Abstract
The burden of congenital gastrointestinal (GI) anomalies is poorly understood. The aim of this systematic review to assess the global prevalence of congenital gastrointestinal (GI) anomalies. We conducted a systematic review of global population-level birth prevalence of gastroschisis, omphalocele, intestinal atresia, esophageal atresia, congenital diaphragmatic hernia, Hirschsprung’s disease, and anorectal malformation. We identified 103 studies from high and middle-income countries. Gastroschisis was the most reported condition. Studies reported geographic heterogeneity in gastroschisis, omphalocele, esophageal atresia, Hirschsprung’s disease, congenital diaphragmatic hernia, and anorectal malformation prevalence. The prevalence of gastroschisis, omphalocele, and Hirschsprung’s disease prevalence was reported to have increased over recent decades. Mean prevalence per 10 000 live and total births ranged from 2.1 to 4.4 cases and 1.4 to 3.6 per 10 000 live or total births, respectively. There is limited surveillance for congenital GI anomalies in LICs and potential temporal and geographic trends that need to be further investigated.
Introduction
The prevalence of major gastrointestinal (GI) congenital anomalies (CAs), such as gastroschisis, omphalocele, intestinal atresia, esophageal atresia, congenital diaphragmatic hernia (CDH), Hirschsprung’s disease, and anorectal malformation (ARM) is largely unknown, particularly in low- and middle-income countries (LMICs). 1 Surveillance for these conditions is challenged by limited surgical resources, lack of national pediatric surgical health planning, barriers to antenatal screening and surgical care, delayed case ascertainment, challenges to case follow-up, and cases being missing from care and surveillance entirely.2,3
The World Health Organization (WHO) supports strengthening and improving access to surgical health systems as a critical part of health system infrastructures. 4 An increasing number of countries are including surgical care in national health plans, but prevalence estimates for most common GI congenital anomalies remains unknown at the country level for most countries. 5 Existing studies have not appraised these prevalence estimates in a systematic fashion, and no study has applied the same systematic review methodology across all of these conditions. Estimates from the handful of studies that have reported prevalence of these conditions at the population-level have yet to be compiled and appraised systematically using the same methodology.6 -17 As well, most existing surveillance systems are predominantly in high-income countries (HICs) and studies often aggregate all disease-specific GI anomaly estimates into one aggregated GI anomaly stratum.18 -24
Extrapolating prevalence estimates from the hospital settings to the larger population would reflect substantial survivor bias, would underestimate true birth prevalence, and is especially problematic in LMICs where mortality from these conditions is substantially higher than in HICs and fewer cases survive long enough to reach care.25,26 Further, prevalence may also be highest in LMIC settings where potential exposures (e.g., insufficient maternal folic acid intake and insufficient maternal weight gain during pregnancy) may be highest. 27
Before justifying the temporal and monetary expense of conducting additional birth prevalence studies, it is important to compile currently available birth prevalence estimates and appraise them systematically. To address these gaps in knowledge of global major GI anomaly birth prevalence and provide a compiled list of birth prevalence estimates for each anomaly, the aim of this systematic review is to identify, compile, and critically assess the population-level birth prevalence of common congenital GI anomalies around the world.
Methods
Our methodology and protocol are registered with PROSPERO (CRD42023460268). We conducted our analysis and organized our findings according to the Preferred Reporting Items for Systematic reviews and Meta-Analyses 2020 (PRISMA 2020; Supplemental Appendix I) guidelines. 28 No changes were made to the original protocol; however, we collapsed reporting of results into only three categories (as described below) for ease of reporting.
