Abstract
Background:
To investigate the effects of personalized DiaBetes TEXT messaging combined with Peer Support Education (DB-TEXT+ PSE) on clinical outcomes in patients with type 2 diabetes.
Methods:
An assessor-blinded, three-arm randomized controlled trial recruited 84 participants between December 2022 and July 2023. Participants were randomly assigned to a DB-TEXT + PSE group, a DB-TEXT group, or a professional education program (PEP) group. Primary outcomes included glycated hemoglobin (HbA1c), fasting blood glucose (FBG) levels, and clinical remission rates, whereas secondary outcomes measured lipid profiles, fatigue, sleep quality, depression, and quality of life (QoL). Outcomes were assessed at baseline (T0), three months (T1), and six months (T2) postintervention.
Results:
Relative to PEP, personalized DB-TEXT + PSE and DB-TEXT alone led to significantly reduced HbA1c levels at T1 and decreased FBG levels at T1 and T2. Type 2 diabetes remission was achieved by 35.7% and 50% of the participants in the DB-TEXT + PSE group, by 17.9% and 25% in the DB-TEXT group, and by 3.7% and 3.7% in the PEP group at T1 and T2, respectively. Relative to the PEP, DB-TEXT + PSE led to significantly reduced TC levels, diastolic blood pressure (DBP), and systolic blood pressure (SBP) and improved sleep quality and QoL over time.
Conclusions:
Personalized DB-TEXT + PSE is more effective at enhancing clinical outcomes, reducing fatigue, improving sleep quality, and improving quality of life in patients with type 2 diabetes than DB-TEXT alone and PEPs are. Educators can incorporate personalized DB-TEXT + PSE as a component of diabetes education programs and thereby integrate digital interventions and supplementary programs.
Keywords
Introduction
Type 2 diabetes has become the most prevalent metabolic disorder in the world, and the incidence rate of the disorder is predicted to increase to 12.2% by 2045 for individuals aged 20 to 79 years. 1 People with diabetes and complications such as neuropathy, nephropathy, retinopathy, and cardiovascular disease 2 are prone to various other complications that can lead to a high financial burden 3 and poor quality of life,4,5 and these complications can cause sleep disorders, 6 fatigue,7,8 and psychological problems. 9 Identifying effective strategies for managing type 2 diabetes is of clinical importance.
Diabetes self-management education and support (DSMES) programs help patients with diabetes make better health decisions and effectively engage in self-care behaviors. Such programs typically consider the clinical, educational, psychosocial, and behavioral dimensions of care.10,11 The Association of Diabetes Care & Education Specialists (ADCES) identified seven self-care behaviors as essential components of DSMES. These behaviors comprise maintaining a healthy diet, exercising, monitoring blood sugar levels, taking prescribed medications, developing problem-solving skills, reducing risk, and engaging in healthy coping. 12 A meta-analysis reported DSMES to be effective in improving glycated hemoglobin (HbA1c) levels, quality of life, and depression outcomes in patients with type 2 diabetes. 13 Therefore, providing DSMES is critical for the management of type 2 diabetes.
Our previous component network meta-analysis revealed that two components—text messaging and peer support education (PSE)—were the most effective strategies within educational interventions, leading to improvements in HbA1c levels in individuals with type 2 diabetes. 14 Despite these promising insights, no randomized controlled trials (RCTs) have yet specifically investigated the effect of combining text messaging with PSE for this population. Moreover, previous studies have suggested that personalized interventions, offering tailored support based on each patient’s unique needs and preferences, may improve outcomes for individuals with diabetes.15,16 Therefore, in the present study, we conducted a randomized controlled trial to validate the findings of our preliminary study 14 and to explore the effects of personalized diabetes text messaging (DB-TEXT) combined with PSE on patients with type 2 diabetes.
Materials and Methods
Study Design
This study is an assessor-blinded, three-arm randomized controlled trial. The research was approved the Joint Institutional Review Board of the Ethical Committee of Medical Research at the Faculty of Dentistry in Jember University (No. 1738/UN25.8/KEPK/DL/2022) and Taipei Medical University (No. N202211055). We referenced the CONSORT guidelines for reporting our findings. 17 The study was prospectively registered on ClinicalTrials.gov under the identifier NCT05629117.
Participants and Study Settings
We recruited participants at a diabetes management center in East Java Province between December 2022 and July 2023. We included individuals who were identified as having uncontrolled type 2 diabetes (HbA1c level >7% in the past three months), who were aged 17 (the legal age for providing informed consent in Indonesia) to 65 years, and who had their own mobile phone. People who could not read or write Indonesian or had a diagnosis of cognitive impairment, a psychiatric disorder, or cancer prior to the study were excluded.
Sample Size Calculation
We estimated the sample size by using G Power 3.1 software, 18 with an effect size of 0.3, power of 0.80, and a two-tailed α of 0.05. We calculated the effect size with reference to a previous study in which text messaging was used as the intervention and the mean changes in HbA1c levels were considered. 19 We anticipated a 20% dropout rate and determined that a sample of 84 participants would be required.
Randomization, Allocation Concealment, and Blinding
Randomization was completed using an automated online system with a 1:1:1 allocation ratio (28 per group). Sealed, numbered envelopes ensured allocation concealment. Participants were assigned to groups after baseline assessment and eligibility confirmation. Outcome assessors were blinded until the study’s end, whereas patients and therapists were aware of group allocation due to the study design.
