Abstract
Aims:
Type 2 diabetes (T2DM), along with menopausal status, can cause depression and anxiety and may also reduce their quality of life (QoL). The aim was to compare the comorbidities, diabetic complications, sleep quality and QoL in overweight and obese pre- versus post-menopausal women with T2DM.
Methods:
388 women were screened between January and October 2023 and divided into groups: G1: pre-menopause, G2: post-menopause. Socio-demographic, clinical, menstruation details, comorbidities and complications were recorded. Their sleeping pattern, anxiety and depression and QoL were assessed using the Pittsburgh Sleep Quality Index, Global Anxiety and Depression scales and WHOQoL, respectively.
Results:
68.3% of women were in post-menopausal status during the screening. Impaired sleep quality score was highly prevalent in G2 (56.1% vs. 32%, P = .001). Anxiety and depression scores were higher in G2 (39.6% vs. 25.2%, P = .006). The multivariable linear regression model revealed that sleep parameters such as subjective sleep quality score (β = 0.59, P = .001), sleep latency (β = 0.41, P = .03), use of sleep medications (β = 0.27, P = .012) and QoL domains including physical health (β = −3.65 P = .001), psychological health (β = −3.15, P = .001), social habit (β = −1.29, P = .004), environmental health (β = −3.88, P = .001) and higher anxiety (β = 1.05, P = .02) and depression scores (β = 1.04, P = .01) were significantly associated with post-menopausal women living with T2DM.
Conclusions:
The burden of comorbidities, complications of diabetes like diabetic neuropathy, impaired sleep quality and overall poor QoL was observed among overweight and obese post-menopausal women with T2DM. Future studies are warranted to understand the comprehensive inter-relationship between post-menopausal status, sleep health and psychological factors among women with diabetes.
Introduction
Type 2 diabetes (T2DM) is a silent epidemic that is on the rise in the current world. 1 Overweight and obese people have become more common in most countries in recent decades due to changes in lifestyle and economic development. The prevalence of diabetes has considerably increased as a result of these changes. 2 T2DM is the most frequent chronic illness among post-menopausal women and a contributing factor to cardiovascular diseases (CVD) and death. 3
Natural menopause, as defined by WHO, is considered a minimum of twelve months of amenorrhea without surgery or any physiological or pathological reason.4,5 Reduction of ovarian function after menopause is linked to increased abdominal obesity. 6 Progesterone and estrogen influence how the cells react to insulin. Those subsequent changes in the composition of the body can cause disruptions in insulin sensitivity and glucose metabolism in post-menopausal women. 7
Obesity has a significant impact on menopause and metabolic profile. An essential aspect of menopause is changes in sex hormones, especially serum estradiol, which might vary in women with and without obesity. 8 The decrease in estrogen during menopause affects metabolism and cardiovascular function, making it a risk factor for CVD. Menopause does, in fact, predispose women to the emergence of numerous conventional CVD risk factors. These include aberrant plasma lipids, impaired glucose tolerance, elevated blood pressure, elevated sympathetic tone, endothelial dysfunction, and vascular inflammation. They also involve alterations in the distribution of body fat from a gynoid to an android pattern. 9
Insomnia and sleep disruptions are frequent problems during the menopausal transition. Various studies found that around 14%–27% of post-menopausal women in developed and developing nations have problems with sleep disruptions.10–12 Women’s quality of sleep during the menopausal transition may also be negatively impacted by hormonal changes. Sleep difficulties are observed in roughly 42% of pre-menopausal women and 60% of post-menopausal women, primarily as a result of hot flashes and night sweats brought on by hormonal changes. 7
Women’s overall vulnerability is increased by the physiological, emotional, and psychological changes, which also lower their quality of life (QoL). Changes that occur during menopause can impede their adaptability to their new psychological and social status and also have an adverse effect on their QoL.13,14
Several studies suggest that people with diabetes are more liable to have feelings of anxiety or depression than those without diabetes.15,16 Additionally, it was reported that post-menopausal women have noticeably more symptoms of anxiety and depression than pre-menopausal women. 17 A multitude of comorbidities are associated with diabetes, such as mental and physical health problems. Diabetes, when coupled with anxiety and depression, can amplify the burden of diabetes and also worsen glycemic control, complications, increase non-adherence to medications, and reduce QoL.18,19 The post-menopausal era is a crucial time for women because of a number of endocrinological changes brought on by the ovaries’ decreased ability to produce estrogen, which can affect their sleep, QoL, mental health and make them prone to comorbidities. Hence, the primary objectives were to compare comorbidities, complications of diabetes and sleep quality among the overweight and obese pre- versus post-menopausal women. In addition, their mental health, with a focus on anxiety, depression and QoL, was also assessed.
Participants
Women with T2DM aged between 25 and 70 years were screened and included, and women with gestational diabetes, Type 1 diabetes, those who had normal BMI (<23 kg/m2), physically disabled, chronic illnesses, those who had undergone hormone replacement therapies, weight loss medications, especially GLP1-RA, as it can cause mood disturbances. Participants who were unwilling to give informed consent or refused to participate in the study were excluded.
Materials and Methods
A total of 388 women who attended a tertiary care center for diabetes in Chennai, South India, for their treatment between January and October 2023, participated in this cross-sectional study. The participants were enrolled based on the inclusion as well as the exclusion criteria of the study.
Sample size was calculated assuming a 50% prevalence of comorbidities among post-menopausal women with diabetes, with a 95% confidence level and 5% margin of error. The sample size was calculated to be 385.
Anthropometric, socio-demographic, clinical and bioch-emical details were collected using a semi-structured questionnaire. The recorded demographic details include age, place of dwelling, education, marital status, religion, occupation and their monthly family income.