Conceptual Framework, Inclusion Criteria, and Exclusion Criteria
We used the Condition, Context, and Population (CoCoPop) framework to guide our search strategy and reporting of results.29,30 The
The literature uses incidence and prevalence interchangeably when referring to birth prevalence. In this review, we searched the literature using both “incidence” and “prevalence” terminology. However, we report all estimates using the term “birth prevalence” (regardless of if the original article uses the word “incidence” or “prevalence”) and use the term “birth prevalence” from here onward. We report three mutually exclusive burden of disease measures: (1) the number of prevalent cases per 10 000 live births, (2) the number of prevalent cases per 10 000 total births (live births and stillbirths [with or without terminations]), and (3) the number of cases per 10 000 pregnancies.
Any study reporting population-level birth prevalence of these major GI anomalies, regardless of the year of data collection or year of publication, was eligible for study inclusion. We define “population-level” as any study in which estimates are calculated at sub-national, national, regional, or global levels and in which the target and study populations are plausibly representative of all births or children with the disease (treated or untreated). We did not apply publication year exclusion criteria given the sparsity of prevalence estimates for these conditions in the literature and the need to see how prevalence (and/or measurement of prevalence) has changed over time in different populations. Only studies in English, Spanish, or Portuguese were included in this review.
Search Strategy
We searched seven comprehensive medical and health databases: PubMed, JSTOR, Academic Search Complete (EBSCO), CINAHL (EBCSO), EMBASE, Global Health (EBSCO), and SCOPUS. Search strategies for each condition are included in Supplemental Appendix II and are summarized version is shown in Table 1. We used Medical Subject Heading (MeSH) terms specific to each GI anomaly and measure of occurrence (incidence or prevalence), and some terms that were broader. For instance, we used terms such as “omphalocele,” “exomphalos,” and “abdominal wall defects” to identify potential studies on omphalocele birth prevalence.
Study Screening, Bias Assessment, and Data extraction
All screening and data management was organized in Covidence systematic review software. 32 KRL first searched each disease’s search strategy in each database (with database specific formatting). KRL extracted and imported into Covidence systematic review software all studies retrieved from each application of each search strategy.32 There, KRL screened all articles on their abstracts for relevance to our study. Articles that had abstracts deemed relevant to our CoCoPop definition were screened by KRL and PEG on their full text and scored for study-level bias assessment using the Joanna Briggs Institute (JBI) Critical Appraisal Checklist for Studies Reporting Prevalence Data (Supplemental Appendix III). 33 Each article’s score was based on yes/no (1,0) points assigned to each of the tool’s nine questions. 33 KRL double-rated bias assessment scores for all articles screened by PEG, with the goal of interrater reliability greater than 0.80.
Data for studies meeting all inclusion criteria were then extracted from Covidence systematic review software for cleaning, organization, and compilation in SAS 9.4® for Windows (Copyright © 2013 SAS Institute Inc.; Cary, NC, USA; protocol under review).32,34 Extracted data included the study’s title, author list, journal, year of publication, exposure condition (gastroschisis, omphalocele, intestinal atresia, esophageal atresia, CDH, Hirschsprung’s disease, and/or ARM), study design (cohort, cross-sectional, case-control, surveillance, other), country/countries, WB geographic region(s) (East Asia and Pacific, Europe and Central Asia, Latin America and the Caribbean, Middle East and North Africa, Sub-Saharan Africa), WB income classification(s) (HIC, MIC, LIC), birth prevalence denominator (per 10 000 live births, total births, or pregnancies), and value of measure of occurrence (including upper and lower confidence intervals (CIs) and P-values if reported).
Reporting of Results
We organize results by surgical condition. For each condition, we list all studies included in the review and their corresponding values for the abstracted variables listed above. We report as a summary the mean (standard deviation [SD]), median (interquartile range [IQR]), minimum, and maximum prevalence per 10 000 live births, total births, and pregnancies. If multiple point or period estimates are reported in a single study for the same study population (e.g., period prevalence from 2000 to 2010, period prevalence in 2000, period prevalence in 2010, etc.), we used the most recent birth prevalence estimate to calculate the mean and median. If estimates are reported for a set of countries, we include the country-specific estimates (not the global, aggregated estimate) in our calculations of these frequency statistics. Finally, we report frequency statistics for JBI bias assessment scores overall and by study condition. 33 We do not conduct any meta-analyses or meta-regressions using the data of any study included in this review, as this is beyond the scope of the current study’s aims.
