Abstract
Glucose-lowering treatment options for type 2 diabetes mellitus patients with chronic kidney disease are limited. We evaluated the potential for linagliptin in combination with insulin in type 2 diabetes mellitus patients with mild-to-severe renal impairment. Data for participants in two phase 3 trials with linagliptin who were receiving insulin were analysed separately (n = 811). Placebo-adjusted mean HbA1c changes from baseline were −0.59% (mild renal impairment) and −0.69% (moderate renal impairment) after 24 weeks and −0.43% (severe renal impairment) after 12 weeks. Drug-related adverse events with linagliptin were similar to placebo (mild renal impairment: 19.9% vs 26.5%; moderate renal impairment: 22.0% vs 25.0%; severe renal impairment: 46.3% vs 43.6%, respectively). Frequencies of hypoglycaemia in patients with mild, moderate and severe renal impairment were 34.9%, 35.6% and 66.7% with linagliptin and 37.5%, 39.7% and 49.1% with placebo, respectively. Episodes of severe hypoglycaemia were low (⩽5.6%). Adding linagliptin to insulin in type 2 diabetes mellitus patients with chronic kidney disease improved glucose control and was well tolerated.
Introduction
Type 2 diabetes mellitus (T2DM) is the leading cause of chronic kidney disease (CKD). 1 A cross-sectional analysis from the fourth National Health and Nutrition Examination Survey further showed that a significant proportion of patients with T2DM were already at advanced stages of CKD. 2 Thus, 11% of patients with T2DM had CKD stage 2 [glomerular filtration rate (GFR) 60 to <90 mL/min/1.73 m2], 18% had CKD stage 3 (GFR 30 to <60 mL/min/1.73 m2) and 2% had CKD stages 4–5 (GFR <30 mL/min/1.73 m2). This is noteworthy because individuals with advanced stages of CKD are generally those at highest risk of progressing to end-stage renal disease (ESRD). Consequently, the prevalence of ESRD in the population with diabetes is continually rising and is estimated to be as high as 716 per million population. 3
Insulin therapy in combination with oral antihyperglycaemic drugs (OADs) is frequently used by patients with T2DM as their glycaemic control deteriorates, which often occurs in parallel with CKD progression. 2 However, commonly used OADs are either contraindicated or should be used at reduced doses in individuals with renal impairment (RI). Potential adverse drug effects include increased risk of developing lactic acidosis with metformin and risk of fluid retention, bone fractures, and congestive heart failure with thiazolidinediones.4,5 In addition, there is limited or no therapeutic experience for the use of glucagon-like peptide-1 receptor agonists in patients with moderate and severe RI.6–8 Importantly, patients with T2DM and CKD also have an increased risk of hypoglycaemia, which is commonly due to decreased clearance of insulinotropic agents (such as sulphonylureas or insulin) and/or their metabolites. Hypoglycaemia is also a concern with insulin therapy regardless of renal function. 9 These safety concerns may partly explain why only 50%–60% of patients with T2DM achieve glycaemic targets even when a structured insulin titration regimen is systematically followed,10,11 confounding the goal of slowing the progression of renal disease through improved glycaemic control. Therefore, additional treatment options suitable for patients with T2DM and CKD and which do not increase the risk of hypoglycaemia could add value to the current treatment armamentarium for diabetes in this challenging population.
Linagliptin is an oral dipeptidyl peptidase (DPP)-4 inhibitor indicated for the treatment of T2DM in a 5-mg once-daily dose as monotherapy or in combination with other OADs 12 or insulin. Linagliptin is predominantly excreted via the entero-hepatic system 13 and can therefore be used without dose adjustment in T2DM patients with RI, 14 including patients with ESRD. Under steady-state conditions, increases in linagliptin exposure in patients with mild, moderate and severe RI were less than twofold relative to patients with normal renal function. Moreover, due to its tight binding to the DPP-4 enzyme, linagliptin is not expected to be eliminated to a therapeutically significant degree by haemodialysis or peritoneal dialysis, and dosing can thus occur independent of those procedures. 14 Linagliptin is an effective glucose-lowering treatment when administered as monotherapy and in addition to common glucose-lowering therapies (including insulin) and is associated with a very low risk of hypoglycaemia and no weight gain.15–18 Recently published trials have demonstrated the efficacy and safety of linagliptin in patients receiving basal insulin 19 and in those with severe RI. 20
Safely achieving glucose targets without increasing the risk of hypoglycaemia is still considered an unmet medical need in patients with T2DM and CKD. The aim of this retrospective analysis was therefore to investigate the efficacy and safety of linagliptin 5 mg in combination with insulin-based regimens in patients with T2DM and renal disease (CKD stages 2–5).
Methods
Study design
This study was a retrospective analysis of data from individuals with T2DM who participated in either of two phase 3 prospective, randomized, placebo-controlled trials. In both trials, participants who met the eligibility criteria were randomized (1:1) to receive double-blind treatment with either linagliptin (5 mg/day) or placebo in addition to their glucose-lowering background therapy for ⩾52 weeks (Table 1). Study 1 (NCT00954447) included patients with T2DM who were receiving stable treatment with basal insulin, alone or in combination with metformin and/or pioglitazone. From Study 1, only patients with prevalent CKD were included in the current analysis. Study 2 (NCT00800683) included T2DM patients with severe RI (CKD stages 4–5) who were receiving treatment with insulin (short- and long-acting), sulphonylurea, glinides, piog-litazone, or α-glucosidase inhibitors. From Study 2, only patients with previous background insulin treatment were included in the current analysis. The dose of insulin remained stable during the first 24 weeks of treatment in Study 1 and the first 12 weeks of treatment in Study 2, after which adjustments were permitted.
Phase 3 trials of linagliptin included in the analysis.
