Abstract
This review highlights challenges in recognition, diagnosis, and management of neonatal sepsis in low-resource emergency settings. It discusses actionable, resource-adapted strategies to reduce neonatal morbidity and mortality. A comprehensive literature review was conducted using peer-reviewed research articles and global health guideline addressing neonatal sepsis. Neonatal sepsis is the leading cause of newborn deaths, with low- and middle-income countries accounting for 99% of cases. Since limited access to blood cultures, biomarker testing, and antibiotic availability delay diagnosis and management. Furthermore, antimicrobial resistance, poor infection control, and socioeconomic barriers worsen outcomes. However, emerging interventions including Point of care diagnostics, Kangaroo Mother Care, telemedicine, and artificial intelligence-based technologies demonstrate effectiveness. Early recognition and evidence-based management should be supported through affordable, feasible diagnostics, refined antibiotic use, and strengthened health infrastructure. Future initiatives must focus on innovations and international collaborations aimed at enhancing neonatal survival and health.
Keywords
Introduction
Neonatal sepsis is a severe, life-threatening condition characterized by a systemic inflammatory response to infection in newborns, yet no universally agreed-upon definition exists. Clinically, the diagnosis of neonatal sepsis depends on a combination of clinical signs such as fever, respiratory compromise, and apnea, in addition to supportive laboratory findings like elevated C-reactive protein (CRP) or a positive blood or cerebrospinal fluid (CSF) culture. A positive culture from either blood or CSF remains the gold standard for diagnosis; however, most neonates with suspected sepsis receive treatment despite negative blood culture results. This is largely due to the suboptimal sensitivity of cultures obtained from small blood volume samples and the precautionary clinical approach of initiating empiric treatment. 1
Globally, neonatal sepsis contributes to significant morbidity and mortality, particularly in resource-limited emergency settings. In 2019, neonatal sepsis was responsible for an estimated 6.31 million cases and approximately 230 000 deaths worldwide, with the heaviest burden in LMICs. In low- and middle-income countries (LMICs), the incidence of clinically suspected sepsis is estimated at 166 per 1000 live births, while laboratory-confirmed sepsis at 46.9 per 1000 live births. 1
This high mortality burden in LMICs is largely preventable. Poverty and inadequate education for women are socio-demographic challenges that contribute to these high mortality rates. Health system challenges related to both service provision and utilization are further hindered by limited resources. Furthermore, early recognition is constrained by limited diagnostics in these environments. Inadequate access to care during the preconception, antenatal, intrapartum, and postnatal periods increases the risk of neonatal sepsis and its adverse consequences. The risk is further increased by poor-quality care, the lack of essential antibiotics, and the rising antimicrobial resistance. 1 Table 1 outlines key differences in neonatal sepsis management between high and low-resource settings, highlighting factors that contribute to the higher burden of neonatal sepsis in LMICs.
Key Differences in Neonatal Sepsis Management: High- Versus Low-Resource Settings.

Despite a 36% increase in the consumption of antibiotics between 2000 and 2010, resource-constrained settings continue to have poor access to essential medications, resulting in increased morbidity and mortality. Accessibility of antibiotics isn’t the only problem; inappropriate prescribing practices has exacerbated resistance, with multidrug-resistant organisms now responsible for approximately 30% of all deaths from neonatal sepsis. 1
Addressing these issues requires urgent adoption of evidence-based strategies tailored to low-resource emergency settings and aimed at enhancing early sepsis recognition, appropriate antibiotic use, and robust healthcare systems to mitigate preventable neonatal fatalities. 1
Methods
This article is a narrative review that aimed to synthesize and summarize the available evidence on neonatal sepsis in low-resource emergency settings. Relevant literature was identified through a non-systematic search of electronic databases (PubMed, Google Scholar, and WHO/UNICEF reports), as well as manual review of reference lists from pertinent articles. Priority was given to studies and reports describing epidemiology, challenges, and management strategies in resource-limited contexts. No formal inclusion/exclusion criteria, quality appraisal, or systematic data extraction procedures were applied, as the intent of this review was to provide a broad overview of existing knowledge and highlight key gaps rather than conduct a systematic synthesis.
Pathophysiology and Early Recognition
The pathophysiology of sepsis in neonates differs from that in adults, with unique host immune responses to septic shock. Figure 1 provides an overview of the pathophysiology of neonatal sepsis. The neonatal adaptive immune system is underdeveloped and relies on B- and T-cell differentiation to neutralize intracellular and extracellular pathogens. Th1 and Th2 responses are distinct T helper cell (Th cell) subsets that play different roles in the immune system. Th1 responses are primarily involved in cell-mediated immunity, fighting intracellular pathogens, while Th2 responses are crucial for humoral immunity, combating extracellular pathogens. However, neonatal B- and T-cell differentiation is relatively immature. Activated B cells are predisposed to IgM isotype switching and demonstrate reduced responsiveness to T cell receptor (TCR)-induced stimuli. This results in a Th2-biased cytokine profile, characterized by higher levels of interleukin-4 (IL-4), interleukin-5 (IL-5), and interleukin-13 (IL-13), leading to an immature humoral immune response. In addition, the emergence of regulatory T (Treg) cells in the neonatal immune system is also underdeveloped. 2

Pathophysiology of neonatal sepsis and immune response.