Height, weight and waist circumference were the anthropometric measurements that were recorded for all the participants. A standard portable digital scale was used to measure the weight. A measuring tape was used to measure the waist circumference. Abnormal waist circumference for women was defined as ≥80 cm. 20 BMI (kg/m2) was calculated. Participants with a BMI between 23 and 24.9 kg/m2 and those with a BMI ≥ 25 kg/m2 were classified as overweight and obese, respectively. 21
Menstruation details included menarche age, history and duration of breastfeeding, number of children, type of menopause and duration of menopause. Information about menopause was assessed through open questions and was considered to be pre- or post-menopausal based on self-reporting of their last menstrual period.
Blood pressure was measured using a mercury sphygmomanometer. A systolic blood pressure >140 mm Hg and a diastolic blood pressure >90 mm Hg, or the usage of antihypertensive drugs, was defined as hypertension. Adult Treatment Panel III guidelines 22 were used to diagnose dyslipidemia; any self-reported history of the disease, which included treatment, was also considered dyslipidemia.
Comorbidities and presence of diabetic complications, treatment details and details on self-monitoring of blood glucose were recorded. Biochemical details like fasting and postprandial glucose, HbA1c, hemoglobin and the lipid profile were measured using fully automated biochemistry analyzers.
The tools for assessment of physical activity, sleep quality, anxiety and depression and QoL were translated to the participants’ local languages and administered by a trained interviewer.
The amount of physical activity of all the participants was determined using the Global Physical Activity Questionnaire (GPAQ) developed by WHO. The frequency, intensity, duration, and domains of PA and sedentary behavior of all the participants were assessed using GPAQ. Participants who performed 150 minutes of moderate-intensity aerobic PA or 75 minutes of vigorous-intensity aerobic PA or a combination of both throughout the week were classified as physically active. Activities that needed hard physical effort, which increase the heart rate or breathing, were considered vigorous-intensity, whereas activities that needed moderate physical effort and caused a mild increase in heart rate or breathing were considered moderate-intensity activities. These included workplace activities, travel to and from places or any recreational activities. The time spent while sitting or reclining on a typical day was assessed for sedentary behavior. 23
The sleep quality and disturbances for a period of one month were assessed using the self-rated Pittsburgh sleep quality index (PSQI) questionnaire. The sleep quality, duration, latency, sleep disturbances, habitual sleep efficiency, daytime dysfunction and the usage of sleeping medication were the nineteen self-reported items under seven subcategories that were used in this questionnaire. The PSQI questions were rated from 0 = no difficulty to 3 = severe difficulty, generating scores that correspond to the domains of the scale. The scores range from 0 to 21, and those with scores ≥5 were considered as having significant sleep disturbance. 24
The level of QoL was determined using the WHO quality of life (WHOQoL), which consists of 26 items. The first two questions are on overall QoL and health, the remaining 24 items are used to determine their perception of the QoL in four domains, which include physical, psychological, social and environmental health. Each item was scored between 1 and 5, and the higher score indicated better QoL. This scale assessed the subjective rather than objective life condition over the past two weeks. 25
Goldberg Anxiety and Depression Scale (GADS) is a tool which assesses the probability of anxiety or depression that might occur. In the two subscales present, the first subscale (questions from 1 to 9) was used for anxiety, and the second subscale (questions 10–18) was used for depression. Questions from 1 to 4 and 10 to 13 were conditioning questions. Two affirmative answers for the subscales 1–4 and one positive answer for subscales 10–13 were required to continue the subscale. The cut-off for the anxiety subscale is 4 or more, and 2 or more points for the depression. Higher point values indicate a more severe problem, with 9 as the highest possible value for each subscale. 26 Cronbach’s alpha was calculated to determine the reliability of the questionnaires and to estimate the internal consistency before performing the analyses. An alpha value of 0.83 was considered acceptable.
A 24-hour dietary recall questionnaire was used to capture detailed information about all food and beverages consumed by the participants in the past 24 hours.
Statistical Analysis
Mean (SD) for continuous variables and median (Min, Max) for skewed data were reported. Numbers and percentages were reported for categorical variables. Independent sample t-test, Mann-Whitney U test and Chi-square test were performed to evaluate the difference between the groups.
The multivariable linear regression model using menopausal status as the predictor variable was used to identify its association with the scores of sleep quality, anxiety, depression and QoL domains, adjusting for the key confounding variables such as age, residence (urban vs. rural), marital status, duration of diabetes, treatment, education and occupation. Regression coefficient (β), P values and 95% confidence intervals were reported. A P value of <.05 was considered statistically significant between the groups. The statistical analyses were performed using SPSS version 30.
Results
Nearly 68.3% of the women were in post-menopausal status during the screening. Table 1 exhibits the demographic and anthropometric measurements of the study groups. As expected, the median age was significantly higher in the post-menopausal group (57 vs. 45 years, P = .001). Most of the participants were from urban locations in both groups (pre vs. post; 78% vs. 74%). Illiterates and completed schooling were higher in the post-menopausal group (13.2 vs. 8.1, P = .146 and 65.3% vs. 52.8%, P = .019). Most of the participants completed graduation in the pre-menopausal group than the post-menopausal group (39% vs. 21.5%, P = .001). The proportion of homemakers, formal and informal workers was almost similar in both groups. Similar to the occupation status, there was no difference in the family income between the groups. Married women were higher in the pre-menopausal group, whereas widows were mainly presented in post-menopausal group (96.7% vs. 88.3%, P = .006 and 11.3% vs. 2.4%, P = .003). Overweight and obesity were similar between the study groups. Median BMI and waist circumference were also similar between the study groups.
Socio-demographic and Anthropometric Details of the Study Participants.
