA functional class compared with those observed in the other groups (both p < 0.001). Laboratory analyses revealed that patients with obstructive HCM had significantly higher serum uric acid levels and lower albumin levels than patients with nonobstructive HCM and controls (both p < 0.001). Consequently, UAR was highest in obstructive HCM (1.56 ± 0.15), intermediate in nonobstructive HCM (1.21 ± 0.08), and lowest in controls (1.15 ± 0.11; p < 0.001). CRP levels, reflecting systemic inflammation, were most pronounced in patients with obstructive HCM (p < 0.001). Echocardiographic assessment showed significantly increased interventricular septal and posterior wall thickness in both HCM groups, with greater hypertrophy observed in the obstructive HCM group compared to that in the nonobstructive HCM group (p < 0.001). Left atrial diameter was significantly enlarged in obstructive HCM (p < 0.001), whereas LVEF remained similar across groups. LVOT gradients at rest and after provocation were significantly higher in the obstructive group (both p < 0.001). The distribution of UAR values across the study groups is depicted in Figure 1. During follow-up, composite adverse events occurred in 32 patients with HCM (25.8%). As shown in Table 2, the incidence of the composite endpoint was significantly higher in patients with obstructive HCM than in those with nonobstructive HCM (34.5% vs 18.2%; p = 0.038). Rates of cardiovascular death and appropriate ICD therapies were low and did not differ significantly between the groups. In multivariable logistic regression analysis, septal thickness (OR, 1.65; 95% confidence interval (CI): 1.30–2.10; p < 0.001) and UAR (per 0.1 increase; OR, 1.50; 95% CI: 1.18–1.90; p < 0.001) were independently associated with the composite endpoint, whereas LVOT gradient and CRP were not independently significant (Table 3). The final model explained approximately 39% of the variance in adverse outcomes (Nagelkerke R2 = 0.388) and demonstrated good calibration (Hosmer–Lemeshow χ2 = 7.41; df = 8; p = 0.494). Correlation coefficients are presented in Table 4. UAR showed a strong correlation with LVOT gradient at rest (r = 0.85; p < 0.001) and a moderate correlation with CRP (r = 0.47; p < 0.001). A weaker but significant correlation was observed between UAR and septal thickness (r = 0.21; p = 0.018). Figure 2 illustrates the strong linear relationship between UAR and LVOT gradient. ROC analysis indicated that UAR predicted the composite endpoint with an area under the curve (AUC) of 0.717 (95% CI: 0.604–0.829; p < 0.001). The optimal cutoff value of 1.19 provided a sensitivity of 87.5% and specificity of 71.7% (Table 5). The ROC curve is presented in Figure 3.
Baseline characteristics of the study population.

Fisher’s exact test.
Baseline demographic, clinical, laboratory, and echocardiographic characteristics of the study population. Data are presented as mean ± standard deviation or n (%).
BMI: body mass index; CRP: C-reactive protein; eGFR: estimated glomerular filtration rate; HCM: hypertrophic cardiomyopathy; IVS: interventricular septum; LA: left atrium; LVEF: left ventricular ejection fraction; LVOT: left ventricular outflow tract; NYHA: New York Heart Association; UAR: uric acid-to-albumin ratio.

Distribution of the uric acid-to-albumin ratio (UAR) across the study groups. UAR values were significantly higher in patients with obstructive hypertrophic cardiomyopathy (HCM) than in those with nonobstructive HCM and controls (p < 0.001).
Clinical outcomes in patients with HCM.

Distribution of clinical outcomes in patients with nonobstructive and obstructive HCM patients. Data are presented as n (%). The composite endpoint included cardiovascular death, hospitalization for heart failure, and appropriate ICD shocks.
HCM: hypertrophic cardiomyopathy; ICD: implantable cardioverter-defibrillator.
Logistic regression analysis of predictors of the composite outcome in patients with HCM.