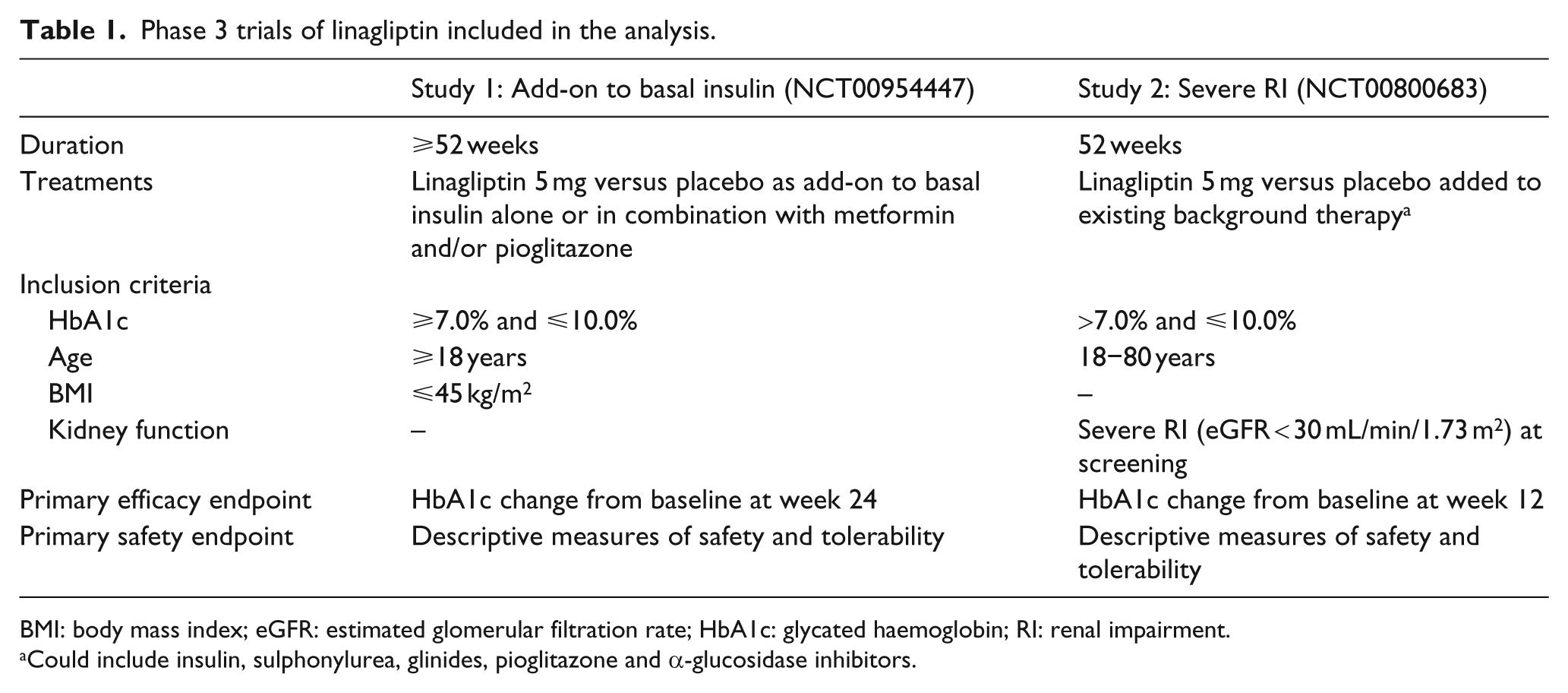
BMI: body mass index; eGFR: estimated glomerular filtration rate; HbA1c: glycated haemoglobin; RI: renal impairment.
Could include insulin, sulphonylurea, glinides, pioglitazone and α-glucosidase inhibitors.
CKD was determined either at baseline (Study 1) or screening (Study 2) by estimated GFR (eGFR) using the Modification of Diet in Renal Disease (MDRD) study group equation and expressed as stages of CKD. Study 1 included patients with mild RI (eGFR: 60 to <90 mL/min/1.73 m2; CKD stage 2) and moderate RI (eGFR: 30 to <60 mL/min/1.73 m2; CKD stage 3). Study 2 included patients with severe RI [eGFR: <30 mL/min/1.73 m2 (while not receiving chronic dialysis); CKD stages 4–5]. The nominal ranges for eGFR are equivalent to the National Kidney Foundation Kidney Disease Outcomes Quality Initiative (KDOQI) guidelines for stages of CKD. Mild RI is equivalent to the KDOQI Stage 2, moderate RI is equivalent to Stage 3 and severe RI is equivalent to Stages 4–5. 21 The design and methodology of these trials have previously been described in detail.19,20
Endpoints
In these trials, the primary efficacy endpoint was the glycated haemoglobin (HbA1c) change from baseline after 24 weeks (Study 1) or 12 weeks (Study 2). Safety and tolerability were assessed by the frequency and intensity of adverse events (AEs), including episodes of hypoglycaemia. AEs of special interest in this CKD population included new onset or worsening of hypertension or heart failure. All AEs were coded using the Medical Dictionary for Drug Regulatory Affairs (MedDRA) version 14.0.
Statistical methods
The two studies were analysed separately. Efficacy analy-ses were performed on the full analysis set, which comprised all randomized patients who were treated with ⩾1 dose of study drug and had a baseline and ⩾1 on-treatment HbA1c measurement. Changes in HbA1c were evaluated using an analysis of covariance model in both trials. In Study 1, the model included continuous baseline HbA1c, concomitant OADs, treatment, subgroup (RI), and treatment × subgroup. In Study 2, the model included continuous baseline HbA1c, continuous baseline creatinine clearance, treatment, and concomitant OADs. Similar models were used to evaluate changes in fasting plasma glucose (FPG), with the addition of baseline FPG. Missing data were imputed using a last observation carried forward approach. Efficacy measurements after start of rescue were replaced by missing values, with the exception of change in insulin background therapy. Safety analyses were performed on the treated set, which comprised all randomized patients who received ⩾1 dose of study drug. The analyses were summarized using descriptive statistics.
