Abstract
Purpose:
This study aimed to evaluate the safety and efficacy of Empagliflozin (Diampa→) alone & with Metformin (Diampa-M→) in patients with Type 2 Diabetes Mellitus (T2DM).
Methods:
This open-label, prospective, observational study was conducted in three hospital-based settings in South Asia. A total of 215 patients aged 18–65 years, with T2DM and HbA1c levels between 7.0% and 10%, were enrolled. The participants were monitored over 6 months, with follow-up visits at 1, 3, and 6 months. Primary outcomes included clinical and laboratory parameters such as weight, blood pressure, HbA1c, fasting blood sugar (FBS), and renal function markers. Secondary outcomes focused on adverse events and treatment discontinuation.
Results:
Significant improvements were observed in clinical and laboratory parameters. Median weight decreased from 75 kg to 74 kg (P = 0.000), and systolic and diastolic blood pressure improved significantly (SBP: P = 0.003, DBP: P = 0.022). Glycemic control also improved, with HbA1c reducing from 8.75% to 7.90% (P = 0.000), and FBS decreased from 149.0 mg/dL to 132.0 mg/dL (P = 0.000). Renal function markers showed mixed results: serum creatinine decreased significantly (P = 0.003), and estimated glomerular filtration rate (eGFR) increased from 109.0 to 112.4 mL/min/1.73 m² (P = 0.000). The frequency of adverse events, including hypoglycemia, dehydration, and urinary tract infections, decreased over time. Serious adverse events were reported in five participants at 1 month, 4 at 3 months, and 3 at 6 months. Treatment discontinuation was observed in four participants at 1 month, 1 at 3 months, and none at 6 months.
Conclusions:
Empagliflozin (Diampa→) alone & with Metformin (Diampa-M→) was effective in improving glycemic control, weight, and blood pressure in South-Asian patients with T2DM. The drug demonstrated a generally favorable safety profile, with adverse events decreasing over time.
Keywords
Introduction
Sodium-glucose cotransporter 2 (SGLT2) inhibitors provide substantial cardiovascular protection and kidney benefits for patients who have Type 2 diabetes mellitus (T2DM) or other similar conditions. Systematic reviews and meta-analyses prove that these drugs lower mortality from all causes as well as cardiovascular disease and heart failure hospitalization risks.1,2 The positive effects of SGLT2 inhibitors match those observed in both patients with kidney issues and cardiac diseases, even if they do not have diabetes. 3 Clinical research shows that these inhibitors help patients lose weight while lowering blood pressure, reducing the risk of acute kidney failure, improving the urinary albumin-creatinine ratio and maintaining generally positive safety results, except they increase genital infection rates.4,5
Empagliflozin exhibits positive safety characteristics when used as an SGLT2 inhibitor to treat patients who have T2DM. Empagliflozin compared to placebo caused equivalent adverse event frequencies among treatment groups in trials which evaluated more than 15,000 patients.6,7 Empagliflozin meets the requirements for daily administration with limited reactions between medicines. 8 It demonstrates a beneficial safety relationship for patients with T2DM based on existing trial results,7,8 while future cardiovascular outcome trials will help assess additional potential benefits.
Research conducted on SGLT2 inhibitors throughout Pakistan reveals that these drugs improve blood sugar control while aiding patients with Type 2 diabetes to reduce their weight and maintain safety. The observational studies demonstrated important findings through their assessment of HbA1c levels, BMI measurements and serum creatinine assessment.9,10 Systematic research with meta-analysis revealed no increased dangers of acute kidney failure or diabetic ketoacidosis (DKA) or urinary tract infections (UTIs) aside from dapagliflozin, nor bone fractures or lower limb amputations relative to placebo-based or active treatment. 11 Post-marketing studies have revealed risks related to SGLT2 inhibitor use, which include DKA alongside UTIs and cancer and bone fractures and foot and leg amputations. 12 Additional research must evaluate how SGLT2 inhibitors affect the long-term safety profile when used by Pakistani patients.