Ethical Guidelines
No financial support or specific funding for this project was received by any author for this work. No authors have conflicts of interests to disclose. This study involves no human research subjects nor the collection of data from any patient population. No results from any study included in this systematic review contain patient health information (PHI) nor can these results be used to identify participants of the respective studies. The review uses only previously published journal articles and, therefore, was deemed exempt from the corresponding author’s Institutional Review Board (IRB) review (IRB Study #23-3083).
Results
Of 2924 studies identified in our search, 1027 were duplicates, 1731 were deemed not relevant, and 63 did not meet inclusion criteria. We identified 103 studies that satisfied our inclusion criteria. Of these studies, 82 (80%) were from HICs, 16 were from MICs (16%), 0 (0%) were from LICs, and 5 (5%) were from both MICs and HICs (Table 2, Figure 1).
Overall Characteristics of Included Studies.
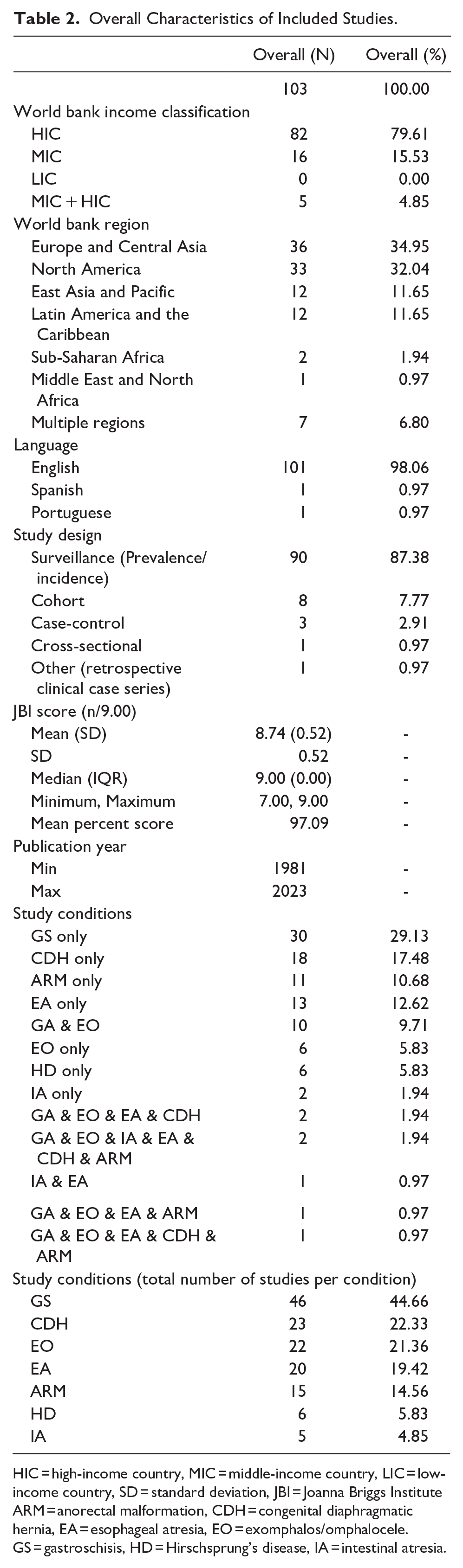
HIC = high-income country, MIC = middle-income country, LIC = low-income country, SD = standard deviation, JBI = Joanna Briggs Institute ARM = anorectal malformation, CDH = congenital diaphragmatic hernia, EA = esophageal atresia, EO = exomphalos/omphalocele. GS = gastroschisis, HD = Hirschsprung’s disease, IA = intestinal atresia.

PRISMA-P diagram.