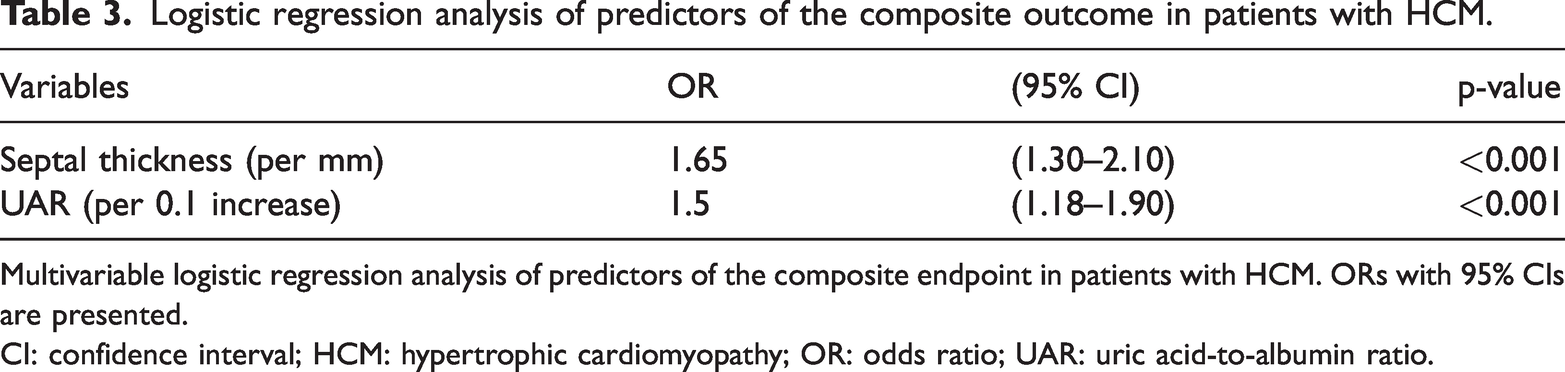
Multivariable logistic regression analysis of predictors of the composite endpoint in patients with HCM. ORs with 95% CIs are presented.
CI: confidence interval; HCM: hypertrophic cardiomyopathy; OR: odds ratio; UAR: uric acid-to-albumin ratio.
Correlations between UAR and clinical and echocardiographic parameters in patients with HCM.

Pearson correlation coefficients between UAR and selected clinical, laboratory, and echocardiographic parameters in patients with HCM (n = 124).
CI: confidence interval; CRP: C-reactive protein; HCM: hypertrophic cardiomyopathy; LA: left atrium; LVOT: left ventricular outflow tract; UAR: uric acid-to-albumin ratio.

Correlations of UAR with (a) left ventricular outflow tract (LVOT) gradient at rest and (b) C-reactive protein (CRP) levels in patients with HCM. UAR showed a strong correlation with LVOT gradient (r = 0.85; p < 0.001) and a moderate correlation with CRP (r = 0.47; p < 0.001). HCM: hypertrophic cardiomyopathy; UAR: uric acid-to-albumin ratio.
ROC analysis of UAR for predicting the composite outcome in patients with HCM.
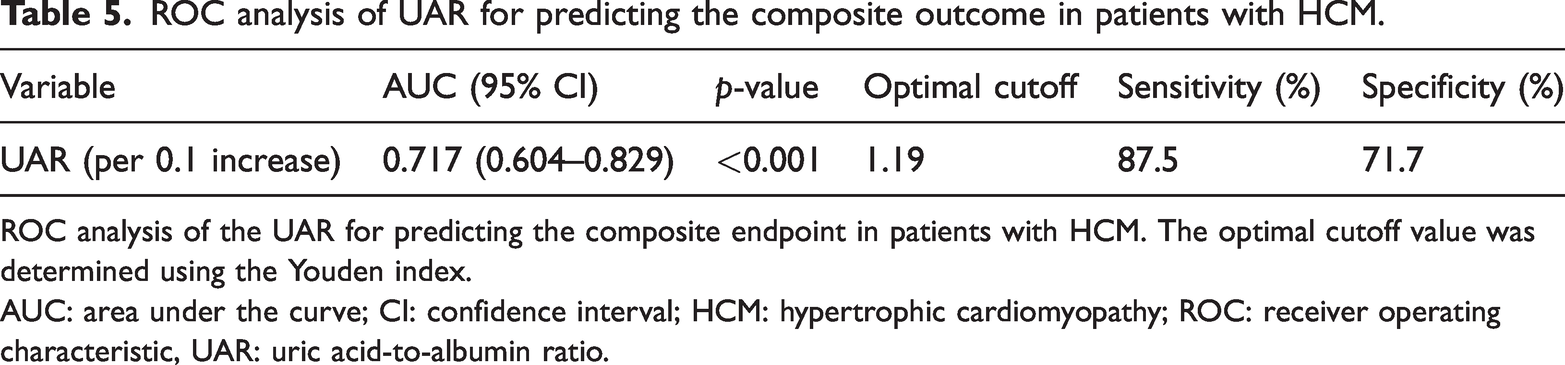
ROC analysis of the UAR for predicting the composite endpoint in patients with HCM. The optimal cutoff value was determined using the Youden index.
