Abstract
Background:
Type 2 diabetes mellitus (T2DM) is a chronic, progressive metabolic disorder with increasing prevalence in India, imposing significant morbidity and healthcare burden. Dipeptidyl peptidase-4 inhibitors (DPP-4i) are established glucose-lowering agents, and Trelagliptin, a novel once-weekly DPP-4i, offers potential advantages in adherence and convenience. However, real-world evidence on its use in routine clinical practice remains scarce.
Objectives:
To evaluate physician perceptions, prescribing patterns, and clinical experience with Trelagliptin in the management of T2DM in India.
Methods:
A multicentre, questionnaire-based survey was conducted among 71 practicing medical professionals across diverse domains in India. The structured questionnaire captured respondents’ demographics, patient load and perceptions of glycaemic control, adherence, tolerability, safety, cost-effectiveness, and factors influencing Trelagliptin prescription. Descriptive statistics were used to analyse responses.
Results:
The results demonstrated that most of the respondents were consultant physicians (64.79%), followed by diabetologists/endocrinologists (18.31%) and other specialities (16.90%). Most respondents (70.42%) identified patients with poor adherence to daily medication as suitable candidates for Trelagliptin. Physicians perceived improved glycaemic control and adherence with Trelagliptin, as rated by 90.14% and 94.37% of respondents, respectively. Additionally, 32.39% of respondents perceived Trelagliptin as superior for glycaemic control, while 70.42% perceived higher adherence and 66.20% perceived greater cost-effectiveness compared with other DPP-4i. A majority of respondents (64.79%) emphasised the importance of reducing pill burden. Improved tolerability was rated by 97.18% of respondents.
Conclusion:
Based on physician perceptions, once-weekly Trelagliptin was considered a safe, effective, and convenient therapeutic option for T2DM, with respondents reporting perceived improvements in adherence and patient-centred care in routine clinical practice.
Introduction
Type 2 diabetes mellitus (T2DM) is a chronic, progressive, and non-communicable disease with a multifactorial aetiology involving genetics, lifestyle, and environmental influences. It is recognised as one of the major global health challenges of the twenty-first century, 1 ranking among the leading causes of morbidity and mortality worldwide alongside cardiovascular, respiratory diseases, and cancer.2,3 According to the International Diabetes Federation (IDF) Diabetes Atlas (2025), the reported global prevalence of diabetes among adults aged 20-79 years reached approximately 589 million across 215 countries and territories. Additionally, diabetes was attributed to an estimated 3.4 million deaths in the year 2024. 2 The burden is particularly higher in low- and middle-income countries such as India. Estimates suggest that over 101 million individuals in India have T2DM, with an additional 136 million classified as pre-diabetic and nearly 40 million undiagnosed cases. 4 Recent surveys indicate that the prevalence of diabetes in India has increased from 7.1% in 2009 to 11.4% in 2023, and is projected to reach 152 million cases by 2045.4,5 This rising prevalence is largely driven by rapid urbanisation, industrialisation, sedentary lifestyles, and dietary transitions.
Pharmacological management of T2DM has advanced considerably over the past two decades, with dipeptidyl peptidase-4 inhibitors (DPP-4i) becoming an important class of glucose-lowering agents. DPP-4 is a serine protease that inactivates incretin hormones, including glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic peptide (GIP), thereby impairing insulin secretion and promoting postprandial hyperglycaemia. Inhibition of DPP-4 enhances the activity of incretins, leading to improved insulin secretion, reduced glucagon release, and better glycaemic control with a low risk of hypoglycaemia. 6 Several DPP-4 inhibitors such as Sitagliptin, Saxagliptin, Alogliptin, and Linagliptin are already in widespread clinical use.7–10
Trelagliptin is a novel, highly selective, once-weekly oral DPP-4 inhibitor developed to improve patient convenience and adherence. Its extended elimination half-life (~54 hours) supports sustained DPP-4 inhibition across the dosing interval. 11 Clinical trials have demonstrated that Trelagliptin 100 mg once-weekly is non-inferior in efficacy to daily Alogliptin, with favourable safety and tolerability profiles. 12 Long-term studies have further confirmed its effectiveness as monotherapy and in combination with other oral antidiabetic drugs. 13
While clinical trials have established the efficacy and safety of Trelagliptin, there is limited evidence regarding its real-world use, particularly in Indian clinical practice. Physicians’ experiences and perceptions can provide valuable insights into their role in the routine management of T2DM. Therefore, the purpose of the present study was to evaluate real-world physician perspectives on the efficacy, safety, and patient adherence associated with Trelagliptin through a structured questionnaire-based survey.