Approximately one-third of studies were conducted in Europe and Central Asia (n = 36, 35%) and one-third in North America (n = 33, 32%). Fewer studies were conducted in Latin America and the Caribbean (n = 12, 12%) and Sub-Saharan Africa (n = 12, 12%; Figure 2, Supplemental Appendix V). Only one study was conducted in the Middle East and North Africa (1%). Seven studies (7%) were from in multiple World Bank regions. The majority of studies (n = 101, 98%) were reported in English while only 2% were in Spanish or Portuguese. Studies excluded based on language were written in German, Icelandic, Czech, Chinese, Norwegian, and Turkish. Most studies were surveillance studies (n = 90, 87%) with fewer being cohort (n = 8, 8%), case-control (n = 3, 3%), cross-sectional (n = 1, 1%), or other (n = 1, 1%) study designs.

Map of studies identified and included in this review.
Bias appraisal scores were high (indicating low risk of bias) for all study conditions regardless of measure reported (e.g., birth prevalence per 10 000 live vs total births). The mean and median bias assessment (JBI) scores (out of 9.00 possible points) across all disease categories were 8.73 (SD: 0.52) and 9.00 (IQR: 0.00), respectively (Table 3; Supplemental Appendix IV). The earliest publication year was 1981 and the most recent 2023, with a decrease in studies after 2011 (Figure 3). Inter-rater reliability on bias assessment was 100%. Most studies did not report 95% confidence intervals (CIs) around birth prevalence estimates (Supplemental Appendix V).
Joanna Briggs Institute (JBI) Scores for Identified Studies by Anomaly Type.

Full tables including JBI score by birth prevalence denominator (live births, total births, pregnancies) available in Supplemental Appendix IV; JBI = Joanna Briggs Institute; SD = standard deviation; IQR = interquartile range; min = minimum; max = maximum.

Number of ascertained major gastrointestinal anomaly birth prevalence studies by publication year.
Gastroschisis had the highest number of studies (n = 46), followed by CDH (n = 23) and exomphalos (n = 22). Intestinal atresia had the fewest number of studies (n = 5), followed by Hirschsprung’s Disease (n = 6). A total of 17 studies (17%) reported birth prevalence for multiple study conditions.
Gastroschisis
Forty-six studies reported gastroschisis prevalence (Table 4, Supplemental Appendix V). Of 33 studies reporting birth prevalence per 10 000 live births, the mean and median prevalence estimates were 2.69 (SD: 1.61) and 2.34 (IQR: 1.31, 3.92) cases per 10 000 live births, respectively. Fifteen studies reported birth prevalence per 10 000 total births (live births and stillbirths, with or without terminations included), with a mean of 3.28 (SD: 2.04) and a median of 2.67 (IQR: 1.90, 3.70). One study reported 0.81 cases per 10 000 live births and one reported 0.31 cases per 10 000 live births, both estimates that were notably lower than all other estimates.8,35 Of 46 studies, only two distinguished between isolated, non-isolated, and total gastroschisis cases.
Birth Prevalence by GI Anomaly.

Not applicable, no studies identified.
Only one estimate identified; therefore, no mean, SD, median, min, or max.
SD = standard deviation, IQR = interquartile range, Min = minimum, Max = maximum.
Omphalocele
Twenty-two studies reported omphalocele birth prevalence (Table 4, Supplemental Appendix V) with considerable variation in estimates and in whether or not the denominator included stillbirths and/or terminations in addition to live births. Twelve studies reported birth prevalence per 10 000 live births (mean: 1.63, SD: 0.70, median: 1.55, IQR: 1.11, 2.08) and twelve reported birth prevalence per 10 000 total births (mean: 2.62, SD: 1.05, median: 2.40, IQR: 2.05, 3.05). Only one study reported an estimate with a different denominator, and reported 5.45 cases per 10 000 pregnancies (including reported perinatal losses). 36
Intestinal Atresia
Five studies reported intestinal atresia (Table 4, Supplemental Appendix V) birth prevalence. Two studies reported the number of cases per 10 000 live births (mean: 2.57, SD: 0.47, median 2.57, IQR: 2.40, 2.73). Three studies reported prevalence per 10 000 total births, with a mean of 2.24 (SD: 0.90) and a median of 2.30 (IQR: 1.58, 2.95). Across studies of intestinal atresia prevalence, mean prevalence per 10 000 live births was higher than prevalence per 10 000 total births. Though, the sample size was small, and studies were conducted in different populations and time periods precluding direct comparison of estimates. Of the five studies, only one study distinguished between cases with and without chromosomal anomalies.