Intervention and Control
DiaBetes TEXT Messaging Combined with Peer Support Education
Patients in the DB-TEXT + PSE group received personalized text messages tailored to Indonesian adults with type 2 diabetes. These messages, crafted by experts, addressed each patient’s preferences and status regarding healthy eating, physical activity, blood glucose monitoring, and medication use. We considered the results of a previous study 20 and incorporated four components of the seven self-care behaviors reported by the ADCES (ie, maintaining a healthy diet, exercising, taking medication, and monitoring blood glucose levels) into the content of the text messages. 12 The content of the text messages was developed based on previous study protocols21,22 and in consultation with diabetes experts. Each message was no longer than 160 characters during the pilot study. The participants received personalized DB-TEXT twice weekly at approximately noon on Monday and Thursday over 12 weeks, manually delivered by our research team. Given that the participants were likely to have different dietary and physical activity preferences and to use different medications, we personalized the DB-TEXT considering their preferences and situations (Table 1).
The content of personalized DB-TEXT.
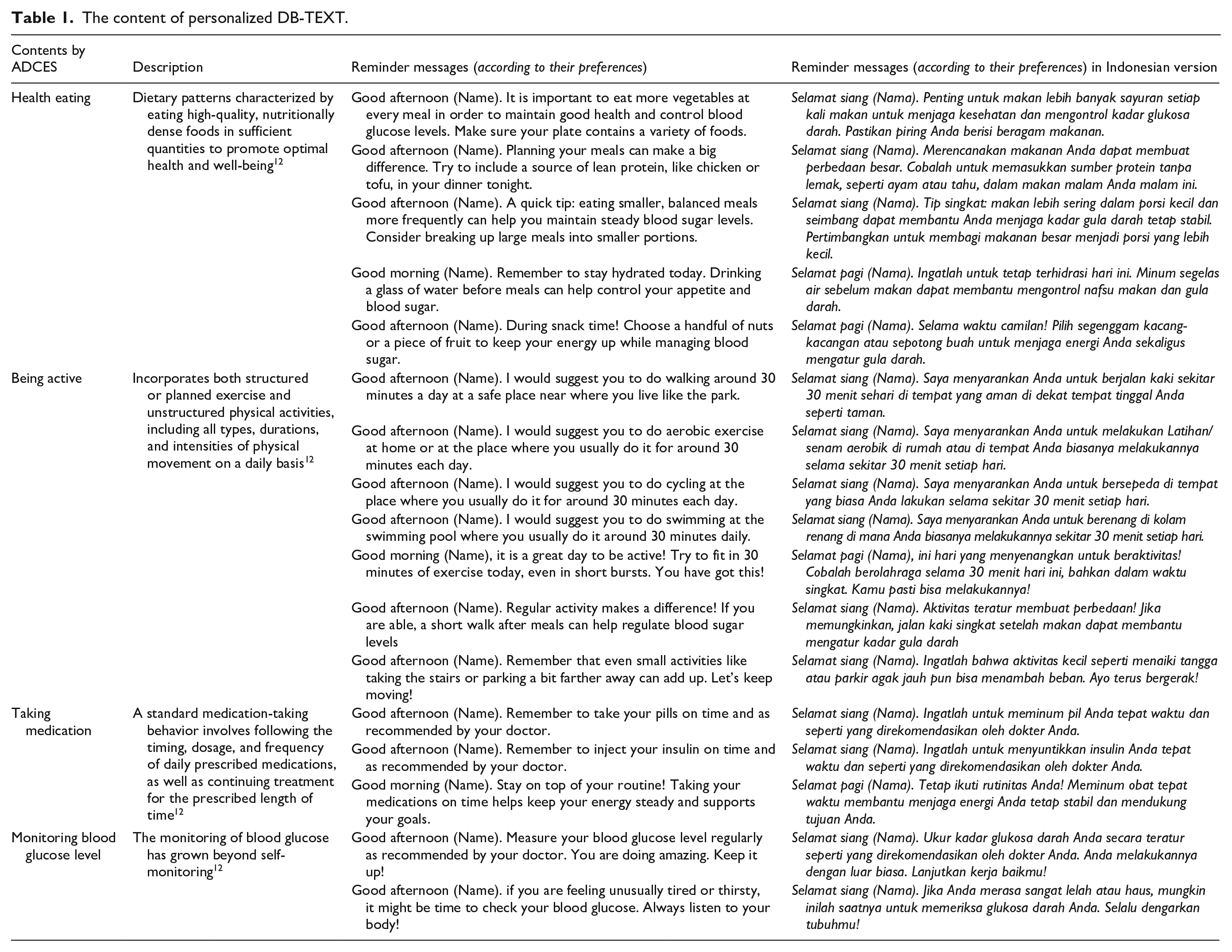
Before the PSE was provided, peer supporter training was implemented. The peer supporter training program was conducted for 30 hours, with the content of the program adapted from previous guidelines.23,24 The program was designed to equip peer supporters with the knowledge, skills, and strategies required to provide three key forms of peer support: (1) assistance in daily self-management, (2) social and emotional support, and (3) linkage to clinical care. Individuals were considered to be eligible to become peer supporters if they (1) had a diagnosis of type 2 diabetes, (2) were aged ≥17 years, (3) spoke Indonesian, (4) had a mobile phone, and (5) had self-reported HbA1c ≤7.0%. They were trained to motivate participants to make positive lifestyle changes, use active listening skills, apply empowerment-based facilitation strategies, set goals, develop action plans, and solve problems. We employed formative (ie, during the training) and summative (ie, after training) assessments. To graduate from the training program, candidates were required to achieve competency in the following five domains: (1) diabetes-related knowledge, (2) empowerment-based facilitation, (3) active listening, (4) goal setting and action planning, and (5) perceived self-efficacy.23,24 Upon successful graduation, the peer supporters received a stipend of $80 as compensation for their time investment and parking fees.