Neonatal innate immunity is underdeveloped, with dendritic cells (DCs) serving as the primary immune cells responsible for pathogen detection and immune activation. DCs produce fewer pro-inflammatory cytokines and decrease the production of type I interferons (IFNs), limiting their effectiveness. Pattern recognition receptors (PRRs) are essential for DC activation and pathogen differentiation. Moreover, neonatal monocytes display a tolerogenic phenotype, characterized by elevated interleukin-10 (IL-10) secretion that suppresses the induction of a protective Th1 response to pathogens. 3
Neonatal sepsis symptoms range from nonspecific findings to hemodynamic collapse, with early symptoms including irritability, lethargy, and poor feeding. Without timely recognition and intervention, the condition can progress to a critical stage, potentially resulting in lifelong cognitive impairments due to imbalances in pro-inflammatory and anti-inflammatory responses. Despite the initial protection provided by the maternal immune barrier, neonatal immunity remains immature. Early diagnosis and effective management are crucial for preventing long-term adverse outcomes. 4
Etiology and Risk Factors in Low-Resource Settings
The late recognition of sepsis contributes to worsened outcomes. Awareness of the unique features of neonatal sepsis in low-resource settings will help direct efforts and research aimed at improving outcomes, particularly by examining the etiology, risk factors, pathogen prevalence, and patterns of antibiotic resistance. 5
Common Bacterial and Viral Causes of Neonatal Sepsis
Neonatal sepsis can be classified into early-onset sepsis (EOS) and late-onset sepsis (LOS), each with distinct etiological agents. EOS, occurring within the first 72 hours of life, is often caused by pathogens acquired during childbirth, while LOS, occurring after 72 hours, is typically linked to nosocomial or community-acquired infections. 6
Bacterial Pathogens
Gram-positive and Gram-negative bacteria are the most common etiological agents of neonatal sepsis in low-resource settings. Group B Streptococcus (GBS) and Escherichia coli are predominant causes of EOS, often transmitted from the mother during delivery. 7 Staphylococcus aureus, Klebsiella pneumoniae, and Pseudomonas aeruginosa are frequently implicated in LOS, particularly in hospital settings where infection control measures are inadequate. 8 In a collaborative study involving 7 LMICs, molecular sequencing identified Klebsiella pneumoniae as the most commonly isolated bacterium causing sepsis, followed by Klebsiella michiganensis, Staphylococcus aureus, Serratia marcescens, and Burkholderia cenocepacia. Meanwhile, Escherichia coli, Enterobacter cloacae, and Acinetobacter spp. were found to be the most widespread pathogens. 9 Similarly, in a single study from Nepal, Klebsiella pneumoniae was the most common isolate in neonatal sepsis. 10
Viral Pathogens
Although bacterial infections are the primary cause of neonatal sepsis, viral sepsis is a major burden in LMICs. Viruses also contribute significantly to neonatal morbidity and mortality. Cytomegalovirus (CMV), herpes simplex virus (HSV), and enteroviruses can cause severe neonatal infections, often leading to complications such as encephalitis and systemic inflammatory responses.11,12 Diagnosis is severely hampered by a lack of adequate microbiological capacity. Consequently, management relies on syndromic approaches and context-specific clinical algorithms. The use of rapid diagnostics and creative, feasible adaptations to standard care are essential for improving outcomes. 13
Risk Factors for Neonatal Sepsis
Preterm Birth and Low Birth Weight
Preterm neonates have an underdeveloped immune system and an increased likelihood of requiring invasive medical procedures, such as mechanical ventilation and central venous catheterization, which elevate their risk of sepsis. A multi-country study by Milton et al confirmed that preterm birth significantly contributes to higher risks for neonatal sepsis in LMICs. 9 Preterm neonates may be unable to resume breastfeeding promptly, and breast milk is crucial for prevention of infections. 10
Maternal Infections
Maternal infections such as chorioamnionitis, urinary tract infections, and sexually transmitted infections increase the risk of vertical transmission of pathogens to the neonate. 7 In settings with limited access to prenatal care, untreated maternal infections contribute to a higher burden of neonatal sepsis. In LMICs, maternal conditions such as previous hospitalizations and hypertension have been shown to be associated with increased risk of sepsis in neonates. 9
Unhygienic Birth Practices
Inadequate hand hygiene, unsterile delivery environments, and the use of contaminated instruments during childbirth significantly increase the risk of neonatal infections. 14 Home deliveries without skilled birth attendants further compound this risk, as newborns are exposed to pathogenic bacteria from the environment or birth attendants. 15 Practicing hygiene by mothers, such as proper handwashing, have been found to be significantly associated with a lower risk of neonatal sepsis in Uganda. 16
Malnutrition and Immunodeficiency
Neonates with poor nutritional status, particularly those suffering from intrauterine growth restriction (IUGR), have weaker immune responses and are more susceptible to infections. 17 Additionally, infants born to HIV-positive mothers are at an increased risk of sepsis due to perinatal exposure and immunodeficiency. 8
Table 2 summarizes the risk factors.
Clinical Risk Factors for Neonatal Sepsis Without Laboratory Access.

Regional Variations in Pathogens
The microbial landscape of neonatal sepsis varies significantly across geographic regions, influenced by local environmental conditions, healthcare infrastructure, and antibiotic usage patterns.
In sub-Saharan Africa, Gram-negative bacteria such as Klebsiella pneumoniae and Escherichia coli are dominant causes of neonatal sepsis, largely due to high rates of hospital-acquired infections. 15 In South Asia, GBS and Staphylococcus aureus are among the leading pathogens, often linked to unhygienic birth conditions and inadequate maternal screening programs. 8
Diagnostic Challenges in Low-Resource Emergency Departments
Rapid and accurate diagnostics are crucial for effective treatment and complication prevention in neonatal sepsis. An important initial step in identifying most conditions is clinical assessment, but this is challenging in neonatal sepsis due to the nonspecific and variable symptoms seen in neonates. Many signs of infection overlap with other neonatal conditions such as respiratory distress syndrome (RDS), congenital heart disease, bowel obstruction, and metabolic disorders. 18 Additionally, early signs of infection may be mistaken for normal transitional changes in preterm neonates, making diagnosis even more difficult. Conversely, the absence of symptoms does not rule out infection, particularly in high-risk neonates. Since clinical assessment alone lacks sensitivity, the diagnosis of neonatal sepsis relies on a combination of clinical presentation, microbiological cultures, and laboratory markers. 19
Limitations in Laboratory Testing
Culture
While blood culture remains the gold standard for diagnosing neonatal sepsis, several challenges make its use less practical in LMICs. The high cost of consumables (e.g., blood culture bottles) along with transport delays and exposure to high temperatures, can compromise sample integrity and hinder timely diagnosis. 1 Additionally, the 24 to 48-hour turn-around time further hinders timely, targeted treatment. Another major issue is culture-negative sepsis, which accounts for a significant proportion of cases. This occurs due to small blood volumes obtained from neonates, low or intermittent bacteremia, and maternal intrapartum antibiotic exposure. 19 These limitations indicate that culture alone is not always a reliable diagnostic tool, highlighting the need for additional complementary methods.