Although the safety and efficacy of Empagliflozin and Metformin have been established in Western and East Asian populations, data from Afghanistan and neighboring South-Asian regions remain scarce. Variations in genetics, diet, and healthcare access may influence treatment outcomes, justifying population-specific evaluation. To our knowledge, no previous post-marketing surveillance or real-world evaluation has been conducted in Afghanistan to assess Empagliflozin with or without Metformin. Such localized evidence is essential for guiding safe and cost-effective diabetes management in resource-limited South-Asian settings. This study aims to determine both the risk profile and the treatment effectiveness of Empagliflozin (Diampa→) alone & with Metformin (Diampa-M→) for T2DM patients.
Materials and Methods
Study Design and Setting
An open-label, prospective, observational, single-arm, multi-center, post-marketing surveillance study was conducted at three hospital-based settings from August 7th, 2023 to December 15th, 2024. Participants were monitored during follow-up visits at 1, 3, and 6 months.
Ethics
The study protocol was approved by the Institutional Review Board (IRB) [Ref# A.0823.416; Dated: August 6, 2023] and complied with the Declaration of Helsinki. Written informed consent was obtained from all participants prior to their inclusion in the study. Participant confidentiality and data protection were maintained in accordance with applicable regulations.
Participants
Inclusion Criteria
Both male and female patients aged 18–65 years, diagnosed with T2DM for at least 1 year, and with HbA1c levels between 7.0% and 10% were included. Patients were required to have been uncontrolled on oral antidiabetic agents and lifestyle modifications for at least 3 months, with an estimated glomerular filtration rate (eGFR) > 60 mL/min/1.73 m², and be naive to Empagliflozin. Additionally, participants had to be willing to provide written informed consent.
Exclusion Criteria
Exclusion criteria included a history of recurrent UTIs or genital fungal infections, severe hypoglycemia (<40 mg/dL), renal or hepatic dysfunctions with abnormal laboratory findings, hospitalization for DKA or hyperosmolar hyperglycemic state in the past 3 months, acute coronary syndrome or stroke within the last 3 months, pregnancy or lactation, and any clinical or social condition compromising patient safety.
Variables
Primary Outcomes
Clinical and laboratory parameters such as weight, blood pressure (systolic blood pressure [SBP], diastolic blood pressure [DBP]), HbA1c, fasting blood sugar (FBS), renal function markers (serum creatinine, blood urea nitrogen [BUN], eGFR).
Secondary Outcomes
Adverse events, treatment discontinuation, and serious adverse events (SAEs).
Data Sources/Measurement
Data were collected at baseline and follow-up visits (1, 3, and 6 months) using case report forms (CRFs). The parameters recorded included demographics, clinical and laboratory parameters. Blood pressure was measured using standard sphygmomanometry, while laboratory parameters were assessed via blood tests (HbA1c, FBS, serum creatinine, BUN, and eGFR). Adverse events were recorded through participant self-reporting and medical review at each follow-up visit.
Bias
To minimize bias, the study employed standardized procedures for patient enrollment, data collection, and laboratory testing. Consecutive sampling ensured uniform participant selection, and investigators were trained to uniformly assess AEs and outcomes.
Study Size
A sample size of 139 was calculated using WHO software Sample Size Determination in Health Studies, with a confidence level of 95%, a margin of error of 5%, and a reported ADR proportion of 8.5%. 13
Statistical Methods
The collected data were analyzed using SPSS version 22.0. Continuous variables were presented as mean and standard deviation, while categorical variables were reported as frequencies and percentages. A test for normality was performed to assess the distribution of variables. The Wilcoxon signed-rank test was applied for non-normally distributed variables to compare changes from baseline to the 6-month follow-up. Statistical significance was set at P < 0.05.
