Abstract
This study was aimed to evaluate in clinical trial settings the psychometric properties of the revised Patient Perception of Migraine Questionnaire (PPMQR), a satisfaction measure for acute migraine treatment. The PPMQ-R was administered 24 h post dosing in 1304 migraineurs randomized to two identical Phase 3, single-attack trials. Reliability, concurrent and construct validity and known-groups validity were evaluated using Cronbach's α, Pearson correlations and analysis of variance, respectively. PPMQ-R scale and Total scores (Efficacy, Functionality and Ease of use) showed very good internal consistency reliability (α 0.84-0.99). Efficacy, Functionality and Total PPMQ-R scores showed large, inverse relationships with migraine pain severity, number of migraine symptoms and work ability (r = −0.62 to −0.75; all P < 0.0001). All scales discriminated among migraine pain severity levels (all P < 0.001). The PPMQ-R has sufficient evidence of validity and reliability for measuring patient satisfaction, an important benchmark of quality and effective care.
Introduction
Treatment satisfaction, defined as the patient's evaluation of important attributes associated with the process and outcomes of the treatment experience, is increasingly assessed in clinical trials and evaluations of disease management programmes (1, 2). For any therapy, key domains associated with treatment satisfaction have been identified in the literature and include treatment effectiveness (i.e. symptom relief, speed of onset, duration of effects), discomfort (i.e. bother, side-effects), regimen characteristics (i.e. convenience), preference for continuing treatment and cost (1–3). These domains are consistent with attributes identified for migraine-specific therapies to treat acute headache attacks, which are: rapid pain relief, complete pain relief, consistent pain relief across headaches, ability to return to normal functioning, relief of migraine-associated symptoms, reduction in headache recurrence and minimal adverse effects (4–10).
Although treatment satisfaction measures are often used in clinical trials, until recently reports describing the development and psychometric validation of these measures have been scarce, limiting clinical study applications and interpretation of study findings. This is particularly true of instruments that measure satisfaction specific to migraine treatments. More recently, however, several well-developed and psychometrically sound measures of satisfaction with specific treatments (e.g. contraception, pain) or general treatment have been developed (11–14). Treatment satisfaction measures with evidence of reliability and validity are needed to evaluate migraine therapies in clinical studies to ensure results are valid and meaningful in this setting. The revised Patient Perception of Migraine Questionnaire (PPMQ-R) is one of the few published questionnaires assessing patient satisfaction with acute migraine treatment and reporting on the development process and psychometric evaluation (15–17).
The PPMQ-R is a 32-item instrument designed to measure patient satisfaction with acute migraine treatment. It measures satisfaction with efficacy, function, ease of use, cost and tolerability of side-effects (i.e. Efficacy, Functionality, Ease of Use, Cost and Tolerability scales). The psychometric properties of the PPMQ-R were previously evaluated in an out-patient population of adult patients from 50 primary care and neurology specialty clinics across the USA (n = 200) (16). Previous research has shown that the measure had good reliability and validity characteristics in both single-attack and multiple-attack evaluations. In addition, the PPMQ-R was responsive to changes in clinical status in multiple-attack evaluations.
Currently, the psychometric properties of the PPMQ-R have not been evaluated in a clinical trial setting. There are also questions as to whether the full range of adverse events for migraine treatment is being captured by the Tolerability domain. The objective of this study was to evaluate the psychometrics properties of the PPMQ-R in a clinical trial setting and the Tolerability scale's coverage of reported adverse events.
Materials and methods
Psychometric evaluation study
This analysis used data from two identical Phase III trials [Trial 1 (TRX101998) and Trial 2 (TRX101999)] for a fixed-dose, single-tablet combination of sumatriptan 85 mg formulated with RT TechnologyTM and naproxen sodium 500 mg. Subjects were eligible to participate in the trials if they (i) were between 18 and 65 years of age; (ii) were male or female (females of non-childbearing potential or of child-bearing potential with a negative pregnancy test at screen and agreement to several conditions); (iii) had at least a 6-month history that met 2004 International Headache Society (HIS) Criteria for migraine with aura (ICHD-II 1.2.1) or migraine without aura (ICHD-II 1.1); (iv) experienced two to six migraine attacks per month in the 3 months prior to screening; (v) typically experienced moderate to severe migraine pain preceded by an identifiable mild pain phase; (vi) were able to distinguish between mild migraine pain and other headache types; (vii) were able to read, comprehend and complete subject diaries and understand that they must treat their migraine during the mild pain phase; and (viii) were able and willing to give written informed consent to participate in the study.
