Abstract
Obesity, traditionally defined by the body mass index (BMI), is now recognized as a chronic, multifactorial disease characterized by excess adiposity that impairs organ function. The 2025 Lancet Commission advocates a function-oriented framework, moving beyond isolated BMI and incorporating measures of fat distribution such as waist circumference (WC) and waist-to-height ratio (WHtR). However, applying global criteria directly to the Indian population presents significant challenges. The prevalent “thin-fat” phenotype among Indians, high visceral adiposity, and insulin resistance at lower BMIs necessitate lower, ethnicity-specific anthropometric cutoffs to accurately predict cardiometabolic risk. Existing global BMI thresholds significantly underestimate obesity prevalence and risk in India, overlooking the metabolically obese normal-weight (MONW) phenotype, which affects 15%–30% of the population. A simplified, primary care-focused classification for India is proposed, combining BMI > 23 WHtR (>0.5) and the presence of complications. This approach aims to enhance early risk stratification and guide interventions, particularly in resource-constrained settings, while acknowledging the need for consensus and validation through large-scale, longitudinal Indian studies to fully optimize diagnostic and prognostic utility.
Keywords
For a practicing clinician, obesity represents a condition that is characterized by (a) excess of adipose tissue, (b) distributed appropriately with consequent (c) dysfunction, and (d) consequences or complications of such excess, distribution, and/or dysfunction (Figure 1).
The Components of Obesity Measurement.
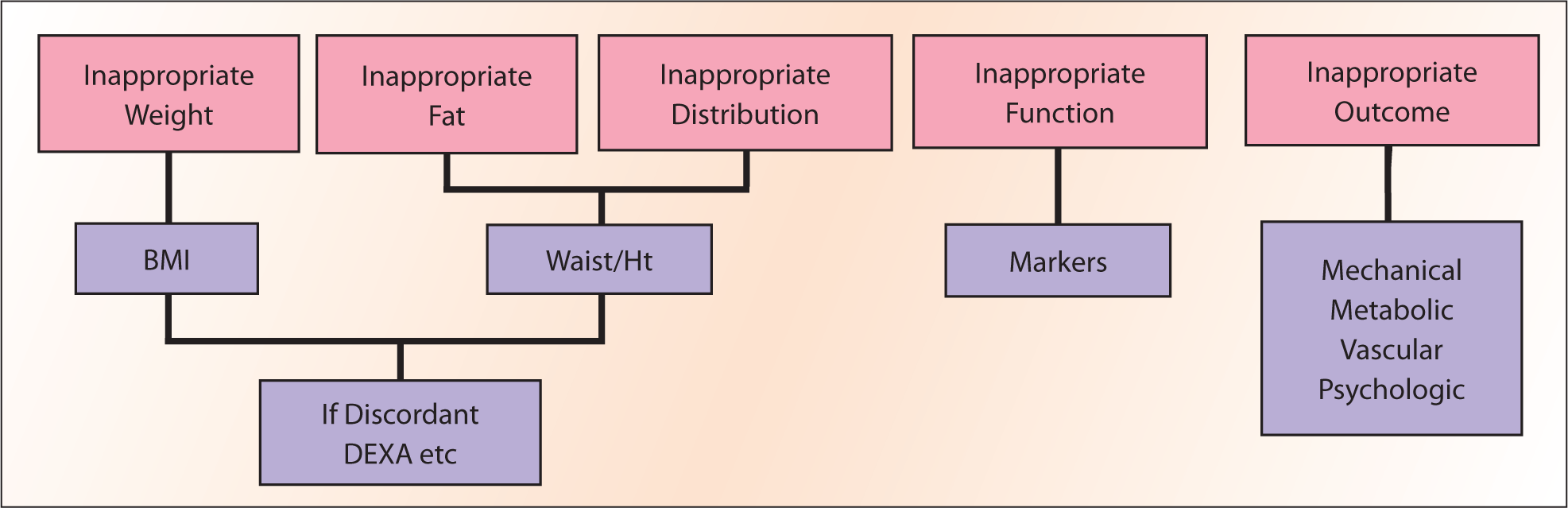
In the clinic, we use a flawed, albeit simple surrogate for excess fat, the body mass index (BMI). 1 While an approximation, the BMI is not a true representation of excess fat; in addition, it does not measure the inappropriate distribution or function. There are more sophisticated methods of measuring fat and its distribution with a reasonable degree of accuracy (such as the dual-energy X-ray absorptiometry), which are generally beyond the reach of the average clinic in most rural and urban settings. Traditional therapies often targeted modest reductions in weight and did little to reverse the consequences of fat excess, inappropriate distribution, and dysfunction; hence, the need to bring better measures of fat into the clinic was less acute. The emergence of therapies that reduce weight and fat by up to a quarter of body weight and alter distribution provides a much-needed impetus for adopting better measures of fat in clinical care. 2