AUC: area under the curve; CI: confidence interval; HCM: hypertrophic cardiomyopathy; ROC: receiver operating characteristic, UAR: uric acid-to-albumin ratio.
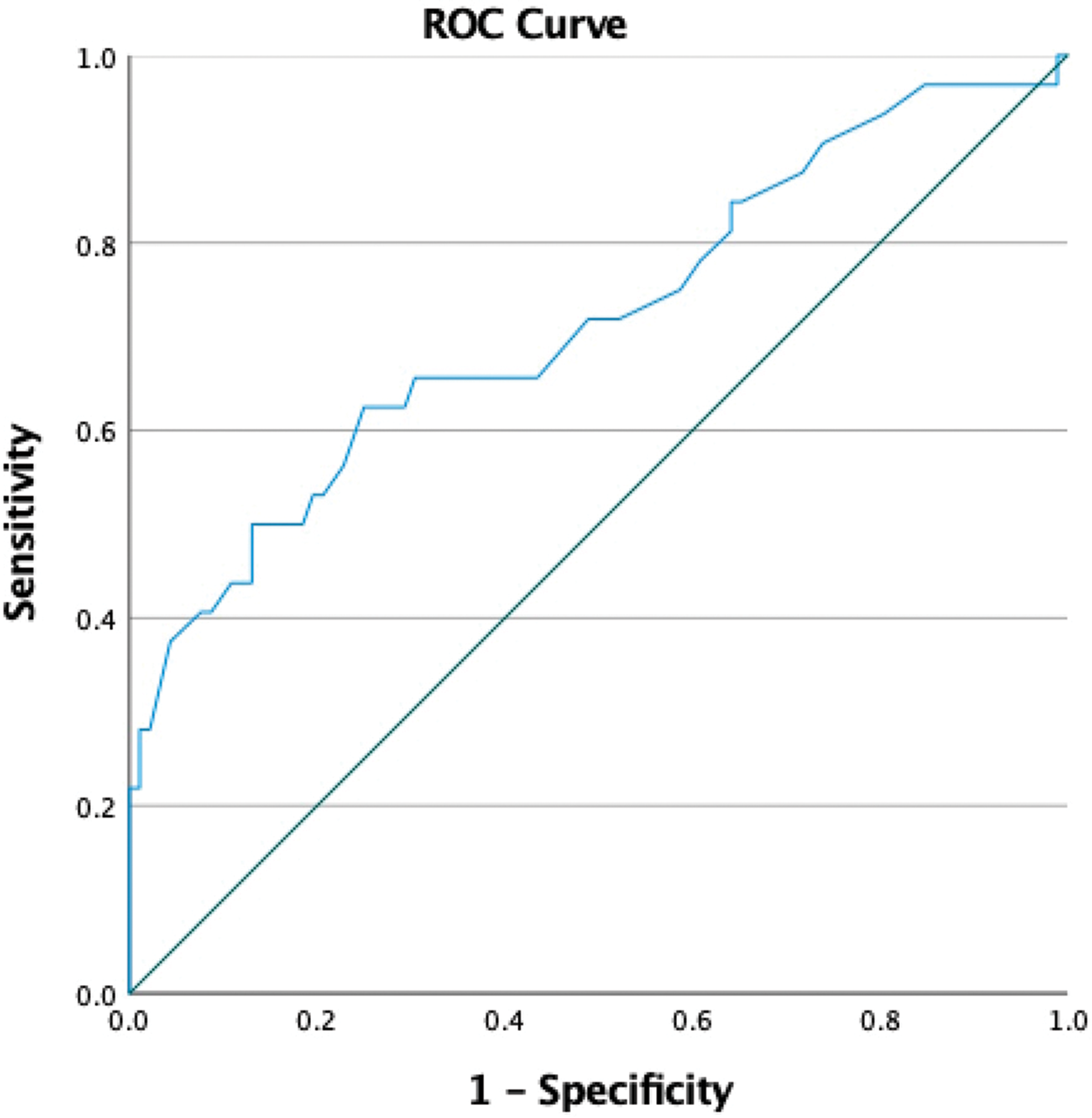
Receiver operating characteristic (ROC) curve of UAR for predicting the composite endpoint in patients with HCM. The area under the curve (AUC) was 0.717 (95% CI: 0.604–0.829; p < 0.001). An optimal cutoff value of 1.19 yielded a sensitivity of 87.5% and a specificity of 71.7%. CI: confidence interval; HCM: hypertrophic cardiomyopathy; UAR: uric acid-to-albumin ratio.
Discussion
In this retrospective analysis, we found that UAR was independently associated with adverse outcomes in patients with HCM. Along with septal thickness, UAR remained significant after multivariable adjustment. Notably, UAR showed a strong correlation with LVOT gradient and a moderate correlation with CRP, suggesting that it reflects hemodynamic severity and systemic inflammation. Serum albumin levels were significantly lower in the obstructive HCM group, indicating that the elevated UAR in these patients is driven not only by higher uric acid concentrations but also by marked hypoalbuminemia. These findings support the role of systemic inflammation and metabolic stress in disease severity.
Our findings build upon earlier studies that focused solely on uric acid. Gao et al. reported that elevated uric acid was associated with cardiac death in patients with obstructive HCM, 14 whereas Wang et al. described a U-shaped relationship between uric acid and mortality. 15 Similarly, Özyılmaz et al. reported a positive correlation between uric acid and the predicted 5-year sudden cardiac death (SCD) risk score. 16 Collectively, these studies highlight the potential role of metabolic and inflammatory disturbances in adverse outcomes in HCM. Consistent with this concept, recent evidence suggests that HCM is characterized by a prothrombotic and hypercoagulable milieu, with intracardiac thrombus formation possible even in the absence of atrial fibrillation. 17