Results
Patient disposition and demographics
In Study 1, 575 and 127 patients inadequately controlled on basal insulin (HbA1c ⩾7.0% and ⩽10.0%) had mild or moderate RI at baseline, respectively. In Study 2, 92 patients with inadequate glycaemic control despite insulin background therapy (HbA1c >7.0% and ⩽10.0%) had severe RI at baseline. Few patients with moderate RI at baseline were included in Study 2 (n = 17) as their respective eGFR values were consistent with severe RI at screening (Figure 1).
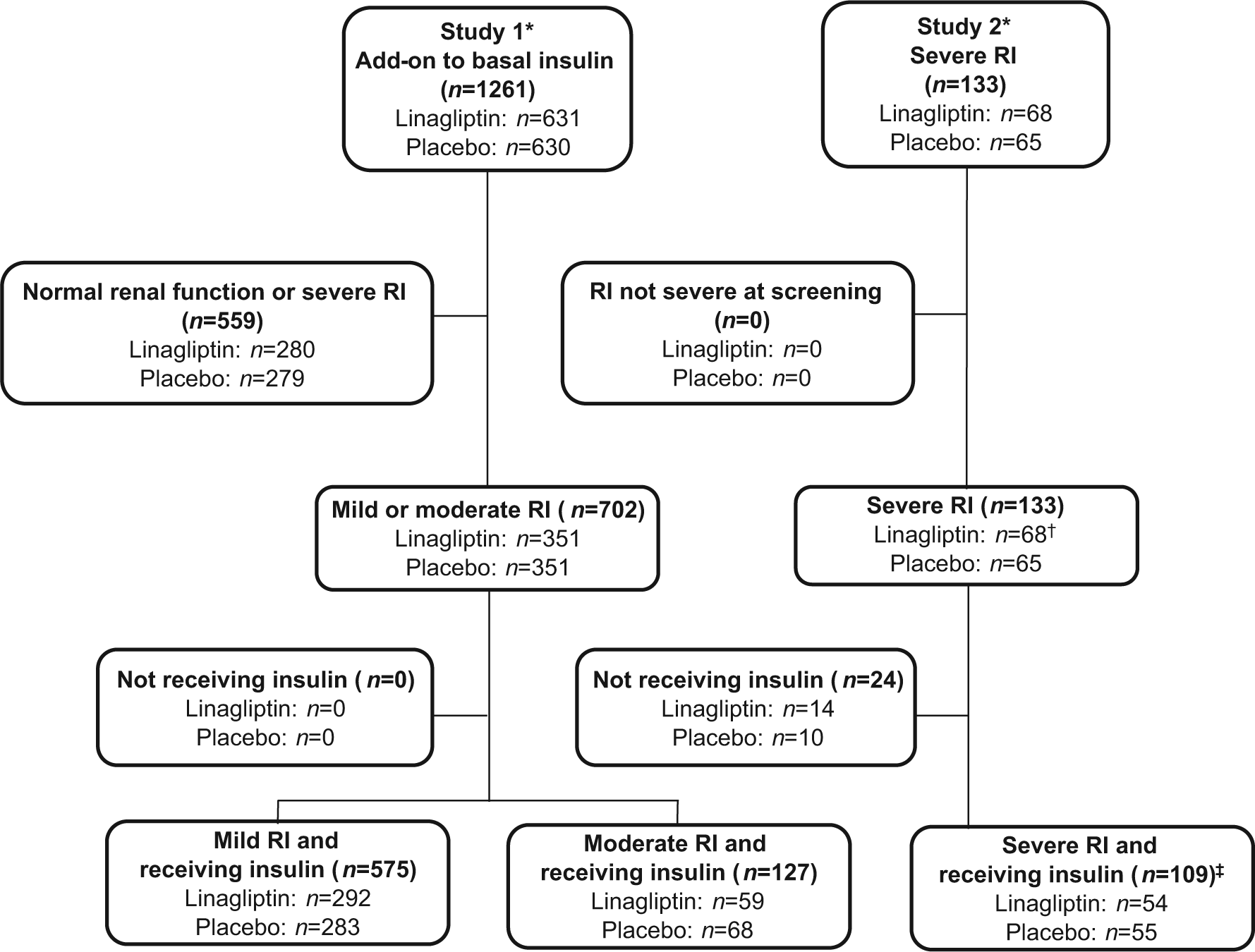
Patient disposition (treated set). eGFR: estimated glomerular filtration rate; RI: renal impairment.
Patient demographics and baseline clinical characteristics are presented in Table 2. Vascular complications were prevalent in this population with 23.5%, 39.4% and 43.1% of patients with mild, moderate and severe RI, respectively, having evidence of coronary artery disease. Hypertension was the most common cardiovascular risk factor and was prevalent in 83.5%, 91.3% and 97.2% of patients with mild, moderate and severe RI, respectively.
Patient demographics and clinical characteristics at baseline.