The 2025 Lancet Commission on the Definition and Diagnostic Criteria of Clinical Obesity represents a paradigm shift by redefining obesity as a chronic, multifactorial disease characterized by excess adiposity that impairs organ function and diminishes quality of life.3,4 This report, formulated through evidence synthesis and expert consensus, critiques the longstanding dependence on BMI as the primary diagnostic metric, noting its inability to account for variations in fat distribution, muscle mass, and associated health risks across diverse populations. Instead of this approach, the commission advocates a function-oriented framework that differentiates preclinical obesity, defined by excess adiposity with intact organ function but heightened susceptibility to non-communicable diseases (NCDs), from clinical obesity, which entails adiposity-induced alterations in tissue or organ function leading to substantial daily limitations. Diagnostic confirmation of excess adiposity relies on anthropometric measures, such as waist circumference (WC) thresholds of ≥102 cm in men and ≥88 cm in women, waist-to-hip ratio >0.9 in men and >0.85 in women, or waist-to-height ratio (WHtR) >0.5, often combined with BMI or advanced techniques like dual-energy X-ray absorptiometry (DXA), whereas risk stratification for clinical obesity incorporates evidence of functional impairment through clinical signs, symptoms, or laboratory assessments. The recommendations of the commission are largely based on systematic reviews, observational studies, and randomized controlled trials (RCTs), with high consensus levels exceeding 80% on core propositions; however, it partially depends on expert opinion to address gaps in pediatric and ethnicity-specific data.3–5
Within the Indian context, where obesity intersects with persistent undernutrition against a backdrop of accelerated urbanization and dietary shifts, this framework provides critical insights yet exposes notable discrepancies in applicability.6,7 Epidemiological projections estimate that by 2040, overweight and obesity will affect 30.5% and 9.5% of Indian men, respectively, and 27.4% and 13.9% of women, with current urban prevalence rates reaching 20%–30% for general obesity and 23%–37% for abdominal obesity; there are significant regional variations, such as elevated figures in southern states. 8 The distinctive “thin-fat” phenotype prevalent among Indians, manifested by greater visceral adiposity, elevated body fat percentage, and insulin resistance at BMIs substantially lower than those observed in Caucasian populations, intensifies risks for type 2 diabetes mellitus (T2DM), cardiovascular disease (CVD), and metabolic syndrome, with these comorbidities clustering at BMIs 5–7 kg/m² below global benchmarks.9–12
