Abstract
Aim & Background:
Lifestyle variables such as dietary habits, physical activity, sleep, and screen time are core to the prevention and control of diabetes. In light of the distinct sociocultural scenario prevalent in the Indian community and the rising cases of metabolic disorders like diabetes, a pressing need exists for a valid and reliable lifestyle measurement tool that takes into account the local culture and psychometric properties. The aim of this research was to validate the content and reliability of the Research Society for the Study of Diabetes in India (RSSDI) Lifestyle Questionnaire.
Methods:
The method of expert review was applied to evaluate the content validity of the questionnaire with 44 items covering a wide aspect of lifestyle questions. Sixty-one physicians with expertise in diabetes care were asked to rate on a four-point ordinal scale. The item-level and scale-level content validity indices (S-CVI) were estimated, and qualitative comments on clarity and relevance to culture were checked, while Cronbach’s alpha values were determined.
Results:
All items had high content validity, with I-CVIs ranging from 0.98 to 1.00. Overall S-CVIAve was 0.94, which is a sign of having high S-CVI. Due to the qualitative feedback, only minor changes in wording were made. There were no items deleted in the qualitative feedback. Cronbach’s alpha value was 0.96, which signified high reliability.
Conclusion:
The RSSDI Lifestyle Questionnaire has adequate content validity and high internal consistency. It is a valid and useful tool to measure lifestyle parameters among diabetics and those at risk of diabetes, according to Indian standards.
Keywords
Introduction
The interplay between lifestyle factors, such as diet, sleep quality, screen time, and physical activity, has garnered significant attention in public health research over the past few decades. These lifestyle components are critical determinants of health outcomes, influencing the prevalence and management of chronic conditions, such as obesity, diabetes, and cardiovascular diseases. 1 Moreover, contemporary challenges, such as the global COVID-19 pandemic, have further emphasized the importance of understanding lifestyle modifications and their implications for health. In India, with its unique sociocultural context and rising concerns about noncommunicable diseases, 2 the development of a validated lifestyle questionnaire is essential for effective public health interventions targeting diet, sleep, screen time, and physical activity.
Importance of Lifestyle Factors in Health Research
Diet
Dietary habits are integral to overall health, with evidence linking poor nutrition to a range of health conditions, including Type 2 diabetes and cardiovascular diseases. A systematic review by Cappuccio et al. highlighted how dietary patterns can influence the incidence of Type 2 diabetes, particularly in vulnerable populations. 3 Moreover, a community-based study in India indicated that lifestyle factors, including diet, significantly contribute to the risk of diabetes. 4 Therefore, the establishment of a reliable questionnaire that accurately assesses dietary habits is foundational to understanding the nutritional implications of health.
Sleep
Sleep quality and duration are associated with various health outcomes. Research has shown that sleep disturbances can lead to adverse metabolic effects, thereby increasing the risk of chronic conditions such as diabetes. 3 Sleep serves not only as a restorative period for the body but also affects mental health and cognitive function. 5 The evaluation of sleep patterns using validated questionnaires can provide insights into how sleep influences lifestyle-related health risks, with emerging evidence demonstrating the interconnectedness of sleep, diet, and physical activity.
Screen Time
The increasing prevalence of digital devices has led to rising concerns regarding screen time and its impact on health status. Prolonged screen time is associated with a sedentary lifestyle, leading to adverse health outcomes, such as obesity and reduced physical fitness. 6 Studies have found that excessive screen time, particularly among children and adolescents, correlates with unhealthy eating habits and decreased physical activity, further complicating public health efforts. 7 By incorporating questions about screen time in lifestyle questionnaires, researchers can better understand the relationship between time spent on digital devices and overall health behaviors.
Physical Activity
Physical activity is a cornerstone of preventive health behavior, as it helps mitigate the risks associated with sedentary lifestyles. Regular physical activity is associated with improved cardiovascular health, reduced obesity rates, and better mental well-being. 8 Research has shown that insufficient physical activity is often coupled with unhealthy dietary patterns, highlighting the need for integrated assessment tools that evaluate both dietary intake and physical activity. 6 Validated questionnaires can accurately capture physical activity habits, providing valuable data for public health strategies aimed at fostering healthier lifestyles.