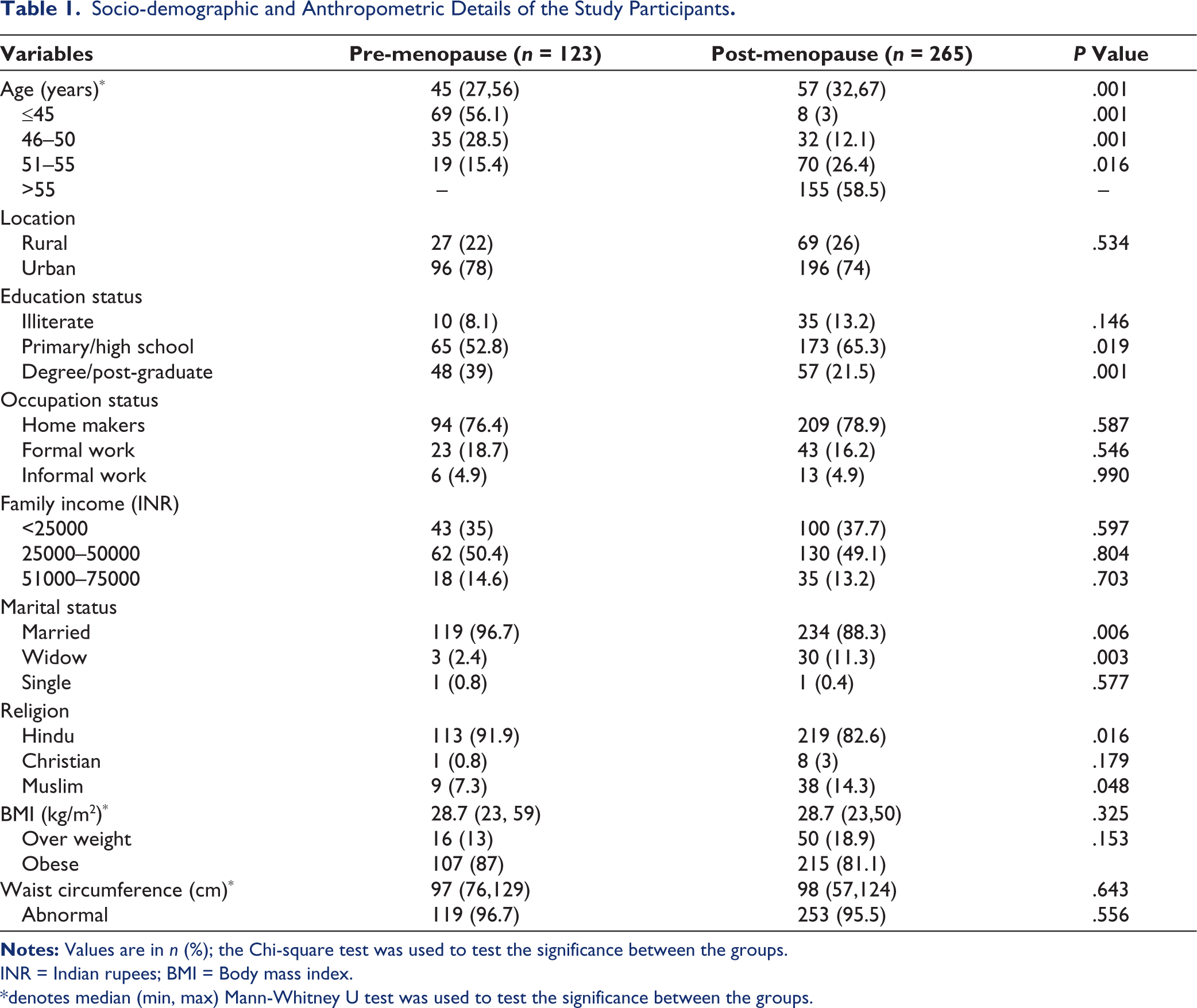
INR = Indian rupees; BMI = Body mass index.
* denotes median (min, max) Mann-Whitney U test was used to test the significance between the groups.
Table 2 depicts the comparison of clinical and biochemical profiles between pre- and post-menopausal women. The median duration of diabetes was significantly higher among the post-menopausal group. A higher proportion of the post-menopause women had a longer duration of diabetes of more than 15 years (22.3% vs. 8.9%; P = .001). The majority of the post-menopausal women were on a combination of OHA plus insulin, whereas those who were on only insulin therapy were significantly higher in pre-menopausal women. Regarding co-morbid conditions, both hypertension and dyslipidemia were significantly higher in post-menopausal women (58.1% vs. 38.2%, P = .001 and 71.3% vs. 57.7%, P = .008). Diabetic complications such as retinopathy, nephropathy, and cardiac problems were also higher among post-menopausal compared to pre-menopausal women, with no significant difference between the groups.
Comparison of Clinical and Biochemical Profiles Between the Study Groups.
