Abstract
Objectives:
Anemia is a prevalent hematological disorder in children infected with human immunodeficiency virus who are receiving antiretroviral therapy. Anemia is increasingly prevalent among children undergoing antiretroviral treatment; however, studies on the incidence and predictors of anemia in this population within Ethiopia remain limited. Therefore, this study aimed to determine the incidence of anemia and its predictors among children who were enrolled in antiretroviral therapy at health facilities in the Wolaita Zone, South Ethiopia, 2022.
Methods:
A facility-based retrospective cohort study was conducted among 352 medical records at health facilities in the Wolaita Zone from February 2022 to January 2023. The medical records were selected using the stratified cluster sampling method. The data were entered into EpiData and analyzed using STATA version 14. The Cox proportional hazard regression model was used. Finally, predictors with p < 0.05 were stated as statistically significant factors. The results were presented using text, tables, and figures.
Results:
The overall incidence rate of anemia among children was 4.29 (95% confidence interval: 3.46–5.34) per 100 person-years. Being female sex (adjusted hazard ratio = 1.79, 95% confidence interval: 1.29–2.47), advanced disease stage (adjusted hazard ratio = 2.26, 95% confidence interval: 1.26–4.06), a zidovudine-based regimen (adjusted hazard ratio = 2.07, 95% confidence interval: 1.16–3.69), and being stunted (adjusted hazard ratio = 3.37, 95% confidence interval: 2.06–5.50) were the predictors of anemia.
Conclusion:
The incidence of anemia in the study setting was relatively low. Females, children at advanced World Health Organization clinical stages, those who were stunted, and those who initiated with a zidovudine-based regimen were the predictors of anemia. Healthcare providers should give special attention to children who are at an advanced disease stage, who were stunted, and who are taking a zidovudine-based regimen.
Background
Anemia is a blood ailment where there are fewer red blood cells than normal or less hemoglobin (HGB) than usual, which results in the blood’s diminished capacity to transport oxygen. 1 Anemia in children under 15 is typically characterized by HGB levels falling below certain age-specific thresholds. In children aged 6 months to 5 years, 5–12 years, and 12–15 years, the threshold of HGB to determine anemia is below 11, 11.5, and 12 g/dL, respectively. 2 It can be categorized as mild (10–10.9, 11–11.4, and 11–11.9 g/dL for children under 5, 5–11, and 12–14 years, respectively), moderate (7–9.9 and 8–10.9 g/dL for children under 5 and above 5, respectively), and severe (<7 and <8 g/dL for children under 5 and above 5, respectively). 3
The signs and symptoms that suggest the presence of anemia include shortness of breathing, heart palpitations, headaches, dizziness or lightheadedness, reduced physical activity or fatigue, sore tongue, pale skin, cold hands and feet, brittle nails, unusual cravings, reduced appetite, and muscle weakness.1,4,5
Anemia was found to have serious consequences for the health and economic status of communities. As adults, children with anemia have irreversible cognitive and developmental delays, as well as lower work-related productivity. 6 It is also a main cause of hospitalization in children, growth impairment, and reduced immune function. Human immunodeficiency virus (HIV) can infect portions of bone marrow producing red blood cells and erythropoietin, which is considered necessary to enhance production of red blood cells in HIV-infected children. Furthermore, children appear to be the most vulnerable to anemia, accounting for a large proportion of HIV-related mortalities every year. 7
Anemia could be brought on mainly by blood loss, an increase in RBC destruction, or a decrease in RBC synthesis. 8 In addition, infections like malaria, intestinal parasites, and HIV/acquired immunodeficiency syndrome (AIDS) add to the burden of anemia in developing countries. 9 Studies suggest that HIV infection increases the risk of anemia occurrence, along with hematological abnormalities in general.10,11
Antiretroviral therapy (ART) in Asian children has a high incidence density of severe anemia, with males having a higher risk compared to females and those using a zidovudine-based regimen. 12 A study from Bangladesh, South Asia, showed there is a strong association between stunting and anemia. 13 Anemia is a major health concern that affects an estimated two billion people around the world; among those, more than 100 million are found in Africa. 14 Similarly, more than half of children living with HIV develop anemia after ART initiation. 15 A study from Senegal, West Africa, showed that anemia was found to be significantly associated with stunting among children. 16 In Ugandan rural clinics, ART is commonly used in children at a younger age, with advanced clinical stages and low cluster of differentiation 4 (CD4) counts being associated with anemia development. 15