While the Lancet commission’s prioritization of adiposity assessment over isolated BMI aligns with Indian physiological needs, discrepancies in measurement thresholds are evident: Global WC cutoffs of ≥102 cm in men and ≥88 cm in women systematically underestimate cardiometabolic risks in Indians, for whom lower thresholds of ≥90 cm in men and ≥80 cm in women more accurately predict adverse outcomes, as substantiated by epidemiological analyses involving more than 10,000 participants.13,14
Lacunae in the Lancet report’s evidentiary base further constrain its direct transposition to India. While the report’s meta-analyses robustly demonstrate BMI’s suboptimal correlation with adiposity across heterogeneous cohorts, its primary reliance on data from high-income countries underrepresents low- and middle-income countries (LMICs) such as India, where the dual burden of malnutrition impacts 9%–10% of children and adolescents. 15 The evidence supporting ethnic adaptations remains at a moderate level, predominantly derived from observational cohorts rather than LMIC-tailored RCTs, thereby potentially overlooking socioeconomic determinants, cultural dietary influences, and rural-urban disparities, including rural obesity rates spanning 8%–38%.8,16 For example, metabolically obese normal-weight (MONW) phenotypes, affecting 15%–30% of Indians and conferring risks comparable to overt obesity, may evade early identification under the commission’s preclinical categorization without localized thresholds.17,18 These evidentiary shortcomings emphasize the imperative for India-specific modifications, as articulated in the revisions suggested. 19 Moreover, T2D in India can even occur at normal BMI or even in underweight individuals with a BMI of 18.5 kg/m2as shown recently in a large Indian study. 20
In terms of risk stratification, the Lancet framework employs functional criteria to predict outcomes, yet its prognostic utility in Indian populations diverges due to phenotypic differences. Anthropometric indices exhibit variable predictive performance: WHtR surpasses BMI in forecasting T2DM and CVD among diabetic individuals, with area under the curve (AUC) values ranging from 0.795 to 0.804. Among Indian adolescents, BMI cutoffs of ≥22.8 kg/m² for boys and ≥20.5 kg/m² for girls, alongside WC thresholds of ≥82.5 cm for boys and ≥80.3 cm for girls, predict prediabetes with AUCs of 0.828–0.838. 