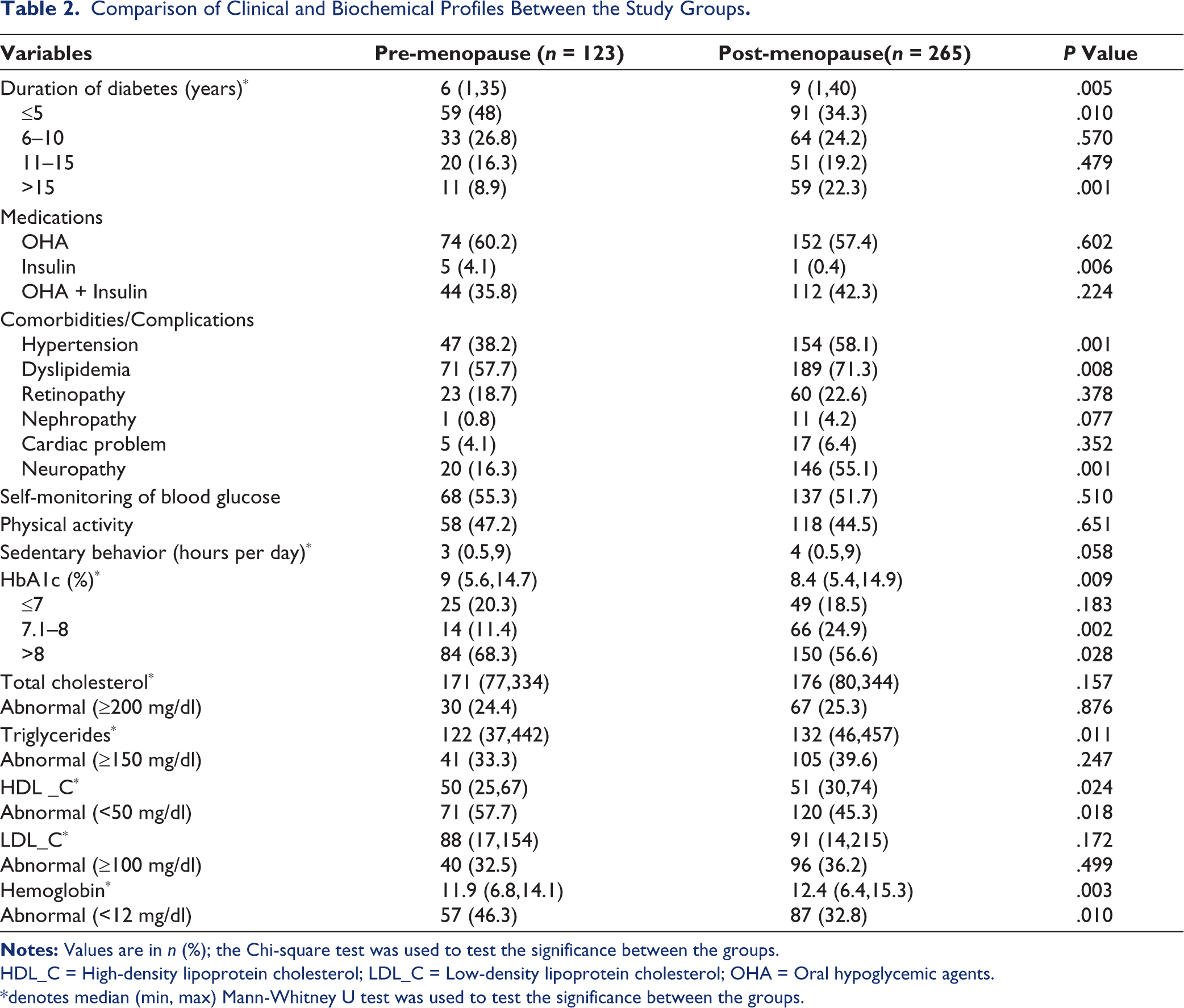
HDL_C = High-density lipoprotein cholesterol; LDL_C = Low-density lipoprotein cholesterol; OHA = Oral hypoglycemic agents.
* denotes median (min, max) Mann-Whitney U test was used to test the significance between the groups.
Neuropathy was significantly higher in post-menopausal women when compared to pre-menopausal women (55.1% vs. 16.3%; P = .001). The level of physical activity and self-monitoring of blood glucose were almost similar in both groups. In contrast, median hours of sedentary behavior per day were higher among post-menopausal women (4 vs. 3 hours, P = .058). Median HbA1c level was higher among pre-menopausal than the post-menopausal group (9% vs. 8.4%, P = .009). Median total cholesterol, triglycerides, and LDL-C were higher in post-menopausal women. Hemoglobin levels were significantly abnormal in the pre-menopausal group (46.3% vs. 32.8%, P = .01).
Mean menarche age was significantly lower in the post-menopausal group when compared to the pre-menopausal group (12.6 vs. 13.1 years, P = .006). The majority of women in the pre-menopausal group had a history of breastfeeding than the other group. The duration of breastfeeding was longer in post-menopausal women (1.5 vs. 1 year, P = .039). A higher proportion of the post-menopausal women were multi-parities and grand multi-parities (82.3% vs. 70.7%, P = .001 and 3% vs. 0.8%) while primi-parities were high in the pre-menopausal group (23.6% vs. 9.4%, P = .001). Among all post-menopausal women, 33.6% had undergone hysterectomy, and the majority (65.7%) of them had late menopause (Table 3).
Menarche, Breastfeeding and Menopause Details of the Study Participants.
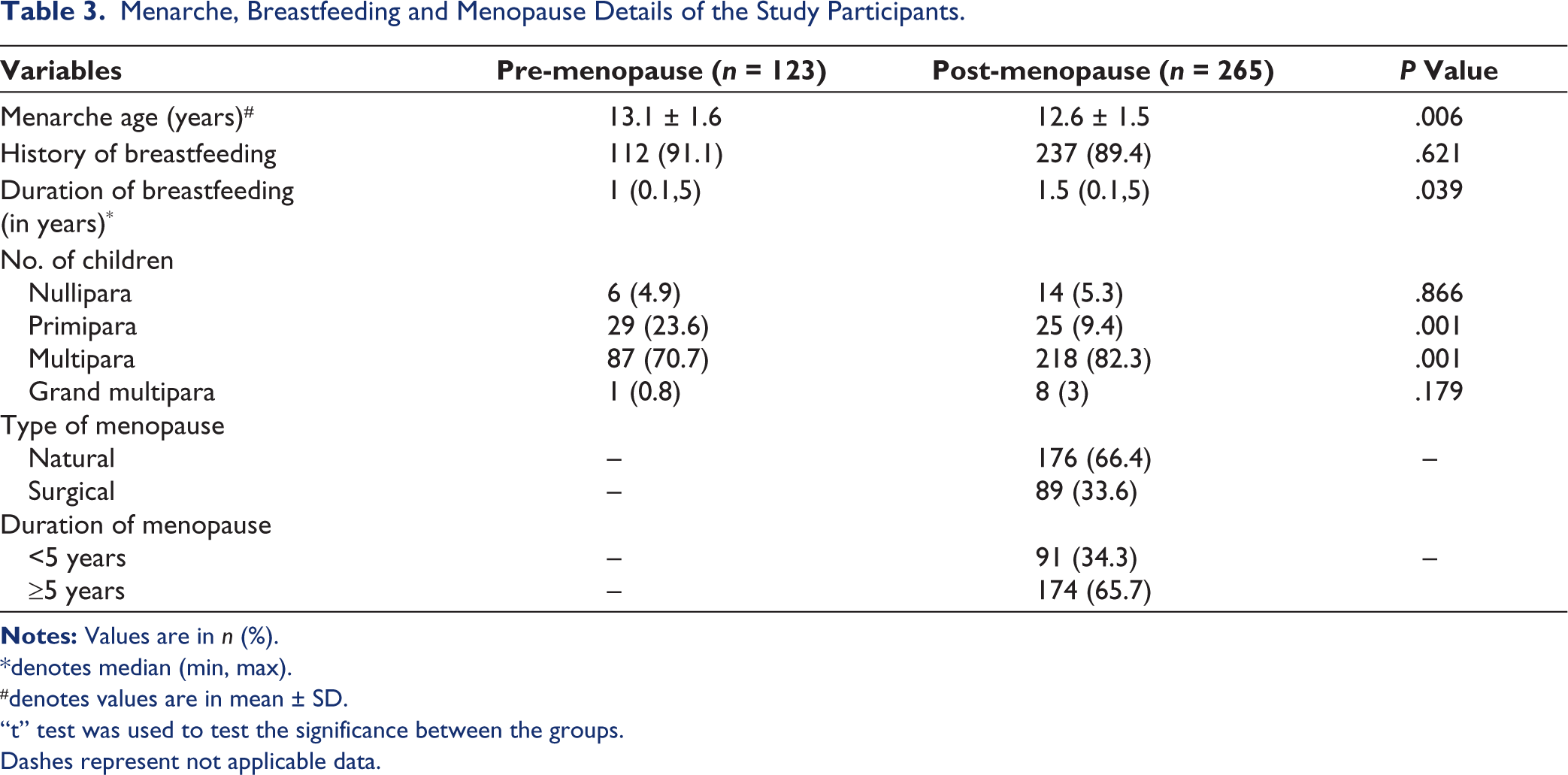
* denotes median (min, max).
