Abstract
Type 2 diabetes mellitus (T2DM) is increasing worldwide. Treatment of T2DM continues to present challenges, with a significant proportion of patients failing to achieve and maintain glycemic targets. Despite the availability of many oral antidiabetic agents, therapeutic efficacy is also offset by side effects such as weight gain and hypoglycemia. Therefore, the search for novel therapeutic agents with an improved benefit–risk profile continues. In the following review we focus on a novel class of oral antidiabetic drugs, the sodium glucose transporter protein 2 (SGLT2) inhibitors, which have unique characteristics. SGLT2 inhibitors focus on the kidney as a therapeutic target, where they inhibit the reabsorption of glucose in the proximal tubule, causing an increase in urinary glucose excretion. Doing this, they reduce plasma glucose independently of the β-cell function of the pancreas. SGLT2 inhibitors are effective at lowering hemoglobin A1c, but also induce weight loss and reduce blood pressure, with a low risk of hypoglycemia. In general, the SGLT2 inhibitors are well tolerated, with the most frequent adverse events being mild urinal and genital infections. Since their primary site of effect is the kidney, these drugs are less effective in patients with impaired kidney function but evidence is emerging that these drugs may also have a protective effect against diabetic nephropathy. This review focuses on the most extensively studied SGLT2 inhibitors dapagliflozin, canagliflozin and empagliflozin. Dapagliflozin and canagliflozin have already been approved for marketing by the US Food and Drug Administration. The European Medicines Agency has accepted all three drugs for marketing.
Keywords
Introduction
Type 2 diabetes mellitus (T2DM) is a disease with a rising incidence, with an estimated 381.8 million patients globally in 2013 [Guariguata et al. 2014]. T2DM is associated with other cardiovascular risk factors, which contribute to micro- and macrovascular complications, causing significant morbidity and mortality. Hyperglycemia in T2DM is caused by a combination of progressive β-cell dysfunction and insulin resistance in muscle, liver and adipocytes. Hyperglycemia in itself also contributes to a further decline in β-cell function and increase in insulin resistance, called ‘glucose toxicity’ [Del Prato, 2009]. Treatment of T2DM focuses on minimizing micro- and macrovascular complications, which is done by maintaining glycemic control and reducing other cardiovascular risk factors. Lifestyle interventions remain the cornerstone of therapy, however many patients will not reach their therapeutic goal and require pharmacological intervention. Many oral antidiabetic agents are available, but therapeutic efficacy is often offset by side effects such as weight gain or hypoglycemia [Inzucchi et al. 2012]. Furthermore most of these therapies rely at least partially on β-cell function, so as the disease progresses and β-cell function declines, these therapies become less effective. Hence, there remains a need for new oral antidiabetic agents targeting a mechanism independent of β-cell function. This review focuses on a novel class of oral antidiabetic agents, the sodium glucose transporter protein 2 (SGLT-2) inhibitors. These drugs represent an attractive new treatment option which allows reduction of glycaemia independent of β-cell function by inducing glycosuria.
The role of the kidney in glucose homeostasis
In the context of diabetes, until recently the kidney was generally seen as one of the end organs which suffered from the complications of hyperglycemia with diabetic nephropathy as the main point of interest. During the last decade, more attention is being paid to the role of the kidney in glucose homeostasis. In a healthy individual about 180 g of glucose is filtered each day at the glomeruli, but because of an active reabsorption system in the proximal tubule, virtually no glucose appears in the urine. As glucose does not penetrate freely through the cell membrane, this transport is facilitated by glucose transporting proteins, of which there are two important families in humans. First there is the SGLT family, which is a sodium glucose cotransporter. This transporter is found on the luminal side and allows for transportation of glucose from the luminal filtrate to the intracellular space of the proximal tubule epithelial cells. The process is driven by a sodium gradient, which is generated by a Na/K pump in the basolateral membrane. This allows for glucose transport against a concentration gradient. The intracellular glucose is then transported through the basolateral membrane to the blood circulation via a GLUT transporter (GLUT2), which is a simple glucose transporter allowing glucose transport down a concentration gradient (Figure 1). Two SGLT isoforms (SGLT1 and SGLT2) have been identified to mediate glucose transport in the renal tubule, SGLT2 being the most important one. SGLT2, which is located in the proximal tubule, is responsible for 90% of reabsorption of glucose in the tubular filtrate. The remaining 10% is reabsorbed by SGLT1 in the S3 segment of the proximal tubule. SGLT2 is only present in the kidney, while SGLT1, although present in the kidney, is mainly found in the small intestine, where it facilitates the absorption of dietary glucose [DeFronzo et al. 2011; Wright et al. 2007]. This transport system is highly efficient, but at glucose concentrations above 180 mg/dl, the transport system becomes saturated and glycosuria occurs. There is little inter-individual variability for this threshold in healthy subjects, though this threshold is remarkably higher in patients with T2DM with increased renal glucose reabsorption, probably because of increased expression of SGLT2 and GLUT proteins [DeFronzo et al. 2011]. Indeed it has been shown that both SGLT2 and GLUT2 expression are increased in cultured renal tubular cells isolated from the urine of patients with T2DM [Rahmoune et al. 2005]. This maladaptive response means that glycosuria occurs only at plasma concentrations well above 180 mg/dl. Theoretically, blocking renal glucose reabsorption and inducing glycosuria could lead to a decrease in plasma glucose and thereby reduce glucotoxicity. This could also theoretically lead to hypoglycemia. However, patients with familial renal glycosuria (FRG), a rare genetic disorder, who have an inactivating mutation in SCL5A2 (encoding SGLT2) do not experience any hypoglycemia or other clinical problems, even though they have significant glycosuria [DeFronzo et al. 2011; Wright et al. 2007]. This led to further research into using SGLT2 inhibition as a therapeutic target for T2DM, mimicking the effect of the inactivating mutation of FRG. SGLT1 inhibition is probably not a reasonable goal since the resulting intestinal malabsorption of glucose and galactose causes debilitating osmotic diarrhea. This is confirmed by subjects with a genetic mutation inactivating SGLT1.