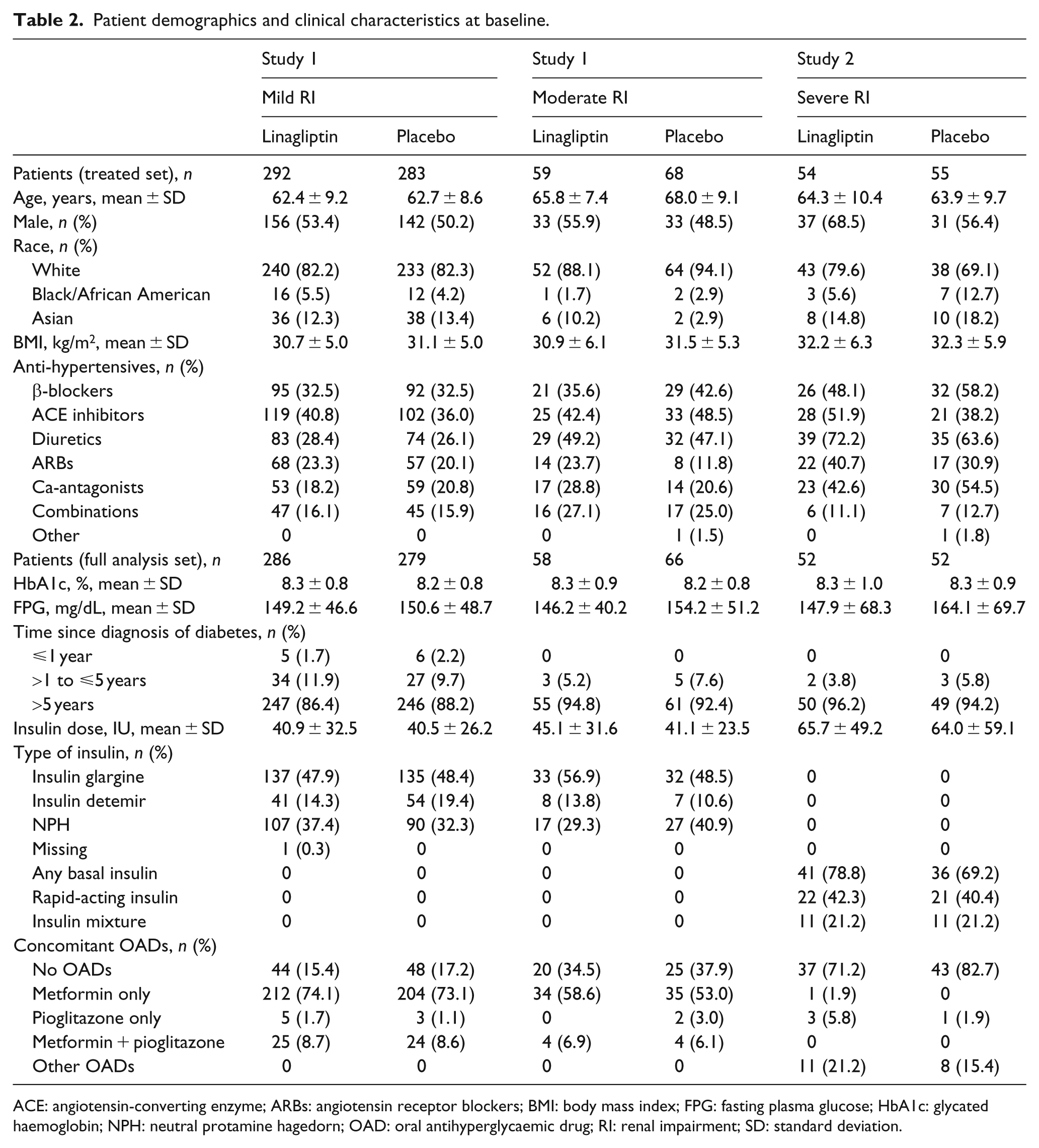
ACE: angiotensin-converting enzyme; ARBs: angiotensin receptor blockers; BMI: body mass index; FPG: fasting plasma glucose; HbA1c: glycated haemoglobin; NPH: neutral protamine hagedorn; OAD: oral antihyperglycaemic drug; RI: renal impairment; SD: standard deviation.
Efficacy
Both studies showed greater reductions in HbA1c after 24 weeks compared with placebo (Figure 2). At week 24, the placebo-adjusted mean changes in HbA1c with linagliptin were −0.59% (95% confidence interval (CI): −0.73, −0.45; p < 0.001) and −0.69% (95% CI: −0.98, −0.40; p < 0.001) in patients with mild and moderate RI, respectively (Study 1). At week 12, the placebo-adjusted mean change in HbA1c with linagliptin was −0.43% (95% CI: −0.75, −0.11; p = 0.008) in patients with severe RI (Study 2). Mean changes from baseline in HbA1c at week 52 were consistent with the primary efficacy endpoint and showed a sustained improvement in glycaemic control with linagliptin across all CKD groups (Figure 2).
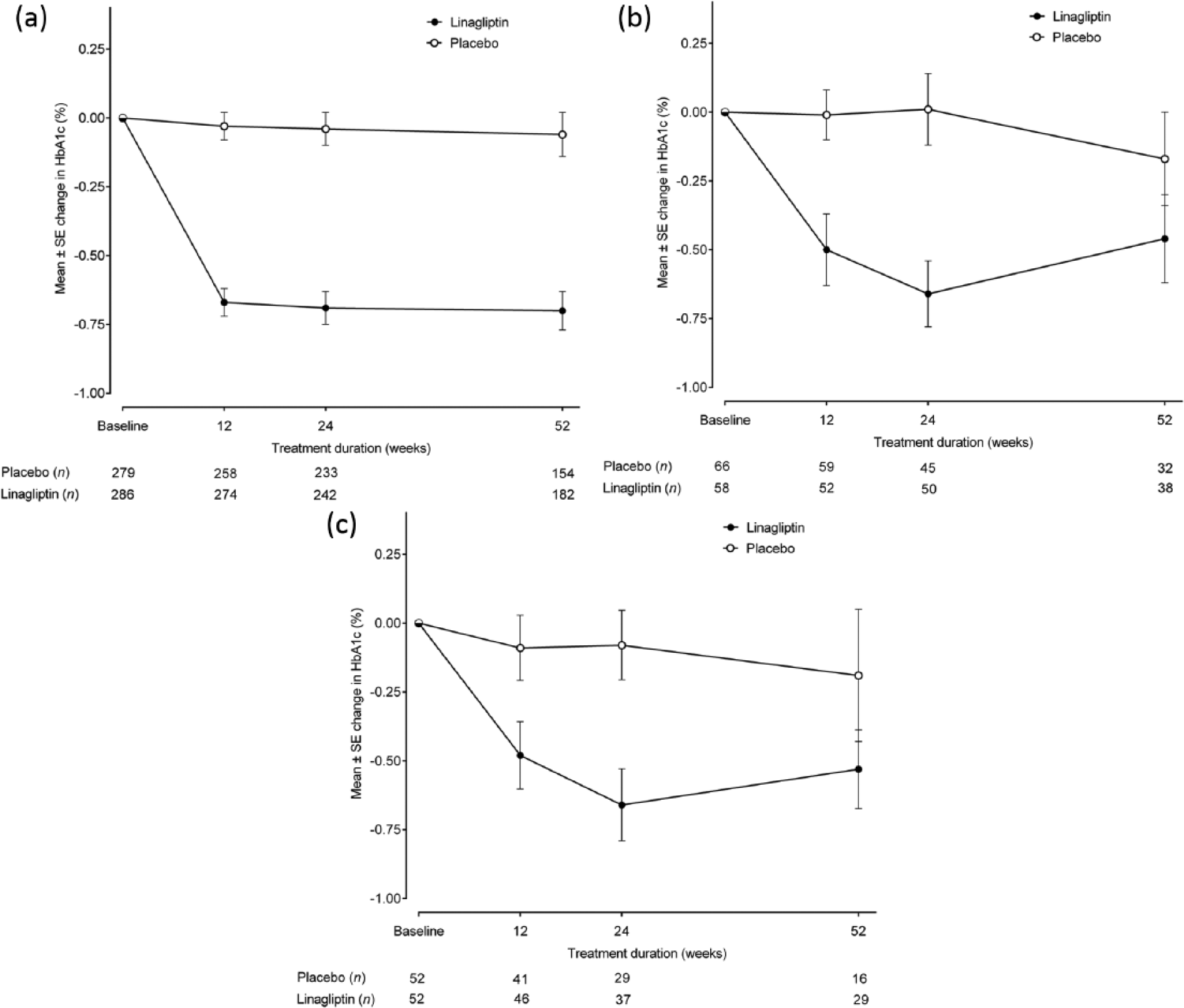
Mean HbA1c change from baseline over time up to 52 weeks for (a) patients with mild RI, (b) patients with moderate RI and (c) patients with severe RI (full analysis set, observed cases).
Change in insulin background therapy