Materials and Methods
The present study employed a structured, questionnaire-based, cross-sectional, multicentre survey designed to evaluate real-world prescribing patterns and clinical perceptions pertaining to the use of Trelagliptin in the management of T2DM. The multicentre nature of the study refers to data collected from 71 independent hospital-based centres, with one response obtained from each participating hospital. These centres were geographically distributed across 23 cities in India, representing diverse regions and clinical practice settings. Practicing medical professionals, including consulting physicians (CPs), diabetologists/endocrinologists, and other specialties (e.g., general practitioners, cardiologists, and surgeons) were invited to participate. Eligible participants were required to be actively practicing medical professionals with prior clinical experience in prescribing or managing patients with T2DM, including the use of Trelagliptin. A convenience sampling approach was adopted, whereby physicians were recruited through established professional medical networks and hospital-based contacts across the participating centres. A total of 85 questionnaires were distributed, of which 71 were completed and returned, resulting in a response rate of 83.5%. The survey was administered in a paper-based, self-filled format over a duration of 1 month period in July 2025.
Study Questionnaire
A structured questionnaire was developed to systematically capture physician-experience on the need, utility, and clinical integration of Trelagliptin. The questionnaire consisted of two sections: (a) demographic and clinical practice characteristics, including respondent specialty, self-reported duration of clinical experience, practice location and self-reported estimated monthly volume of patients with T2DM, and (b) drug-related domains addressing perceived treatment need, ideal pharmacological characteristics, efficacy, adherence, safety, tolerability, and determinants influencing prescription preference. The questionnaire incorporated predefined multiple-choice responses, rating scales, and optional free-text fields to allow elaboration. Both closed-ended (multiple-choice, rating scales) and open-ended questions were included to ensure comprehensive responses and minimise reporting bias. Multiple responses were permitted for the selected questionnaires to accurately capture the range of physician perspectives and allow respondents to reflect all applicable considerations, ensuring comprehensive and representative data. It is acknowledged that the questionnaire was author-developed and did not undergo formal psychometric validation or independent content validity assessment prior to administration. However, questions were reviewed iteratively by the study authors for face validity and clarity prior to distribution, and the free-text fields provided an opportunity to capture perspectives beyond the predefined response options. The complete study questionnaire is provided in Appendix A.
Data Collection and Statistical Analysis
All responses were compiled and entered into Microsoft Excel for analysis. Internal quality checks, including cross-verification of entries and consistency assessment, were performed to ensure data accuracy and completeness. Descriptive statistical methods were applied; no inferential analyses were planned. Categorical variables summarised as absolute frequencies (n) and percentages (%). Continuous parameters, where applicable, were reported using measures of central tendency, including mean and average values. Graphical representations (bar charts and pie charts) were prepared to illustrate key findings.
Ethical Considerations
An ethics committee review was waived for this study, as it involved anonymised, non-interventional physician respo-nses without patient-level data collection. Although formal ethics approval was waived, verbal informed consent was obtained from all physician participants prior to questionnaire administration. Participation was entirely voluntary, physicians were informed of the study purpose and their right to withdraw at any time, and all responses remained anonymous. Confidentiality of respondent identity and submitted information was strictly maintained throughout data handling and reporting.