Results
A total of 215 participants were enrolled in the study, with a mean age of 50.82 years (SD = 9.11). The majority of participants were female (62.3%, n = 134), while males comprised 37.7% (n = 81). The smoking status indicated that 97.2% (n = 209) were non-smokers, and only 2.8% (n = 6) were smokers. Regarding education, 3.7% (n = 8) had completed graduate or higher education, 61.9% (n = 133) had secondary or below education, and 34.4% (n = 74) were uneducated or had no formal education. Co-morbid conditions were prevalent among participants, with hypertension observed in 40.5% (n = 87), dyslipidemia in 34.0% (n = 73), ischemic heart disease in 0.9% (n = 2), and chronic kidney disease in 0.5% (n = 1). Other co-morbidities were present in 2.8% (n = 6) of the participants (Table 1).
Baseline Characteristics of Study Participants (N = 215).

Medication details were recorded for all participants. Among those receiving Empagliflozin with Metformin (Diampa-M→) (n = 180), the most common dose was 12.5 mg + 500 mg, accounting for 35.8% (n = 77). Other dosages included 12.5 mg + 1000 mg (25.1%, n = 54) and 12.5 mg + 850 mg (10.7%, n = 23). The majority of these participants took the medication once daily (46.0%, n = 99), while 37.7% (n = 81) took it twice daily. For participants on Empagliflozin alone (n = 35), 13.5% (n = 29) were prescribed a 10 mg dose, and 2.8% (n = 6) received a 25 mg dose (Table 1).