In Study 1, the mean change from baseline in basal insulin dose increased to a lesser extent with linagliptin compared with placebo at week 52 (mild RI: linagliptin +1.51 IU (95% CI: 0.55, 2.47), placebo +2.03 IU (95% CI: 1.05, 3.0); moderate RI: linagliptin +1.69 IU (95% CI: −0.21, 3.59), placebo +3.12 IU (95% CI: 0.53, 5.71)). In Study 2, the mean change from baseline in insulin dose at week 52 was −6.15 IU (95% CI: −15.32, 3.02) for linagliptin and −0.31 IU (95% CI: −4.41, 3.79) for placebo.
Safety and tolerability
The overall incidence of AEs and drug-related AEs with linagliptin were similar to placebo (Table 3). Regarding AEs of special interest, the frequency of new or worsening hypertension was comparable between the linagliptin and placebo groups in patients with mild and severe RI. Among patients with moderate RI, more patients in the placebo group experienced hypertension. In addition, the incidence of heart failure was similarly low for both linagliptin and placebo groups, across renal stages (Table 3). In Study 1, renal AEs, determined by narrow standardized MedDRA queries (SMQ) occurred in low percentages (Figure 3). Acute renal failure was a rare event and increased numerically by baseline CKD status. Respective incidence rates in patients with mild, moderate and severe RI were 0.3%, 0.4% and 0.0% with linagliptin and 1.5%, 3.7% and 7.3% with placebo.
Safety of linagliptin in T2DM patients with CKD (treated set).
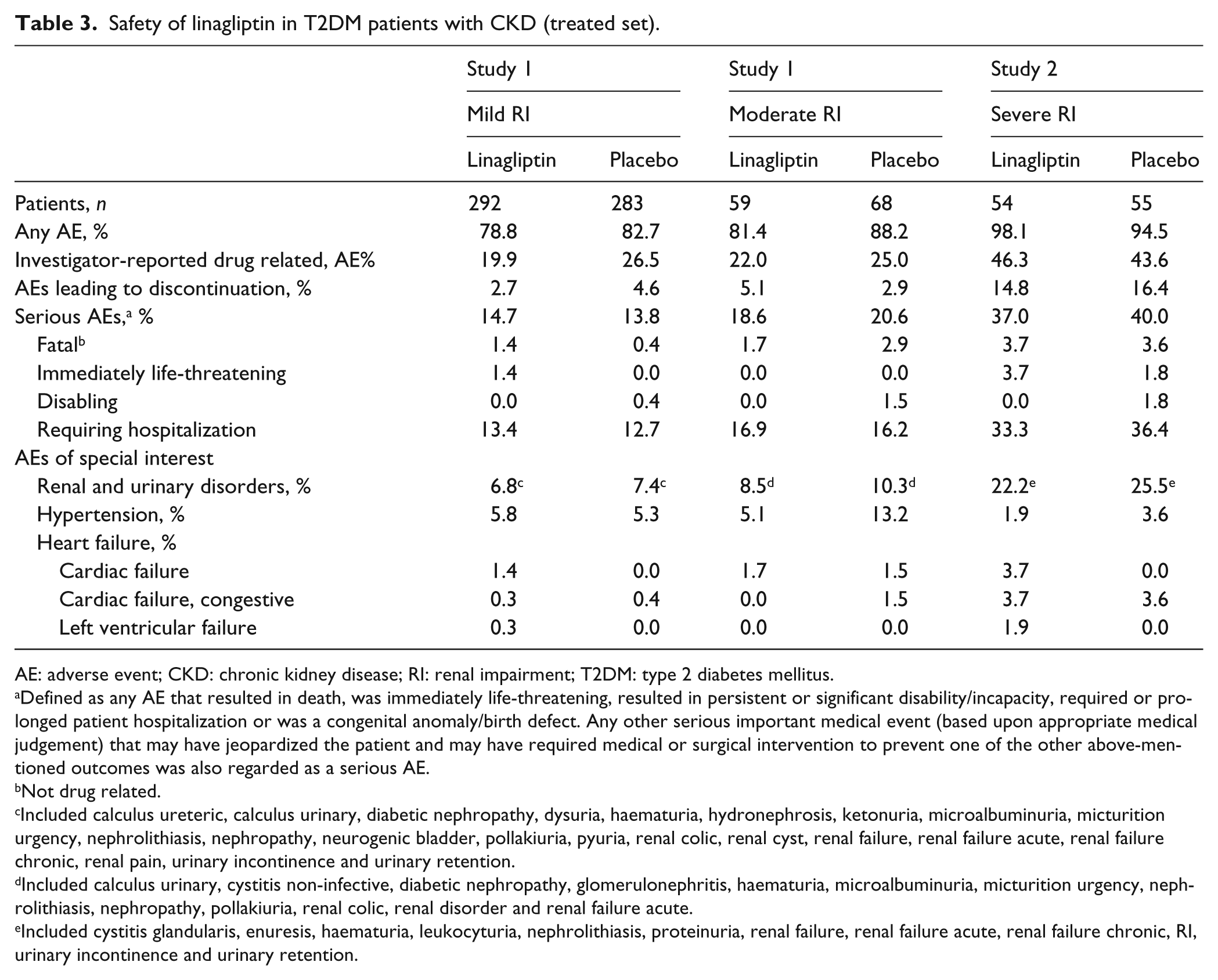
AE: adverse event; CKD: chronic kidney disease; RI: renal impairment; T2DM: type 2 diabetes mellitus.