Results
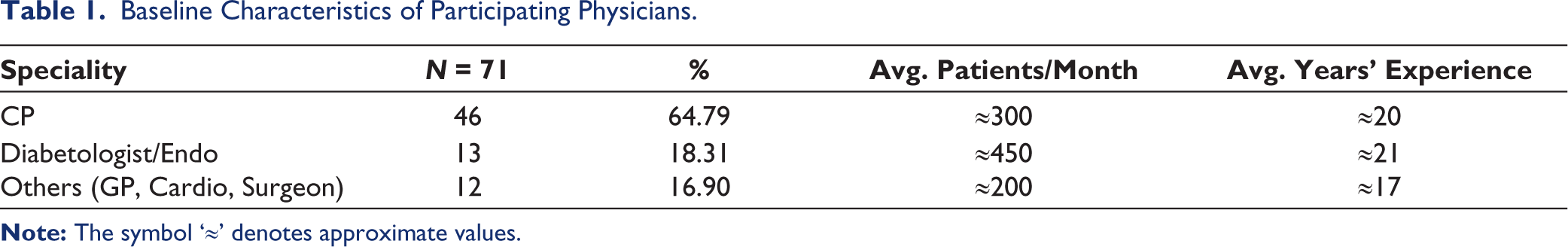
In the present study, a total of 71 respondents from diverse practice areas were included. Among these respondents, the majority were CPs (64.79%), followed by diabetologists or endocrinologists (18.31%) and other specialties (e.g., general practitioners, cardiologists, and surgeons) (16.90%). A detailed description is presented in Table 1. Most respondents (64.79%) rated reduction in pill burden as very important in diabetes management, while a small proportion (2.82%) regarded pill burden reduction as having limited importance in clinical decision-making (Figure 1A). Regarding ideal patient profiles for Trelagliptin therapy, a substantial proportion of respondents (70.42%) identified individuals with poor adherence to daily antidiabetic medication as the most suitable candidates. Working professionals or frequent travellers were the next commonly identified group, as indicated by approximately half of the respondents (43.66%). Smaller proportions of respondents recommended Trelagliptin for patients with renal impairment (18.31%) or those with intolerance to other DPP-4 inhibitors (14.08%) (Table 2).
Baseline Characteristics of Participating Physicians.
Patient Categories Considered Ideal for the Use of Trelagliptin.

(A) Pie Chart of Physicians’ Views on the Need for Pill Burden Reduction in Diabetes Management. (B) Pie Chart of Physicians’ Views on Adherence Improvement. (C) Pie Chart of Physicians’ Switching Preference to Trelagliptin. (D) Bar Chart of Influencing Factors to Prescribe Trelagliptin.