Esophageal Atresia
Twenty articles reported esophageal atresia birth prevalence (Table 4, Supplemental Appendix V). Over half of studies (n = 11) reported the number of prevalent cases per 10 000 live births (mean: 2.46, SD: 0.53, median: 2.45, IQR: 2.08, 2.92). Nine studies reported prevalence per 10 000 total births (mean: 2.53, SD: 0.71, median: 2.51, IQR: 1.97, 2.89). As with studies identified for intestinal atresia, only one study stratified estimates by presence or absence of chromosomal anomalies.
Hirschsprung’s Disease
Six studies reported population-level birth prevalence of Hirschsprung’s disease (Table 4, Supplemental Appendix V). Of the four studies reporting prevalence per 10 000 live births, there was a mean of 2.09 (SD: 0.21, median: 2.13, IQR: 1.99, 2.22). Two studies, across 32 countries and regions collectively, reported birth prevalence per 10 000 total births (mean: 1.38, SD: 0.70, median: 1.52, min: 0.07, max: 0.79, 1.84).
Congenital Diaphragmatic Hernia
At total of 23 studies reported CDH birth prevalence (Table 4, Supplemental Appendix V) and less than half (n = 11) reported the number of prevalent cases per 10 000 live births (mean: 2.61, SD: 0.89, median: 2.80, IQR: 2.52, 3.10). Thirteen studies reported birth prevalence per 10 000 total births, collectively having a slightly higher mean and an equal median compared to estimates of prevalence per 10 000 live births (mean: 2.85, SD: 1.09, median: 2.80, IQR: 2.30, 3.35).
Anorectal Malformation
Fifteen studies reported ARM prevalence at birth (Table 4, Supplemental Appendix V), most of which (n = 11) reported prevalence per 10 000 total births (mean: 3.61, SD: 1.61, median: 3.47, IQR: 2.82, 4.42). The mean of estimates reporting prevalence per 10 000 live births was lower than the mean of estimates reporting prevalence per 10 000 live births (mean: 4.44, STD: 3.74, median: 3.00, IQR: 2.33, 5.11). There was one estimate far higher than any other in this review (9.94 incident cases per 10 000 live births) but this estimate was stable in the study population during the 13 years of study. 37
Discussion
Few studies have systematically reviewed the literature for birth prevalence of major GI anomalies, and no single study has systematically reviewed the literature for the birth prevalence of a large set of these anomalies using the same methodologies and search strategies. We identified articles reporting gastroschisis, omphalocele, intestinal atresia, esophageal atresias, CDH, Hirschsprung’s disease, and ARM birth prevalence. The number of studies addressing the prevalence of common GI anomalies increased over time, but seems to have plateaued since 2011, with few studies on intestinal atresia and Hirschsprung’s disease. The locations of identified studies largely reflect the locations of established surveillance systems in Europe and Central Asia and North America.18 -20,22,23,38 -40 The summary statistics of birth prevalence of each anomaly are, therefore, more representative of HICs and MICs than LICs. Importantly, outside of these surveillance systems, clinical cohort and hospital settings that may not be representative of the larger, general population of interest are often relied on for surveillance. 1 This is problematic because (1) many births and infant deaths are not registered, (2) not all GI anomaly cases will receive surgical care at the given hospital(s), and (3) not all cases at the given hospital(s) will be reported to a centralized surveillance network.41 -47
The lack of LIC specific estimates is unsurprising given the sparsity of surveillance programs for pediatric surgical conditions in LICs but remains alarming. Although it is possible that LICs were included in Di Tanna et al study, it is unclear what countries are included in the South America region in this study. 48 This disparity in GI anomaly research and surveillance in LICs is a key finding that underscores the need for study of these conditions and their prevalence in LIC settings.18 -20,22,23,39,40
Of 103 studies, only two stratified prevalence estimates by public versus private health sector, which is important where health insurance and socioeconomic statuses may be proxies or predictors for hypothesized exposures related to these anomalies or for access to specialized surgical care.49,50 No studies included in the review were written in French, which represents a possible limitation in identifying studies conducted in French-speaking LMICs and/or a lack of studies in these settings where we anticipate inequitably high mortality from these anomalies.26,51 Methodologically, there was substantial variation in reporting terminology for birth prevalence across the literature, both in whether incidence or prevalence terminology was used and the denominator used (number of live births, number of total births, or number of pregnancies).