CBC
Interpreting a CBC count in neonates is challenging due to its significant variability based on gestational age, postnatal age, and circulatory changes. Markers such as WBC count, immature/total neutrophil ratio (I/T ratio), and absolute neutrophil count (ANC) help assess the risk of EOS, but they have several limitations. Neutrophil counts can be influenced by maternal conditions (e.g., hypertension, fever), intrapartum events (e.g., asphyxia, mode of delivery), and perinatal complications (e.g., periventricular hemorrhage, hemolytic disease, pneumothorax). 19 Additionally, interpretation of CBC parameters requires careful consideration, as their values can vary depending on the type of sepsis. In EOS, low WBC, low ANC, and an elevated I/T ratio are commonly observed. In contrast, LOS may present with either high or low WBC, elevated ANC, increased I/T ratio, and thrombocytopenia. 20
Biomarkers
When combined with clinical scoring systems, biomarkers offer significant advantages over blood cultures by eliminating the waiting period for results. 21 Among the most extensively studied acute-phase reactants are CRP and PCT. 19 CRP rises within 8 to 12 hours and peaks at 24 to 48 hours after infection onset. 18 A single CRP measurement has low sensitivity but taking serial measurements 24 to 48 hours after symptom onset improves sensitivity to 74% to 89%. However, CRP values can be affected by factors such as premature rupture of membranes, maternal fever, meconium aspiration, fetal distress, and infection etiology. 20 PCT levels, in contrast, increase more rapidly in 2 to 4 hours and peaks at 6 to 12 hours. PCT has a slightly higher sensitivity (81%) than CRP at the early stages of infection, as it peaks within 6 to 12 hours. However, its specificity is lower. 18
Presepsin has emerged as a promising specific biomarker for neonatal sepsis, as it is less affected by variables such as mode of delivery, maternal fever, or stained amniotic fluid. Studies suggest that presepsin has the highest diagnostic accuracy, with a sensitivity of 88.9% and specificity of 85.7% for detecting EOS. 18 Studies show that hsCRP, PCT, and Serum Amyloid A (SAA) provide greater accuracy when combined. 21 CD64 neutrophil marker is another useful indicator with high sensitivity and specificity and requires only small blood volumes, making it particularly useful in neonates. IL-6 and IL-8, which are among the most widely studied inflammatory markers, rise rapidly in response to bacterial infections but normalize within 24 hours, limiting their clinical utility. 20
Role of Point-of-Care (POC) Testing in Diagnosis
Point-of-care testing (POCT) involves performing diagnostic tests near the patient, delivering quick results that help reduce treatment delays, enhance patient outcomes, and shorten hospital stay. 1 These are particularly valuable in low-resource settings, where access to advanced laboratory facilities is often limited. Many health centers and clinics in such areas lack the infrastructure to transport samples to centralized laboratories, making POC diagnostics a practical solution. 22 They are also especially valuable in neonates, where the minimal sample volume required for POCT provides a distinct clinical advantage.
Various biomarkers, including PCT, presepsin, hsCRP, IL-6, and neutrophil gelatinase-associated lipocalin (NGAL), have POC testing methods available. 21 However, some diagnostic techniques, such as PCR-based tests, face implementation challenges at the point of care because they require specialized equipment, constant power supply, and controlled storage conditions.
A commonly used POC diagnostic tool is the lateral flow assay (LFA), also known as a dipstick test. Blood-based LFAs have been developed to detect bacterial infections and sepsis using CRP. However, due to the high levels of CRP in the blood, these tests may suffer from the Hook effect, which can reduce their accuracy. 22 Table 3 presents the diagnostic utility, availability, feasibility, and limitations of some POC biomarkers.
Summary of Point-of-care Biomarkers in Neonatal Sepsis: Diagnostic Utility, Availability, Feasibility, and Limitations for Use in Low-Resoiurce emergency Settings.