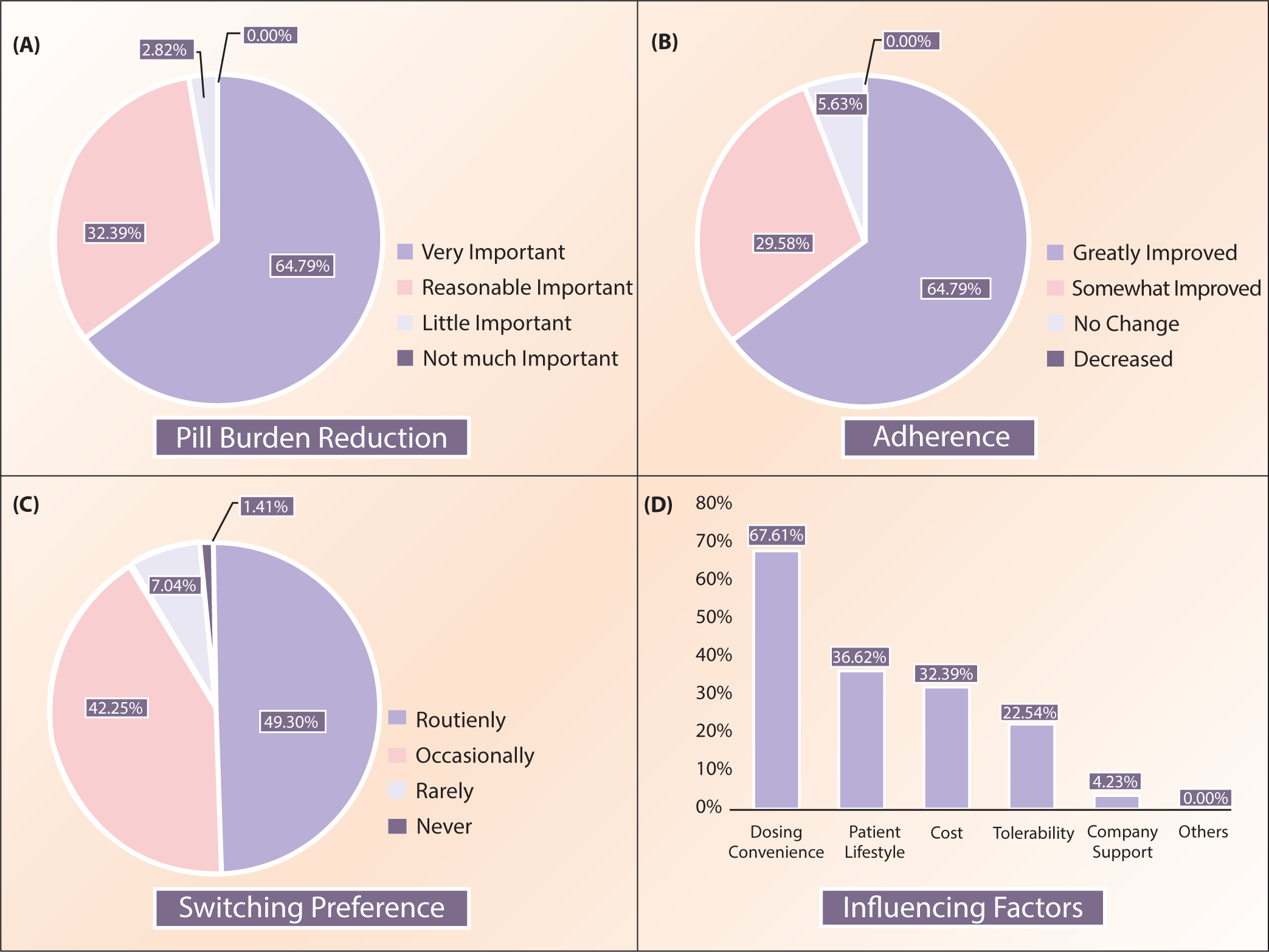
In terms of treatment efficacy, more than half of the respondents (56.34%) rated Trelagliptin’s overall glycemic control as good, while 33.80% perceived it as excellent. With respect to Glycated haemoglobin (HbA1c) reduction specifically, 74.65% of respondents noted the effect as moderately effective, while 19.72% rated it as highly effective. Only 5.63% viewed its HbA1c reduction capacity as minimal (Table 3).
Physicians’ Views on Efficacy of Trelagliptin.
As illustrated in Figure 1B, perceptions of treatment adherence were strongly favourable. A once-weekly dosing regimen was perceived to positively influence adherence, with 64.79% of respondents stating it led to substantial improvement. Only 5.63% rated no observable change in adherence patterns.
Comparative evaluations demonstrated that a majority of respondents (70.42%) considered adherence outcomes with Trelagliptin superior to other available therapeutic options. Cost-effectiveness was also observed positively, with 66.20% rating the drug as more cost-effective and only 4.23% perceiving it as less favourable in this regard. In relation to glycaemic efficacy, most respondents (67.61%) considered Trelagliptin comparable to other DPP-4 inhibitors, although 32.39% believed its performance to be superior (Table 4).
Comparative Assessment of Trelagliptin Versus Other DPP-4 Inhibitors.