The frequency of adverse events varied across follow-up visits. During the first follow-up (1 month), the most common adverse events included hypoglycemia (7.4%, n = 16), flatulence (7.9%, n = 17), and nausea (7.4%, n = 16). By the third follow-up (6 months), the frequency of adverse events such as dehydration, nausea, and UTIs decreased significantly (Table 2). SAEs were reported as follows: Five cases during the first follow-up, four cases during the second, and three cases during the third follow-up (Figure 1). Treatment discontinuation was observed in four participants at the first follow-up, 1 participant at the second follow-up, and none at the third follow-up (Figure 2).
Frequency of Adverse Events During Study Follow-up.
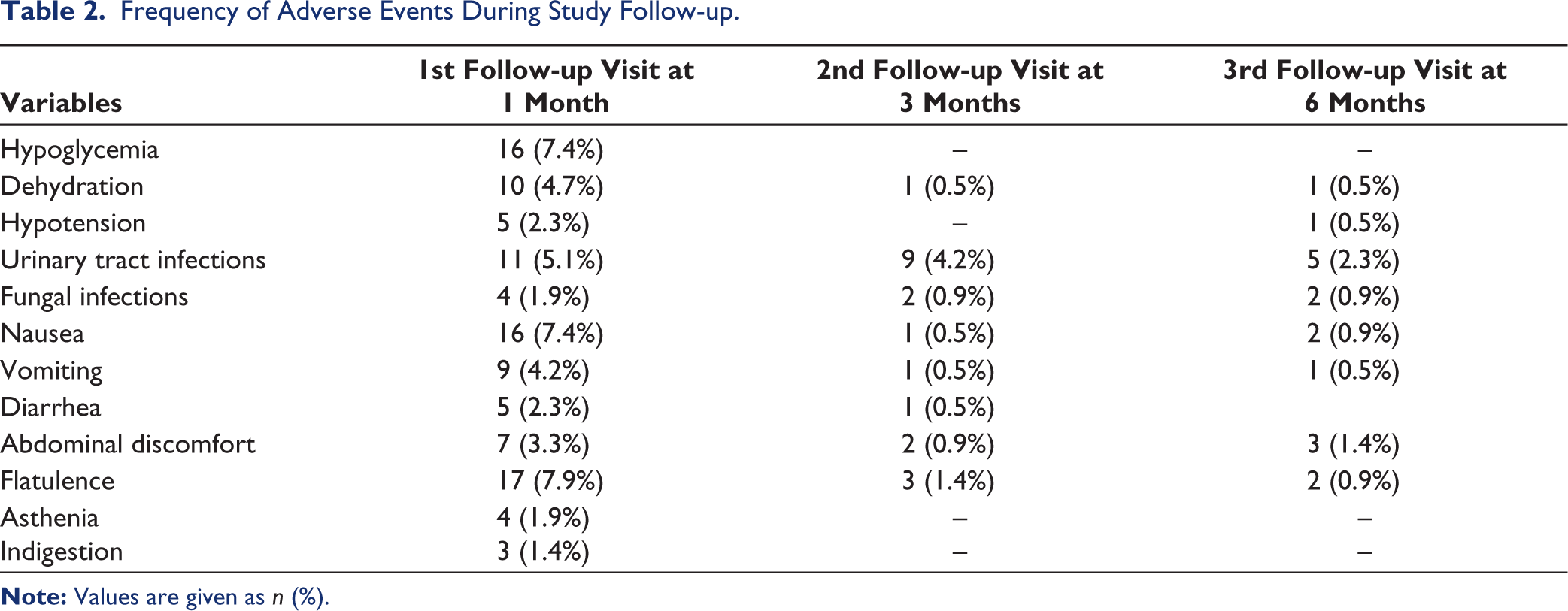
Frequency of Serious Adverse Event (SAE) Across Study Visits.
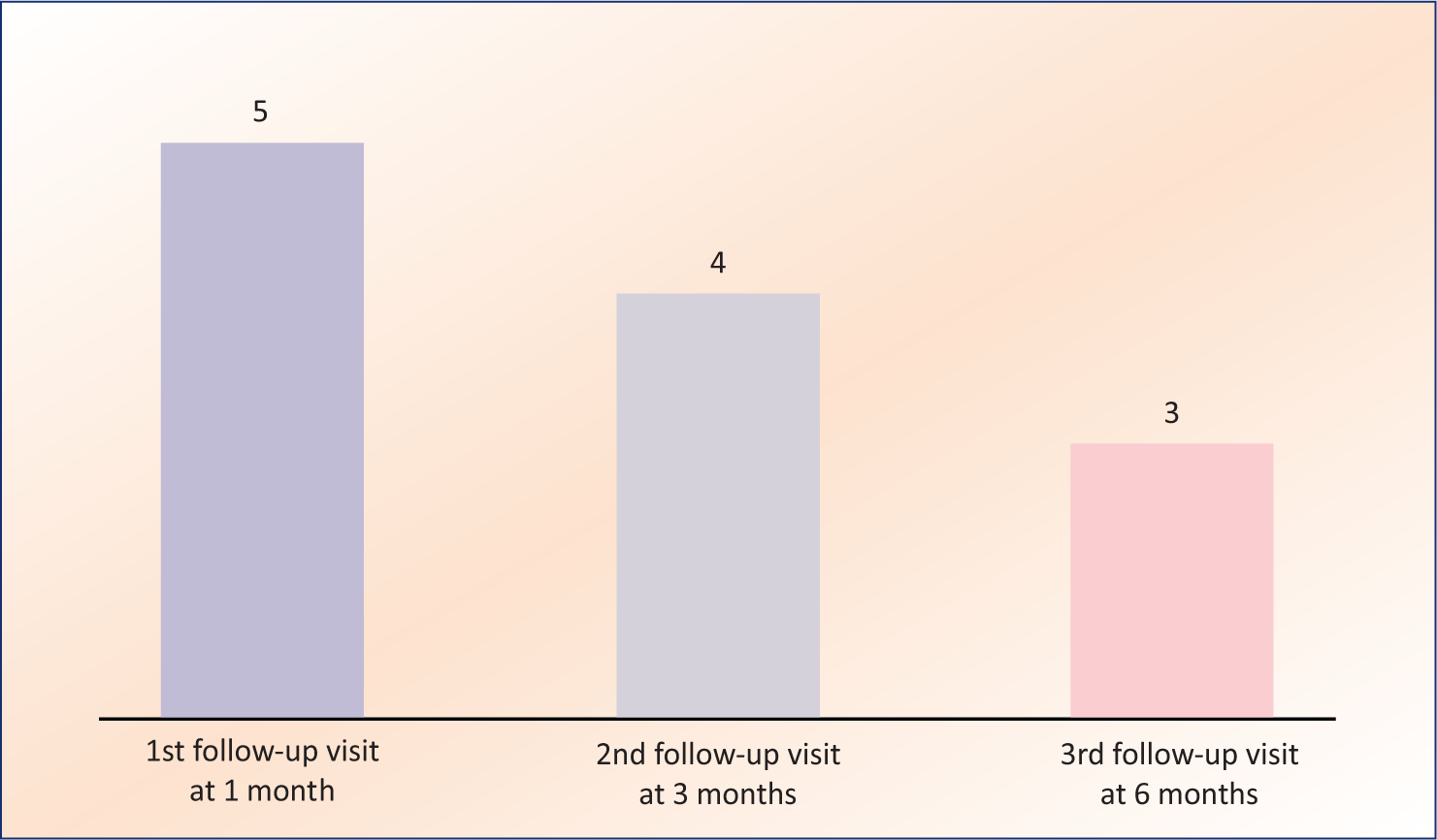
Frequency of Treatment Discontinuation Across Study Visits.
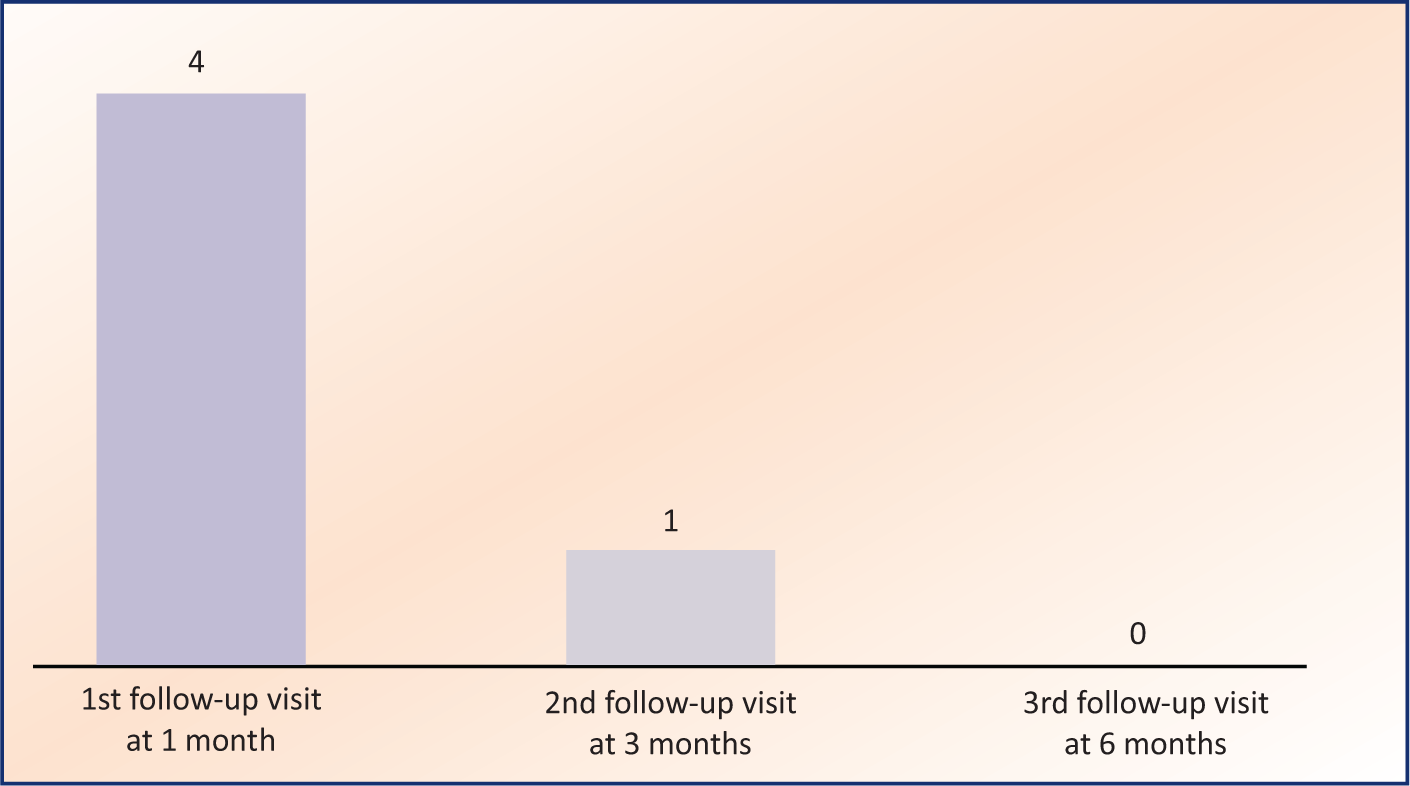