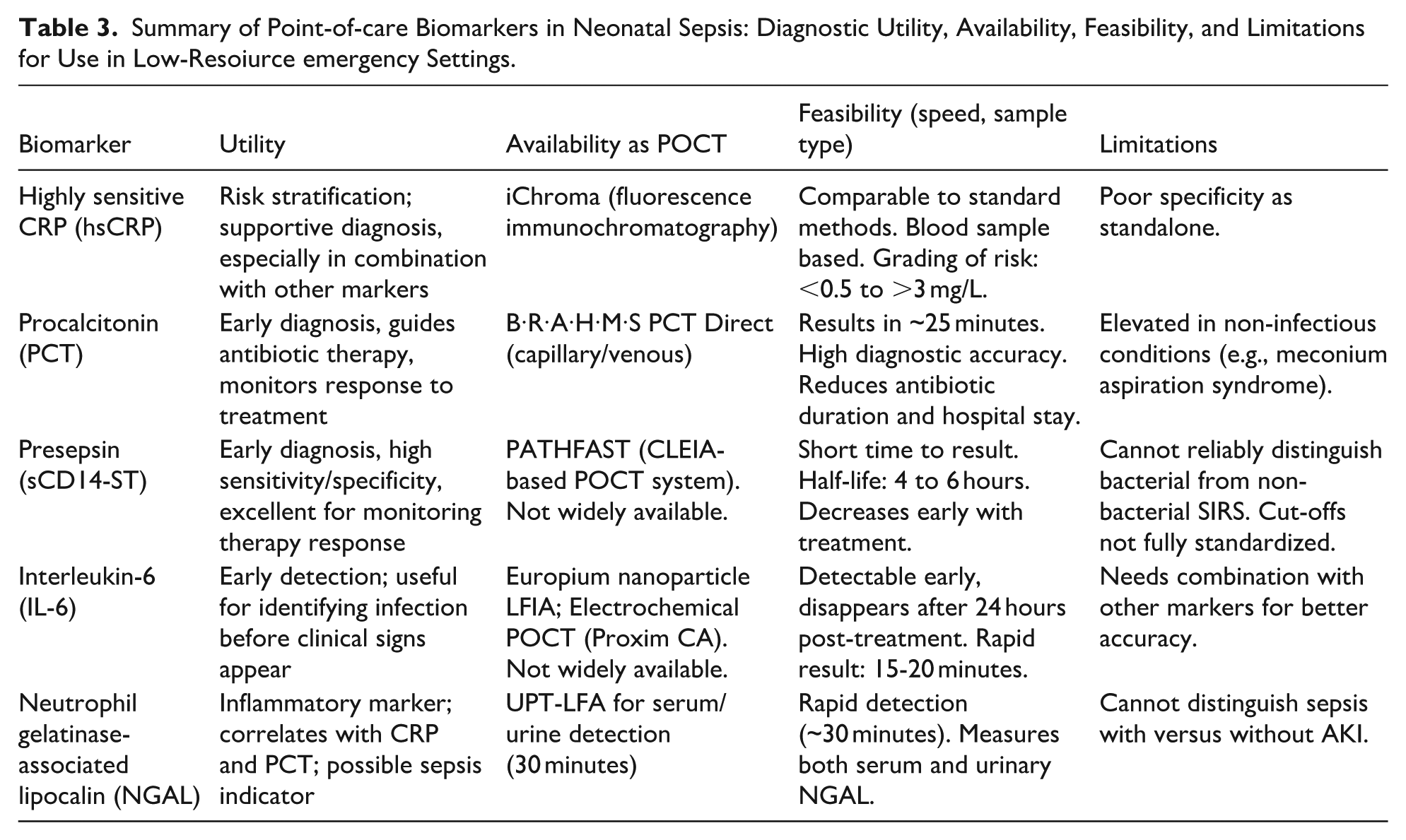
Incorporating POC diagnostic tools into pediatric emergency departments in LMICs could enable early intervention and improve outcomes for neonates with sepsis. However, POCT is not without its limitations; It is generally more costly on a per-test basis compared to conventional laboratory methods, largely due to the single-use nature of most POCT devices, which increases overall expenditures. Furthermore, the feasibility and widespread availability of POCT in LMICs remain major concerns, as many healthcare facilities lack the necessary expertise, consistent supply chains, and personnel to implement and sustain these technologies. In addition, challenges related to documentation and the potential for recording errors can compromise the accuracy and traceability of results, further limiting their reliability in routine clinical practice. 22
Alternatives to Lumbar Puncture
Meningitis is a serious complication of neonatal sepsis, occurring in approximately 6% to 12% in EOS and 1% to 12% in LOS. 18 LP remains the gold standard for diagnosing meningitis; however, pre-antibiotic administration often leads to negative CSF cultures, reducing its diagnostic accuracy. In LMICs, additional challenges include limited access to laboratory diagnostics and a shortage of trained personnel, making LP even more difficult to perform. 23
To address these limitations, alternative diagnostic methods have been explored. Blood biomarkers, such as PCT, CRP, and cytokine levels, have been proposed to help differentiate bacterial from viral infections, but they lack sufficient sensitivity and specificity. Unlike LP, PCR-based tests remain unaffected by prior antibiotic administration, making them a promising alternative. However, their high cost and resource-intensive requirements limit their feasibility in LMIC settings. Another potential non-invasive alternative is spinal ultrasound, which can detect inflammatory changes in the spinal canal without requiring an invasive procedure. Although not 100% accurate, combining these alternative diagnostic tools can aid clinical decision-making, particularly when LP is not feasible. 23
Figure 2 presents a diagnostic flowchart for neonatal sepsis in a low-resource emergency setting.

Diagnostic flowchart for neonatal sepsis in a low-resource emergency setting. Abbreviations: WBC: white blood cells; ANC: absolute neutrophil count; CRP: C-reactive protein; PCT: procalcitonin; SAA: serum amyloid A; NGAL: neutrophil gelatinase-associated lipocalin.