Gastroschisis
The mean and median number of prevalent cases per 10 000 total births were higher than the mean and median number of cases per 10 000 live births for gastroschisis (inclusion of stillbirths and terminations in the denominator should capture more cases than using a denominator counting only live births). 52 Two studies reported low estimates, 0.50 (Taiwan, 1996-2013) and 0.81 (Canada, 1981) cases per 10 000 total births, though the latter is likely outdated.8,35 Alternatively, there was one high estimate using National Registry of Congenital Anomalies (RENAC) data in Argentina from 2009-2013 (8.53 (95% CI: 7.68, 9.24)). 49 Collectively, results indicate potential geographic and temporal variation with increasing prevalence since the 1980s (or changes in maternal age structure and/or differences in detection and reporting in recent years).8,53 -57
Omphalocele
The mean and median prevalence per 10 000 live births were lower than the mean and median prevalence per 10 000 total births as expected. Grover et al reported a particularly high prevalence, which is over double the other reported estimates (5.45 cases per 10 000 pregnancies, UK, 1994-2015) and which may be inflated by inclusion of pregnancies not resulting in live births. 36 Nembhard et al also reported also reported a high birth prevalence (5.80 cases per 10 000 total births, France, 2000-2012), and results for omphalocele were sensitive to inclusion versus exclusion of terminations in the denominator.58,59 Overall, there is likely geographic variation in omphalocele birth prevalence. 58 However, there does not appear to be consensus in the current literature on whether or not omphalocele birth prevalence has increased or decreased over recent decades (such as increasing prevalence in the United States from 2015-2018 vs stable prevalence in the Liaoning province of China from 2006-2015).55,60 -66 Given increased gastroschisis prevalence over recent decades, reported changes in omphalocele birth prevalence should be investigated further and in the context of potential geographic variation in prevalence.
Intestinal Atresia
Few studies were identified for intestinal atresia birth prevalence. Reporting birth prevalence in Argentina using RENAC data, Bidondo et al reported a substantially higher estimate of 3.10 (95% CI: 2.70, 3.54) compared to Bidondo et al (2021; 1.69 (1.52, 1.88)), suggesting that birth prevalence might have decreased in this population over recent years.49,50 However, much more investigation of trends in intestinal atresia birth prevalence is warranted, as evidence for temporal and geographic trends remains inconclusive and population-level estimates remain sparse.
Esophageal Atresia
For esophageal atresia, the mean and median number of prevalent cases per 10 000 live births were lower than the mean and median number of prevalent cases per 10 000 total births as expected, and some studies reported similar estimates over time.62,67 Others, however, reported temporal trends. 68 Further, results from two studies suggest possible geographic variation, though stillbirth definitions and years of data collection differed across studies.11,69 Beyond the studies included in this review, there is little literature on geographic and temporal trends for esophageal atresia. Investigation of geographic variation, controlling for reporting practices and years of data collection, is warranted. Further, only a handful of studies stratified estimates by esophageal atresia cases with tracheoesophageal fistula (TEF) or without TEF, which could contribute to variations in estimates and impact mortality predictions from these estimates.
