Abstract
Objectives:
To describe the burden of anemia at 6 to 11 months and the change in anemia prevalence after 1 year of multi-micronutrient supplementation.
Methods:
We analyzed hemoglobin concentration twice in 569 children. First when they were 6 to 11 months old and second after 1 year (at ages 18-23 months). All children were given a lipid-based micronutrient formulation daily for a year.
Results:
The prevalence (95% CI) of anemia at 6 to 11 months was 65% (61%-68%) and decreased to 25% (21%-28%) after 1 year. Among the 367 infants with anemia at baseline, 29% continued to be anemic during follow-up. There were also 18% infants who developed anemia during follow-up out of 202 infants with normal hemoglobin concentration at baseline.
Conclusions:
The anemia prevalence was substantially reduced after 1 year with micronutrient supplementation. However, even after 1 year of multimicronutrient supplementation, one-fourth of toddlers had anemia which merits further integrated research and comprehensive intervention.
Keywords
Introduction
Nutritional anemia is the most common micronutrient deficiency among children in low- and middle-income countries. 1 The global pooled prevalence of iron deficiency and iron deficiency anemia (IDA) were 17.9 and 16.4%, respectively, and figures are particularly high in Asia (23.3% IDA) among children <2 years of age. 2 When the prevalence of anemia is >40%, it is considered a severe public health concern by the WHO prompting regular screening and universal iron supplementation during the first 2 years of life. 3 Currently, 56 countries, including Nepal, have an anemia prevalence of >40% among children 6 to 59 months of age4,5 and the figures are stagnant or have increased in the last 5 years.6,7 As per the SDG target 2.2, the Nepal government is determined to end all forms of malnutrition, including anemia. 8 However, there are no routine screening in Nepal. An analysis of a representative sample of 1942 children 6 to 59 months of age based on the 2016 Nepal Demographic Health survey (NDHS) showed that not only younger age, underweight or poor maternal education are associated with increased burden of anemia in children, but it also varies significantly between different regions within Nepal. 9 Data on patterns of changes in anemia prevalence with longitudinal follow-up during the first 2 years of life are not available.
Reducing anemia prevalence by iron or micronutrient supplementation among children is one of the main priority programs in most government and WHO strategies. 10 However, the implementation and compliance of iron supplementation to control anemia are often poor. 11 Due to the mild severity of anemia, it is also considered that most of these anemia cases could be physiologically normalized with increasing age without any intervention.12,13 Similarly, diagnosis of iron deficiency anemia based only on Hb measurement may overestimate the prevalence figure. 14
Globally, nutritional anemia, mainly due to iron deficiency, was responsible for 422 years of living with disability per 100 000 population in 2021. 5 Its effect during the first 2 years of life is not limited to short-term consequences such as infections, 15 but it is also negatively associated with growth, neurodevelopment, and school performance. 16 For instance, a review concluded that iron deficiency anemia was related to poor cognitive and socio-emotional function. 17
Apart from iron, deficiencies in other micronutrients such as zinc, 18 vitamin B12, 19 folate 20 and vitamin A 21 also may contribute to anemia. If the mother during pregnancy or lactation is suffering from anemia, their children are at risk for anemia.7,22 The intake of animal-based foods, which are rich sources of iron and other micronutrients, is low among mothers and children in resource-poor settings. According to the last Nepal Micronutrient Status Survey (NMSS), only 48% of the women 15 to 49 years of age achieved a minimum food diversity, and only 38% of the children received a minimum acceptable diet. 23
To combat micronutrient deficiencies, including the control of anemia and stunting, the Government of Nepal launched a micronutrient supplementation (MNS-Baal Vita) program for children 6 to 23 months of age as per the WHO recommendation 24 but observed limited adherence and is lacking data on its impact on anemia prevalence. 25 The etiology behind anemia and stunting overlap thus it is not surprising that the prevalence of anemia was more common among children with stunting (and vice versa).26,27 MNS, particularly in the lipid-based form, has been shown to reduce the anemia prevalence and other forms of malnutrition among under-five children. 28 However, most of the available data on anemia are based on one-time measurements, including the NDHS, which lacked information on natural physiological changes in hemoglobin (Hb) concentration during different stages of life. 29 Our objective in this secondary analysis of a community-based longitudinal follow-up study in Bhaktapur, Nepal, is to estimate the prevalence of anemia at 6 to 11 months before and after 1 year of multimicronutrient supplementation.