Empirical Management Strategies and Antibiotic Stewardship
The World Health Organization (WHO) classifies infants aged 0 to 59 days into 3 distinct risk categories based on mortality likelihood: critical illness (highest mortality risk), severe clinical infection (moderate mortality risk), and isolated rapid breathing in infants aged 7 to 59 days (lowest mortality risk). 14
Outpatient Settings
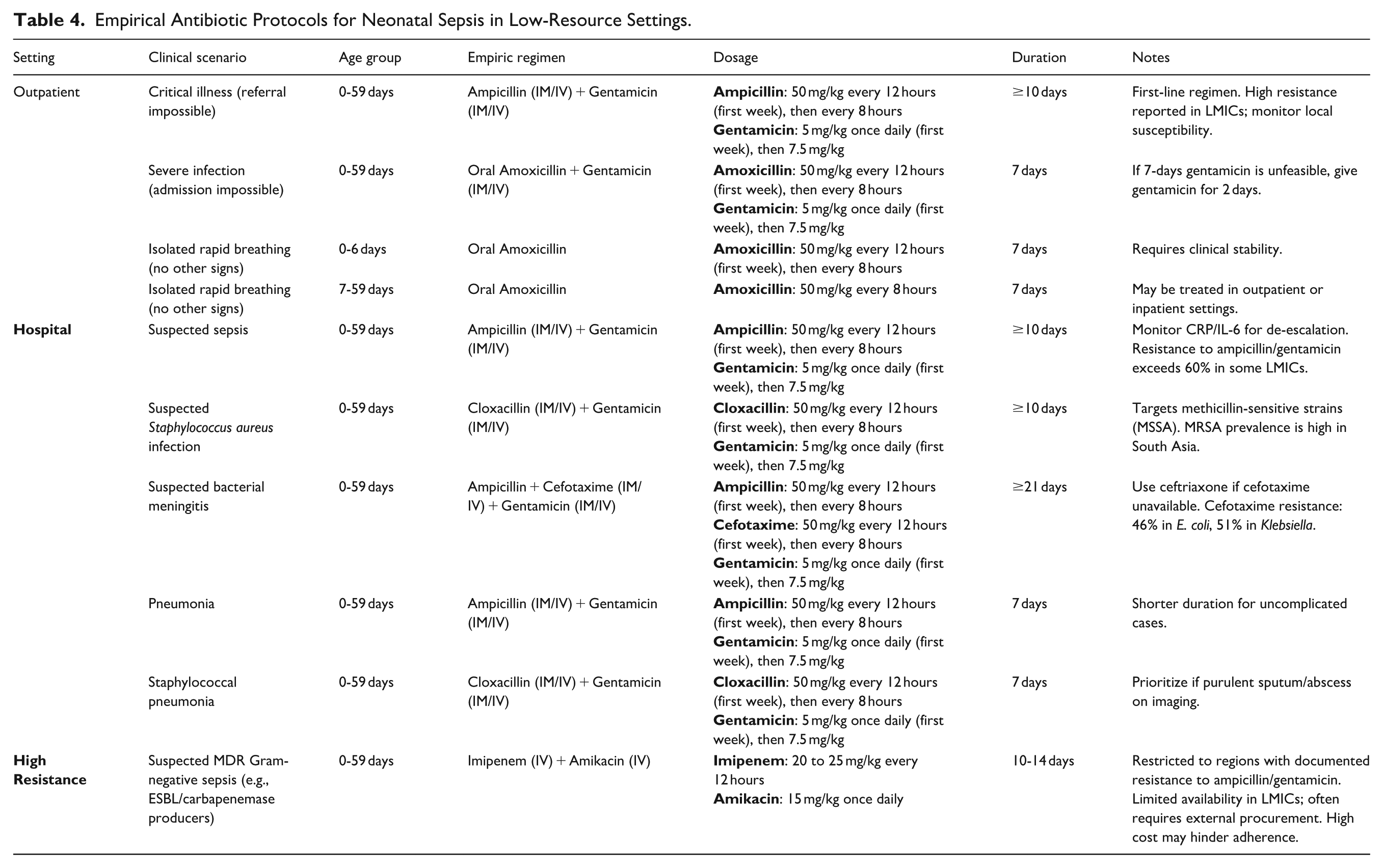
According to the 2024 WHO guidelines for neonatal sepsis management, critically ill infants under 2 months of age (0-59 days) exhibiting clinical signs of critical severe illness, as defined by the Integrated Management of Childhood Illness (IMCI) criteria, should be referred to receive urgent hospital care. In settings where hospital transfer is unfeasible, the recommended empiric antibiotic regimen is intramuscular (IM) or intravenous (IV) ampicillin combined with gentamicin, administered for a minimum of 10 days. 14
Newborns and infants up to 2 months old (0-59 days) displaying clinical signs of severe infection, as per IMCI criteria, require immediate hospitalization. When hospital admission is unattainable, a dual antibiotic regimen is recommended: oral amoxicillin administered daily for a minimum of 7 days combined with IM/IV gentamicin for the same duration. If the 7-day course of gentamicin is not feasible, an alternative regimen involves oral amoxicillin for 7 days combined with IV/IM gentamicin for 2 days. 14
Newborns (0-6 days old) presenting with rapid breathing as the only clinical indicator of illness also require urgent hospital assessment. However, when hospital transfer is not possible, a 7-day course of oral amoxicillin is advised as an alternative. Similarly, infants aged 7 to 59 days presenting with rapid breathing as the sole clinical sign may be treated in outpatient or inpatient settings. 14
Hospital Settings
For hospitalized infants under 2 months of age (0-59 days) with suspected sepsis, the standard first-line therapy consists of IM/IV ampicillin combined with IM/IV gentamicin for at least 10 days. For those with comorbid pneumonia, 7 days are recommended. If
For infants with suspected bacterial meningitis, broader empiric coverage is advised. A combination of ampicillin, cefotaxime, or ceftriaxone (IM/IV) alongside IM/IV gentamicin should be administered for at least 3 weeks to ensure adequate penetration into the central nervous system. 14
Table 4 summarizes empirical Antibiotic Protocols for Neonatal Sepsis in Low-Resource Settings
Empirical Antibiotic Protocols for Neonatal Sepsis in Low-Resource Settings.
Stewardship Interventions
To support treatment standardization and promote antibiotic stewardship, the WHO introduced the AWaRe classification of antibiotics, outlined in the 2021 evaluation and monitoring spreadsheet, and later published the WHO AWaRe Antibiotic Book in 2022. The AWaRe classification provides recommendations for first- and second-line antibiotics for common infections in both high-income countries and LMICs. It categorizes antibiotics into 4 groups: “Access,” “Watch,” “Reserve,” and a fourth category, “Not Recommended.” 14 “Access” antibiotics typically have a limited spectrum of activity, are more affordable, have a favorable safety profile, and are less likely to contribute to resistance. They are commonly prescribed as first- or second-line treatments for routine infections. In contrast, “Watch” antibiotics are broader in spectrum, often more expensive, and are usually reserved for more severe cases or infections where the pathogens are suspected to be resistant to “Access” antibiotics. “Reserve” antibiotics are considered last-resort options, typically used to treat infections caused by multidrug-resistant pathogens when other antibiotics have failed. 14
Moreover, there is an online risk stratification calculator for neonates with suspected early-onset sepsis, which has been shown to reduce early antibiotic use significantly in neonates. (https://neonatalsepsiscalculator.kaiserpermanente.org). It is also very important to depend on clinical evaluation for excluding sepsis and deciding not to start antibiotics, because it has a very good negative predictive value. 24
Antibiotic Resistance
Frequent antibiotic use in newborns carries significant risks, such as disturbing the delicate balance of their gut microbiota, increasing the likelihood of medication-related complications, and contributing to the emergence of drug-resistant bacteria. 24
Antibiotic resistance can emerge due to the use of inappropriate classes of antibiotics, inappropriate duration of therapy, or inappropriate dosing. Multidrug-resistant strains, including those that produce extended-spectrum β-lactamase (ESBLs) and carbapenemases, are increasingly prevalent in both healthcare facilities and community settings worldwide.