Defined as any AE that resulted in death, was immediately life-threatening, resulted in persistent or significant disability/incapacity, required or prolonged patient hospitalization or was a congenital anomaly/birth defect. Any other serious important medical event (based upon appropriate medical judgement) that may have jeopardized the patient and may have required medical or surgical intervention to prevent one of the other above-mentioned outcomes was also regarded as a serious AE.
Not drug related.
Included calculus ureteric, calculus urinary, diabetic nephropathy, dysuria, haematuria, hydronephrosis, ketonuria, microalbuminuria, micturition urgency, nephrolithiasis, nephropathy, neurogenic bladder, pollakiuria, pyuria, renal colic, renal cyst, renal failure, renal failure acute, renal failure chronic, renal pain, urinary incontinence and urinary retention.
Included calculus urinary, cystitis non-infective, diabetic nephropathy, glomerulonephritis, haematuria, microalbuminuria, micturition urgency, nephrolithiasis, nephropathy, pollakiuria, renal colic, renal disorder and renal failure acute.
Included cystitis glandularis, enuresis, haematuria, leukocyturia, nephrolithiasis, proteinuria, renal failure, renal failure acute, renal failure chronic, RI, urinary incontinence and urinary retention.
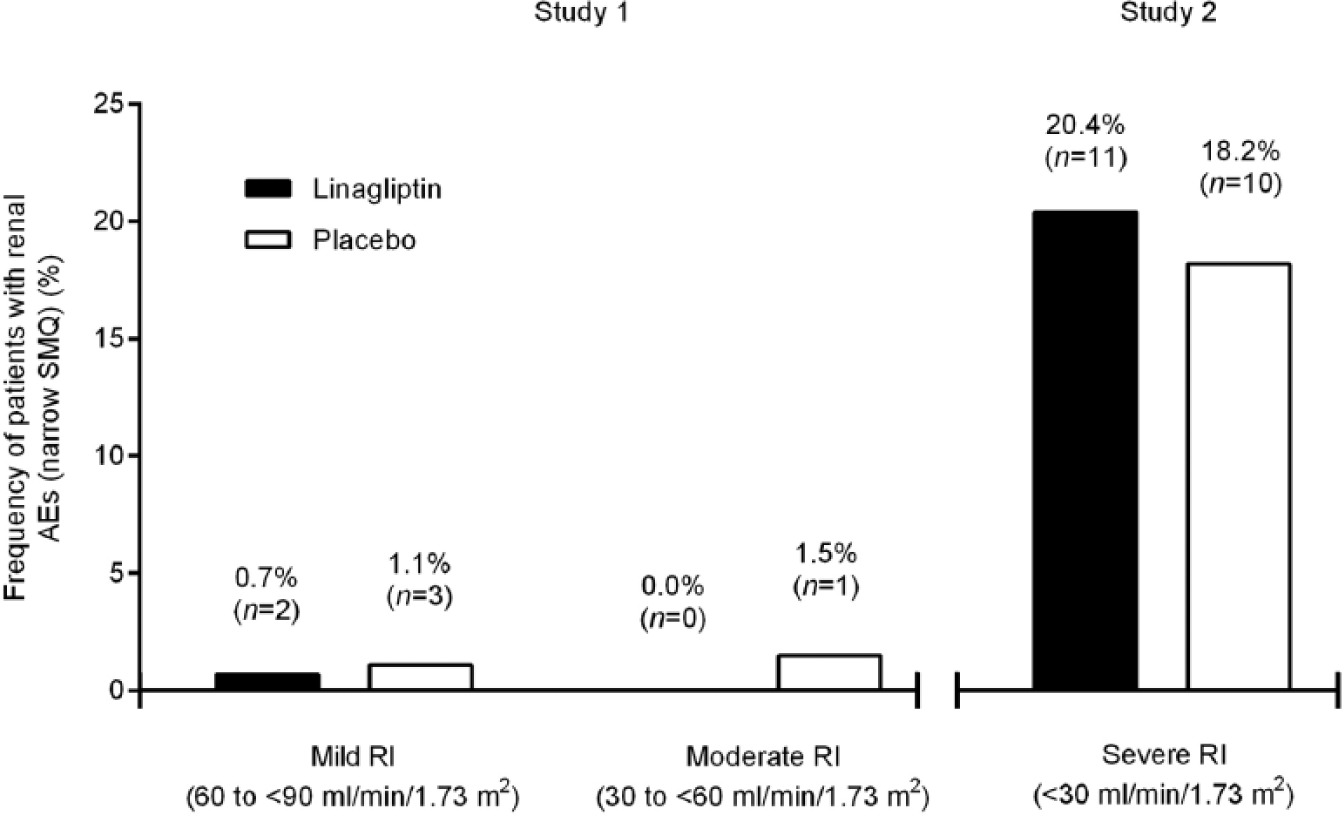
Incidence of renal AEsa in Study 1 and Study 2 as determined by narrow SMQ during the study periods (treated set).