21 For estimating 10-year CVD risks exceeding 10%, alternative metrics such as body shape index, body roundness index, and WHtR demonstrate superior AUCs of 0.80–0.85 compared to BMI in South Asian cohorts. Furthermore, metabolic obesity subtypes delineate distinct prognostic profiles: MONW (prevalence 43.3%) and metabolically obese obese (MOO; 28.3%) phenotypes correlate with heightened odds ratios for T2DM (1.5–2.0), coronary artery disease (1.3–1.8), and chronic kidney disease (1.4–2.1), underscoring MONW as a key driver of elevated T2DM incidence in Indians despite normal BMI ranges.18,22 Such stratification enhances outcome predictability, facilitating targeted interventions.
If adopted without adaptation, the global criteria would exert profound effects on the Indian population. Reclassification implications are substantial: Reliance on World Health Organization (WHO) BMI thresholds of ≥30 kg/m² underestimates obesity prevalence by 20%–30%, overlooking 15%–30% of MONW individuals who harbor equivalent cardiometabolic risks. 23 Comparative analyses indicate that Indians experience T2DM risks equivalent to those in White populations at BMIs of 23–25 kg/m² versus 30 kg/m², potentially reclassifying an additional 40%–50% of the population as obese and expanding the affected cohort to encompass 254 million individuals. 24 Therapeutic ramifications include deferred interventions for preclinical cases misidentified as low-risk, thereby exacerbating the NCD burden, while conversely risking overtreatment in metabolically healthy obese (prevalence 1.8%) subgroups through unwarranted pharmacotherapy or surgical referrals. 24 Moreover, the stipulated use of resource-intensive tools such as DXA would strain India’s primary care infrastructure, where 70% of healthcare encounters occur at grassroots levels with constrained diagnostic capabilities, thereby compromising access. 25
Consequently, tailoring the framework to Indian contexts is paramount, as exemplified by the 2025 revised guidelines from Misra et al. which incorporate staged definitions attuned to the thin-fat phenotype via Delphi consensus. 16 These guidelines designate stage 1 obesity as increased adiposity without dysfunction at BMI >23 kg/m², progressing to stage 2 with the addition of abdominal markers (e.g., elevated WC or WHtR) and either functional limitations (e.g., dyspnea, arthralgia) or comorbidities (e.g., T2DM, hypertension). This adaptation mitigates Lancet discrepancies by reducing BMI thresholds from ≥30 kg/m², bolstered by evidence that WHO criteria overlook 20%–30% of cases and early metabolic derangements. 26 To enhance predictability, ethnic-specific cutoffs are integrated, such as BMI 27 kg/m² for Asians equating to 32–35 kg/m² in Whites for comorbidity onset. 27