Significant improvements were observed in clinical and laboratory parameters from baseline to the 6-month follow-up (Table 3). Median weight decreased from 75 kg to 74 kg (P = 0.000), while SBP and DBP both improved significantly (SBP: P = 0.003, DBP: P = 0.022). Glycemic control also improved, with HbA1c reducing from 8.75% to 7.90% (P = 0.000) and FBS decreasing from 149.0 mg/dL to 132.0 mg/dL (P = 0.000). Renal function markers showed mixed results: serum creatinine levels decreased significantly (P = 0.003), while the eGFR increased from 109.0 to 112.4 mL/min/1.73 m² (P = 0.000). However, BUN levels did not change significantly (P = 0.073).
Changes in Clinical and Laboratory Parameters from Baseline to 6-month Follow-up Visit.
Wilcoxon Signed-Rank Test is applied.
*P < 0.05 is considered statistically significant.
Discussion
Empagliflozin has consistently demonstrated safety and efficacy across various treatment regimens for T2DM. Unlike prior large-scale randomized trials conducted in high-income or East-Asian cohorts, the present study documents real-world treatment outcomes in an under-represented Afghan population, thereby expanding the global pharmacovigilance evidence base for Empagliflozin (Diampa→) alone & with Metformin (Diampa-M→). Notably, when used as an add-on therapy to pioglitazone with or without Metformin, Empagliflozin led to sustained reductions in HbA1c and body weight. 14 Furthermore, a study in the South Indian population found that daily doses of 25 mg Empagliflozin were well tolerated, with minimal adverse events such as hypoglycemia, hyponatremia, and UTIs. 15
A comprehensive meta-analysis further affirmed the safety and effectiveness of Empagliflozin, both as monotherapy and in combination with other diabetes medications.15,16 Our study adds valuable insights into the safety profile of Empagliflozin during longer follow-up periods. By 6 months, the incidence of the adverse events significantly declined, with persistent but less frequent occurrences of UTIs (5.1% at 1 month, 2.3% at 6 months), followed by dehydration and abdominal discomfort. Serious events, such as hypotension and fungal infections, were rare and manageable, further supporting the overall safety of Empagliflozin. Moreover, the stratification as per study treatments showed that patients administering Empagliflozin with Metformin experienced a higher frequency of adverse events, including hypoglycemia, dehydration, UTIs, nausea, vomiting, and flatulence. However, these events had significantly reduced by the last follow-up (6 months). In contrast, those on Empagliflozin without Metformin faced fewer adverse events throughout the study, with minor issues such as UTIs at the final follow-up.