# denotes values are in mean ± SD.
“t” test was used to test the significance between the groups.
Dashes represent not applicable data.
Table 4 depicts the sleep pattern of the study participants. Impaired sleep quality was higher in post-menopausal women when compared to pre-menopausal women (56.1% vs. 32%, P = .001). Global sleep index mean score was also significantly high among post-menopausal women (7.3 vs. 4.3, P = .001). Similarly, the average score of all seven components of sleep quality was significantly greater in post-menopausal women when compared to pre-menopausal women (sleep quality:1.5 vs. 0.7, sleep latency: 1.8 vs. 1.2, sleep duration: 1.2 vs. 0.9, habitual sleep efficiency: 0.8 vs. 0.3, sleep disturbance: 1 vs. 0.7, use of sleeping medications: 0.3 vs. 0.2, daytime dysfunction: 0.6 vs. 0.5).
Sleep Pattern Among the Study Participants.
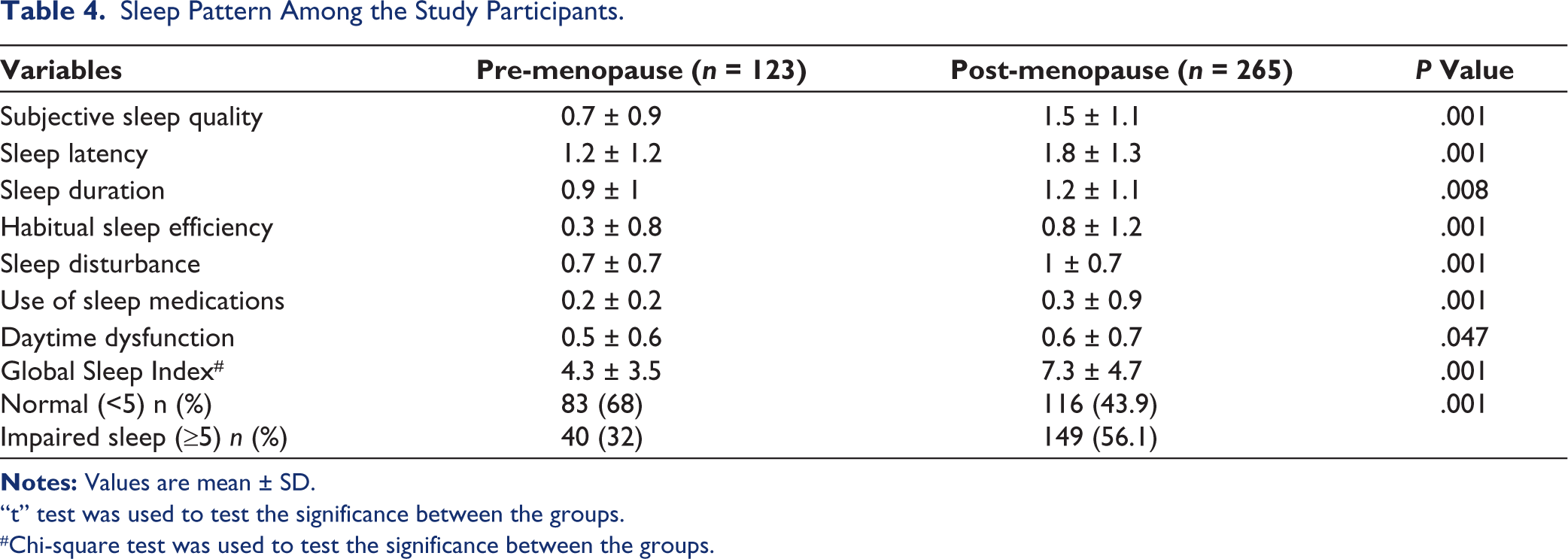
“t” test was used to test the significance between the groups.
#Chi-square test was used to test the significance between the groups.
The anxiety and depression status of the participants was shown in Figure 1. It was noted that a higher proportion (39.6%) of the post-menopausal women were anxious and depressed, in particular, 54.7% had anxiety and 52.8% had depression, while in the pre-menopausal group, only 25.2% were anxious and depressed (46.3% had anxiety and 37.4% had depression).