The Need for Validated Assessments
The significance of developing a tailored lifestyle questionnaire, specifically in India, cannot be overstated. Previous efforts to establish lifestyle assessment tools in India have often faced challenges regarding their cultural relevance and contextual applicability. 4 Hence, it is paramount to devise a comprehensive tool that resonates with the unique lifestyle patterns and health concerns ofthe Indian population. The TNRSSDI Lifestyle Questionnaire, which focuses on diet, sleep, screen use, and physical activity, addresses these unique needs.
Methodological Framework for Validation
The validation process for the TNRSSDI Lifestyle Questionnaire involved rigorous evaluation methods to ensure the tool’s accuracy and relevance. A critical component of this validation was assessing content validity, which ensured that each questionnaire item adequately reflected the construct being measured.
Content Validity Index
For the TNRSSDI questionnaire, we adopted the content validity index (CVI) methodology, which leverages expert insights to determine the relevance of the questionnaire items. Following the recommendations of Polit and Beck, we consulted a panel of 50 physicians specializing in fields relevant to lifestyle assessment. 9 Each item was rated for clarity and relevance, with the CVI calculated to quantify the overall validity of the questionnaire.9,10 A high CVI indicates that the questionnaire items are suitably drafted to reflect the intended constructs. 11 Notably, this structured approach ensured that the items were grounded in expert consensus, thereby enhancing the credibility of the questionnaire.
Reliability Testing
In addition to the content validity assessment, we performed reliability tests employing metrics such as Cronbach’s alpha to analyze internal consistency. 12 Studies highlight that a Cronbach’s alpha value of 0.70 or higher is generally acceptable for social science and healthcare-related instruments, ensuring that the items within the questionnaire are interrelated and consistently reflect the underlying concept.13,14 This multifaceted approach ensures that the TNRSSDI Lifestyle Questionnaire is not only valid in measuring relevant constructs but also reliable across different populations and settings.
Significance of Validation
The validation of the TNRSSDI Lifestyle Questionnaire serves several essential purposes. First, it bolsters the scientific rigor of future research efforts by providing an empirically grounded tool for measuring lifestyle factors. Second, it enables clinicians and public health officials to identify at-risk populations and implement appropriate interventions in a culturally sensitive manner. Finally, a validated instrument enhances researchers’ ability to assess the effectiveness of lifestyle interventions, paving the way for evidence-based approaches to public health that can mitigate the growing burden of lifestyle-related diseases in India and beyond.
Materials & Methods
Study Design
This methodological validation study aimed to establish the content validity of a structured lifestyle questionnaire developed under the auspices of the Research Society for the Study of Diabetes in India (RSSDI). The validation process adhered to standard recommendations for instrument development in clinical research. A physician expert review served as the primary validation step before conducting reliability assessments, ensuring the clinical relevance and conceptual adequacy of the questionnaire items.
Questionnaire Description and Domains
The RSSDI Lifestyle Questionnaire was designed to comprehensively assess lifestyle determinants influencing glycemic control and metabolic risk in adults with diabetes. The instrument comprises 44 items distributed across predefined domains that reflect key behavioral and clinical constructs pertinent to diabetes management. These domains include sociodemographic and clinical profiles, dietary practices, physical activity and sedentary behavior, screen time and sleep health, and social and substance use history. The sociodemographic and clinical profile domain captures age, sex, educational status, occupation, anthropometric measurements, duration of diabetes, co-morbid conditions, and glycemic parameters, including fasting blood sugar, postprandial blood sugar, and glycated hemoglobin levels. The dietary practices domain evaluates both the quality and frequency of food intake, encompassing fiber-rich foods, fermented foods, fruits, nuts, protein sources, carbohydrate load, processed foods, sugar consumption, cooking oils, portion size, and eating-out behavior. Physical activity and sedentary behavior items assess the frequency and duration of vigorous and moderate physical activity, resistance training, yoga or stretching practices, and daily sitting time. The screen time and sleep health domains examine screen exposure, sleep duration, sleep quality, sleep consistency, and daytime fatigue. The social and substance use history domain evaluates tobacco smoking, alcohol intake, and the use of smokeless tobacco. All items were framed using simple and clinically interpretable language. Response formats included dichotomous (yes/no), categorical, and frequency-based Likert-type scales to ensure the feasibility of administration in routine clinical practice and community-based settings.