Furthermore, Empagliflozin (Diampa→) alone & with Metformin (Diampa-M→) demonstrated improvements in glycemic control and weight reduction, with minimal group-based differences. The reduction in HbA1c was slightly more pronounced in the Empagliflozin (Diampa→) (from 8.4% to 8.31%) compared to the Empagliflozin with Metformin (Diampa-M→) (from 8.85% to 8.34%). Similarly, both Empagliflozin (Diampa→) alone & with Metformin (Diampa-M→) experienced weight reduction, that is, 76.97 kg to 76.51 kg, and 73.97 kg to 73.85 kg, respectively. This finding aligns with previous studies, such as Ferrannini et al. (2013), which reported sustained glycemic control with Empagliflozin alone. 17 Studies also confirm that Empagliflozin, when added to Metformin, improves glucose control with minimal hypoglycemia risk while reducing body weight and blood pressure. 18 Pooled data from multiple clinical trials indicate that Empagliflozin is well tolerated, with no increased risk of adverse events compared to placebo. 19 A comparative study found that Empagliflozin as an add-on therapy to Metformin and sitagliptin significantly reduced body weight, HbA1c, and blood pressure in obese T2DM patients without significant adverse effects. 20
Our research registered significant advancements in renal biomarkers through lower serum creatinine levels, together with diminished urinary albumin-to-creatinine ratios that match previous study results. 21 This dual action shows that Empagliflozin can effectively treat the renal elements as well as the metabolic features of T2DM.
The mean change in HbA1c levels in Afghanistan seems lower than in other trials, possibly because patients in Afghanistan struggle to follow treatment recommendations because of limited health care structure and financial restrictions and difficult access to medical professionals. The high economic expenses for diabetes management, such as prescription drugs and diagnostic appointments, along with healthcare facility transportation costs, diminish treatment compliance among patients. People who fast during Ramadan experience blood glucose level changes because their dietary patterns alter during fasting periods, which causes intermittent high or low blood glucose levels, though they typically consume more high-carbohydrate meals before dawn and after sunset. A large number of Afghan patients demonstrate inadequate understanding about ideal dietary maintenance, including carbohydrate amounts and portion sizes and glycemic index-related choices that negatively affect blood sugar control. Metabolic outcomes together with treatment adherence in Afghanistan depend strongly on enhancing healthcare systems and implementing diabetes management strategies tailored to local cultures while providing proper patient education.
Limitations
The current study has several limitations that need to be considered. The study’s specific location with South-Asian participants reduced the ability of researchers to apply their results beyond this population. This 6-month period may not define the complete safety and effectiveness capabilities of Empagliflozin with or without Metformin treatment. The study’s reporting bias should be acknowledged since some data rely on participant self-reporting, especially adverse events. The study’s results about Empagliflozin alone are restricted because of the small patient sample size, thus impacting the study’s power to draw definitive conclusions.
Conclusion
Empagliflozin (Diampa→) alone & with Metformin (Diampa-M→) shows promising safety combined with efficacy results for patients diagnosed with T2DM according to this research. The study data show that it effectively enhances blood pressure levels and reduces weight while improving glycemic control. Additional research must analyze both long-term medical results and the specific effects of using Empagliflozin independently or combined with additional treatment options among various patient groups.
Footnotes
Acknowledgements
The authors would like to acknowledge the Medical Affairs department of Getz Pharma for their technical support and assistance in the publication process.
Authors’ Contribution
MWN, A, GMA, JK, MNT: Substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work. MNT, AN, JK: Drafting the work or reviewing it critically for important intellectual content. MWN, HM, JK, AN: Final approval of the version to be published. MWN, HM, A, GMA, JK, MNT, AN: Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Data Availability Statements
The data that support the findings of this study will be provided by the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study protocol was approved by the Institutional Review Board (IRB) of the Afghanistan National Public Health Institute [Ref# A.0823.416; Dated 6 August 2023].
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Human Ethics and Consent to Participate
The study was developed and conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all participants prior to their inclusion in the study. Participant confidentiality and data protection were maintained in accordance with applicable regulations.