Moreover, contemporary data indicate that currently used SCD prediction models demonstrate suboptimal accuracy in real-world cohorts, highlighting the need for adjunctive biomarkers to enhance risk stratification. 18 Notably, adverse events have been reported among patients with HCM classified as low risk by conventional SCD algorithms, underscoring the relevance of additional clinical markers such as UAR. 19 Collectively, these findings emphasize the potential value of UAR within a broader framework linking metabolic dysregulation, inflammation, and hemodynamic burden to arrhythmic vulnerability.
Beyond uric acid, albumin and albumin-based indices have increasingly been recognized as prognostic markers in HCM. Low albumin reflects systemic inflammation and impaired nutritional status. Previous studies have demonstrated the prognostic value of the albumin-to-fibrinogen ratio, 20 albumin–bilirubin (ALBI) score, 21 and prognostic nutritional index. 22 By integrating uric acid and albumin into a single ratio, UAR may serve as a more comprehensive marker of metabolic stress, inflammation, and nutritional reserve. To the best of our knowledge, this is the first study specifically assessing UAR in HCM, and our results suggest that it may function as a novel prognostic marker. Supporting this mechanistic interpretation, recent studies have demonstrated that uric acid–related inflammatory markers, including UAR, can predict atrial fibrillation recurrence following ablation procedures, highlighting the link between oxidative stress, inflammation, and arrhythmic vulnerability.23,24
In ROC analysis, UAR demonstrated acceptable discriminatory capacity (AUC, 0.717), and the logistic regression model explained approximately 40% of the variance in adverse outcomes. Although this explanatory power may appear modest, it is comparable to that of other established clinical biomarker studies, where Nagelkerke R2 values typically range from 0.2 to 0.4. Notably, UAR remained significant in the multivariable model even when LVOT gradient lost significance, suggesting that it may provide prognostic information beyond traditional hemodynamic parameters. This observation raises an important mechanistic question: Does UAR primarily reflect the severity of LVOT obstruction, or does it provide independent prognostic information? The persistence of UAR in the multivariable model supports the latter, suggesting that the metabolic and inflammatory pathways captured by UAR extend beyond hemodynamic burden alone.