The participants were first introduced to the peer supporters at the diabetes management center, and the participants and supporters were matched on the basis of their sex and geographic location. The PSE was delivered once a week, lasting approximately 45 to 60 minutes, for a total of 12 sessions. Both the first and final sessions were conducted in-person at the management center, whereas sessions 2 through 11 were facilitated by peer supporters over the phone with their assigned partners. During the calls, the peer supporters shared their experiences and provided support related to managing diabetes regarding maintaining a healthy diet, exercising, taking medication, and monitoring blood glucose levels (Supplementary Table 1). The peer supporters were instructed to not answer questions regarding medical or clinical matters. Research assistants called each peer supporter weekly to evaluate and ensure the quality of the peer support program. Peer supporters informed the research team that all participants successfully completed all 12 sessions of the PSE program.
DiaBetes TEXT Messaging Group
The participants in DB-TEXT group received text messages twice weekly, on Monday and Thursday, over 12 weeks.
Professional Education Program Group
Health education related to diabetes management was provided to the participants in the Professional Education Program (PEP) group once per month for three months. This group received booklets regarding healthy eating and physical activity in the first month, medication adherence (eg, oral hypoglycemic agents and insulin injections) in the second month, and blood glucose monitoring in the third month.
Outcomes
Demographic and Disease Characteristics
Data on demographic and disease characteristics, including age, sex, education level, body mass index, marital status, and income level as well as current diabetes treatment, were collected using predesigned information sheets. These characteristics were reported to be associated with glycemic control in patients with type 2 diabetes. 25
Primary Outcomes
The primary clinical outcomes were HbA1c and fasting blood glucose (FBG) levels at three and six months after commencement of the study. The participants were instructed to measure their FBG level after not eating or drinking anything (except water) for at least 8 hours. Type 2 diabetes remission was defined if HbA1c levels were <6.5% for three months. 26
Secondary Outcomes
The participants’ total cholesterol (TC), low-density lipoprotein, high-density lipoprotein, and triglyceride levels were assessed at the diabetes management center during the study period. Systolic blood pressure (SBP) and diastolic blood pressure (DBP) were measured using a standard protocol with a standard digital sphygmomanometer. All outcomes were assessed at three and six months postintervention.
The participants’ fatigue levels were assessed using the Indonesian version of the Multidimensional Fatigue Inventory-20 (IMFI-20). 27 The IMFI-20 comprises 20 items and 4 subscales, namely general and physical fatigue, reduced motivation, reduced activity, and mental fatigue. Each subscale comprises four items rated on a five-point Likert scale with endpoints ranging from 1 (strongly agree) to 5 (strongly disagree). We reverse-scored 10 items with positive wording (ie, items 2, 5, 9, 10, 13, 14, 16, 17, 18, and 19). The score for each subscale (range = 4-20 points) was calculated as the sum of the scores of the items, and the total fatigue score (range = 20-100 points) was calculated as the sum of the scores of the subscales, with higher scores indicating a greater level of fatigue. The IMFI-20 had high validity and reliability (Cronbach’s α = 0.92).
Sleep quality was measured using the Pittsburgh Sleep Quality Index (PSQI). The PSQI can be used to assess self-reported sleep quality and sleep disturbance in the preceding month. The scale comprises 19 items and the following seven dimensions: (1) subjective sleep quality, (2) sleep latency, (3) sleep duration, (4) sleep efficiency, (5) sleep disturbance, (6) sleeping medication use, and (7) daytime dysfunction. The items are rated on a four-point Likert scale with endpoints ranging from 0 to 3, and the overall score ranges from 0 to 20. A score of less than five indicates favorable sleep quality. 28 The Indonesian version of the PSQI exhibited high validity and reliability (Cronbach’s α = 0.720). 29
Depression was assessed using the Beck Depression Inventory–Second Edition (BDI-II). The BDI-II comprises 21 items that are used to measure subjective depressive symptoms over the preceding two weeks. 30 Each item is scored on a four-point Likert scale with endpoints ranging from 0 to 3, with the total score ranging from 0 to 63. 30 The BDI-II includes cognitive, emotional, and somatic components. 31 The Indonesian version of the BDI-II exhibited good convergent validity as well as high internal consistency (Cronbach’s α = 0.90). 32
Quality of life was measured using the Diabetes Quality of Life–Brief Clinical Inventory (DQoL-BCI). 33 The DQoL-BCI comprises 15 items, each of which is assessed on a five-point Likert scale (1 = very dissatisfied/all the time, 2 = moderately dissatisfied/sometimes, 3 = neither/sometimes, and 4 = moderately satisfied/very seldom). The total score of the DQoL-BCI is calculated as the average scores of all items. Total scores range from 1 to 5, with 1 indicating worse quality of life and 5 indicating better. The Indonesian version of the DQoL-BCI exhibited good reliability, with a Cronbach’s α of 0.74. The content validity index scores for the items and scale were 0.80 to 1.00 and 0.90, respectively. 34
Statistical Analysis
The SPSS 18.0 was used for analysis, with significance set at 0.05. We used the Kolmogorov-Smirnov to test the normality of the distribution of the data; the data were normally distributed. To compare the baseline characteristics between the groups, one-way analysis of variance was conducted for examining continuous variables, and the chi-square test was conducted for analyzing categorical variables.