Anemia prevalence in Ethiopia ranges from 22.3% to 57.5% among children living with HIV/AIDS. Geospatial inequality and non-random distribution of anemia are prevalent across regions. Factors such as rural residency, maleness, lack of education, unemployment, younger age, intestinal parasitic illnesses, poor CD4 cell count, and lack of Highly Active Antiretroviral Therapy (HAART) are associated with anemia. Children on cotrimoxazole therapy are two times more likely to develop anemia. HIV-infected children on zidovudine-based ART regimens, World Health Organization (WHO) clinical stages III and IV, and low CD4 cell counts are at higher risk.17–20
Becoming immune-compromised, using a zidovudine-based ART, and having tuberculosis (TB) were all independent factors that increased the likelihood of anemia in children. 21 According to a study conducted in Bahir Dar city in Northwest Ethiopia and Tikur Anbesa Hospital in Addis Ababa, study participants who were underweight were more likely to appear anemic.22,23 According to a study conducted in North Wollo, Ethiopia, having a weight-for-age Z (WAZ) score between −2 and −3 was strongly associated with the occurrence of anemia. 19
The Ethiopian government has been attempting to lower the rate of anemia in children. As a result, it created guidelines for the prevention and control of micronutrient deficiencies, adopted micronutrient fortification, and supported the national nutrition program and bimanual school deworming. However, research from many regions of the nation has demonstrated that pediatric anemia remains a significant public health issue in this nation. 18
Even though anemia is one of the six WHO worldwide nutrition targets by 2025 and diet-related global targets of the World Health Assembly, which were approved in 2012 and 2013, its incidence is increasing today. 24 Tackling anemia has a significant contribution to achieving the second goal of Sustainable Development Goal 2 (SDG2: zero hunger), which targets ending all forms of malnutrition to reach zero global hunger by 2030. Furthermore, anemia is on the global reference list of 100 core health indicators endorsed in 2018, particularly aiming to tackle the prevalence of anemia to achieve health-related SDG. 25
To the best of our knowledge, there is no study on the incidence of anemia and its predictors among children on ART in Ethiopia, despite the fact that anemia is becoming more prevalent in these children. Therefore, this study aimed to investigate the incidence and predictors of anemia among children receiving ART at health facilities in the Wolaita Zone, South Ethiopia.
Methods and materials
Study area and period
A facility-based retrospective cohort study was conducted at health facilities in the Wolaita Zone among children enrolled in ART from February 2022 to January 2023. Wolaita Zone is one of the zones in the South Ethiopia Region. It is located about 328 km southwest of Addis Ababa. Currently, there are 20 health facilities which provide HIV care and treatment in the area (seven are public hospitals namely Wolaita Sodo University Comprehensive Specialized Hospital, Bale Primary Hospital, Halala Primary Hospital, Bitena Primary Hospital, Gesuba Primary Hospital, Bombe Primary Hospital, and Tebela Primary Hospital; two private hospitals namely Dubo St. Mery Primary Hospital and Sodo Christian Hospital; and 11 health centers namely Sodo Health Center, Ade-Shanto Health Center, Gurumo-Koysha Health Center, Areka Health Center, Boditi Health Center, Bedessa Health Center, Hobicha Health Center, Abala Faracho Health Center, Tome-Gerera Health Center, Amacho-Kodo, and Gununo Health Center). Among those, eight health facilities, of which four were hospitals, namely, Wolaita Sodo University Comprehensive Specialized Hospital, Halala Primary Hospital, Dubo St. May Primary Hospital, and Sodo Christian Hospital, and four were health centers, which are Sodo Health Center, Areka Health Center, Boditi Health Center, and Gununo Health Center, were included in this study. According to the Wolaita Zone health service delivery report, a total of 735 children below 15 years had been on ART within health facilities in Wolaita Zone in general, and 387 children in selected health facilities from January 1, 2012 to December 30, 2021. 26
Source and study population
All records of children aged <15 years who had been on ART at health facilities in Wolaita Zone. The study populations were all randomly selected records of children <15 years old who had been on ART at selected health facilities in Wolaita Zone and sorted from January 1, 2012 to December 30, 2021.