Content Validation Procedure
Expert Panel Selection
Content validation was performed using a physician expert review, widely regarded as the gold standard for establishing clinical relevance during instrument development. A panel comprising 50 physician-experts was constituted based on predefined eligibility criteria. Experts were required to hold formal qualifications of Doctor of Medicine in internal medicine, endocrinology, diabetology, or family medicine, have a minimum of three years of independent clinical practice, and be actively involved in diabetes care and lifestyle counseling. Experts were recruited from diverse clinical settings to ensure a broad representation of real-world practice perspectives.
Validation Materials and Process
Each expert received the complete RSSDI Lifestyle Questionnaire, a structured item validation rating sheet, and written instructions explaining the objectives of the validation exercise and the item-rating procedure. The experts were instructed to independently assess each questionnaire item for relevance to the intended construct. An independent evaluation was emphasized to minimize potential bias and ensure unbiased expert judgment.
Item-rating Scale
The relevance of each item was assessed using a four-point ordinal scale, aligning with best practices for evaluating content validity and designed to prevent neutral responses. The scale ranged from 1 (not relevant) to 4 (highly relevant). Besides numerical ratings, experts were invited to offer qualitative feedback on the clarity, wording, redundancy, and clinical applicability of each item.
CVI Calculation
Item-level Content Validity Index
The I-CVI was determined for each questionnaire item by calculating the proportion of experts who rated the item as either quite relevant or highly relevant (ratings of 3 or 4). Items with an I-CVI of 0.78 or above were deemed acceptable. Items with I-CVI values between 0.70 and 0.77 were marked for revision, while those with values below 0.70 were considered for possible removal.
Scale-level Content Validity Index (S-CVI/Ave)
The overall content validity of the questionnaire was evaluated using the scale-level content validity index, calculated by the average method (S-CVI/Ave). This index was derived by averaging the I-CVI values across all items in the questionnaire. An S-CVI/Ave of 0.90 or higher was considered indicative of excellent content validity at the scale level.
Handling of Qualitative Expert Feedback
All qualitative feedback from experts was systematically reviewed and documented. Suggested changes were categorized to improve clarity of wording, reduce ambiguity, eliminate redundancy, and enhance cultural or clinical relevance. Items that met statistical acceptability criteria but received consistent qualitative concerns were revised to enhance interpretability while maintaining the underlying construct.
Post-validation Item Refinement
Final decisions on item retention, revision, or removal were based on a combined evaluation of quantitative CVI metrics and qualitative expert feedback. Items showing strong statistical validity and clinical relevance were retained without changes. Items needing improvement were revised in wording or structure, while those lacking both adequate CVI values and expert agreement were flagged for removal. The revised instrument became the final content-validated version of the questionnaire.
Statistical Analysis
Content validity analysis was descriptive. Item-level and scale-level CVI values were calculated and presented in tabular form for transparency. After completing content validation and item refinement, the internal consistency reliability of the revised questionnaire was planned to be assessed using Cronbach’s alpha. Values of 0.70 or higher were considered acceptable, with values of 0.80 or higher preferred for applied clinical research. All statistical analyses were conducted using standard statistical software SPSS.
Ethical Considerations
As this phase of the study involved expert review without using patient-level data, formal ethical approval was not required. Expert participation was voluntary, and all responses were anonymized to ensure confidentiality.
Results
Expert Participation
A total of 61 physician-experts took part in the content validation process, providing item-level relevance ratings for the RSSDI Lifestyle Questionnaire. Each expert met the established eligibility criteria and independently assessed the relevance of every questionnaire item.