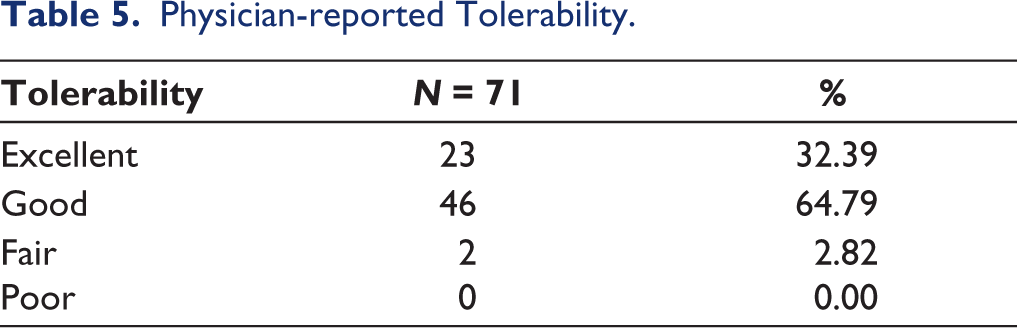
With respect to safety and tolerability, 64.79% of participants rated tolerability as good, while 32.39% rated it as excellent. Only 2.82% considered tolerability merely fair (Table 5). Adverse events (minor allergy) were rated by 2.82% of respondents.
Physician-reported Tolerability.
Prescribing intentions showed that nearly half of the respondents (49.30%) anticipated routinely switching suitable patients to Trelagliptin, while a small number of respondents rated that they would rarely (7.04%) or never (1.41%) consider switching existing therapy to Trelagliptin (Figure 1C). The most frequently reported factor influencing the decision to prescribe Trelagliptin was dosing convenience, as identified by more than two-thirds of respondents (67.61%). Other key considerations included alignment with patient lifestyle (36.62%), cost implications (32.39%), and perceived tolerability (22.54%). Only a small proportion of respondents (4.23%) identified company-provided support as an influencing variable (Figure 1D).
Discussion
This survey provides exploratory insights into prescribing patterns and perceptions of Trelagliptin in routine clinical care. The majority of respondents rated poor adherence to daily therapy as the primary reason for selecting Trelagliptin. Physicians perceived improved adherence and good glycaemic control following initiation of Trelagliptin therapy. Respondents perceived that, when compared with other DPP-4 inhibitors, Trelagliptin was superior in convenience and adherence, and comparable or better in cost-effectiveness and tolerability. These findings reflect physician attitudes and perceptions, not objective clinical outcome data.
Nearly two-thirds of respondents (64.79%) highlighted a reduction in pill burden as a critical consideration in diabetic patients who are receiving multiple medications. Respondents primarily considered patients with poor adherence to daily medications, working professionals, and elderly individuals with polypharmacy as suitable candidates for Trelagliptin therapy. These findings align with literature emphasising that poor adherence is prevalent in chronic conditions like Type 2 diabetes, often due to regimen complexity and frequent dosing. 14 Correspondingly, in the present survey substantial proportion (94.37%) of the respondents perceived that once-weekly dosing of the Trelagliptin was associated with improved adherence. These findings are consistent with evidence demonstrating that Trelagliptin improves adherence and reduces stigma associated with daily medication use, particularly in real-world populations such as those in Kenya.15,16
Regarding clinical efficacy, both glycaemic control and reduction in HbA1c were key parameters evaluated. The majority of respondents (90.14%) rated that Trelagliptin offered good to excellent glycaemic control, while only a small proportion of respondents indicated merely satisfactory control, and none reported poor glycaemic control. Furthermore, 94.37% respondents consider Trelagliptin as either highly or moderately effective in HbA1c reduction. These findings are consistent with the literature, highlighting the importance of glycaemic variability, beyond HbA1c reduction alone, is essential in T2DM management. DPP-4 inhibitors, including once-weekly Trelagliptin, have demonstrated efficacy in reducing glucose fluctuations while providing a convenient and well-tolerated option for long-term management. 17
Respondents further compared Trelagliptin therapy with other DPP-4 inhibitors currently used in clinical practice. While most respondents rated comparable results in glycaemic control, superior in adherence and cost-effectiveness. Clinical evidence supports that once-weekly Trelagliptin achieves sustained DPP-4 inhibition comparable to Alogliptin 12 or Vildagliptin, 13 with favourable long-term safety. It is convenient that weekly dosing likely improves adherence and patient satisfaction, without compromising glycaemic outcomes. 11