Inclusion and exclusion criteria
All ART records of children under 15 years old who were patients enrolled on ART at selected health facilities in the Wolaita Zone from January 1, 2012 to December 30, 2021, were excluded, and children who already had anemia at the start of the follow-up, who had a follow-up of <6 months, and children with incomplete data recording at baseline and during the follow-up time were excluded from the study.
Sample size determination
The sample size was computed using OpenEpi version 3.01 using the sample size calculator for cohort study by considering the following assumptions: 95% two-sided significance level (1-alpha), 80% power (1-beta, % chance of detecting), 1:1 ratio of unexposed/exposed, proportion of unexposed with outcome = 57.14%, and adjusted hazard ratio = 2, which was nearest to one from a previous study. 20 The estimated sample size was 320, and adding 10% incomplete data (320 × 10/100 ≈ 32), the final sample became 352.
Sampling procedure
For this study, the participants were selected using the stratified cluster sampling method. At first, health facilities were stratified by considering hospitals as the first stratum and health centers as the second stratum. Each health facility was considered a cluster. Then, clusters were selected randomly from each stratum, and the participants’ records that were consecutively selected were included in the study. Hence, out of 20 health facilities that provide ART service, 8 health facilities were selected for this study, namely Wolaita Sodo University Comprehensive Specialized Hospital, Dubo St. Mary Primary Hospital, Sodo Christian Hospital, Halala Primary Hospital, Sodo Health Center, Areka Health Center, Boditi Health Center, and Gununo Health Center (Figure 1).

Sampling procedure to select the final sample size.
Data collection tool and techniques
After reviewing the follow-up chart, client intake form, medical record, and laboratory requests, we prepared a checklist in English. The data extraction format includes socio-demographic characteristics, nutritional status, and clinically related information. The data were collected using a pretested questionnaire. The pretest was done in 5% of participants outside of the study area, which was not included in the actual study. The data were collected by eight health professionals and supervised by two health professionals who have Bachelor of Science degrees and who trained on comprehensive HIV care to ensure the quality of data. The data were collected by reviewing the follow-up chart, client intake form, medical record, and laboratory requests.
The study was conducted in accordance with the Helsinki Declaration of 1975 as revised in 2024, and the reporting of this study conforms to STROBE guidelines. 27 We have de-identified all the patients’ details while we conducted this study.
Data quality assurance
Data quality was assured by carrying out careful design of data extraction formats and recruitment and by giving adequate training and follow-up for data collectors and supervisors. Before the actual data collection commenced, the checklist was pretested on 5% of the sample size at Bedessa Health Center to ensure the completeness and consistency of the data-collecting tool. The Cronbach’s alpha test result for pretest data was found to be >0.7 (i.e., the assumption of internal consistency across the data collection tool was fulfilled). 28 Hence, no modification was applied to variables after the pretest. The data gathered from the pretest were not incorporated into the final analysis. Two days of training were provided for data collectors and supervisors. Intensive supervision was done by the principal investigator and supervisors during the whole period of data collection. There was random cross-checking for data completeness, accuracy, and consistency.
Operational definitions
Anemia: described as a HGB concentration of lower than 11 g/dL in children under the age of 5 years, 11.5 g/dL in children between the ages of 5 and 11 years, and 12 g/dL in children aged 12–14 years, after adjusting for altitude. 2 The adjustment to individual observed HGB value (g/dL) for altitude was conducted based on the following information: no adjustment (<1000 m), −0.2 g/dL (⩾1000 and <1250 m), −0.5 g/dL (⩾1250 and <1750 m), −0.8 g/dL (⩾1750 and <2250 m), −1.3 g/dL (⩾2250 and <2750 m), −1.9 g/dL (⩾2750 and <3250 m), −2.7 g/dL (⩾3250 and <3750 m), −3.5 g/dL (⩾3750 and <4250 m), −4.5 g/dL (⩾4250 and <4750 m), −5.5 g/dL (⩾4750 and <5250 m), and −6.7 g/dL (⩾5250 m). 29
Event: the development of anemia throughout the study period.