Methods and Materials
Participants and Enrollment Procedure
This is a secondary data analysis based on children who participated in a double-blind, randomized, placebo-controlled trial measuring the effect of vitamin B12 supplementation during infancy on growth, neurodevelopment, and Hb concentration. Details on the study protocol and results of main outcomes including the impact of B12 supplementation on anemia prevalence, have been published elsewhere.30,31 Briefly, for enrollment in the study, we screened eligible infants at a vaccine clinic and through a community survey in the study area. The inclusion criteria were age between 6 and 11 months, a length-for-age <−1 z-score, and the family intended to reside in the study area at least for the next 12 months. We focused on mildly stunted infants for the main study as we wanted to include a population that was at risk of vitamin B12 deficiency. 32 We did not enroll children in the study if they had one of the following conditions; severe malnutrition (weight for length z-score < −3), severe anemia (Hb <7/g/dL), acute or chronic illnesses, or if they were taking multivitamins that contained vitamin B12. A total of 600 children were enrolled in the main study, for which we obtained baseline and demographic information. For this analysis, we included 569 children for whom we have Hb measurements at two time points, that is, at enrollment (age 6-11 months) and at the end of supplementation (age 18-23 months; Supplemental Figure 1). Peri-natal characteristics, household and parental information were obtained at or within 1 week of enrollment. Mother’s Body Mass Index (BMI) was calculated as weight in kg divided by height in meter square and value <18.5 kg/m2 is considered undernutrition. Low birth weight (birth weight <2.5 kg), preterm birth (<37 weeks of gestation), exclusive breastfeeding and categorization of nutritional status (underweight, stunting or underweight) were defined as per the WHO guidelines. 33
Study Site Characteristics
Enrollment as well as blood sampling and testing were done at Siddhi Memorial Hospital, Bhaktapur. Children were recruited mainly from the Bhaktapur municipality, and from the surrounding communities of nearby municipalities. According to the last census of Nepal, the total population of Bhaktapur district was 432 132, and among them, 28 007 were below 5 years of age. The average population growth rate of Bhaktapur was 3.35% in 2021 which is the highest out of total 77 districts in Nepal. 34 Bhaktapur municipality is known for its close clusters of agricultural-based communities. Many people live in joint families and belong to the Newar ethnic group, however due to recent socio-demographic changes the population is increasingly more heterogeneous, and more people are now engaging in business and service at private or government jobs including remittance from abroad. 34
Composition of Micronutrient Supplementation and Compliance Assessment
The MNS was administered daily in paste form (eeZee20), equivalent to approximately 1 RDA of 15 vitamins and minerals. The list of these vitamins and minerals per 20 g sachet (daily dose) is presented in Table 1. The supplement contained 8.6 mg iron daily, which is 1 to 2 RDA for children aged 6 to 23 months as per the Dietary Reference Intake, Institute of Medicine recommendations. 35 Due to the nature of the main clinical trial, only the intervention group received an vitamin B12 (cyanocobalamin) at a dose of 2 µg (2-3 RDAs) along with the other nutrients. 30 The MNS was produced by GC Rieber Compact (http://www.gcriebercompact.com) as per the WHO’s multiple micronutrient formulations. 24 Shipment and storage were done with air tight sachets as per the manufacturer instruction.
Composition of Micronutrients in the Supplementation Sachets.

Content per 20 g sachet is equivalent to one daily RDA for children as per the WHO
Only children in the intervention groups (50%) received vitamin B12
The compliance on MNS supplementation were recorded during the weekly visits by field workers. Day wise information covering for the last 7 days regarding vomiting, missing, partial or full sachet MNS supplementation were recorded, and all empty sachets were also verified and counted. At the end of 1-year MNS supplementation, we asked to mothers regarding any missing and challenges on MNS supplementation.