We utilized intention-to-treat (ITT) principle for outcome analyses. The ITT approach was applied to mitigate potential biases caused by nonrandom dropouts. Missing data were addressed using multiple imputation, assuming the data were normally distributed and missing at random. 35 Linear mixed models (LMMs) were used to compare changes in the primary and secondary outcomes among the three groups at baseline (T0), three months postintervention (T1), and six months (T2) postintervention. Repeated measures analysis of variance (ANOVA) and chi-square tests were utilized to examine changes in body mass index (BMI) and type of medication over time. Because BMI and medication usage change over time, we have included adjustments for both BMI and medication type in the LMM analyses.
Type 2 diabetes remission was evaluated at three months and six months postintervention by using the chi-square test and multivariable logistic regression. The completion rate was calculated by dividing the number of respondents who completed the three-month intervention by the number of respondents who started the intervention.
Results
Participant Characteristics
A total of 84 individuals with type 2 diabetes participated in the trial and were randomly allocated to the DB-TEXT + PSE group (n = 28), DE-TEXT group (n = 28), or PEP group (n = 28). Figure 1 presents the participant recruitment flowchart reported in accordance with CONSORT guidelines. One participant in the PEP group was excluded from the study due to traffic accident-related death; 83 participants completed the intervention.
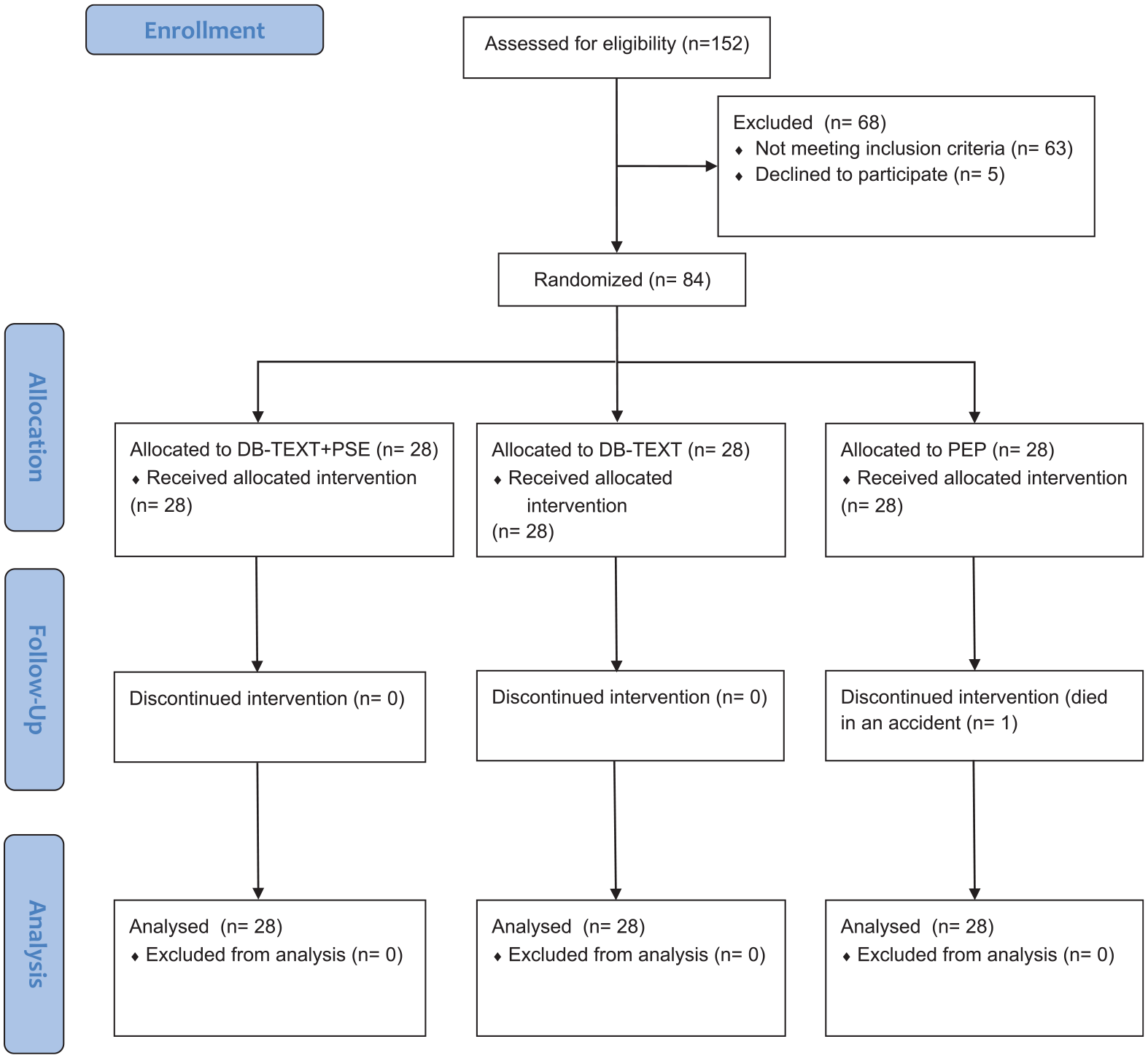
Consort flow diagram of the study. DB-TEXT + PSE; DiaBetes TEXT messaging combined with Peer Support Education, DB-TEXT; diabetes text messaging, PEP; professional education program.