In each single-attack trial, approximately 600 subjects were randomized to receive sumatriptan 85 mg/naproxen sodium 500 mg or placebo, to be taken orally within 1 h of onset of migraine head pain if the pain was mild at the onset and while the pain remained mild. At the screening visit, demographic data were collected and subjects completed the Headache Impact Test (HIT)-6 (Table 1). Using a diary, subjects recorded details of their migraine attack. Migraine pain severity and the presence of migraine-associated symptoms were recorded at dosing and 30 min and 1, 2 and 4 h after the first dose of the study medication. Work ability was recorded at dosing with the study medication and 2 and 4 h after dosing, while use of additional medications and migraine pain reoccurrence were recorded throughout the study. The PPMQ-R and productivity questionnaires were completed 24 h after dosing with the study medication. Clinical variables, including adverse events, were recorded by clinical personnel throughout the study.
Timetable for measures used in Trials 1 and 2

During the 24 h following dosing with investigational product if migraine pain returned.
The psychometric analyses combined data from both treatment groups and were performed blinded to treatment group status. Repeated observations over time for each subject (e.g. migraine pain severity) were not aggregated.
Clinical and patient-reported outcome measures
Patient perception migraine questionnaire
The PPMQ-R consists of 29 questions that assess a patient's satisfaction with migraine medication in terms of its Efficacy (11 items), Functionality (four items), Ease of use (two items) and Cost (two items), as well as the degree to which side-effects were tolerated (Tolerability; 10 items). In addition, three global items measuring subject satisfaction in terms of medication effectiveness, side-effects and overall satisfaction are included. Items that evaluate treatment satisfaction are scored on a seven-point Likert-type scale from 1 (very satisfied) to 7 (very dissatisfied); whereas items that evaluate tolerability of side-effects (i.e. bother due to side-effects) are scored on a five-point Likert-type scale from 1 (not at all) to 5 (extremely). Data from the PPMQ-R are analysed as four subscale scores (Efficacy, Functionality, Ease of use and Tolerability) and a Total score (composite of the Efficacy, Functionality and Ease of use subscales). Scale scores are transformed to range from 0 to 100, with higher scores indicating better satisfaction or tolerability. For this study, the two items that comprise the cost subscale were not used because subjects did not pay for the study medication; therefore, 27 questions and three global items were used.
Migraine diary
The details of the single migraine attack were captured in a self-administered subject diary. Specifically, subjects recorded data on headache pain severity (none, mild, moderate, severe); the presence of migraine symptoms [nausea, vomiting, sensitivity to light and sound, neck pain/discomfort, sinus (facial) pain/pressure]; ability to work or perform normal or usual activities (normal, mildly impaired, moderately impaired, severely impaired, required bedrest); the presence of a recurrent migraine pain; the time and severity level (mild, moderate, severe) of pain recurrence; and additional medications taken, including the date and time taken and the primary reason for taking.
Productivity questionnaire
The productivity questionnaire included: (i) the number of hours a subject missed from work and non-work activities; (ii) the number of hours a subject worked with symptoms and continued to participate in non-work activities with symptoms; and (iii) the subject's rating of effectiveness while continuing work and non-work activities with symptoms during the 24 h after taking the study drug.
Demographic and clinical characteristics
Demographic and clinical characteristics such as the IHS headache classification (migraine with and without aura), migraine history (e.g. number of migraine attacks typically experienced per 30-day calendar month), previous migraine treatments and satisfaction with previous treatments were collected at screening. Information on non-serious adverse events (AEs) and serious adverse events (SAEs) were collected throughout the study and included: description of event, start and stop date, outcome, maximum intensity, action taken with the study medication as a result of the AE or SAE, subject withdrawal from the study, and the relationship of the AE or SAE to the study medication (i.e. reasonable possibility).
Headache impact test
The HIT-6 consists of six questions used to measure the impact headaches have on a person's ability to function at work, school, home, or social situations. Scores range from 36 to 78, with higher scores indicating a greater impact of headaches on the person's life (18).
Psychometric analysis
Analyses were performed to examine item characteristics, factor structure and scoring, reliability, and validity (concurrent and construct) of the PPMQ-R in measuring satisfaction with an acute migraine treatment for a single attack. The data from both trials were combined for the psychometric analysis of the PPMQ-R and the statistical analyses were performed blinded to treatment group. All patients randomized to treatment were included in this analysis.
Sociodemographic characteristics
Sample sociodemographic characteristics at screening such as age, gender and race were summarized (e.g. means, frequencies) for the study sample to describe the sample population. Clinical factors such as medication characteristics (e.g. previous/current treatment for migraine) and migraine characteristics (e.g. headache type, migraine history) were also summarized for the screening evaluation.
Item characteristics and factor structure
To evaluate whether PPMQ-R items are capturing the full range of response options and the associations between items, characteristics of individual items were examined by calculating the mean, minimum possible and maximum possible response (i.e. floor and ceiling effects), percent missing and item–item correlations.