Hemoglobin Measurement and Definition of Anemia
Blood was obtained from one of the antecubital veins into a polypropylene tube containing EDTA (Sarstedt, Germany). Immediately after the blood sampling, Hb was measured by HemoCue Hb 201+ (Ångelholm, Sweden). According to the WHO, anemia among children <5 years of age is defined as Hb <11 g/dl. 36 We also categorized anemia severity as mild, moderate and severe as 10-10.9 g/dl, 7-9.9 g/dl, and <7 g/dl, respectively. As the study site is 1400 m above sea level, we also present anemia prevalence adjusted for altitude (adding 0.3 and 0.7 g/dl to the standard cutoff values of Hb) as per CDC 37 and the revised recommendation by Sharma et al 38 Altitude adjustment of Hb is required due to adaptive body responses to lower oxygen saturation of the blood. We have used different cut-off values of Hb to define anemia during infancy as this is the period of life when there is physiological variation of Hb. Because of this there is also uncertainty regarding the cut-offs which may over or underestimate the anemia prevalence.
Statistical Analysis
The baseline socio-demographic and clinical assessment forms were filled during enrollment interviews. All forms were manually checked by field supervisors and data were double entered in Microsoft Access databases. All the analyses were performed using Stata® version 16 (Stata Corporation, College Station, Texas, USA). The differences on anemia prevalence at enrollment (6-11 months) and follow up (18-23 months) were estimated by using chi-square test, and a P-value < .05 is considered statistically significant. The anthropometric measurements were categorized as underweight (weight-for-age < −2 SD), wasting (weight-for-length < −2 SD) and stunting (length-for-age < −2SD) based on the WHO growth standards. The distribution of Hb concentrations at 6 to 11 months and 18 to 23 months is depicted in a scatter plot. We report the change in anemia prevalence between the 2 measurement time points and graphically display these in a Sankey diagram. 39 Sankey diagrams are commonly used to visualize how participants change between categories over time.
Ethical Approval and Informed Consent
The study obtained approval from the Nepal Health Research Council (Reg.no.2333/2014) and Regional Committee for Medical and Health Research Ethics (REC) Norway (2014/1528). After explaining all the details about the study, we obtained written informed consent from the parents or thumbprint from those who are illiterate (in the presence of impartial witness). During the study period, we provided services to all enrolled children for common childhood infections whenever required. For the children with mild anemia, we provided nutritional counseling; for children with moderate anemia (Hb 7-9.9 g/dl), we also supplemented with oral iron in the syrup/drop form daily for 1 to 2 months.
Results
Baseline Characteristics
The mean (SD) age of infants at the enrollment was 8.0 (1.7) months; 187 children (33%) were stunted (<−2SD) at baseline (6-11 months of age) and 183 (32%) at 18 to 23 months of age. During enrollment, 14 infants (2.5%) were reported to not being breastfed anymore and 288 (51%) had been started on solid or semisolid complementary feeding within 4 months of age. The mean (SD) age of mothers was 27.5 (4.6) years. A total of 28 mothers (5%) were underweight (BMI < 18.5 kg/m2), and 200 (35%) were overweight (BMI > 25 kg/m2; Table 2).
Characteristics of Mothers and Children in Bhaktapur, Nepal at Enrollment and 1-year Follow-Up.

values are n (%) unless otherwise specified (total n = 569)
Information on birth weight were available from 549 newborns
Salaried jobs defined as jobs with regular daily or monthly payment including government or private sectors
Compliance With Micronutrient Supplementation
Based on weekly supplementation compliance records, all the recommended sachets of MNS were successfully given on 81% of the total supplementation days (159,741 out of 197,212 days). The supplements were not given in 6% (11 533 out of 197 212) of the days, mainly due to illness or travel. Also, only less than half of the sachet was given in 9% (17 749 out of 197 212) of the days.