Time-to-event: the time period between initiation of follow-up and the occurrence of anemia.
Censored: when HIV-positive children withdraw, transfer out, are lost to follow-up, die, or the investigation period ends before the development of anemia.
WHO clinical stage: was categorized as early stage (WHO clinical stages I and II) and advanced stage (WHO clinical stages III and IV). 30
Nutritional status: based on weight-for-height Z (WHZ), nutritional status was categorized as normal (⩾−2 SD) and thinness (<−2 SD). Based on WAZ (for ⩽10 years) and Body Mass Index-for-Age Z-Score (BAZ) (for ⩾5 years), the nutritional status was categorized as not underweight (⩾−2 SD) and underweight (<−2 SD). Based on height-for-age Z (HAZ), nutritional status was categorized as not stunted (⩾−2 SD) and stunted (<−2 SD). 31
Data processing and analysis
The data were checked for completeness and consistency and then coded and entered into EpiData version 4.6.0.2. Then, it was cleaned and analyzed using STATA version 14. Descriptive statistics were computed to determine the frequencies and summary statistics. WHO AnthroPlus software (for ages under 5 years) and WHO AnthroPlus software (for ages 5–14 years) were used to create Z scores to classify nutritional category in view of WAZ, WHZ, body mass index for age, and HAZ. The Kaplan–Meier survival curve was used to estimate survival time after initiation of ART, and the Log-rank test was computed to compare survival functions of categorical variables. The Cox regression model was used to assess predictors of anemia. Those variables with p ⩽ 0.2 in the bi-variable analysis were selected as candidates for the multivariate Cox regression model. The Cox proportional hazard model assumption was checked graphically with log-log Cox adjusted survival estimates for each categorical variable and statistically using the scaled Schoenfeld residual test. Checking for multicollinearity was conducted using the variance inflation factor and tolerance. The model fitness of the final model was checked using the Cox–Snell residuals test. Variables with a p < 0.05 were considered statistically significant.
Results
Overview of the follow-up
A total of 387 medical records of children enrolled in ART were reviewed. Out of these, 23 (5.94%), 4 (1.03%), 5 (1.29%), and 3 (0.77%) were excluded due to lack of baseline HGB test results, developing anemia at baseline, lack of baseline CD4 count, and having <6-month follow-up period, respectively. Hence, observations from 352 medical records were included in the analysis of this study. The total analysis time at risk and under observation was 1932 person-years. Throughout the study period, 83 (23.58%) of the study participants developed the event of interest (anemia). The remaining 269 (76.42%) were censored. Among those who were censored, 168 (47.73%) continued until the end of the study without developing the outcome event, 52 (14.77%) were transferred, 29 (8.24%) were lost to follow-up, and 20 (5.68%) were dead before developing the outcome event.
Socio-demographic features
The median age at baseline was 7, with an interquartile range of 4. More than half of the participants (190 (53.98%)) were females. The majority of participants (253 (71.88%)) were from urban centers. The median age of caregivers was 35 with an IQR of 6. Most, 335 (95.17%), of the caregivers of the children enrolled in ART were married (Table 1).
Incidence density of anemia computed for socio-demographic-related features.

Baseline clinical-related features
Almost half of the children (177 (50.28%)) initiated ART at the first WHO clinical stage. The majority, 294 (83.52%), of the HIV-infected children were in the category of WHO clinical stage of one or two at baseline. The median CD4 count was 467 with an interquartile range of 269 (343, 612). More than two-thirds (257 (73.01%)) of the children’s CD4 counts were higher than 350 cells/µL.
The majority of the children enrolled in ART (293 (83.24%)) were free from opportunistic infections at baseline. Most of the HIV-infected children (330 (93.75%)) had no previous or current history of TB infection at baseline (Table 2).
Incidence of anemia stratified for baseline clinical characteristics.

CD4: cluster of differentiation 4; CPT: cotrimoxazole prophylactic therapy; TB: tuberculosis; WHO: World Health Organization.