Approximately half of the respondents (49.30%) indicated a preference for switching eligible patients from daily DPP-4 inhibitors to a once-weekly regimen. In clinical practice, Trelagliptin is perceived to be suitable to use as a switchover therapy from agents such as Sitagliptin, maintaining treatment effectiveness and demonstrating favourable short- and long-term outcomes both as monotherapy and in combination therapy. 14
The majority of respondents (97.18%) predominantly rated tolerability as either good or excellent. Only a small proportion reported adverse events, primarily mild allergic reactions. Clinical trials reported similarly low incidences of mild adverse effects, comparable to daily DPP-4 inhibitors such as Vildagliptin. 13 The combination of efficacy, safety, and once-weekly dosing highlights Trelagliptin’s potential to improve adherence, patient satisfaction, and long-term glycaemic outcomes.17,18
Dosing convenience emerged as the most influential factor of prescribing decisions, as rated by a substantial proportion of respondents (67.61%). Patient lifestyle and cost were also frequently cited considerations (36.62% and 32.29%, respectively), whereas tolerability was noted as a comparatively less influential factor by 22.54% of respondents. Once-weekly dosing aligns with these priorities, providing a practical, patient-centred option that balances efficacy, safety, and convenience in routine care.
This study has certain limitations that warrant consideration. The relatively small sample size (n = 71) and use of convenience sampling limit representativeness and increase the risk of selection bias, restricting generalizability across India. Outcomes were based solely on physician perceptions without objective patient-level data (e.g., HbA1c values, adherence measures, prescription records, or adverse event reports); therefore, findings reflect perceived rather than actual clinical efficacy or safety. The author-developed questionnaire was not formally validated or pilot-tested, which may affect reliability and internal consistency. Potential recall and social desirability biases, as well as the influence of pharmaceutical promotion or drug availability, may have affected responses. Additionally, unmeasured regional variations in practice patterns, affordability, and medication access could have influenced results. As the survey was conducted exclusively among physicians in India, the findings may not be generalisable to other healthcare systems. International comparative studies would enhance external validity and provide broader insight.
Conclusion
This exploratory survey captures physician perceptions of once-weekly Trelagliptin as a valuable option in the management of T2DM, with respondents perceiving favourable efficacy, tolerability, and adherence outcomes. Physicians perceived it as particularly beneficial for patients with poor medication compliance or polypharmacy. The perceived advantages in adherence, convenience, and cost-effectiveness over other DPP-4 inhibitors suggest its potential role as a practical, patient-centred therapeutic option in routine clinical practice. It must be emphasised that these findings are based exclusively on physician-reported perceptions and do not constitute objective clinical evidence of efficacy, safety, or adherence improvement. Future prospective and longitudinal studies incorporating patient-reported outcomes and objective adherence measures are warranted to further evaluate the long-term clinical benefits of once-weekly DPP-4 inhibitor therapy.
Footnotes
Acknowledgements
None.
Authors’ Contributions
Harsh J. Shah: Conception, design, data collection, data validation, data analysis, manuscript review and guarantor.
Jacky K. Pariyani: Data analysis and manuscript review.
Kalyani V. Shinde: Drafting/revising content and manuscript review.
Dharmil P. Pandya: Data validation, data analysis, drafting/revising content, manuscript preparation and manuscript review.
Bhakti A. Dave: Drafting/revising content and manuscript review.
Data Availability Statement
The datasets generated and/or analysed during the current study are not publicly available; however, they may be obtained from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Not applicable.