Prevalence of Anemia and Its Changes at 18-23 Months of Age
The mean (SD) Hb concentrations were 10.6 (0.93) at 6 to 11 months of age and 11.6 (0.91) g/dL at 18 to 23. Hb concentrations at the third, fifth and 97th percentile stratified by age (in months) and sex are presented as Supplemental Table 1. The overall anemia prevalence (95% CI) was 65% (61-68%) at 6 to 11 months of age and decreased to 25% (21-28%) at 18 to 23 months of age, (p-value = .004; Supplemental figure 1). The prevalence (95% CI) of altitude-adjusted anemia was 74% (71-78%) and 88% (86-91%) at 6 to 11 months, and 38% (34-42%, p-value .003)) and 55% (51-59%, p-value .001) at 18-23 months when we used cut-off values of 11.3 g/dl and 11.7 g/dl, respectively (Table 3). By using different cut-off values of Hb, the prevalence of anemia varied 65-88% at 6-11 months and 25%-55% at 18-23 months of age. Most of the observations were clustered at right lower quadrant of the scatter plot, which indicates improvement of Hb concentration at 18-23 months (Figure 1). Among the 367 anemic infants at 6-11 months of age, 106 (29%) remained anemic at 18-23 months; among 202 infants who had normal Hb concentration 36 (18%) developed anemia at 18-23 months of age (Figure 2). Most of the anemia (40% at 6-11 months of age and 24% at 18-23 months of age) were categorized as mild severity (Hb 10-10.9 g/dl).
Mean Hemoglobin Concentration and Prevalence of Anemia at 6 to 11 and 18 to 23 months of age (n = 569).

chi-squared test
Values are n (%) and 95% confidence interval
Values are adjusted for the local altitude of 1400 m as per the recommendation by CDC *
Values are adjusted for the local altitude of 1400 m as per the recommendation by Sharma et al 38
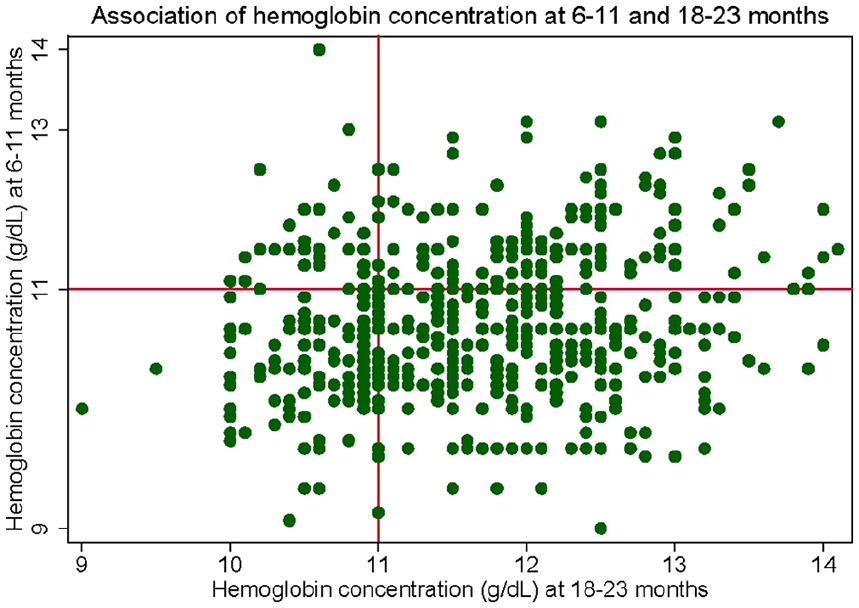
Association of hemoglobin concentration at 6 to 11 months and 18 to 23 months in Bhaktapur Nepal.

Changes in the severity of anemia prevalence (no, mild or moderate) from 6 to 11 months (left yaxis) to 18 to 23 months (right y-axis) in Bhaktapur, Nepal. The diagrams were constructed by using Sankey plot. The figures and presented as total %.
Discussion
In this community-based cohort study, we describe the anemia prevalence among mildly stunted children before and after a year of WHO-formulated MNS. According to the WHO criteria, 36 the prevalence of anemia in our study at baseline indicates that this is a severe public health problem in infancy. Universal iron supplementation along with regular screening is recommended when anemia is a severe public health problem 4 as it may cause irreversible changes in the growing brain 40 and predispose to increased mortality. 41 With 1 year of MNS in our study, anemia was decreased to 25%. Most anemia cases at 18 to 23 months of age and 40% at 6 to 11 months of age were mild. Despite excellent compliance with the MNS, 29% of the anemic children remained anemic and 18% with normal Hb during infancy developed anemia at 18 to 23 months of age.