Confirmatory factor analysis was used to substantiate the findings on factor structure and item groupings based on previous research conducted on the PPMQ-R (16) and was performed with Mplus software (Version 4.1). To determine the acceptability of the factor structure and item loadings, fit indices were evaluated for two models—a two-factor (Efficacy/Functionality and Ease of use) and a three-factor (Efficacy, Functionality and Ease of use) solution. The following fit indices were used to assess the fit of each model: Bentler's (19) comparative fit index (CFI; values > 0.90 represent acceptable fit), root mean square error of approximation (RMSEA; values > 0.05 to ≤ 0.08 represent adequate fit) (20) and standardized root mean square residual (SRMR; values < 0.05 represent acceptable fit) (21).
Reliability
Internal consistency reliability of multi-item scales was assessed using Cronbach's α, with values ≥ 0.70 considered acceptable for group-level comparisons (22, 23).
Validity
Correlations between the PPMQ-R and other measures that are considered conceptually or clinically related were evaluated, including pain severity, number of symptoms, work ability, recurrence of migraine pain, number of additional medications taken and productivity. Concurrent validity was supported when a specific scale was substantially correlated (> 0.40) with a generic scale measuring the same or a similar concept. Conversely, scales measuring different concepts should be less strongly correlated (< 0.40). For interpretation, guidelines suggested by Cohen (24) were used, where absolute correlation values between 0.10 and 0.29 are considered small, 0.30–0.49 are considered medium, and 0.50–1.00 are considered large.
The ability of the PPMQ-R scores to discriminate between groups of patients according to clinical severity was assessed using analysis of variance (
Completeness of the Tolerability scale
Completeness of the Tolerability scale was assessed by cross-classifying side-effects listed in the Tolerability domain with the (i) severity of AEs (mild, moderate, severe) and (ii) AE ratings related to study medication (possibly related to study medications—yes/no).
Results
Sample characteristics
A total of 1304 patients suffering from migraine attacks participated in this study. Patient characteristics are summarized in Table 2. Study participants were primarily White (85.5%), women (87.8%), with an average age of 40 years. On average, participants were 22.5 years old when first experiencing migraine attacks. Based on a 6-month history that meets IHS criteria for headache with or without aura at screening, the majority of participants had experienced migraine without aura (80.6%), while just over a third (33.6%) had experienced migraine with aura. Since patients can experience both types of migraine headaches, these categories are not mutually exclusive. At screening, patients reported that they had experienced an average of 3.8 migraine attacks per month during the past year and that they averaged 7.3 days per month with any headache. Participants scored an average 53.9 points on the HIT-6, indicating that their migraines had some impact on their lives.
Demographics at screening for all eligible patients

Scores range from 36 to 78, with higher scores indicating a greater impact of headaches on the person's life.
The most commonly used medications to treat acute migraine attacks—prior to enrolment in the trials—were sumatriptan (39.5%) and over-the-counter non-steroidal anti-inflammatory drugs that do not contain naproxen (43.9%). Of all patients in the study (n = 1304), 53% reported that they were satisfied with sumatriptan, which was the highest rate of satisfaction among the 12 medications or medication classes listed.
At the time patients initiated treatment for their attack, most (74.2%; 968 of 1304) rated their average migraine pain as mild (Table 3). These patients reported an average of 2.3 migraine symptoms per attack, having experienced light sensitivity (60.2%), sound sensitivity (52.8%) and neck pain or discomfort (52.0%) most often. Approximately three-quarters of patients experienced some level of impairment (mildly impaired to requiring bedrest) in their ability to perform work or usual activities.
Migraine attack characteristics (n = 1304)

Symptoms include nausea, vomiting, light sensitivity, sound sensitivity, neck pain/discomfort, sinus pain/pressure.
Improvements in pain and ability to perform work or usual activities were observed after treatment, with more than two-fifths of patients reporting no migraine pain (40.8%) and no work impairment (45.4%) 4 h after dosing with study medication (sumatriptan 85 mg/naproxen sodium and placebo groups combined). On average, patients experienced one less symptom 4 h after dosing. Light sensitivity, sound sensitivity and neck pain or discomfort—the three most common symptoms—were reduced by 34.1%, 29.3% and 27.4%, respectively. Compared with being as effective as usual in performing activities (i.e. 100% effective), patients reported being 71.3% effective performing paid work activities and 62.2% effective performing activities outside of paid work 24 h after treatment. The clinical and health outcome effects of sumatriptan 85 mg/naproxen sodium compared with placebo are reported elsewhere (25, 26).
Of 576 patients who reported being pain free at 2 h, 78% did not have a return of migraine pain 2–24 h after dosing (i.e. 22% recurrence rate). Approximately half of all patients took an additional medication to treat their migraine attack.