Nutritional-status-related features
Nearly one-third of HIV-infected children (102 (28.98%)) had normal weight at ART initiation, followed by 146 (41.48%), 81 (23.01%), and 23 (6.53%) of children enrolled in ART who were mildly, moderately, and severely underweight, respectively. Regarding thinness, more than half (54.55%) of children were within the normal range of WHZ score at baseline. The remaining 29.09%, 10%, and 6.36% were found to experience mild, moderate, and severe thinness, respectively. About 38.35% of children enrolled in ART were found to be within the normal range of HAZ score. While the rest, 27.27%, 25.28%, and 9.09%, belong to the HAZ score category of mild, moderate, and severe stunting, respectively.
Follow-up-related characteristics
Nearly two-thirds of HIV-infected children (219 (62.22%)) initiated ART with a non-zidovudine-based regimen. Most of the children (321 (91.19%)) had good adherence to the ART regimen. More than two-thirds (267 (75.85%)) of the study participants’ ART regimens were changed (Table 3).
Incidence rate of anemia stratified for follow-up-related characteristics.

ART: antiretroviral therapy.
Incidence rate of anemia
The overall incidence rate of anemia throughout the follow-up period was 4.29 (95% confidence interval (CI): 3.46–5.34) per 100 person-years of follow-up. During the first 24 months of observation, the incidence rate was 10.17 (95% CI: 3.81–27.09) per 100 person-years. Then it increased to 13.05 (95% CI: 7.41–22.99) per 100 person-years within 36 months of observation. And then decreased back to 12.91 (95% CI: 8.33–20.02), 10.98 (95% CI: 7.76–15.52), and 1.11 (95% CI: 0.67–1.84) per 100 person-years of observation in 48, 60, and more than 60 months of follow-up, respectively. The cumulative proportion of anemia was 23.58% (95% CI: 19.42–28.32).
As the Kaplan–Meier survival estimates showing time to developing anemia, the cumulative probability of being anemia-free at 12, 24, 48, 60, 90, and 120 months of follow-up was 99.72%, 98.82%, 87.99%, 75.61%, 69.26%, and 65.11%, respectively (Figure 2).
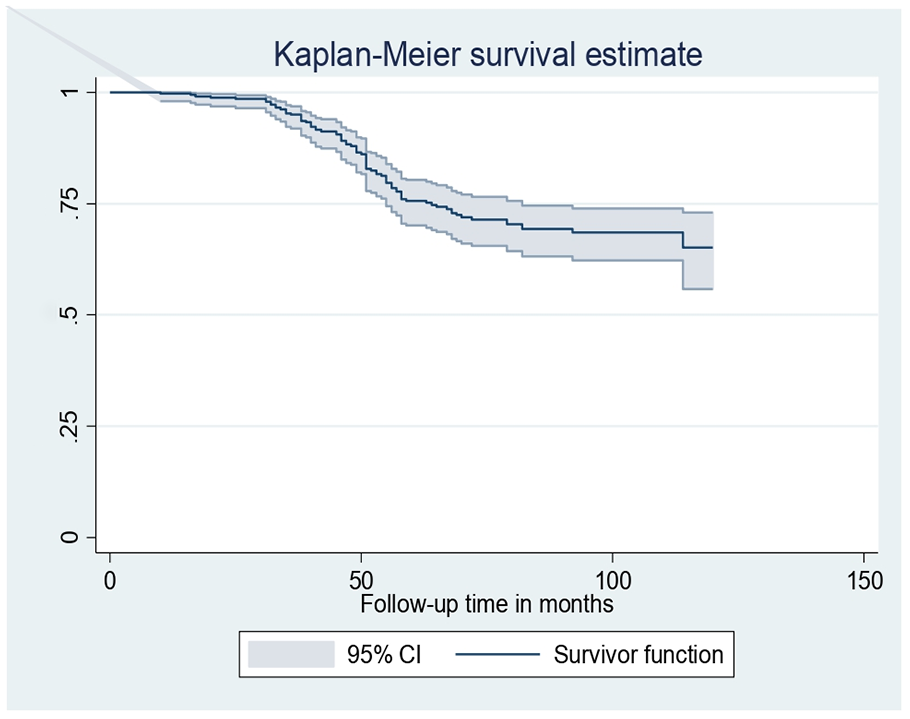
Kaplan–Meier survival estimate of children on ART at health facilities.