Hirschsprung’s Disease
As with intestinal atresia, estimates of measures of occurrence for Hirschsprung’s Disease were sparse. Opposite of what is expected, the mean and median birth prevalence 10 000 live births were higher than mean and median birth prevalence per 10 000 total births. However, we remind the reader that we can only broadly compare these estimates, as they are calculated from different study populations and contexts. Best et al (2012) found an increase in prevalence in England from 1990-1994 to 2005 to 2008, though this increase could be due to a true increase in burden of disease and/or due to increased surveillance or varying diagnostic methods for the disease. 70 Best et al, again studying prevalence in 20 European countries, later reported potential geographic variation in Hirschsprung’s disease prevalence and, again, found a minor increase in prevalence from 1990-2009. 12 Data collection years, and potentially other surveillance, diagnostic, and data collection factors, varied slightly; though, the trend remained the same with inclusion and exclusion of chromosomal anomaly cases. 12 Further, stillbirth and losses may occur at different proportions with Hirschsprung’s disease compared to other anomalies under study. 71 These few studies warrant further investigation of Hirschsprung’s disease birth prevalence over time and between geographic regions as we identified no consensus on these trends.
Congenital Diaphragmatic Hernia
We identified a large number of studies reporting CDH birth prevalence, including one study which reported prevalence separately in 19 countries and regions. 72 Paoletti et al’s systematic review of CDH found a pooled prevalence of 2.3 CDH cases per 10 000 live births, which is similar to what is reported in a handful of studies included in this review.15,62,72 -74 Of note, some studies stratified estimates by isolated and non-isolated cases while others did not, and trends in prevalence may be different between these types of cases. 75 Mean and median prevalence and incidence estimates were similar, regardless of denominator (live or total births) and were similar to what we expected based on prior evidence.75,76 Some studies reported a slight increase in prevalence over time, but evidence for temporal trends remains unclear.62,73,77,78 Finally, Chen et al, a study published after we ascertained review articles and therefore not included in this review, reported geographic trends in prevalence and increasing CDH prevalence in recent years suggesting investigation of CDH birth prevalence in different geographic populations is warranted. 79
Anorectal Malformation
Most studies reporting estimates for ARM reported prevalence per 10 000 total births, which was lower that prevalence per 10 000 live births. One study (using data from Saudi Arabia from 1998-2010) reported a high birth prevalence of 9.94 cases per 10 000 live births that remained stable over the study period. 37 It is possible that there is a higher burden of disease in this setting while it is also possible that this setting has better surveillance or diagnostic practices and/or more false positives. 37 Regardless, such a high prevalence compared to other regions should be prioritized for investigation. Further, multiple studies in different countries reported no statistically significant changes in birth prevalence over their respective study periods.13,37,80 Overall, the studies included in this review on ARM suggest potential geographic patterns that should be further explored, and do not point to significant temporal trends in these study populations.13,37,50,80 -82
Recommendations
Overall, beyond extreme rigor and assessment of which prevalence estimates are (and are not) comparable to each other (non-standardized terminology for reporting birth prevalence can lead to difficulty comparing and interpreting different measures of occurrence), our recommendations for next steps in research and practice are to:
1. Further investigate the following:
a. Birth prevalence of these major GI anomalies in LIC settings, as no studies identified estimates drawn from LICs alone. Mortality from these conditions remains inequitably high in LIC settings compared to HIC settings, but it remains unclear if baseline prevalence is also higher. 26
b. Birth prevalence of intestinal atresia and Hirschsprung’s disease in all country income settings. These conditions had the least comprehensive research on prevalence/incidence compared to other conditions of interest in this review.
c. Temporal trends, controlling for reporting, diagnostic, and surveillance practices, particularly for intestinal atresia, esophageal atresia, CDH, ARM, and Hirschsprung’s disease.
d. Geographic variation, controlling for reporting, diagnostic, and surveillance practices, for these anomalies. If trends remain, investigation potential exposures related to increased prevalence in some geographic regions compared to others should be considered.