PPMQ-R item and subscale characteristics
Item characteristics
Scores for PPMQ-R items rated on the seven-point satisfaction scale (i.e. items 1A-1Q; 3A-3C) covered the full range of response options, with mean scores ranging from 2.0 (satisfied) to 4.3 (neither satisfied nor dissatisfied). The percentages of patients scoring at the floor (the lowest possible score: ‘very dissatisfied’) ranged from 1.6% (how convenient the medication is to use) to 23.1% (how well the medication prevents the need for other medications to treat the migraine attacks). The percentages of patients scoring at the ceiling (the highest possible score: ‘very satisfied’) ranged from 7.6% (how fast the medication relieves other migraine symptoms) to 38.1% (how convenient the medication is to use). Skewness statistics for the individual PPMQ-R items ranged from −0.0004 to 0.198 for the satisfaction items and from 0.090 to 0.112 for the overall satisfaction items. Except for Ease of use items, scores across response categories appear to be relatively evenly distributed.
Scores for PPMQ-R items rated on the five-point tolerability scale (i.e. items 2A-2J) covered the full range of response options, with mean scores ranging from 1.1 (not at all bothered) to 1.6 (slightly bothered). Floor effects, reporting ‘extremely [bothered]’, were low and ranged from 0.5% (nervousness, chest pain) to 2.5% (nausea). In contrast, high ceiling effects, reporting ‘not at all [bothered]’, were observed for all items, ranging from 55.9% (tired or drowsy) to 81.0% (vomiting). Skewness statistics for individual tolerability items ranged from 1.807 to 5.982. Scores across response categories were not evenly distributed, since not everyone experienced side-effects.
Evaluation of structural validity
Confirmatory factor analysis of the PPMQ-R satisfaction items showed evidence for both a two- and three-factor model: for the three-factor model [Efficacy (11 items), Functionality (4 items) and Ease of use (2 items)], loadings for all items within each factor exceeded 0.40, with the majority being > 0.90 (range 0.880–0.995). Fit indices were as follows: CFI = 0.922, RMSEA = 0.148 [90% confidence interval (CI) 0.143, 0.153] and SRMR = 0.027. The correlation between the Efficacy and Functionality factors was 0.938 (Table 4).
Confirmatory factor analysis of PPMQ-R at 24 h: factor loading for ‘satisfaction’ items

Correlation of Efficacy and Function scales = 0.939.
Items 3a−3c are single-item scales and are not included in the analysis.
For three-factor solution: comparative fit index (CFI) = 0.922; root mean square error of approximation (RMSEA) = 0.148. For two-factor solution: CFI = 0.978; RMSEA = 0.081.
Similar loadings were observed for the two-factor model [Efficacy/Functionality (15 items) and Ease of use (two items)], with the majority being > 0.90 (range 0.829–0.958). Fit indices were as follows: CFI = 0.978, RMSEA = 0.081 (90% CI 0.076, 0.086) and SRMR = 0.013.
The final scoring algorithm for the 27 core PPMQ-R items therefore consisted of four scale scores (Efficacy, Functionality, Ease of use and Tolerability). Each scale score is calculated by summing the value of individual items within a scale. A Total score, representing the average of the Efficacy, Functionality and Ease of use scales, also can be calculated. All scale scores were transformed to a 0–100 scale, with higher scores reflecting more satisfaction or better Tolerability.
Subscale characteristics
Each subscale was scored on a 0–100 scale, with higher scores reflecting greater satisfaction or Tolerability. Forty per cent of patients scored at the ceiling (100% satisfaction or tolerability) for the Ease of use and Tolerability scales, with the mean scores being 82.4 and 92.1, respectively. In addition, none of the patients scored at the floor (0% tolerability) for the Tolerability scale. Except for correlations with the Tolerability scale (Tolerability and Efficacy; Tolerability and Functionality), all subscale to subscale correlations were statistically significant (all P < 0.05), ranging from 0.08 (Tolerability and Ease of use) to 0.94 (Efficacy and Functionality) (Table 5). Total to subscale correlations were also significant (all P < 0.0001), except for the Tolerability scale, ranging from 0.65 (Ease of use) to 0.96 (Efficacy).