As presented in Table 2, the mean ages of the participants in the DB-TEXT + PSE, DE-TEXT, and PEP groups were 54.5, 53.4, and 52.8 years, respectively. More than 60% of the participants were women. We did not identify intergroup differences in the clinicodemographic characteristics of the three groups at baseline (all P > .05, Table 2).
Demographic and disease characteristic of the participants (n = 84).
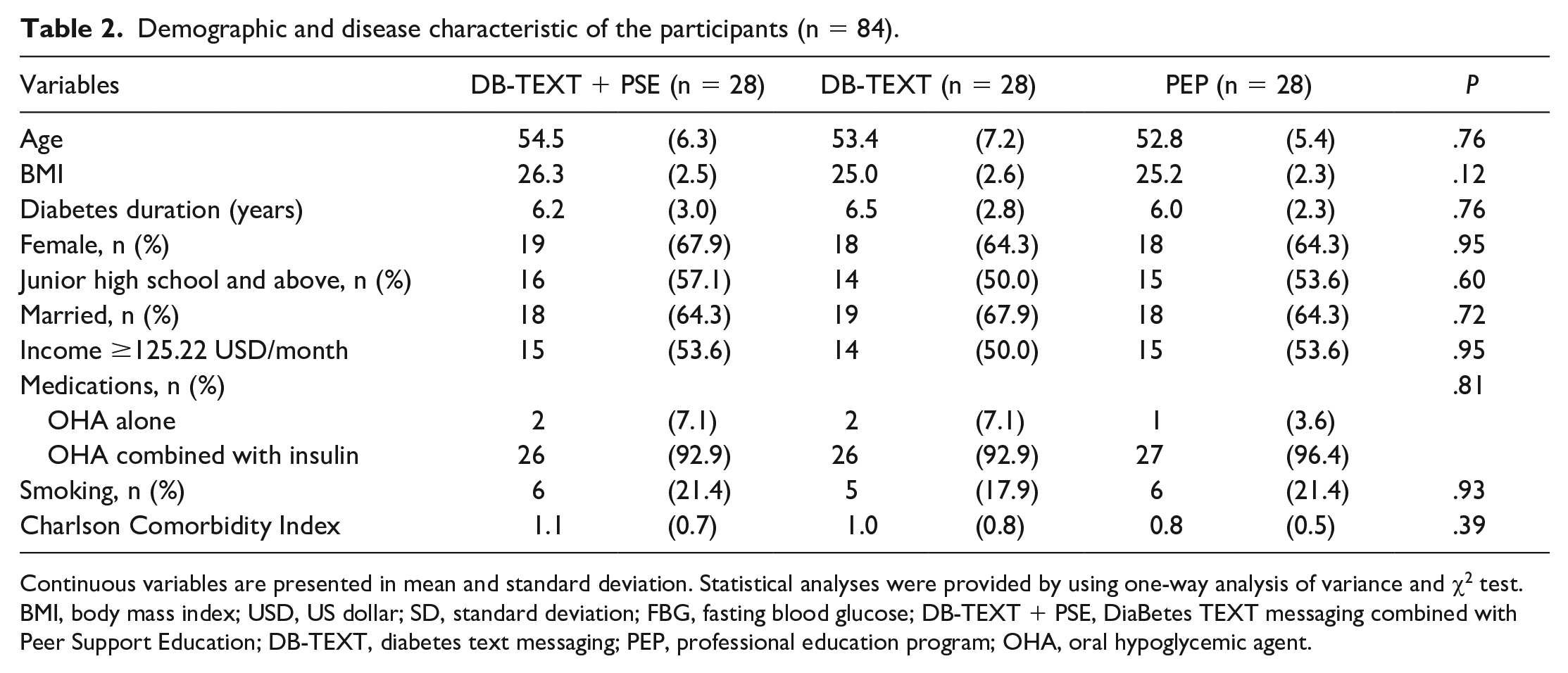
Continuous variables are presented in mean and standard deviation. Statistical analyses were provided by using one-way analysis of variance and χ2 test. BMI, body mass index; USD, US dollar; SD, standard deviation; FBG, fasting blood glucose; DB-TEXT + PSE, DiaBetes TEXT messaging combined with Peer Support Education; DB-TEXT, diabetes text messaging; PEP, professional education program; OHA, oral hypoglycemic agent.