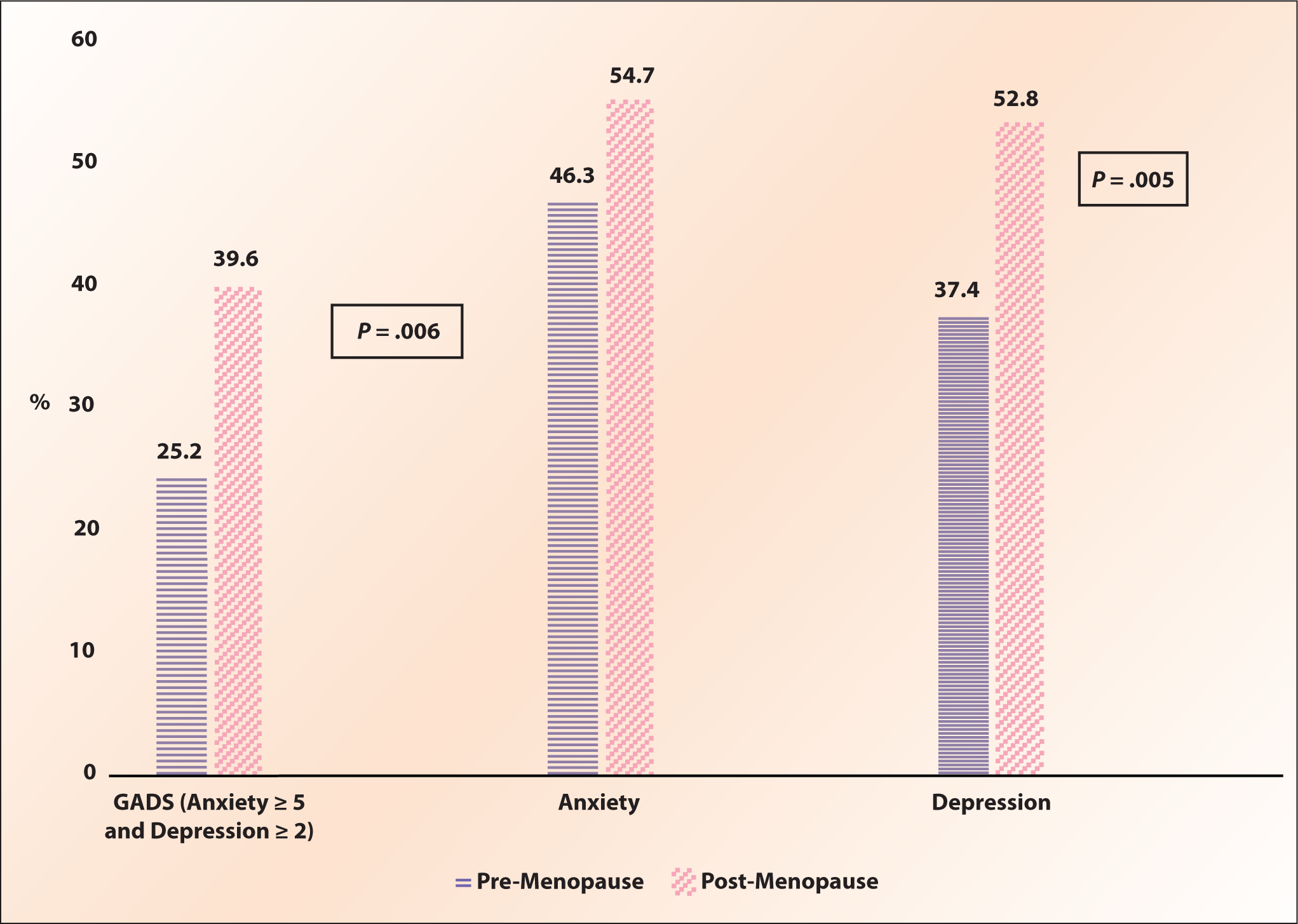
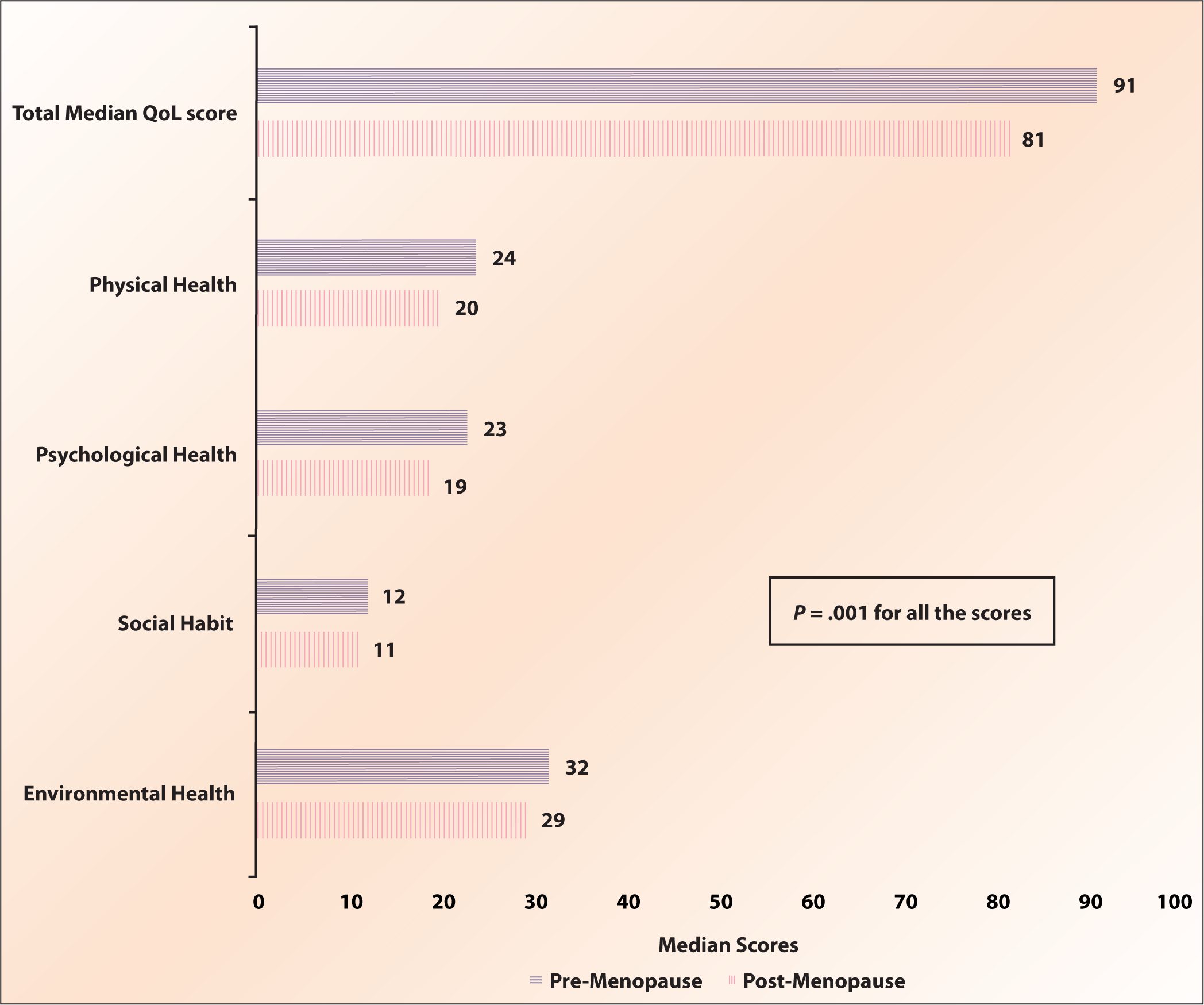
Figure 2 shows the comparison of QoL among the groups. According to the findings, there was a large difference observed in the median total QoL score and in all domains of QoL among post-menopausal women. The median total QoL score showed significantly lower scores in post-menopausal women than in pre-menopausal women. There were significantly lower median scores in all the QoL domains among post-menopausal women than their counterparts.
Quality of Life Between the Study Groups.
The results of the multivariable linear regression model are shown in Table 5. The sleep parameters such as subjective