PPMQ-R subscale to subscale correlations† 24 h after investigational product
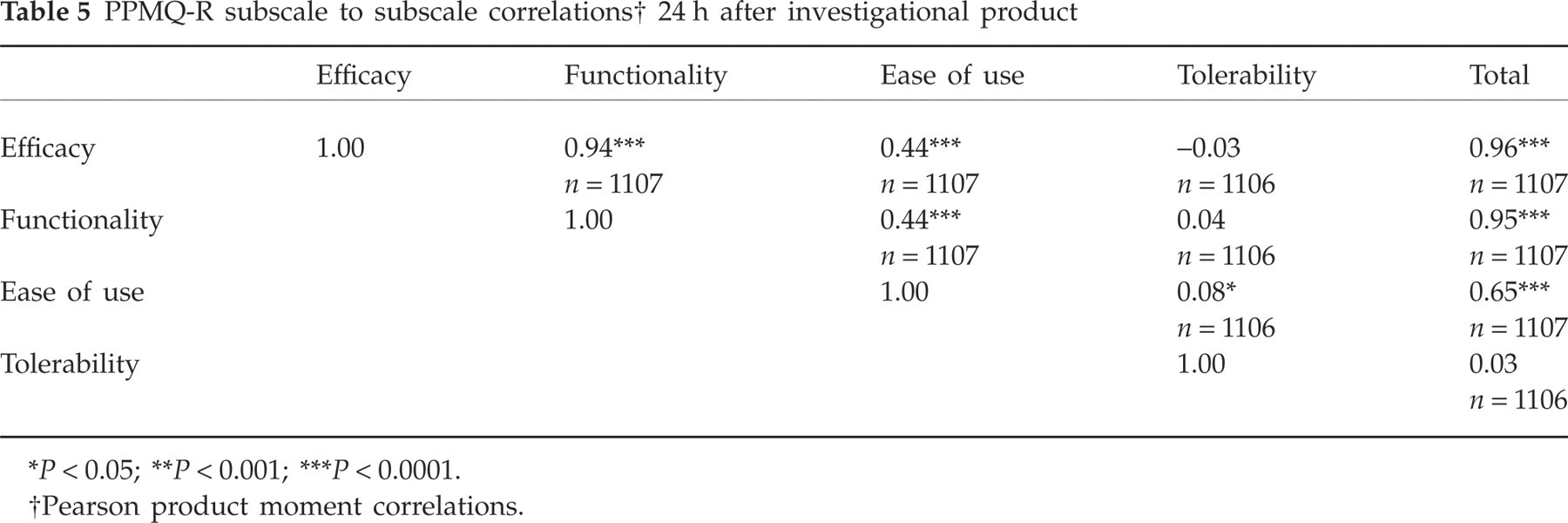
P < 0.05;
P < 0.001;
P < 0.0001.
Pearson product moment correlations.
Reliability
The four PPMQ-R scale scores and Total score demonstrated good internal consistency reliability (Cronbach's α = 0.84–0.99) 24 h after treatment (Table 6).
Distributional and scale characteristics of the PPMQ-R

Validity
Concurrent and construct validity
Table 7 summarizes the correlations between the PPMQ-R and indicators of severity such as migraine pain severity, the number of migraine symptoms and work ability (i.e. concurrent validity). All PPMQ-R scores were negatively correlated with migraine pain ratings at 4 h after the migraine attack (P < 0.0001). Migraine pain, number of migraine symptoms, work ability and the number of additional medications to treat migraine pain had large, significant negative correlations with the Efficacy and Functionality subscales and the Total scores (e.g. the less pain, symptoms, impairment, or medications, more treatment satisfaction), with correlations ranging from −0.49 to 0.75 (all P < 0.0001). Percent effectiveness at work (i.e. paid work) and hours missed from non-work activities had medium, significant correlations with the Efficacy and Functionality subscales (r = 0.36–0.42; all P < 0.0001). The Ease of use scale was most highly correlated with migraine pain severity (r = 0.34, P < 0.0001), whereas the Tolerability scale was most highly correlated with percent effectiveness at work (r = 0.29, P < 0.0001).
Concurrent and construct validity at 24 h: correlation between PPMQ-R, migraine severity and symptoms at 4 h, work ability and the productivity questionnaire

P < 0.05;
P < 0.001;
P < 0.0001.
Pearson product moment correlations or Spearman rank correlations.
4 h after treating with study medication.
Between 2 and 24 h after treating with study medication.
24 h after treating with study medication.
In addition, the three global satisfaction items of the PPMQ-R (Overall Satisfaction, Overall Efficacy and Side-effects) had significant correlations with each PPMQ-R subscale and Total score (r =−0.19–0.93; all P < 0.0001) (i.e. construct validity). In particular, the Overall Satisfaction and Efficacy items had large correlations with the Efficacy and Functionality subscales and the Total scores (ranging from 0.89 to 0.93; all P < 0.0001). For the Tolerability scale, the highest correlation was with the global Side-effects item (r = 0.28, P < 0.0001).
Known groups validity
All PPMQ-R scale scores discriminated among pain severity levels 4 h after dosing, with those experiencing severe pain generally having the lowest PPMQ-R satisfaction scores (most P < 0.0001) (Fig. 1). PPMQ-R scores differed significantly among patients categorized based on their ability to work 4 h after treatment for their migraine attack (most P < 0.0001) (Fig. 2). Except for the Tolerability scale, all PPMQ-R scores also differentiated between the number of migraine symptoms 4 h after treatment (all P < 0.0001), with those experiencing more symptoms reporting the lowest scores (least satisfaction). In general, PPMQ-R scale scores and Total scores were highest for patients who were not impaired and lowest for those who required bedrest following treatment initiation.