Our findings of a reduction from 65% prevalence at 6 to 11 months to 25% anemia at 18 to 23 months indicate positive effects of MNS supplementation for a year. The use of a WHO-formulated MNS for children 6 to 23 months of age reduced anemia and iron deficiency by 26% (RR; 0.74, 95% CI; 0.66, 0.83) and 52% (RR; 0.48, 95% CI; 0.36, 0.62), respectively. 24 Based on this and other evidence, it is recommended that MNS be used for children in this age group where anemia prevalence is a public health problem. 42 Reduction in anemia, iron deficiency, and stunting were found in 2 districts of Nepal (Achham and Kapilvastu) where post-pilot scale-up of MNS was carried out for 3 years. 43 The prevalence of 65% anemia in our study at 6 to 11 months of age was in line with the 70% found among the same age group in NDHS 2021. 44 Anemia was still high (63.9%) at 12 to 23 months of age in the NDHS survey. Thus, the current study further supports the recommended use of MNS for children in this age group where anemia prevalence is a public health problem. However, a direct comparison of our findings of anemia prevalence of 25% at 18 to 23 months may not be possible due to the age difference between these studies. 44
Our study is unique in that we had community-based recruitment, and we obtained detailed weekly information on MNS compliance for 1 year. Most of the studies on anemia prevalence are based on one time point, a wide age range (eg, 6-59 months of age) and commonly during hospital visits which are prone to over or under represent of certain age groups or influenced due to infection or inflammation. 45 As our analysis was based on participants included in a randomized control trial, 30 this study inherited some of the limitations such as the restriction of participants with the length of age z score <−1 SD which compromises the generalizability of our findings. Although one of the exclusion criteria for the main study was severe anemia defined as Hb < 7 g/dl, we did not have any children in this category during screening. We gave MNS with vitamin B12 to 50% of our participants (intervention arm) but there was no effect of vitamin B12 on hemoglobin concentration at any time point.30,31 Our analysis is an observational design without a control group, with which we cannot ascribe the changes in anemia prevalence to the MNS. Furthermore, we gave additional iron supplementation (in syrup) for 1 to 2 months for those who were moderately anemic at 6 to 11 months, which most likely influenced the prevalence of anemia at 18 to 23 months. Another limitation to note is that we lacked information on anemia among mothers of children enrolled in our study which could be also associated with anemia among offspring. 46 Thus, our study findings should be interpreted in light of these limitations along with results based from a pre-post analysis in a vulnerable population from 1 municipality in Nepal.
It is recommended to adjust Hb concentration according to the altitude for the population residing above 1000 m sea level for a better estimation of anemia. 37 An even higher Hb adjustment (0.7 g/dl) was recently recommended for people living at 1000 to 1499 m sea level. 38 Based on this recommendation, our prevalence of anemia increased to 88% during infancy and 55% at 18 to 23 months of age. It should be noted that among 14 studies included for the establishment of this new altitude-specific recommendation, none of them were from South Asia. 38
Hb measurement by the cyanmethemoglobin method is considered a gold standard and recommended by the WHO, 47 and automated hematology analyzers are commonly used to analyze Hb in clinical laboratory settings. 48 Due to its simplicity, quickness, portability, and cost-effectiveness, the Hb photometer (HemoCue) is commonly used for anemia screening, including in the NDHS surveys, and also for estimating intervention outcomes. 49 We used venous blood, which is less prone to error than capillary blood in a field settings. 50 The Hb concentrations are consistently higher when capillary blood is compared with venous blood 51 and for the precision of Hb test venous blood is recommended.23,52