sleep quality score (β = 0.59, P = .001), sleep latency (β = 0.41, P = .03), use of sleep medications (β = 0.27, P = .012) and QoL domains including physical health (β = −3.65 P = .001), psychological health (β = −3.15, P = .001), social habit (β = −1.29, P = .004), environmental health (β = −3.88, P = .001) and higher anxiety (β = 1.05, P = .02) and depression scores (β = 1.04, P = .01) were significantly associated with post-menopausal status of women with T2DM.
Results of Multivariable Linear Regression Model.
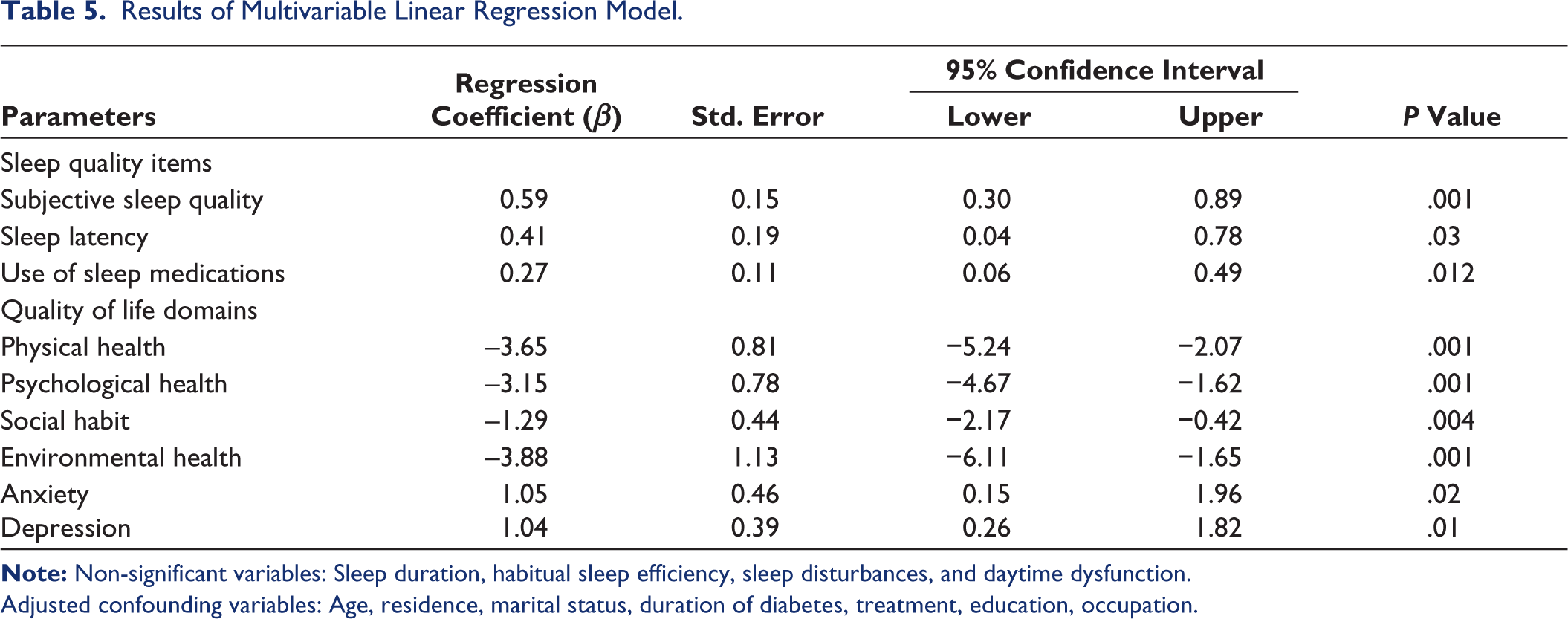
Adjusted confounding variables: Age, residence, marital status, duration of diabetes, treatment, education, occupation.