PPMQ-R scores discriminate among pain severity levels. ∗∗P < 0.0001,
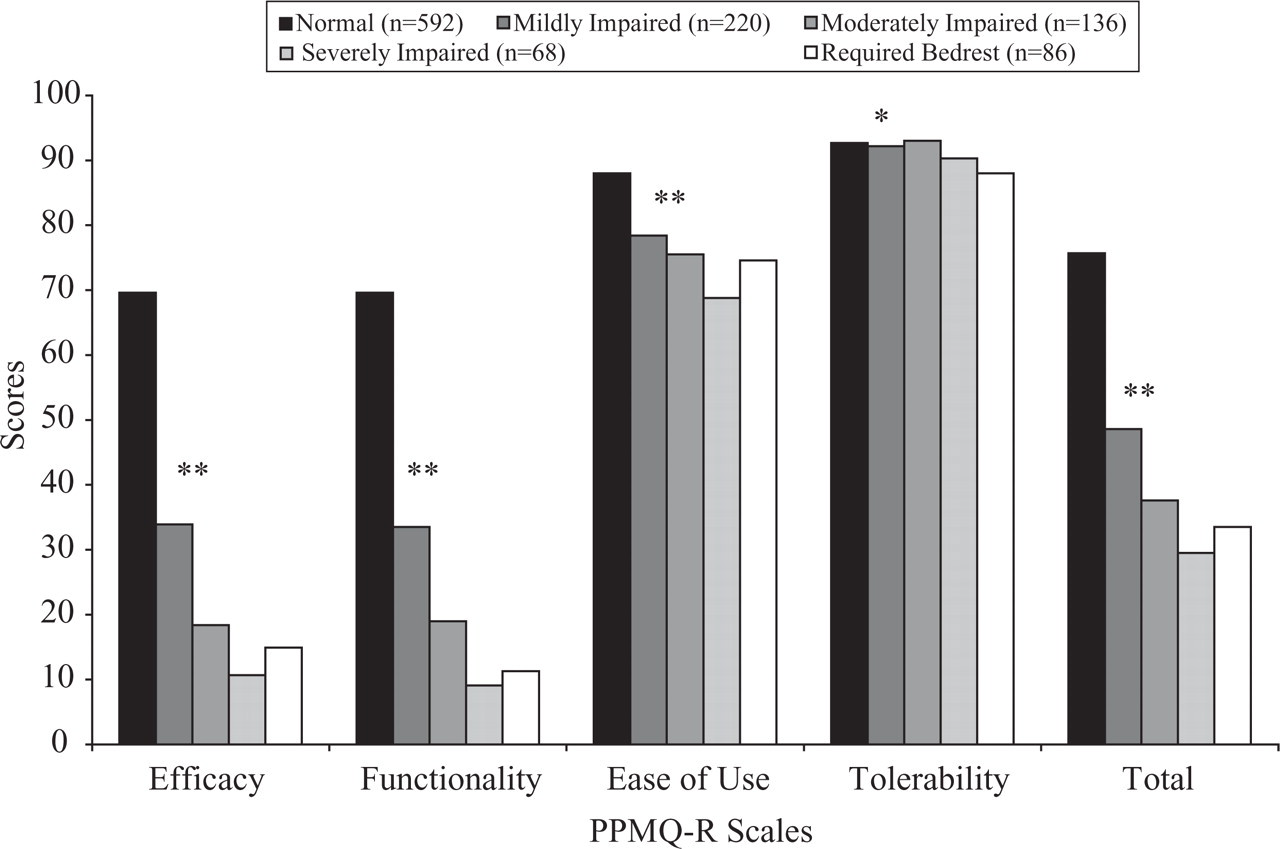
PPMQ-R scores discriminate among work ability levels. ∗∗P < 0.0001,
Adverse events
Of 1304 patients, 114 experienced 180 AEs. There were no SAEs reported. Ninety-three of the 180 AEs reported (51.7%) were categorized as one of the 10 side-effects included in the PPMQ-R Tolerability scale (Table 8). Nausea was the most frequently reported side-effect that was captured in the Tolerability scale, occurring 34 times (18.9% of all AEs). Being tired or drowsy, experiencing dizziness, having chest pains or pressure, and having a dry mouth were the next most frequently reported side-effects that were captured in the Tolerability scale (7.8%, 6.7%, 5.6% and 5.0%, respectively, for all AEs). The most frequently reported side-effects that were not captured by the Tolerability scale were: experiencing sensations or pain (e.g. pressure, paraesthesia); having an infection; or experiencing ear, nose and throat symptoms (e.g. congestion) (21.7%, 9.4% and 4.4%, respectively).