In addition, we tracked changes in BMI, as well as adjustments in medication types and dosages among participants at three and six months postintervention, to assess their progress following the intervention (Supplementary Table 2). Our findings revealed that BMI and type of medications exhibited significant changes over time among three groups (P < .05).
Supplementary Table 3 presents the primary and secondary outcomes at baseline, three months postintervention, and six months postintervention. No significant differences were observed among the three groups regarding primary and secondary outcomes at baseline, supporting the homogeneity of the study population.
Treatment Effects on Primary Outcomes
The values of HbA1c, FBG, TC, SBP, DBP, fatigue, and quality of life significantly differed at the different time points (Supplementary Table 3, all P < .05). Table 3 presents the results of the LMM for HbA1c and FBG. Compared with the PEP group at baseline, the DB-TEXT + PSE group exhibited significantly lower HbA1c levels at three and six months postintervention (β = −1.14 and −1.37, respectively; both P < .001). The DB-TEXT group exhibited significantly lower HbA1c levels at three and six months postintervention (β = −0.51 and −0.62, respectively; both P < .001) relative to the PSE group at baseline.
Results of linear mixed models in the effects of DB-TEXT + PSE on primary outcomes (diabetes clinical outcomes) over the study period (n = 84).
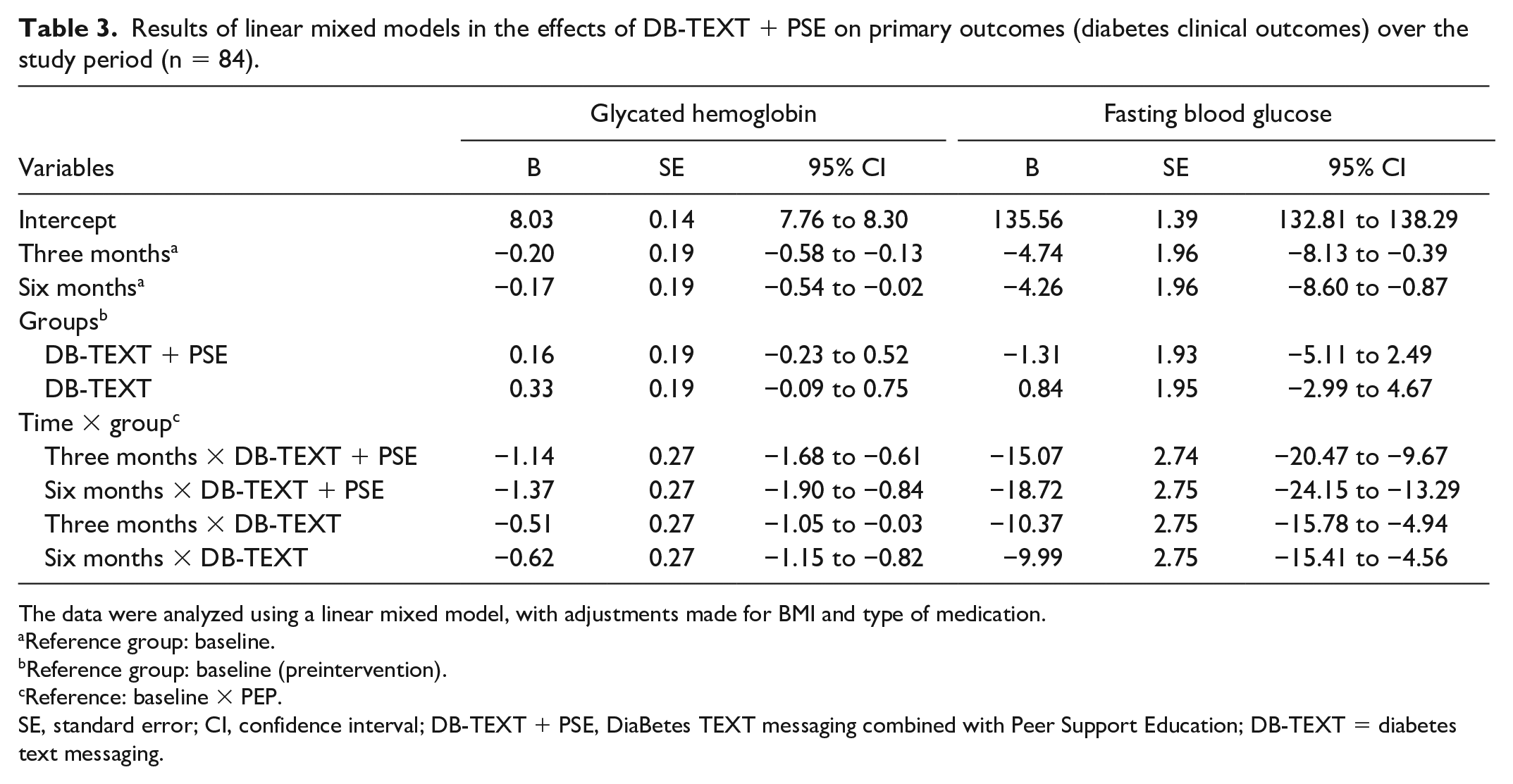
The data were analyzed using a linear mixed model, with adjustments made for BMI and type of medication.