Item-level Content Validity
Item-Level content validity analysis of item-level content validity showed strong expert consensus across all questionnaire items. The I-CVI values ranged from 0.98 to 1.00, indicating that individual items were perceived as highly relevant. Most items achieved perfect agreement, with all experts rating them as either quite relevant or highly relevant. Specifically, several items related to dietary and lifestyle behaviors—such as those evaluating meal portion size, carbohydrate contribution to meals, protein intake, fiber-rich food consumption, and dairy intake— achieved an I-CVI of 1.00, reflecting unanimous expert approval. The remaining items recorded I-CVI values of ≥0.98, all surpassing the recommended acceptability threshold of 0.78. No item had an I-CVI below the revision or deletion thresholds, indicating that all items were deemed clinically relevant and representative of the intended constructs (Table 1).
Item-level and Scale-level Content Validity Indices of the RSSDI Lifestyle Questionnaire.
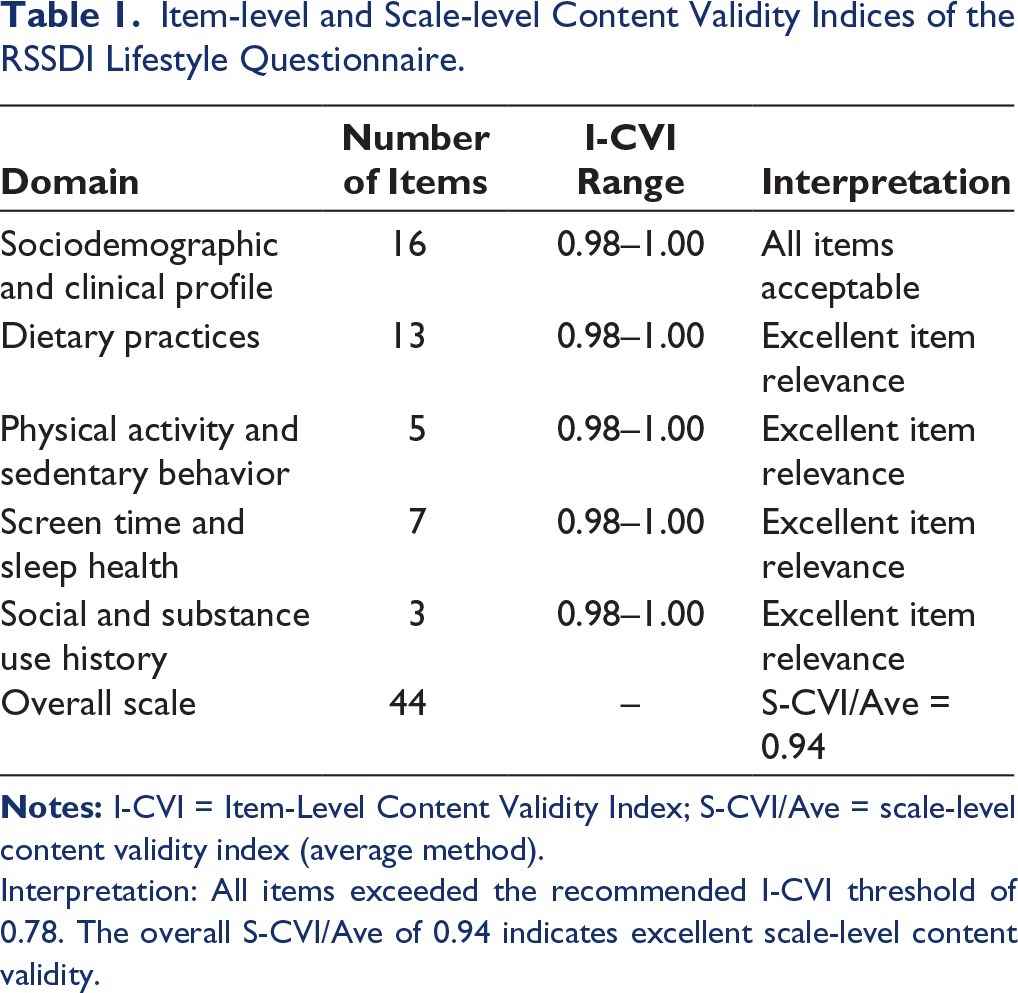
Interpretation: All items exceeded the recommended I-CVI threshold of 0.78. The overall S-CVI/Ave of 0.94 indicates excellent scale-level content validity.