Cross classification: reported adverse event (AE) (categorized based on PPMQ tolerability items) by relationship to study medication

For AEs that occurred at any time during the trial; n = 180.
Included chest discomfort and chest pain AE terms.
Included confusional state and mental impairment AE terms.
Included dizziness and vertigo AE terms.
Included fatigue and somnolence AE terms.
Included dyspepsia and nausea AE terms.
Included agitation, anxiety, nervousness and psychomotor hyperactivity AE terms.
Included heart rate increased and palpitations AE terms.
ENT, Ear, nose and throat.
Of the 93 reported AEs captured by the Tolerability scale, the majority were rated as (i) either mild (48.4%) or moderate (41.9%) in severity and (ii) having a reasonable possibility of a relationship to the study medication (63.8%). Forty-six (36.2%) of the AEs not included in the PPMQ-R were considered as having a reasonable possibility of a relationship to the study medication.
Discussion
The PPMQ-R was specifically designed to measure satisfaction with pharmacological therapy in patients who experience migraine headaches. Previous work evaluating the psychometric properties of the PPMQ-R in a prospective study of 200 migraine patients visiting neurologists and primary care clinics suggests that the PPMQ-R has five scales measuring satisfaction: Efficacy, Functionality, Ease of use, medication cost and tolerability of medication side-effects. The PPMQ-R scale scores and Total score demonstrated good reliability (internal consistency and test–retest), validity (construct and discriminant) and responsiveness to change (16). A difference of five points was recommended as the minimally important difference (MID) for the Efficacy, Functionality and Ease of use scale scores, as well as the PPMQ-R Total score (16). Findings from the current study, which assessed treatment satisfaction 24 h after dosing, provides further confirmation of the psychometric characteristics of the PPMQ-R (i.e. factor structure, internal consistency reliability and validity) in a clinical trial setting.
Individual responses for the satisfaction-related items for the Efficacy and Functionality subscales did not demonstrate significant ceiling or floor effects. The responses for the two questions on satisfaction with Ease of use demonstrated ceiling effects. However, these results may reflect the convenience of using oral medications and the popularity of this formulation among the study sample. Given that skewed responses were a common problem in most previously published treatment satisfaction measures (i.e. more positive ratings of satisfaction) (2, 3), these results for the PPMQ-R are encouraging. The responses for the items in the Tolerability subscale were highly skewed and demonstrated large ceiling effects (i.e. little bother with the listed side-effects). This finding was not unexpected, given that only a small number of patients in this study experienced side-effects (8.7%). The PPMQ-R subscale and Total scores have acceptable ceiling and floor effects, except for Ease of use and Tolerability. For example, the Total score had only 1.4% of scores at the lowest score and 3.3% at the highest possible score.
The confirmatory factor analyses provide evidence supporting either a two-factor or three-factor solution for the Efficacy, Functionality and Ease of use domain scores. These factor analysis results are consistent with earlier research, where a strong correlation was observed between the Efficacy and Functionality scales (r = 0.87) (16). Although the two-factor solution fits these data somewhat better, there are reasons for keeping the Efficacy and Functionality subscales separate. Previous research on treatment satisfaction suggests differentiating effectiveness and functional impact (1, 2), and these two outcomes provide more comprehensive understanding of patient perceptions of satisfaction. The qualitative data from migraine patients also suggested that functional impact and efficacy were considered separate and conceptually distinct attributes. Finally, it is important, and of interest to clinicians, to be able to understand differences between satisfaction with efficacy and function. Although these two satisfaction concepts are expected to be strongly correlated, they do capture different aspects of satisfaction with migraine treatment. Regardless, the confirmatory factor analyses also provide strong support for combining the three subscales into a Total score. Future research needs to explore the interrelationship between satisfaction with the effectiveness of treatment and satisfaction with its functional impact.
The PPMQ-R demonstrated acceptable internal consistency reliability for group level comparisons. The findings indicated that the subscale and Total scores are internally consistent (Cronbach's α range 0.84–0.99). The reliability results were comparable to those found in Revicki et al. (16).
The PPMQ-R scores discriminated among various levels of pain and work and usual activity impairment. Except for Tolerability, significant differences in satisfaction scores were observed among patient-rated migraine pain severity levels for all PPMQ-R subscale and Total scores. The results demonstrated that patients with less pain were more satisfied with their treatment. Similar findings were observed for differences in satisfaction scores among levels of work ability and levels of effectiveness for paid work and non-paid activities for all PPMQ-R subscale and Total scores. Patients reporting less work and activity impairment were more satisfied with their treatment. These findings are consistent by previous work on the PPMQ-R (16).