Investigator-reported hypoglycaemic events (any asymptomatic or symptomatic hypoglycaemia reported as AEs) were lower with linagliptin compared with placebo in Study 1 (mild RI: 34.9% vs 37.5%; moderate RI: 35.6% vs 39.7%, respectively). In Study 2, 66.7% and 49.1% of linagliptin and placebo patients experienced investigator-reported hypoglycaemic events, respectively. Importantly, however, the incidence of severe hypoglycaemic events (requiring the assistance of another person to actively administer carbohydrate, glucagon or other resuscitative actions) was similar between the linagliptin and placebo groups across renal stages (Figure 4).
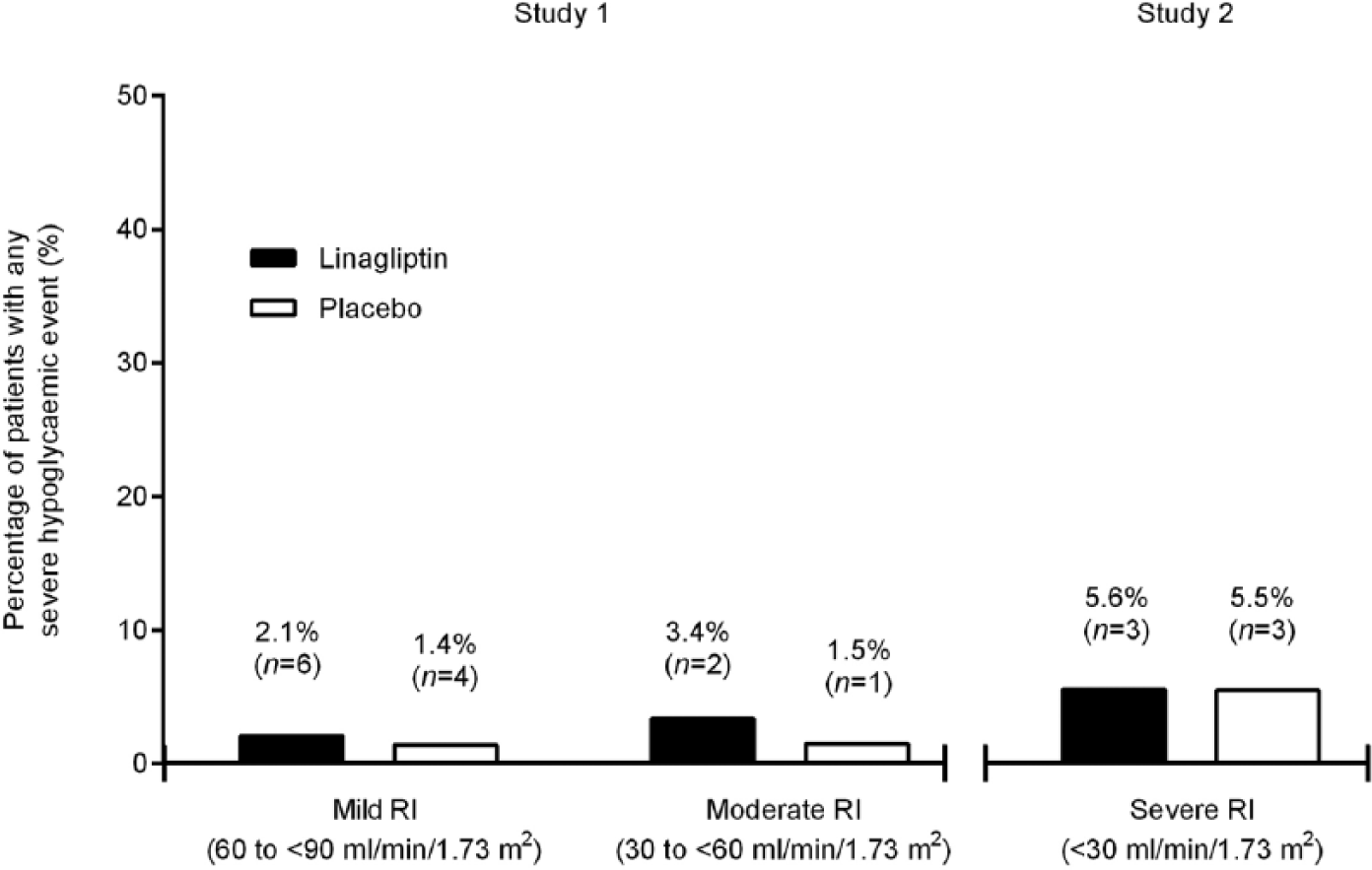
Patients with any severe hypoglycaemic AEa in Study 1 and Study 2 (treated set). AE: adverse event; RI: renal impairment.
There were no significant changes from baseline in mean body weight in either study groups. In patients treated with linagliptin, the placebo-adjusted mean changes in body weight were −0.35 kg (95% CI: −0.82, 0.12; p = 0.1452) and 0.21 kg (95% CI: −0.84, 1.26; p = 0.6912) in patients with mild and moderate RI, respectively, at week 24. At week 12, the placebo-adjusted mean change in body weight was −0.22 kg (95% CI: −1.10, 0.65; p = 0.6106) in patients with severe RI. Mean changes from baseline in body weight at week 52 were also small and showed no significant between-group differences.