Scale-level Content Validity
Scale-level content validity analysis using the average method revealed an S-CVI/Ave of 0.94, confirming the questionnaire’s excellent overall content validity. This value exceeded the recommended benchmark of 0.90 for robust scale-level content validity, indicating strong expert agreement on the instrument’s adequacy and comprehensiveness. The scale-level content validity index using universal agreement (S-CVI/UA) was 0.50, reflecting partial universal agreement across all items, which is acceptable given the large number of items and experts involved.
Qualitative Expert Feedback
In addition to quantitative ratings, experts provided qualitative feedback primarily related to minor wording refinements and clarity enhancements. Suggestions focused on improving item phrasing to enhance interpretability in routine clinical settings and ensuring the cultural appropriateness of dietary examples. No expert raised concerns regarding the redundancy or irrelevance of domains. Based on this feedback, select items underwent minor wording modifications without altering the underlying constructs. No items were removed as part of the content validation process.
Post-validation Item Refinement
Post-validation item refinement following the integration of quantitative CVI findings and qualitative expert suggestions, all 44 items were retained in the final version of the questionnaire. Minor editorial refinements were implemented to improve clarity and consistency across items. The resulting instrument constituted the final content-validated version of the RSSDI Lifestyle Questionnaire.
Internal Consistency Reliability
Following content validation, internal consistency reliability was evaluated using Cronbach’s alpha. The reliability analysis included 61 complete response sets, with incomplete cases being excluded. The questionnaire achieved an overall Cronbach’s alpha of 0.96, signifying excellent internal consistency (Table 2). This value surpassed the widely accepted threshold of 0.80 for applied clinical research, indicating the coherence and uniformity of the retained items.
Internal Consistency Reliability of the RSSDI Lifestyle Questionnaire.

In summary, the RSSDI Lifestyle Questionnaire showed strong content validity at both the item and scale levels, backed by high expert consensus and positive qualitative feedback. The tool also demonstrated outstanding internal consistency, confirming that the retained items effectively measure related lifestyle constructs. These results endorse the questionnaire’s appropriateness for use in clinical research, epidemiological studies, and future prospective validation efforts.
Discussion
The findings from the content validation and reliability analysis of the RSSDI Lifestyle Questionnaire provide evidence of its capacity to evaluate dietary and lifestyle behaviors relevant to diabetes management. The high item-level and scale-level content validity indices (I-CVI and S-CVI) reflect significant agreement among the panel of physician-experts, reinforcing previous studies that emphasize the importance of expert evaluations in establishing the validity of health-related questionnaires.9,15 The results show I-CVI values ranging from 0.98 to 1.00 and an overall S-CVI/Ave of 0.94, indicating that the items are relevant and comprehensive for the intended target population, in alignment with guidelines advocating for I-CVI scores above 0.78 for content validity.9,10
Studies have demonstrated that rigorous psychometric evaluations yield high reliability and validity scores similar to those achieved in this validation process. Batista et al. validated the Portuguese version of a healthy lifestyle questionnaire, achieving strong internal consistency (Cronbach’s alpha of 0.92) alongside substantial content validity. 16 This reinforces the idea that meticulous development and evaluation processes contribute to effective lifestyle assessment tools. Moreover, research by Kowalkowska et al. mirrored these outcomes, reporting excellent reliability and test-retest reproducibility for their dietary habits questionnaire, highlighting the importance of instrument validation in varied cultural contexts. 17
However, contrasting considerations emerge from studies that reveal challenges in achieving broad expert consensus, particularly within multifaceted health behavior assessments that require heightened specificity to correlate effectively with health outcomes, Odunaiya et al. criticized the inadequacies in validation methods for cardiovascular disease (CVD) risk factor questionnaires, indicating that many tools lacked comprehensive content validity due to insufficient expert input or neglect of certain lifestyle factors. 18 This points to the necessity of engaging various expert stakeholders to enhance the questionnaire’s robustness and ensure it captures the diverse dimensions of health and lifestyle pertinent to different populations, and this was addressed in the study by involving 50 physician-experts from diverse clinical domains, thereby strengthening the robustness of the questionnaire and ensuring comprehensive representation of lifestyle and health dimensions across populations.