Reference group: baseline.
Reference group: baseline (preintervention).
Reference: baseline × PEP.
SE, standard error; CI, confidence interval; DB-TEXT + PSE, DiaBetes TEXT messaging combined with Peer Support Education; DB-TEXT = diabetes text messaging.
The DB-TEXT + PSE group (β = −15.07 and −18.72, respectively; P < .001) and the DB-TEXT group (β = −10.37 and −9.99, respectively; P < .001) exhibited significantly lower FBG levels at three and six months postintervention relative to the PSE group at baseline.
Type 2 diabetes remission was achieved by 35.7% and 50.0% of the participants in the DB-TEXT + PSE group at three and six months postintervention, respectively. Only 17.9% and 25.0% of the participants in the DB-TEXT group achieved remission at three and six months postintervention, respectively, and only 3.7% and 3.7% of participants in the PEP group achieved remission at three and six months postintervention, respectively. Multivariable logistic regression revealed that the participants in the DB-TEXT + PSE group were more likely to achieve type 2 diabetes remission at three months and six months than those in the PEP group were (odds ratios = 14.44 and 26.01; 95% confidence intervals = 1.69-122.97 and 3.09-218.84, respectively). The DB-TEXT group did not achieve a significantly higher type 2 diabetes remission rate than the PEP group at three and six months postintervention (odds ratios = 5.65 and 8.67; P = .13 and .06, respectively).
Treatment Effects on Secondary Outcomes
Our results revealed that only the DB-TEXT + PSE group exhibited significantly reduced TC (β = −13.05 and −12.60, respectively), SBP (β = −5.49 and −6.39, respectively), and DBP (β = −2.15 and −2.71, respectively) at three and six months postintervention relative to the PEP group. The results also indicate a significant improvement in sleep quality from baseline to three months postintervention in the DB-TEXT + PSE group relative to the PEP group (β = −0.43). The DB-TEXT group exhibited significantly reduced SBP and DBP at three months, but not at six months, postintervention (β = −2.52 and −1.58, respectively; Supplementary Table 4).
The DB-TEXT + PSE group exhibited lower levels of fatigue (β = −5.58 and −4.51, respectively; both P < .001) and improved quality of life (β = 0.16 and 0.29, respectively; both P < .001) at three and six months postintervention relative to the PEP group at baseline. The DB-TEXT group had significantly lower levels of fatigue and significantly improved quality of life at three months, but not six months, postintervention (β = −1.35 and 0.05; P = .002 and .01, respectively; Supplementary Table 5).
Discussion
To the best of our knowledge, our study is the first randomized controlled trial to explore the effects of personalized DB-TEXT + PSE on patients with type 2 diabetes. Our findings revealed that, relative to the PEP group, the DB-TEXT + PSE group exhibited substantially improvement over time in HbA1c levels, FBG levels, TC, SBP, DBP, fatigue, sleep quality, and quality of life. In contrast, the DB-TEXT group only exhibited reductions in HbA1c and FBG levels at three and six months postintervention as well as reduced SBP, DBP, and fatigue at three months postintervention relative to the PEP group. Note that both intervention groups, but not the control group, exhibited downward trends in BMI and reduced diabetes-related medication dosages, which may further contribute to glycemic control.
These findings indicate that sending pertinent information and reminders through text messaging can promote a patient’s motivation to continue learning about their disease and to maintain the behaviors recommended by diabetes management guidelines.19,36 This study demonstrated that providing personalized DB-TEXT, in which the patient is referenced by name and the content is adapted to the patient’s preferences and diabetes management status, can substantially improve glycemic control. The personalization of the messages led the participants to pay closer attention to the reminder messages they received and to maintain their diabetes care, which helped them to maintain glycemic control.
This study provides robust evidence indicating that the DB-TEXT + PSE intervention is more likely to have immediate and lasting effects on remission among patients with type 2 diabetes than DB-TEXT alone, revealing that PSE has a critical influence on the outcomes of type 2 diabetes. Peer supporters having similar life experiences to those of their matched partners (patients with type 2 diabetes) may influence the success of PSE because it leads to mutually beneficial relationships. 37 In the present study, by providing consistent PSE, the participants received positive support from peers with similar disease backgrounds and conditions, which made the participants more confident in continuing their diabetes care. This may be because the peer supporters shared their own strategies for effectively managing their diabetes and discussed the challenges they faced in maintaining glycemic control.
Peer support is crucial to maintaining diabetes self-management. 38 The participants in the DB-TEXT + PSE group had lower TC, SBP, and DBP levels at three and six months postintervention than those in the PEP group did. This finding indicates that adding a PSE component to DB-TEXT is effective for improving TC and blood pressure control in individuals with type 2 diabetes. In previous studies, PSE alone was reported to lead to similar results.39,40 Managing blood lipid levels and blood pressure is essential for reducing the risk of diabetes complications.41,42 The findings of the present study indicate that improving glycemic control can lead to a better lipid profile and more favorable blood pressure in people with type 2 diabetes, which is in line with the findings of previous studies.43,44 Further studies are required to investigate the exact mechanism connecting glycemic control, lipid levels, and blood pressure among patients with type 2 diabetes.