2. Distinguish between estimates drawn from public health sectors and private health sectors where plausible differences in mortality and/or prevalence exist amongst populations accessing care through each health sector.
3. Explicitly define the following:
a. If chromosomal anomaly and/or non-chromosomal anomaly cases are included in the denominator.
b. If isolated and/or non-isolated anomaly cases are included in the case definition.
c. Stillbirth (e.g., number weeks, number of grams, etc.) where stillbirths are included in the denominator, and if terminations were or were not included in this denominator.
Limitations
There are several limitations to our analysis. First, as with any systematic review, publication bias is a risk as is the risk of not identifying all potentially relevant articles, particularly from databases not searched. 83 However, our comprehensive search of seven major medical, population health, and global health databases helps mitigate this risk. Further, we cross-checked our review with systematic reviews for these conditions with different research questions to ensure we did not include studies that do not fully meet our inclusion criteria.14 -17 Use of PRISMA-P guidelines, a priori PROSPERO registration, and peer reviewed publication of our study protocol support transparent reporting of all essential measures and methodological decisions, and a key strength of this study is that we applied the same methodologies to our search for all seven major GI anomalies of interest. Use of the validated JBI checklists for all articles included in the review increases consistency of bias assessment across all studies.
Second, exclusion of studies not reported in English, Spanish, or Portuguese is a limitation (studies excluded based on language were written in German, Chinese, Czech, Icelandic, and Turkish). Identification of no studies written in French represents a potential limitation in our search strategy and/or a lack of publication of research from French-speaking countries. Third, we cannot observe and measure the exact incident event in which these conditions develop, only those that survive long enough (in utero or to birth) to be detected. 52 Changes in prevalence over time may be due to true changes in prevalence, changes in populations under study (e.g., the population’s age structure and number at risk for the condition), changes in case detection, and/or changes in exposures. Finally, it is important to note that these estimates can only be broadly compared to each other as they are from different studies and different populations at risk.
Conclusions
This review identifies key population-level birth prevalence estimates for a set of major GI anomalies and identifies gaps in current knowledge of the prevalence of these conditions. There is still a paucity of described birth prevalence of major GI anomalies requiring surgery in LMIC settings and for intestinal atresia and Hirschsprung’s disease prevalence in particular. These data are essential to pediatric surgical planning, financing, and resource allocation and should be prioritized for future study.
Supplemental Material
sj-docx-1-gph-10.1177_30502225251304582 – Supplemental material for The Global Burden of Major Gastrointestinal Anomalies: A Global, Systematic Review of Gastrointestinal Anomaly Birth Prevalence
Supplemental material, sj-docx-1-gph-10.1177_30502225251304582 for The Global Burden of Major Gastrointestinal Anomalies: A Global, Systematic Review of Gastrointestinal Anomaly Birth Prevalence by Kelsey R. Landrum, Pamela Espinoza, Cesia Cotache-Condor, Brian W. Pence, Henry E. Rice, Mark G. Shrime, Chris B. Agala, Emily R. Smith and Jessie K. Edwards in Sage Open Pediatrics
Footnotes
Author Contributions
KRL conceptualized this study and led the systematic review, the study analysis, and the study’s manuscript preparation. KRL conducted all searches and abstract screenings. KRL and PE conducted full-text screening and bias assessment together. ERS, HER, JKE, MGS, and BWP supervised this study. ERS, HER, MGS, and CCC provided content and methodological expertise. CBA, along with all co-authors, contributed substantially to methodology and manuscript review.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