Our findings of 25% anemia prevalence at 18 to 23 months of age despite 1 year MNS and treatment with iron for those with moderate anemia may indicate other causes of anemia such as parasitic infections (hookworm) and hereditary hemoglobinopathies. 53 However, available literature suggests that these other causes of anemia are not common in our study area, and a 6-monthly deworming program has been rolled out for more than 3 decades.23,54 The persistent of anemia also could be due to poor absorption of MNS related with gut inflammation and enteropathy which are common among children residing in resource poor settings. 55 Although anemia and stunting in children may coexist as an outcome of chronic malnutrition, 26 the co-occurrence may be independent as well as shown in a study conducted in Peru and India. 56 In this study, the co-occurrence of anemia and stunting among 6 to 18 months children was found only 21.5% in India and 30.4% in Peru indicating that separate interventions are probably required to address these 2 common conditions. Besides nutrition, early life exposure to ambient air pollution 57 lead toxicity, 58 inflammation, or infection, 59 which also may predispose anemia in children. There has been a debate for a long time about the usefulness of the universal use of 11 g/dL Hb to define anemia for 6 to 59 months of age. Data to establish this cut-off were mainly from European and North American populations.60,61 Hb is lower in infancy than later in childhood, this may result in an overestimation of anemia prevalence when we used the same cut-off of Hb in the older age groups. 29 A lower cut-off compared to the WHO anemia definition was proposed for Qatari children at 1 year of age (9.8 g/dl) 62 and Australian infants (10.5 g/dl). 63 Recently, using international data sources from 1998 to 2020, the cut-off of 10.4 g/dl based on the fifth percentile to define anemia in 6 to 23 months children was recommended. 64 With this cut-off, 251 (44%) children at 6 to 11 months and 49 (9%) at 18 to 23 months would have been classified as anemic in our study.
Conclusion
The prevalence of anemia was very high during infancy and decreased to 25% following 1 year of MNS. Daily supplementation of MNS starting during the second half of infancy could have had an important role in this reduction and potential to contribute to achieving the SDG target 2.2, that is, to end all forms of malnutrition, including anemia. Our findings suggest that the nutritional programs in Nepal need to go beyond micronutrient supplementation at around 1 RDA to reduce the burden of anemia in early childhood. However, the persistence of anemia prevalence at 18 to 23 months of age even after 1-year MNS reflects the diverse etiology of anemia in young children which merits further research with integrated approaches such as reduction on infection, air pollution and environmental contamination of lead and other toxic substances.
Supplemental Material
sj-docx-1-gph-10.1177_30502225251346286 – Supplemental material for One-Year Daily Micronutrient Supplementation and Anemia Prevalence Among Mildly Stunted Infants in Bhaktapur, Nepal
Supplemental material, sj-docx-1-gph-10.1177_30502225251346286 for One-Year Daily Micronutrient Supplementation and Anemia Prevalence Among Mildly Stunted Infants in Bhaktapur, Nepal by Ram Krishna Chandyo, Suman Ranjitkar, Manjeswori Ulak, Mari Hysing, Catherine Schwinger and Tor A. Strand in Sage Open Pediatrics
Supplemental Material
sj-tiff-2-gph-10.1177_30502225251346286 – Supplemental material for One-Year Daily Micronutrient Supplementation and Anemia Prevalence Among Mildly Stunted Infants in Bhaktapur, Nepal
Supplemental material, sj-tiff-2-gph-10.1177_30502225251346286 for One-Year Daily Micronutrient Supplementation and Anemia Prevalence Among Mildly Stunted Infants in Bhaktapur, Nepal by Ram Krishna Chandyo, Suman Ranjitkar, Manjeswori Ulak, Mari Hysing, Catherine Schwinger and Tor A. Strand in Sage Open Pediatrics
Footnotes
Authors’ Note
Catherine Schwinger is now affiliated to Department of Disease Burden, Norwegian Institute of Public Health, Norway.
Author Contributions
TAS, MH, CS, RKC and MU designed the research; MU, RKC and SR conducted the research; RKC, SR, MU and TAS analyzed the data, wrote the paper and taken feedback from all co-authors. RKC and TAS had the primary responsibility for the final content and all authors read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Thrasher Research Fund (award # 11512), the GC Rieber foundation, the University of Bergen (UiB). The funder had no role in the design of the study, data collection, analysis, and interpretation of the result, or in writing of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