This study has provided evidence supporting the concurrent and construct validity of the PPMQ-R in migraine patients and findings were generally consistent with the previous work for the PPMQ-R. Except for Tolerability, all the subscale scores were significantly related to reports of migraine pain severity, number of migraine symptoms, work ability, migraine recurrence and the need for additional medications. The PPMQ-R subscale and Total scores, except for Tolerability, varied significantly by number of symptoms, where patients with fewer symptoms 4 h after treatment reported higher treatment satisfaction. The PPMQ-R subscale and Total scores also varied significantly by pain severity, where patients reporting less pain severity also reported more satisfaction with treatment. The relationship between migraine symptoms and PPMQ-R scores suggests that this measure of treatment satisfaction may be sensitive to outcomes in clinical trials. In addition, the subscale and Total scores were significantly associated with work impairment levels, with those patients with the most impairment reporting the least treatment satisfaction.
The PPMQ-R global satisfaction items (Side-effects, Overall efficacy and Overall satisfaction) were not included in the subscale scoring of the PPMQ-R. However, strong correlations were observed between these items and the PPMQ-R subscale and Total scores. For example, the global rating of satisfaction with efficacy was correlated 0.93 with Efficacy subscale scores and 0.91 with the Total score on the PPMQ-R. Correlations between the global rating of satisfaction with side-effects and the subscale and Total scores were lower than correlations between other global ratings and the subscale and Total scores, perhaps due to the small number of AEs reported. These results suggest that the single-item scores may be useful as simple measures of treatment satisfaction for clinical practice or other studies where briefer measures may be needed. For clinical trial situations, it is recommended that the complete PPMQ-R be used, since multi-item subscales have better reliability, validity and responsiveness.
Findings from the AE analyses suggest that the PPMQ-R captures the side-effects most often associated with migraine treatment. Although AEs were not often reported, half of those reported were captured by the PPMQ-R Tolerability scale, particularly the nausea item, which was the most frequent AE reported. The Tolerability scale covers 64% of those AEs related to treatment, with an additional 23% related to pain, which, if due to headache, is covered by the Efficacy scale. Therefore, the PPMQ-R covered up to 87% of the AEs reported by migraine patients.
The PPMQ-R was developed for application in randomized clinical trials comparing different medication treatments for acute migraine headaches. Findings from this research confirm previous recommendation for use in clinical trials, where it is recommended that the Total score is used as the primary treatment satisfaction end-point because of its evidence on reliability, validity and responsiveness (from previous work), and because it covers key domains of importance to patients (16). The subscale scores can then be used to examine reasons for any observed differences in PPMQ-R Total scores, and to provide more insight into treatment effects. The PPMQ-R may be used in practice-based studies to gain information about satisfaction with different migraine treatments, and is also recommended for end-point evaluations in multiattack clinical trials of acute migraine therapy. The single item ratings of Overall Satisfaction, Efficacy and Side-effects can be used in practice settings or in naturalistic studies (i.e. large simple trials) to gather information on patient satisfaction with treatment. These single-item measures are not recommended as the only measures for clinical trials, since they are less sensitive to change and have less reliability than the multi-item subscale and Total scores.
Several potential limitations should be considered when interpreting the results of this study. First, the patient sample was taken from two clinical trials, therefore generalizability to the general population of migraineurs and to those with chronic headaches may be limited. The sample is representative of those migraineurs participating in clinical trials on migraine treatment, which is different from the general population of migraineurs, where approximately 50% do not consult a physician and only 10% receive triptan therapy (27). Second, study participants were receiving either placebo or sumatriptan/naproxen sodium, limiting generalizability across different treatments for migraine. However, previous research in community samples, which reflects standard treatment patterns, supports the psychometric characteristics of the PPMQ-R (16), and the current findings are comparable to those from a community setting. Although participants in this study were required to have had two to six migraines per month and previously used triptans and over-the-counter analgesics, there is no reason to believe that these characteristics would introduce any bias in the perceptions of the study participants regarding their ratings of treatment satisfaction for study medication.
In conclusion, the PPMQ-R demonstrated excellent internal consistency reliability and good concurrent and construct validity in measuring satisfaction with migraine treatment. These findings are consistent with previous psychometric research on this measure (16). Evidence of the psychometric properties of the PPMQ-R is important to support claims of treatment benefit for the Food and Drug Administration (28–30). Since this study was conducted only in the USA, research is needed to confirm these findings with migraine patients in other countries. The PPMQ-R is a useful measure for evaluating satisfaction with pharmacological therapy among migraine patients, particularly in clinical trials.
Footnotes
Acknowledgements
This study was funded by GlaxoSmithKline. The authors thank Frederick Derosier, DO for help developing adverse event categories.