Log-rank test to compare the survival functions of categorical variables
The log-rank test for sex groups showed the difference in the pattern of time to event occurrence, p = 0.0257 (Figure 3(a)). Again, the log-rank test showed a significant difference between early and advanced stages of WHO clinical stage in terms of the pattern of time until the event occurs, p < 0.001 (Figure 3(b)). Similarly, the log-rank test showed a significant difference regarding the pattern time until the event between stunted and not stunted groups, p < 0.001 (Figure 3(a)). Furthermore, the log-rank test revealed the difference between zidovudine-based and non-zidovudine-based ART regimen groups in terms of distribution of time until the event occurs, p = 0.0214 (Figure 3(b)).

Kaplan–Meier survival curve in terms of sex (a) and WHO clinical stage (b).
Predictors of anemia
On bivariable Cox regression, sex, residence, WHO clinical stage, regimen change, ART regimen, ART adherence, and HAZ were identified as predictors of anemia occurrence. The multivariable analysis showed that only sex, WHO clinical stage, ART regimen category, and HAZ were identified as statistically important factors associated with the incidence of anemia.
The finding of this study shows that the hazard of developing anemia at any given time among females was 1.78 (95% CI: 1.29–2.47) times higher than males. Again, the hazard of developing anemia at any given time among children who belong to the advanced WHO clinical stage was 2.26 (95% CI: 1.26–4.06) times higher than those who belong to the early WHO (Figure 4).
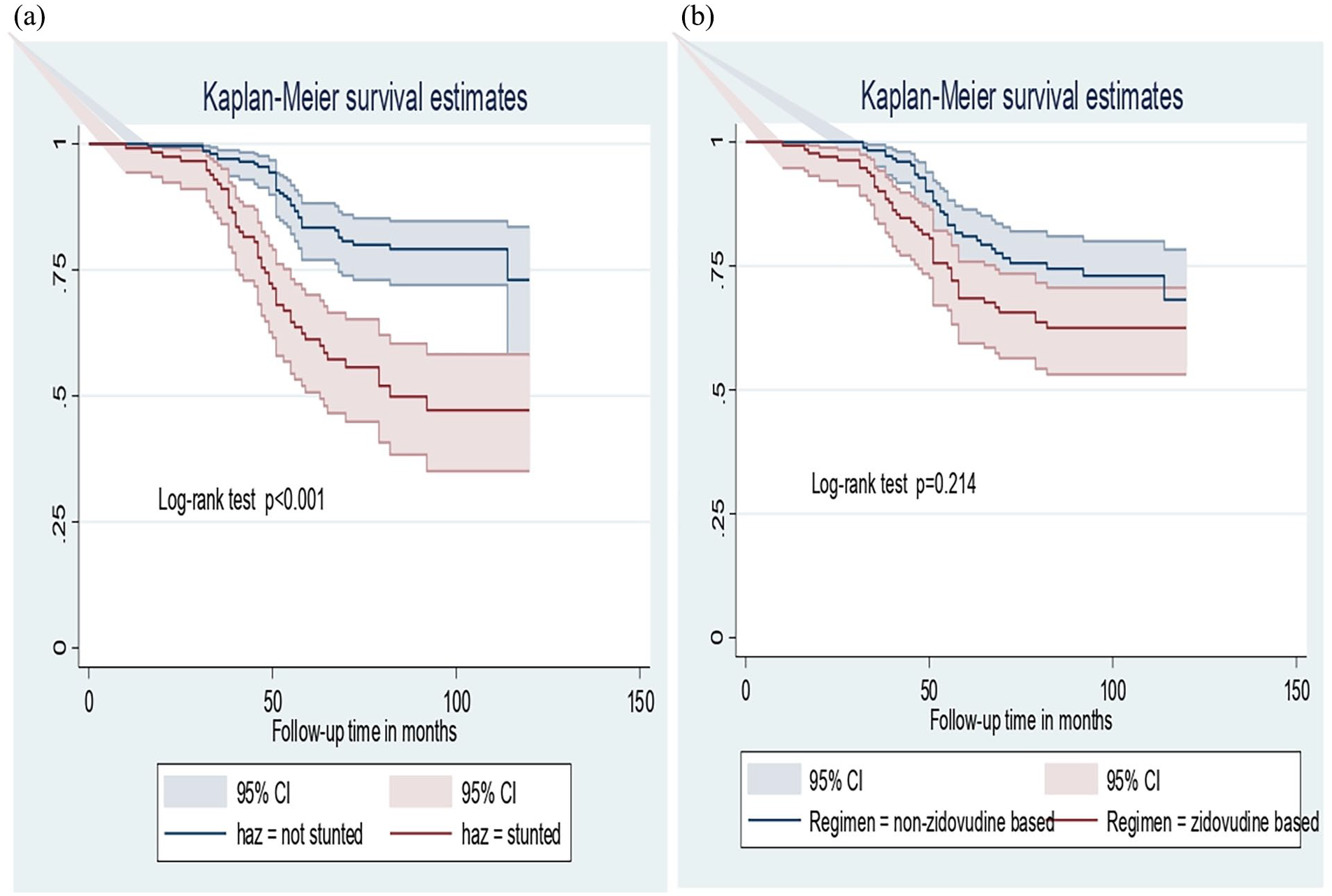
Kaplan–Meier survival curve of height-for-age Z score (a) and ART regimen (b).
The finding of this study also shows that the hazard of developing anemia at any given time among children on an AZT-based regimen is 2.07 (95% CI: 1.16–3.69) times higher compared to those who are on a non-AZT-based regimen. Furthermore, the hazard of developing anemia at any given time among HIV-infected children who were stunted at baseline was 3.34 (95% CI: 2.03–5.49) times higher when compared to their counterparts (Table 4 and Supplemental Appendix).
Result of bivariable and multivariable Cox regression analyses.

AHR: adjusted hazard ratio; ART: antiretroviral therapy; CHR: crude hazard ratio; CI: confidence interval; WHO: World Health Organization.
p < 0.05. **p < 0.01. ***p < 0.001.
Discussion
According to this study, the overall incidence density of anemia among children on ART was 4.29 (95% CI: 3.46–5.34) per 100 person-years of observation. The hazard of anemia among children was found to be 1.78 (95% CI: 1.29–2.47), 2.26 (95% CI: 1.26–4.06), 3.34 (95% CI: 2.03–5.49), and 2.07 (95% CI: 1.16–3.69) times higher among females, those who were at an advanced clinical stage (WHO clinical stage III or IV) at baseline, those who were stunted at baseline, and those who initiated ART with a zidovudine-containing regimen, when compared to their counterparts, respectively.
The overall incidence density of anemia identified in this study is in line with the study from West Africa, which showed that the incidence rate of anemia among HIV-infected children was 2.47–4.25/100 person-years. 32 However, the overall incidence density of anemia is lower when compared to studies conducted in Asia and Gondar, Northwest Ethiopia, which showed that the incidence density of anemia was found to be 5.4 and 10.5/100 person-years among children following the start of ART, respectively.12,21