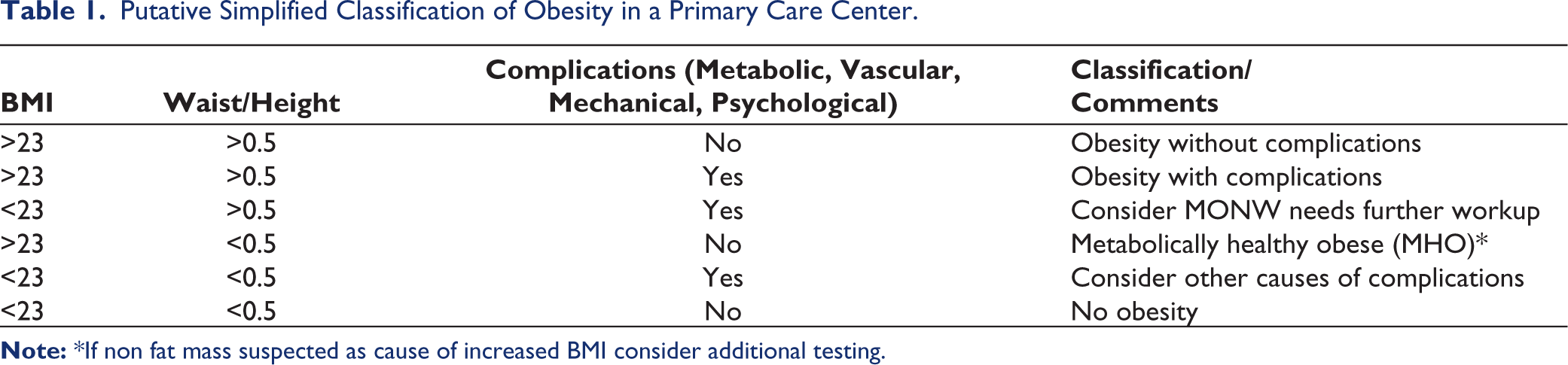
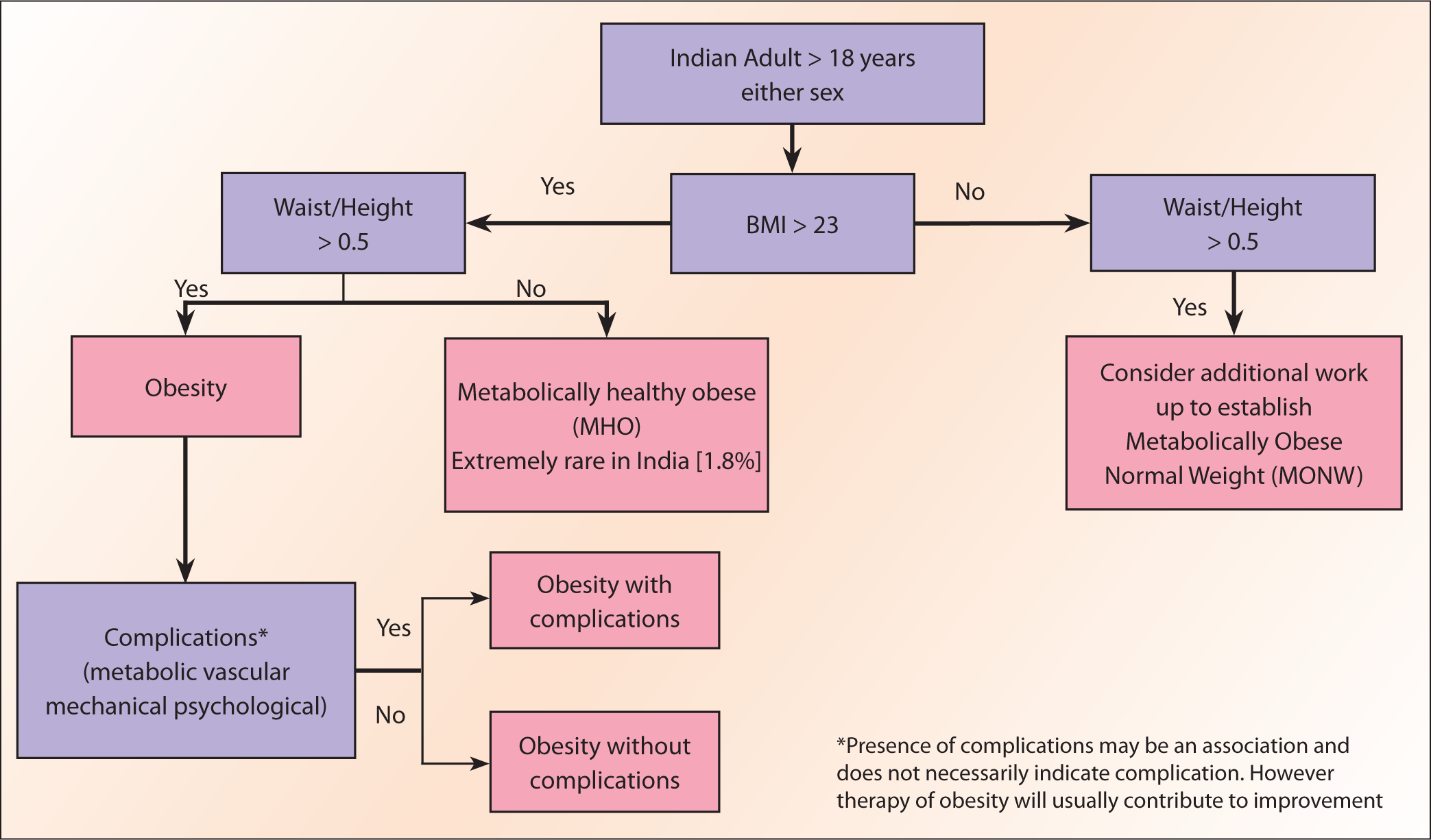
We believe that this could be further simplified for a primary care approach in India. Simplification for primary care involves BMI combined with WC or WHtR, eschewing DXA, and incorporating the presence or absence of comorbidities or complications. Metabolic profiling (e.g., lipid and glucose assessments) is advocated to detect MONW, thereby refining risk stratification. Building on Misra et al., a person with a BMI of >23 kg/m2 in association with a WHtR >0.5 should be classified as obese. A discrepancy in the above should trigger efforts at quantifying obesity with other testing. A patient with obesity could be further classified as one with complications (which could be metabolic, vascular, mechanical, or psychological), warranting mitigating efforts beyond lifestyle, or without complications, where lifestyle and attempts in prevention of progression to complications should be the thrust of care (Table 1 and Figure 2). Needless to add, this requires a consensus and a plan to validate such an approach with well-defined studies.
Putative Simplified Classification of Obesity in a Primary Care Center.
A Simplified Approach to Obesity for Primary Care.
Research gaps impede the full optimization of any of these approaches outlined. Existing data predominantly comprise cross-sectional and urban-centric studies, with scant large-scale RCTs evaluating the longitudinal predictability of staging systems in Indian cohorts, particularly neglecting rural MONW prevalence at 46%. Longitudinal investigations are required to assess anthropometric indices’ capacity to forecast long-term CVD and T2DM outcomes across diverse demographic strata. In LMICs, evidence deficiencies persist regarding ethnic adaptations and post-reclassification therapeutic efficacy. To address these, funding should prioritize national cohorts akin to the ICMR-INDIAB survey, emphasizing rural-urban and gender disparities, alongside studies on simplified tools’ comparative AUCs (e.g., WHtR versus BMI in primary settings) and gene-environment interactions.
In the meantime, professional societies such as the RSSDI should take a lead in simplifying the obesity conundrum for practice. Since the clinical care of obesity (in the presence or absence of diabetes) is increasingly likely to move into primary care settings, a consensus based on Indian data and evidence is essential if the obesity epidemic in India is to be nipped in the bud.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