In a study involving over 36 000 infants from tertiary hospitals across 7 LMICs, up to 60% of bacterial isolates were resistant to both ampicillin and gentamicin. Klebsiella species display inherent resistance to ampicillin, with research indicating that resistance to gentamicin can occur in up to 71% of cases. 25
Research estimates that approximately 70% of pathogens causing bloodstream infections in neonatal units within developing countries may fall outside the coverage spectrum of the WHO-recommended empirical therapy (ampicillin combined with gentamicin) for neonatal sepsis.
26
Escalating resistance to costlier later-line antimicrobial agents poses a significant concern. For instance, studies report resistance rates to cefotaxime as high as 46% in
Special Considerations for Neonates in Low-Resource Settings
Management of Preterm Neonates and Those With Low Birth Weight
Every year, 15 million infants are born prematurely around the world. More than 1 in 10 of the world’s births are premature (<37 completed weeks of gestation) according to a survey conducted in 138 LMICs in 2010. 27 WHO recommends Kangaroo Mother Care (KMC) and breastfeeding for all preterm and low-birth weight (LBW) infants. 28
KMC as an Adjunct to Improving Outcomes
KMC is a practical intervention that provides immediate care for preterm and LBW infants. WHO defines KMC as “the care of preterm and LBW infants where the mother keeps the baby in skin-to-skin contact (SSC) on her chest continuously.” 29
Even though many preterm infants may need to receive prolonged medical care, the WHO recommended starting KMC as soon as possible after birth for preterm and LBW infants (weighing ≤ 2.0 kg). It is also recommended to introduce immediate KMC sessions at home for infants who have no danger signs and at healthcare services unless the infant is unable to breathe spontaneously without additional oxygen or is in shock and requires mechanical ventilation (MV). 28
A recent Cochrane Review stated that KMC was associated with a 32% lower risk of mortality and a 43% lower risk of severe infection/sepsis compared to infants who received conventional neonatal care after analysis of 9 randomized clinical trials, including 1952 infants (LBW < 2500 g). This review also supported the use of KMC as an appropriate and safe substitute to conventional neonatal care in LBW infants, mainly in resource-limited countries. 30
Another quasi-experimental study was performed to investigate the effects of KMC and incubator care on body temperature, weight gain, exclusive breastfeeding, and physiological measures of 130 low-birth-weight infants (1200-2490 g) admitted to NICU during 2019 to 2020 at one of the hospitals in Tehran. The study revealed that there were significantly higher risks of sepsis, apnea, and mortality in the incubator group. Also, in the KMC group, exclusive breastfeeding was started earlier and achieved better outcomes. 31
Emergency Sepsis Management Protocols in Resource-Limited Settings
The WHO clarifies that a possible serious bacterial infection (PSBI) is an infection that has at least 1 of 7 clinical symptoms: low body temperature (<35.5°C), high body temperature (≥38°C), severe chest indrawing, fast breathing (≥60 breaths/minute) in newborns aged 0 to 6 days, symptoms of convulsions in newborns aged 0–59 days, inadequate feeding or unable to feed at all, and movement only when stimulated, or no movement generally. 14
According to the WHO recommendations, infants with suspected sepsis or PSBI should be sent to hospitals immediately for medical supervision and antibiotic treatment. However, hospital access in LMICs may be restricted. Therefore, WHO and UNICEF (United Nations International Children’s Emergency Fund) have developed the 7-sign IMCI algorithm for managing infants with PSBI both in hospitals and in situations where referral is not possible, especially in resource-limited settings. 14 Table 5 outlines the simplified management algorithm. The following recommendations for simplified sepsis management pathways could be appropriate for application in settings with limited resources:
Simplified Neonatal Sepsis Management Algorithm.

Risk Stratification
Risk stratification or scoring systems are important for identifying high-risk neonatal sepsis, helping physicians decide when to initiate treatment, and reducing antibiotic usage. A new validated risk stratification model should be designed for use in resource-limited settings, with consideration to the included risk indicators, in order to apply the model in standard clinical practice. 1
Antimicrobial Treatment
WHO recommends using ampicillin (or penicillin) and gentamicin as first-line in hospitals. In cases where there is a lower suspicion of sepsis and families cannot access hospital care, oral amoxicillin is recommended. Additionally, WHO encourages antimicrobial stewardship and classifies all of these antibiotics as “Access” antibiotics and included them in the Essential Medicines List. 32
Prevention
Initiation of breastfeeding during the newborn’s early days of life can decrease neonatal sepsis and total mortality. Additionally, neonatal sepsis can be reduced by preventive practices like handwashing and maintaining good hygiene before and after delivery. Chlorhexidine also reduces neonatal sepsis when used in vaginal washes during labor, cleaning the umbilical cord stump, or as a skin antiseptic for newborns. 20
Use of Oral versus IV Antibiotics in Extremely Low-Resource Environments
Existing evidence from studies performed in resource-limited settings shows that many young infants with signs of PSBI did not receive the recommended inpatient treatment because such treatment was not accessible, acceptable, or affordable to families. 32
When treating newborn infants (0-59 days old) with PSBI, oral antibiotics may be considered as an alternative to IV antibiotics in extremely resource-constrained areas, especially when hospitalization is not practical. An oral antibiotic regimen is equally effective as an injectable regimen but simpler, avoids injections, and is more acceptable and preferable to families. Oral treatment is also more accessible and likely to have greater treatment completion rates. Oral amoxicillin is less expensive than injectable antibiotics, thereby reducing the economic burden on families and health systems. It is also routinely available at most health facilities for treating pneumonia in children 2 to 59 months old. 32
Fluid Resuscitation Challenges and Prevention of Overhydration
Fluid resuscitation is a vital component of supportive care for neonates with sepsis, but it poses substantial challenges in resource-limited settings, particularly in avoiding fluid overload.