The combination of quantitative and qualitative feedback from experts facilitated the refinement of the RSSDI Lifestyle Questionnaire, reflecting standard practices in questionnaire development. 19 While experts proposed minor adjustments for clarity and cultural relevance, none contested the core relevance or completeness of the items. This is consistent with findings by Hamid et al., which emphasized the criticality of expert judgment in establishing content validity and cultural appropriateness within health education materials. 20 Such iterative assessments substantiate that instruments remain attuned to evolving health paradigms, exemplifying effective participatory methods in health intervention research.
The overall internal consistency reliability of the questionnaire, evidenced by a Cronbach’s alpha of 0.96, aligns with high standards for psychometric evaluations in health-related surveys, surpassing the commonly accepted threshold of 0.80. 21 This degree of reliability enhances confidence in the RSSDI Lifestyle Questionnaire’s ability to yield consistent results over time. These findings resonate with prior studies that highlight the potential for validated questionnaires to reliably inform intervention strategies and assessments in clinical practice. 22 The RSSDI Lifestyle Questionnaire stands as a well-validated instrument capable of accurately capturing lifestyle behaviors essential for diabetes management. The sustained alignment with stringent reliability and validity benchmarks, alongside expert recommendations for improvement, underscores its viability for future prospective studies and its role in clinical applications.
Conclusion
The RSSDI Lifestyle Questionnaire showed excellent content validity and internal consistency, confirming the suitability of this comprehensive tool in assessing lifestyle behaviors relevant to diabetes and metabolic health in the Indian population. Strong consensus by experts in all domains underscores clinical relevance, cultural appropriateness, and conceptual clarity of the instrument. This questionnaire can be reliably applied in clinical practice and also in epidemiological studies and studies on lifestyle interventions. External validation, construct validity, test-retest reliability, and responsiveness to lifestyle interventions across diverse Indian populations and care settings are further studies that need to be done.
Supplemental Material
Supplemental material for this article is available online.
Footnotes
Acknowledgements
The authors would like to acknowledge the physician-experts who participated in the content validation process of the RSSDI Lifestyle Questionnaire by providing item-level relevance ratings and constructive qualitative feedback. Their expert insights were instrumental in refining the clarity, cultural appropriateness, and clinical relevance of the questionnaire items.
The authors also acknowledge the administrative and institutional support received during the coordination of expert reviews and data collation. All contributors acknowledged in this section do not meet the criteria for authorship as defined by the International Committee of Medical Journal Editors (ICMJE).
Authors’ Contributions
Author contributions were defined in accordance with the CRediT Contributor Roles Taxonomy.
Vijay Viswanathan: Conceptualization; Methodology; Supervision; Validation; Writing – review & editing.
Vijay Kumar: Investigation; Data curation; Validation; Writing – review & editing.
Krishna G. Seshadri: Conceptualization; Validation; Critical review of manuscript; Writing – review & editing.
Shanmugavelu M: Methodology; Validation; Investigation; Writing – review & editing.
Balamurugan: Investigation; Validation; Data curation; Writing – review & editing.
Kumaravel: Methodology; Validation; Formal analysis; Writing – review & editing.
V. Rajendran: Investigation; Validation; Writing – review & editing.
Ashwin Karuppan Viswanathan: Project administration; Data curation; Formal analysis; Writing – original draft; Writing – review & editing.
All authors reviewed and approved the final version of the manuscript and agree to be accountable for all aspects of the work in ensuring its accuracy and integrity.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Disclosure of Artificial Intelligence Use
In accordance with the journal’s Artificial Intelligence policy and the guidelines issued by SAGE Publications, the authors disclose that AI-assisted tools were used solely for language editing, grammar correction, and improvement of clarity and readability of the manuscript. No generative AI tools were used for study design, data collection, data analysis, interpretation of results, or generation of scientific content. The authors retain full responsibility for the integrity, originality, and accuracy of the work.
Ethical Approval
Ethical approval was not required, as the study involved expert validation without patient-level data. This has been clearly stated in the manuscript under “Ethical Considerations.”
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Informed consent was obtained from all participants and non-author contributors prior to their inclusion in the study.
Patient Consent
Patient consent was not required, as no patient data or identifiable information was used in this study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