The current study discovered that compared with PEP, personalized DB-TEXT combined with PSE led to significantly reduced fatigue levels at three and six months postintervention and improved sleep quality at three months postintervention. By contrast, compared with PEP, DB-TEXT alone only significantly reduced fatigue levels at three months postintervention. This finding supports that PSE components are crucial to managing fatigue levels and sleep quality in patients with type 2 diabetes. Glycemic control may be associated with the fatigue level and sleep quality.27,45,46 Therefore, achieving glycemic control might improve fatigue symptoms and sleep quality in patients with type 2 diabetes.46-48 In the present study, the personalized DB-TEXT + PSE intervention yielded greater improvement in glycemic control than the other interventions did, indicating that this intervention may improve fatigue symptoms and sleep quality.
In the present study, we further discovered that the DB-TEXT + PSE group exhibited significantly improved quality of life at three and six months postintervention relative to the PEP group. The DB-TEXT alone intervention improved quality of life at three months, but not at six months, postintervention. Patient empowerment involving diabetes self-management education and self-care behaviors is essential for enhancing quality of life among patients with type 2 diabetes. 49 Improvements in diabetes care knowledge were reported to positively affect quality of life in patients with diabetes. 50 Furthermore, providing PSE had the positive effect of improving quality of life in the patients with type 2 diabetes. 51 This may be because both diabetes knowledge and psychological support, such as that from peers, were required for improving the patients’ quality of life, which may explain why DB-TEXT alone did not lead to significantly improved quality of life at six months postintervention. By combining a text messaging intervention with a peer support program, we were able to provide our participants with a comprehensive understanding of diabetes.
Strength and Limitation
The current study had several strengths. First, our study was the first RCT in evaluating the effect of DB-TEXT + PSE in improving diabetes clinical outcomes, fatigue, sleep, depression, and quality of life in patients with type 2 diabetes. Second, this study had high completion rate of the participants. In addition, this study had a number of limitations. First, since our participants were recruited from a single diabetes management center, the external validity of the study findings was restricted. Second, given the constraints of limited budget and manpower, we were unable to carry out follow-up assessments to determine the long-term effects of DB-TEXT + PSE on clinical outcomes. It is recommended that further research be undertaken to comprehensively examine the long-term impact of DB-TEXT + PSE on diabetes clinical outcomes, fatigue, sleep, depression, and quality of life among patients with type 2 diabetes.
Conclusions
Compared with DB-TEXT alone and PEP alone, personalized DB-TEXT + PSE is more likely to lead to type 2 diabetes remission and to enhance glycemic control in patients with type 2 diabetes. Furthermore, compared with PEP, personalized DB-TEXT + PSE effectively enhances TC, SBP, sleep quality, fatigue, and quality of life in patients with type 2 diabetes. Diabetes educators can incorporate personalized DB-TEXT + PSE into diabetes education programs. This approach can assist individuals with type 2 diabetes in maintaining optimal glycemic control, reducing fatigue, improving sleep quality, and improving their overall quality of life.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251314501 – Supplemental material for Effects of Personalized DiaBetes TEXT Messaging Combined with Peer Support Education on Patients With Type 2 Diabetes: A Randomized Controlled Trial
Supplemental material, sj-docx-1-dst-10.1177_19322968251314501 for Effects of Personalized DiaBetes TEXT Messaging Combined with Peer Support Education on Patients With Type 2 Diabetes: A Randomized Controlled Trial by Debby Syahru Romadlon, Hui-Chuan Huang, Yu-Chi Chen, Sophia H Hu, Rudy Kurniawan, Tri Juli Edi Tarigan, Safiruddin Al Baqi, Faizul Hasan and Hsiao-Yean Chiu in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
None.
Abbreviations
ADCES, the Association of Diabetes Care & Education Specialists; BDI-II, the Beck Depression Inventory–Second Edition; BMI, body mass index; DB-TEXT, DiaBetes TEXT messaging; DB-TEXT + PSE, DiaBetes TEXT messaging combined with Peer Support Education; DBP, diastolic blood pressure; DQoL-BCI, the Diabetes Quality of Life–Brief Clinical Inventory; DSMES, Diabetes Self-Management Education and Support; FBG, fasting blood glucose; HbA1c, glycated hemoglobin; HDL, high-density lipoprotein; IMFI-20, the Indonesian version of the Multidimensional Fatigue Inventory-20; LDL, low-density lipoprotein; LMM, linear mixed models; PSE, peer support education; PSQI, the Pittsburgh Sleep Quality Index; QoL, quality of life; RCT, randomized controlled trial; SBP, systolic blood pressure; TC, total cholesterol.
Author Contributions
DSR and H-YC designed the study. DSR, H-YC, H-CH, Y-CC, SHH, RK, TJET, SAB, and FH performed the interventions, investigations, and project administration. DSR and H-YC were responsible for data analysis, drafting, and editing the manuscript. All authors have reviewed and approved the final version of the manuscript. DSR and H-YC are accountable for the integrity of the project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants awarded to HYC from the National Science and Technology Council, Taiwan (NSTC 113-2628-B-038-003-MY3and NSTC 111-2314-B-038-033-MY3).
Data Availability Statement
The data of this study are available from corresponding author upon request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