Discussion
Control of hyperglycaemia in patients with T2DM and CKD can be challenging as many available OADs are either contraindicated or have treatment limitations. In order to avoid some of the complexity associated with OADs, physicians often prefer to prescribe insulin exclusively as first-line therapy in this population. However, even 40%–50% of patients with normal renal function fail to reach glycaemic targets despite initiation of insulin treatment, with hypoglycaemia being a major clinical concern.10,11 Therefore, alternative oral treatment options, ideally suitable for combination with insulin-based regimens in patients with T2DM and CKD, are needed. Such agents should effectively lower glucose levels without increasing risk of hypoglycaemia. In addition, a benign renal safety profile, low propensity for relevant drug–drug interactions, and a low potential for clinically relevant drug accumulation under conditions of impaired renal function need to be considered. The novelty of the current analysis is based on its clinical value for health-care providers to improve informed treatment decision-making in patients with T2DM and CKD. Our results are based on the largest population of T2DM patients with CKD studied so far for novel glucose-lowering agents in combination with insulin. The results support a positive benefit–risk profile for linagliptin in combination with insulin. Drug treatment was overall well tolerated and provided clinically meaningful improvements in glycaemic control without increasing the risk of severe hypoglycaemia or body weight gain. Linagliptin had a benign renal safety profile with renal function remaining stable throughout the 52-week study periods.
The efficacy results of this analysis are consistent with previous studies, in which linagliptin significantly lowered HbA1c levels in patients with T2DM and normal renal function in combination with common OADs.15–18 The placebo-adjusted mean changes from baseline in HbA1c in the present population are also comparable with glycaemic improvements reported in trials of other DPP-4 inhibitors in patients with RI.22,23 The present analysis, however, is the first to specifically assess the combination of a DPP-4 inhibitor with exogenous insulin in patients with T2DM and CKD. We therefore believe that this analysis has a clinically relevant application because most patients with CKD require insulin therapy. 24
A positive benefit–risk ratio with linagliptin has recently been reported from a trial in elderly patients with T2DM (aged >70 years), a population that commonly experiences declining renal function. 25 In the present analysis, the mean age of individuals was >60 years and the majority had long-standing diabetes, both of which are concomitant risk factors for progression of CKD. Linagliptin was well tolerated, and no new clinically relevant safety signals or drug–drug interactions arose. This is particularly important in light of previously reported potential interactions between the DPP-4 inhibitor sitagliptin and high-dose acute angiotensin-converting enzyme (ACE) inhibition with enalapril. 26 In this trial, sitagliptin increased sympathetic nervous system activity and heart rate, and the blood pressure-lowering effect of enalapril was diminished. In our analysis, although 40%–52% of individuals across the linagliptin groups were treated with concomitant ACE inhibitors, the frequency of new or worsening hypertension was low. These observations add further safety evidence to a previous pooled analysis in which linagliptin significantly reduced albuminuria in patients with T2DM and renal dysfunction (all being treated with renin angiotensin aldosterone system inhibitors). 27
In the current analysis, the addition of linagliptin to basal insulin was associated with a low frequency of severe hypoglycaemia, which was similar to placebo. Reducing hyperglycaemia with low risk of severe hypoglycaemic events would be a clinical advantage for T2DM patients with CKD. The low risk of severe hypoglycaemia with linagliptin observed in our analysis, even when given in combination with insulin-based regimens, may partly reflect incretin-enhanced regulation of counter-regulative glucagon secretion from pancreatic α-cells under conditions of DPP-4 inhibition. 28 Additionally, treatment with linagliptin in patients with mild, moderate and severe RI did not significantly increase body weight. This finding is important considering that treatment with insulin has been associated with significant weight gain. 29
An important limitation of this study is that, although all efficacy and safety endpoints reported are in accordance with the respective protocol definitions of the two trials, analyses specifically addressing T2DM patients with CKD on insulin background therapy were retrospective in nature. It should also be noted that Study 1 and Study 2 were analysed separately. This was based on different renal function statuses between the two studies at baseline. In addition, different protocol-defined time points for the evaluation of the primary efficacy endpoint (24 and 12 weeks for Study 1 and Study 2, respectively) had to be considered. The large sample size, however, did allow for thorough and comprehensive safety evaluations across all relevant categories of advanced CKD, which is considered to be a strength of the available clinical data.
In conclusion, patients with T2DM and CKD represent a highly vulnerable patient population who often do not reach glycaemic targets even when treated with insulin. Adding linagliptin to short- and long-acting insulin regimens in patients with CKD stages 2–5 achieved clinically meaningful improvements in glycaemic control and was well tolerated. Risk of severe hypoglycaemia did not increase with linagliptin and, overall, treatment was not associated with increased renal-related side effects.
Footnotes
Acknowledgements
The authors were fully responsible for all content and editorial decisions, were involved at all stages of manuscript development, and have approved the final version. Medical writing assistance, supported financially by Boehringer Ingelheim, was provided by Claire Stevens of Envision Scientific Solutions during the preparation of this manuscript.
Declaration of conflicting interests
Janet B McGill has served as a speaker and consultant for Boehringer Ingelheim and Merck. Hannele Yki-Jarvinen has received research support from Lilly-Amylin and Boehringer Ingelheim. She has also received honoraria for consultancy for Sanofi, Novartis, Astra-Zeneca, MSD, Bristol-Myers Squibb, and for lectures at meetings sponsored by Sanofi and MSD. Susanne Crowe, Hans-Juergen Woerle, and Maximilian von Eynatten are employees of Boehringer Ingelheim. No other potential conflicts of interest relevant to this article were reported.
Funding
This study was supported by Boehringer Ingelheim, the manufacturer of linagliptin.