There was no difference in the total calorie intake between the two groups (1108 vs. 1099 Kcal). The percentage of carbohydrates, protein and fat was also similar between the groups. A higher percentage of post-menopause women followed an appropriate meal schedule as compared to pre-menopause women (36.1% vs. 27.8%). Adequate meal composition was similar between the groups (81.4% vs. 82%).
Discussion
Women, by entering the post-menopausal period, may experience several physiological and psychological changes that influence their QoL and general well-being. Comorbidities, diabetic complications, sleep quality, mental health and QoL in overweight and obese post-menopausal women were compared with pre-menopausal women with T2DM in this study. Around 68.3% of the women were in post-menopausal status during the screening process.
Almost all the women had abnormal waist circumference in both groups. The duration of diabetes was longer among post-menopausal women, with a higher proportion of them on combination therapy of oral hypoglycemic agents and insulin. This study found that the presence of co-morbid conditions such as obesity, hypertension (58%) and dyslipidemia (71%) and diabetic neuropathy was significantly higher in post-menopausal women. It was observed that pre-menopausal women had poor glycemic control. Our study findings highlighted that post-menopausal status was significantly associated with sleep health scores, anxiety, depression scores and domains of QoL, which included physical, psychological, social and environmental health.
Our study demonstrated that a higher proportion of the post-menopausal women had anxiety (54.7%) and depression (52.8%). Around 40% of the post-menopausal women had both anxiety and depression, whereas only 25.2% of pre-menopausal women had both anxiety and depression. Our findings were in agreement with a previous study done in Malaysian women. It was reported that around 11.8% and 8.4% of post-menopausal women with diabetes had depression and anxiety, respectively, 27 but the proportion of women with these symptoms was lower when compared with our study. In a very recent study, a significant association between depression and T2DM was observed in a large national data sample from South Korea. 28 Various studies suggested that during post-menopausal status, women are at an increased risk of depression and anxiety.27,29 The reason behind this would be psychosomatic symptoms. Hormonal changes associated with reduced estrogen have been found to be directly related, and depression may also be associated with decreased estrogen. Moreover, post-menopausal women no longer benefit from the protective properties of estrogen, and the symptoms are linked to estrogen deficiency.30,31
In addition to anxiety and depression symptoms, our study also revealed that post-menopausal women experienced poor sleep quality and decreased QoL. The sleep quality was highly impaired in post-menopausal women in our study. Around 56.1% of the post-menopausal women had poor sleep quality as compared to 32% of pre-menopausal women. To support our results, it was found that 56.3% of women in post-menopausal status were poor sleepers in an Iranian study. 32 Sleep quality was also observed among 30.2% post-menopausal women in the South Korean population. 33 The variations in the findings could be attributed to ethnic variations, biological mechanisms and psychosocial factors.
In our study, only 7.5% were on sleep medications in the post-menopausal group. Although the history of sleep medications was recorded, we did not collect data on specific drug classes like TriCyclic Antidepressants (TCA), Gamma Amino Butyric Acid (GABA), etc. Future studies stratifying participants based on the use of these medications may better delineate their effect on sleep and psychological disturbances. There is a link between higher BMI and poor sleep quality. 34 In the present study, the participants were obese and half of them had poor sleep quality. Changes in the hormonal levels and body fat distribution, along with psychological factors, can cause weight gain among middle-aged women. Among post-menopausal women in our study, the median time spent on sedentary behavior in a day was higher. Lack of physical activity may be another reason that causes obesity among them.
Lower median scores of all QoL domains in post-menopausal women indicated impaired QoL. Impaired QoL was significantly associated with post-menopausal status of women in the regression model. Overall QoL, psychological health and sleep health were affected in post-menopausal women in our study. Hormonal changes and other physical symptoms together affect their sleep pattern, QoL and make them more vulnerable to psychological illness and sensitive to negative emotional status. All these factors may lead women to experience comorbidities, depression, anxiety, and low QoL. 35 Another study conducted in South India compared QoL in post-menopausal women in urban and rural communities and reported no significant difference in the QoL. 36
The present study has a few limitations. Due to the cross-sectional nature of the study design, we could not evaluate the cause-and-effect association between diabetes and menopausal status. At the time of assessment, it is not necessary for all the participants to experience an anxiety attack since anxiety is considered episodic. We could not compare the findings among control women without diabetes. The selection of participants was from a tertiary care center, limiting the generalizability of the findings. The screen exposure before sleeping can have adverse effects on the onset and quality of sleep. However, we did not collect data on the use of gadgets or screen time in the present study, which may impact the sleep quality. Further research is needed to understand the inter-relationship between diabetes, obesity, post-menopausal status, depression, anxiety and sleep quality. It is necessary to explore potential causal relationships among the various factors studied, but it was not possible due to the cross-sectional design of the study. Another limitation is self-reporting of menopausal status without hormonal confirmation. Despite these limitations, the study has the strength of an overall assessment of post-menopausal women with diabetes in a larger sample size.
In conclusion, the study highlights the burden of comorbidities and the presence of diabetic neuropathy among overweight and obese post-menopausal women with diabetes, as compared to pre-menopausal women. Impaired sleep quality, anxiety, depression and overall poor QoL were observed in post-menopausal women. Pre-menopausal women had poor glycemic control. Our findings emphasize the burden of psychological disturbances and sleep among post-menopausal women living with diabetes, highlighting the need for future interventional studies to evaluate the need for targeted screening and strategies that may improve the QoL.
Footnotes
Acknowledgements
We thank all the participants who spent their valuable time and extended cooperation to participate in this study. The abstract of this article has been accepted for oral presentation and was presented at the 3rd RSSDI Research Retreat 2024, held in Jaipur on April 13–14, 2024. The accepted abstract has been published in the International Journal of Clinical Metabolism & Diabetes, August 2024, pp. 1–14. DOI: 10.1177/30502071241264885.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Institutional Ethics Committee (IEC/N-008/01/2023).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Written informed consent was procured from all the women who were willing to participate in the study. A legally acceptable representative signed the informed consent for illiterate participants.