To prevent hyponatremic fluid overload and hypernatremic dehydration, clinicians must ensure that patients are well-fed and allow for Extracellular Fluid (ECF) volume contraction. Neonates are especially susceptible to dehydration due to a larger body surface area, thin epidermis, and limited water conservation capacity. 33
There are some issues facing fluid resuscitation that arise in resource-limited settings. Some resource-limited hospitals or healthcare facilities are not equipped efficiently to provide premixed IV fluid and total parenteral nutrition (TPN) or lack fluid pumps, increasing the risk of infection upon fluid preparation. 1 Transportation issues are a vital concern in resource-limited environments, as neonates born at home or in smaller facilities develop sepsis; they often need to be transferred to more specialized facilities; however, this is frequently a long trip by a bus or a motorbike, which can result in complications that involve hypothermia and hypoglycemia. 1 Additionally, infrastructure that supports interventions—such as access to electronic resources and equipment, clean water and sanitation, energy for refrigeration, and dedicated spaces for personalized care and counseling—is often lacking. 14 Absence of resources such as access to medical equipment, supplies, medicines, and nutritional supplements further complicates care. Furthermore, inadequate healthcare human resources with the required knowledge and abilities to carry out, supervise, and support approved clinical practices pose a significant challenge to fluid resuscitation in resource-limited environments. 14
Fluid overload is associated with higher mortality rates, more requirements for intensive and prolonged ventilatory care due to pulmonary and chest wall edema, congestive heart failure, acute kidney damage (AKI), and longer hospital stays. New information on fluid overload in neonates highlights a higher chance of death, bronchopulmonary dysplasia (BPD), intraventricular hemorrhage (IVH), necrotizing enterocolitis (NEC), and AKI. 33
Several strategies aid in preventing overhydration and improving outcomes in resource-limited settings. Neonates with sepsis should receive carefully titrated fluid resuscitation, be monitored daily for overhydration, and be assessed for phenotypes of fluid non-responders or those who need early de-resuscitation practices. 34 Moreover, healthcare professionals should be professionally trained for updated recommendations and should receive training on practices when using intravenous fluids, especially in the first few days of life. 14 Co-infections and comorbidities, including malnutrition, should be carefully managed according to the WHO guidelines. 14
Global Health Perspectives: Low-Cost Interventions for Sepsis Reduction
Community-Based Strategies for Early Recognition and Referral
Traditional healers are more likely to treat neonates with detected sepsis outside the home than those taken directly to the healthcare system. Preventive strategies, including education on genital hygiene, antiseptic washes, clean delivery kits, hygienic practices, and neonatal care, are crucial in responding to maternal and neonatal sepsis. 35
Ethical studies suggest that outcomes may improve if treatment outside of health facilities is permitted. In rural sub-Saharan Africa, the pre-referral delays for neonates with sepsis occur substantially outside the health system. In low-income countries, home deliveries are common, with community workers and doulas playing a crucial role in healthcare services. Early postnatal care is recognized, with WHO recommending home visits. 36
A community-based strategy against severe neonatal sepsis has been proposed, embedding greater capacity in the community through traditional birth attendants. These attendants are responsible for identifying, administering, and referring septic neonates and are provided with a bicycle and mobile phone. This strategy aims to reduce the overall delay in recognition and treatment for sepsis in African neonates. 36
Impact of Midwife-Led Neonatal Care in Preventing Sepsis-Related Deaths
The WHO has published a consultation report on postpartum and postnatal care, highlighting the need for skilled health workers to provide adequate postnatal care. The report emphasizes the importance of postnatal review on the first day after delivery and recommends community-based maternal care, including immediate referral if a mother’s temperature rises above 38°C or there are observable symptoms of sepsis. 37
Midwives can significantly reduce maternal and neonatal mortality and stillbirths in LMICs, particularly through family planning, periconceptual, antenatal, childbirth, and postnatal interventions. However, they require adequate skills, a multidisciplinary team, and an enabling environment. Regular evidence review and increased coverage of midwife-delivered interventions can enhance health systems’ effectiveness. 38
Cost-Effective Interventions Such as Cord Care Antiseptics and Chlorhexidine Washes
Neonatal cord infections are a significant risk due to pathogens accessing viable tissue through exposed vessels. As the cord falls off, the umbilical cord and surrounding skin transition toward baseline immunity. Protective barriers to infection, particularly proper umbilical cord care, are cost-effective interventions. 39 Cord care using an elemental antiseptic has consistently reduced infection and neonatal morbidity. Chlorhexidine skin treatment after birth may reduce cord complications. A triple intervention of chlorhexidine skin treatment, cord treatment, and dry cleansing after cord falls off has been suggested to decrease complications associated with cord separation. 39
Future Directions and Research Gaps
Need for Affordable Point-of-Care Diagnostics for Neonatal Sepsis
The development and adoption of affordable, reliable POCT diagnostics in low-resource emergency settings is one of the most pressing needs in addressing neonatal sepsis. Certain technical specifications (sensitivity, specificity) and an affordable price are necessary for feasible implementation. 40
Chevalier et al reported that the implementation of POCT could reduce neonatal mortality, averting up to 19% at the hospital level and up to 76% at the community level. It is also suggested that healthcare costs could be reduced substantially with evidence of 17% to 43% of baseline costs saved in hospital and 48% to 81% saved in primary healthcare settings. This proves that the integration of POCT for sepsis detection in neonates can decrease overall morbidity and mortality, remaining cost-neutral when priced effectively. Additionally, Chevalier et al. concluded that a test with a sensitivity of 85% and specificity of 80% for hospitalized neonates, and 90% sensitivity and 70% specificity for community-presenting infants is expected to reduce mortality. 40