Several mechanisms may account for our findings. Hyperuricemia promotes oxidative stress, endothelial dysfunction, and renin–angiotensin system activation, all of which contribute to myocardial fibrosis and remodeling.6,25 Albumin, a negative acute phase reactant, decreases in states of inflammation and malnutrition, both known to worsen outcomes. 26 Experimental and clinical studies have also suggested that uric acid may exert direct detrimental effects on cardiomyocytes, promoting mitochondrial dysfunction, oxidative cellular injury, and impaired contractility.27–29 These mechanisms support the biological plausibility of UAR as an integrated indicator of myocardial stress and vulnerability.
A high UAR therefore reflects the coexistence of elevated oxidative stress and impaired systemic resilience, a condition that may predispose patients to arrhythmic events, heart failure progression, and increased mortality. The strong correlation between UAR and LVOT gradient further suggests a link between metabolic stress and obstructive physiology.
Clinically, risk stratification in HCM currently relies on structural, hemodynamic, and arrhythmic factors. 1 Incorporating biomarkers such as UAR may offer incremental prognostic value and help identify patients at higher risk who could benefit from more intensive monitoring or earlier therapeutic interventions. Given its simplicity and low cost, UAR may be particularly useful in clinical settings where advanced imaging or genetic testing is not readily available.
Limitations
Several limitations must be acknowledged. First, this was a retrospective and single-center study with a relatively small sample size and a limited number of clinical events, raising the possibility of residual confounding and model overfitting. Second, outcomes were obtained from existing patient records rather than prospective follow-up, which may have led to an underestimation of event rates. Third, although patients with advanced renal dysfunction and those on uric acid–modifying or diuretic therapy were excluded, unmeasured factors, such as dietary intake, concomitant medications, or subclinical inflammatory conditions, may still have influenced the results. Fourth, the limited number of events may have constrained the robustness of multivariable analyses and the inclusion of additional covariates. Finally, left ventricular mass index and related structural parameters were not systematically available in our dataset and therefore could not be included in the analyses. Consequently, their potential influence on the observed associations cannot be excluded. In addition, the optimal UAR cutoff value was derived from the present cohort and has not been externally validated, warranting confirmation in larger, prospective, multicenter studies.
Conclusion
In this retrospective study, UAR was independently associated with adverse outcomes in patients with HCM, alongside septal thickness. UAR showed a strong correlation with LVOT gradient and a moderate correlation with CRP, reflecting hemodynamic burden and systemic inflammation. However, the absence of left ventricular mass and left ventricular mass index data limits comprehensive adjustment for structural disease severity; therefore, the results should be interpreted cautiously. Overall, these findings suggest that UAR may serve as a simple, inexpensive, and clinically accessible biomarker for risk stratification in HCM. Future prospective, multicenter studies are warranted to validate its prognostic utility and to clarify its role in routine clinical practice.
Footnotes
Acknowledgments
Not applicable.
Authors’ contributions
A.Ü.K and T.K contributed to study conception and design; data collection, analysis, and manuscript preparation. Both authors read and approved the final manuscript.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare no conflicts of interest.
Ethical approval and consent to participate
The study was approved by the Akdeniz University Scientific Research Ethics Committee (Approval No. 782; 28 August 2025) and was conducted in accordance with the principles of the Helsinki Declaration of 1975, as revised in 2024. Written informed consent was waived due to the retrospective and anonymized nature of the data.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