The finding of this study shows that females are nearly twice as likely to develop anemia compared to males. This finding is supported by cross-sectional studies conducted in our country, which state that females reported that being female is almost two times more risky for becoming anemic when compared to being male.13,16 However, this finding contradicts some studies conducted before, which showed that males are more prone to develop anemia. Literature shows that due to menstrual bleeding, iron deficiency seems to be more frequent among females in the reproductive age group.13,33
The current study shows that the hazard of developing anemia at any given time is increased by twofold among children with advanced WHO clinical stage (WHO stage III or IV) when compared to those who are at an early stage of disease. This finding is in line with the study conducted in Eastern and Northern Ethiopia, which showed that HIV-infected children who belong to WHO clinical stage III or IV are at higher risk of developing anemia.22,34
The current study revealed that the hazard of anemia at any given time among children who were stunted was increased by threefold at baseline compared to their counterparts. This strong positive association could be taken as a sign of long-term malnutrition that may result in iron deficiency. 34 This finding is in line with the study from Senegal, which stated that anemia was found to be significantly associated with stunting. 16 This may be explained as nutritional deficiency increases the risk of health problems in general, and these conditions result in diminished HGB levels, which increase the risk of anemia. 13 Stunting and the incidence of anemia have a strong positive relationship, that is, supporting the notion that these two conditions should be treated as connected health issues in children since they are more likely to cohabit and impact one another. 35
This study also revealed that the hazard of developing anemia at any given time among children on zidovudine-containing ART is two times higher than those who are on non-zidovudine-containing ART regimens. This finding is supported by the findings from studies from Asia and Northern Ethiopia, which were conducted on HIV-infected children on ART.12,21,22 The possible explanations are that Zidovudine causes anemia; however, the actual mechanism through which it does so is still unclear. It was postulated that AZT might suppress erythroid stem cells or decrease erythropoiesis, raising erythropoietin levels and boosting mean corpuscular volume. 36 Furthermore, HIV directly impacts bone marrow, and the viral load can be suppressed by ART; in turn, this decreases the occurrence of anemia. 37
Strength and limitations
One of the strengths of the study is the use of a cohort study design, which gives more evidence-based findings. On the other hand, its limitation is that variables like intestinal parasite infection, household wealth index, inflammation, or C-reactive protein, and dietary score were not assessed. Exclusion of participants due to incomplete data may introduce selection bias.
Conclusion