Based on a model by Chevalier et al (2024), implementing point-of-care tests (POCTs) for the management of neonatal sepsis in low-resource settings faces major practical challenges. The main barrier is cost; for example, for the test to be cost-neutral for the global healthcare system, it would need to cost no more than $21 in hospitals and $3 in community settings. Significant indirect costs for training, supply chain logistics, and quality control must also be accounted for. Furthermore, infrastructure limitations complicate implementation, as blood culture services are often unavailable and community-level referrals to hospitals are frequently not feasible. These combined financial and infrastructural obstacles must be addressed for POCTs to be successfully implemented. 40
Future research should aim to address the need for affordable POCT diagnostics in LMICs for sepsis diagnosis, while evaluating its impact on patient outcomes and antimicrobial resistance (AMR). Moreover, to facilitate access to diagnostic solutions for susceptible populations, urgent investments in diagnostics development are required. 40
Feasibility of Telemedicine and AI-Assisted Screening in Low-Resource Emergency Departments
Telemedicine and AI-assisted screening are potential solutions for effective management of neonatal sepsis in low-resource settings. The adoption of mobile health (mHealth) technologies, clinical decision support systems (CDSS), and wearable devices are examples of innovative solutions that could help overcome some of the challenges in providing high-quality patient monitoring in low-resource settings. 41
In a study by Garbern et al conducted in an inpatient setting in urban Bangladesh, it was demonstrated that a wearable-enabled, mobile monitoring system was practical for continuous, remote physiologic monitoring among children with sepsis. 41
This system, which relies on minimal clinician input and bypasses the need for laboratory tests, was able to effectively identify advanced sepsis cases, offering a significant advantage in low-resource settings where timely diagnosis is often delayed. Moreover, machine learning (ML) algorithms can harness large datasets collected from these devices to predict sepsis severity, enabling clinicians to make faster and more accurate decisions regarding treatment. 41
Additionally, the Smart-NICU model, which integrates telemedicine and remote patient care, has demonstrated success in India’s neonatal intensive care units (NICUs). This model allows specialists to remotely monitor and guide local healthcare providers in managing neonatal patients. The hub-and-spoke framework reduces the need for neonatal transfers, expedites specialty care, and improves triage speed, ultimately decreasing mortality rates. 41
Evaluating Modified Antibiotic Regimens for Different Settings
Research has identified more than 200 empirical antibiotic combinations currently in use, highlighting the absence of standardized protocols and highlights the critical need for evidence-based strategies. Future studies must aim to address the need for modified antibiotic regimens for neonatal sepsis in LMICs and must prioritize pragmatic clinical trials to determine optimal empiric treatment approaches to combat the rising AMR and variable compliance with WHO guidelines. A crucial focus is the assessment of carbapenem-sparing treatment approaches, including piperacillin-tazobactam, ceftazidime, and regimens based on quinolones, especially when used alongside amikacin, with an emphasis on their effectiveness in neonates. Moreover, use of colistin, a last-resort antibiotic, requires optimization through studies focusing on dosing, combination therapies, and methods of CSF penetration. 42
International collaboration is essential as it permits collecting data on antibiotic resistance patterns from various regions, resulting in a broader understanding of the worldwide AMR state. For instance, the NeoOBS study is a successful global alliance, involving 19 sites in 11 countries; it facilitated gathering data on antibiotic use, pathogens, and clinical outcomes, contributing to the creation of NeoSep Severity and Recovery Scores, which can guide treatment decisions in LMICs. 42
Affordability and feasibility must be taken into consideration when developing novel antibiotic regimens. To maintain affordability, new treatment protocols should focus on off-patent antibiotics and require fewer costly diagnostic tools. Feasibility necessitates that treatment protocols are easy to implement, need less training for medical personnel, and align with the current systems in LMIC healthcare facilities. Healthcare providers, policymakers, and pharmaceutical companies must collaborate to address challenges related to access, cost, and sustainability to ensure the successful implementation of innovative treatment plans. 42
Addressing socioeconomic conditions by enhancing healthcare accessibility, reducing poverty, and encouraging informed health behavior is also essential. This encompasses initiatives such as programs that provide cash transfers based on specific conditions, community health worker initiatives, and health education campaigns. Furthermore, investments are needed to overcome healthcare infrastructure challenges by enhancing healthcare systems, improving diagnostic capacity, and ensuring a consistent availability of essential medications. 42 A prime example of a successful community-based intervention, like Ethiopia’s successful Health Extension Program, is vital for improving health knowledge, care-seeking behavior, and equitable access to services. Crucially, overcoming these challenges depends on a collaborative approach that integrates health services with efforts to improve socioeconomic conditions. 43
Conclusion
Although preventable, neonatal sepsis remains a life-threatening condition in LMICs. This review showcases the contributing factors, including immature neonatal immunity, maternal risk factors, suboptimal hygiene, and healthcare infrastructure gaps. Advancements such as POCT diagnostics, KMC, and midwife-led interventions are potential solutions for enhancing outcomes. Developing affordable, feasible antibiotics is also crucial. Moreover, global research efforts should focus on evaluating telemedicine and AI-assisted monitoring in LMICs. To reduce the global burden of neonatal sepsis and enhance overall outcomes, a collaborative, interdisciplinary approach is essential. Therefore, we issue a call to action for global health funders and policymakers to prioritize and substantially increase investment in the implementation and scaling of these critical, evidence-based interventions.
Footnotes
Authors Note
All cited studies are presumed to have adhered to contemporary ethical standards for research involving human participants.
Author Contributions
Salma Tamer Abdelrahman is the first author and contributed to the conception, design, formulation, drafting, and revision of the manuscript. Mohammed Alsabri is the corresponding author and contributed to the conception, design, and revision of the manuscript. Nourhan M. Kamal, Sohaila Mohamed, Mohamed Abdelbar, Rana Al Juhmani, Chibuike Daniel Onyejesi, and Almotazbellah Attalla contributed to the interpretation and drafting of the manuscript. All authors provided final approval of the version to be published and agree to be accountable for all aspects of the work, ensuring its integrity and accuracy.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
No datasets were generated or analyzed during the current study.