The overall incidence density of anemia in the study setting was found to be relatively lower compared to incidence densities reported from other regions and countries. Females, children who were at an advanced WHO clinical stage at baseline, and children who were stunted at baseline may increase the hazard of anemia. This study showed a statistically significant difference in treatment effect on the occurrence of anemia among children who initiated ART with zidovudine-based and non-zidovudine-based regimens.
Healthcare providers should give special care to HIV-infected children who are at WHO clinical stage III or IV, those who are stunted during ART initiation, and those who initiate ART with a zidovudine-based regimen. Programs and nutrition interventions should emphasize children living with HIV/AIDS, who are stunted. A prospective cohort study is recommended to address the limitations mentioned in this study.
Supplemental Material
sj-docx-1-smo-10.1177_20503121251371898 – Supplemental material for Incidence and predictors of anemia among children with antiretroviral therapy at health facilities in Southern Ethiopia: A retrospective follow-up study
Supplemental material, sj-docx-1-smo-10.1177_20503121251371898 for Incidence and predictors of anemia among children with antiretroviral therapy at health facilities in Southern Ethiopia: A retrospective follow-up study by Legesse Tesfaye Gina, Yoseph Halala Handiso, Abebe Sorsa Badacho, Workineh Shume Hailu and Abiyot Wolie Asres in SAGE Open Medicine
Supplemental Material
sj-docx-2-smo-10.1177_20503121251371898 – Supplemental material for Incidence and predictors of anemia among children with antiretroviral therapy at health facilities in Southern Ethiopia: A retrospective follow-up study
Supplemental material, sj-docx-2-smo-10.1177_20503121251371898 for Incidence and predictors of anemia among children with antiretroviral therapy at health facilities in Southern Ethiopia: A retrospective follow-up study by Legesse Tesfaye Gina, Yoseph Halala Handiso, Abebe Sorsa Badacho, Workineh Shume Hailu and Abiyot Wolie Asres in SAGE Open Medicine
Footnotes
Acknowledgements
We would like to thank study participants, data collectors, supervisors, and hospital records registration officers for their great cooperation.
Ethical considerations
Ethical approval was obtained from the Wolaita Sodo University Institutional Review Board with the ethical reference number CRC SD132/02/14. The study was conducted in accordance with the Declaration of Helsinki.
Consent to participate
Written informed consent was obtained from the patients’ records following relevant guidelines and regulations. All data obtained from records was kept confidential using codes instead of any personal identifiers.
Author contributions
A.W.A., A.S.B., L.T.G., and Y.H.H.: participated in conception, data curation, formal analysis, investigation, funding acquisition, methodology, project administration, software, supervision, validation, visualization, writing – original draft preparation, writing – review and editing. A.W.A., A.S.B., L.T.G., Y.H.H., and W.S.H.: contributed to supervision, validation, visualization, writing – review and editing. All authors read and approved this article submission.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