Glucose reabsorption in the proximal tubule of the kidney.
The first molecule which allowed the effectiveness of inhibition of the SGLT system to be tested was phlorizin. Phlorizin, isolated from the bark of apple trees in 1835 and initially studied as an antipyretic agent, induced major glycosuria. This is caused by blocking both SGLT1 and SGLT2. Because of its blocking effect on SGLT1, it is not suitable for use in humans, but experiences with phlorizin in animal models were promising. Phlorizin administered to diabetic (90% pancreatectomized) rats reduced glycaemia and reduced insulin resistance, while β-cell function improved [Rosetti et al. 1987, 1990]. The promising results with phlorizin led to the development of drugs that are highly selective for SGLT2, allowing use in humans.
Effect of SGLT2 inhibitors
Several molecules were developed that are selective inhibitors of SGLT2 (Figure 2). Only two of them, dapagliflozin (marketed as Forxiga in the USA and Farxiga in Europe by AstraZeneca - London, United Kingdom) and canagliflozin (marketed as Invokana by Janssen Pharmaceuticals - Beerse, Belgium), are approved for use in humans by the US Food and Drug Administration [FDA, 2013, 2014] and the European Medicines Agency [EMA, 2012, 2013]. Empagliflozin, (marketed as Jardiance by Boehringer Ingelheim - Ingelheim, Germany), was recently approved by the EMA [EMA, 2014] . This review focuses on these three molecules. All of these molecules are highly selective for SGLT2 over SGLT1 (respectively >2500 fold for empagliflozin, >1200 fold for dapagliflozin and >250 fold for canagliflozin) [Grempler et al. 2012].

Pleiotropic effects of sodium glucose transporter protein 2 (SGLT2) inhibitors.
Efficacy, glycemic control and hypoglycemia risk
Dapagliflozin, canagliflozin and empagliflozin have been proven to be effective molecules in several large trials. Tables 1–3 summarize the results of the largest trials with at least 24 weeks of duration for dapagliflozin, canagliflozin and empagliflozin respectively.
Main studies and outcomes regarding glycaemia and weight for dapagliflozin.

N = number of patients.
Effect on HbA1c with highest dose of SGLT2 inhibitor minus effect from placebo or control (standard error).
Additional weight change versus placebo or control with highest dose SGLT2 inhibitor (standard error).
HbA1c, hemoglobin A1c; SGLT2, sodium glucose transporter protein 2.
Main studies and outcomes regarding glycaemia and weight for canagliflozin.

N = number of patients.
Effect on HbA1c with highest dose of SGLT2 inhibitor minus effect from placebo or control (standard error).
Additional weight change versus placebo or control with highest dose SGLT2 inhibitor (standard error).
HbA1c, hemoglobin A1c; SGLT2, sodium glucose transporter protein 2; SU, sulfonylurea.
Main studies and outcomes regarding glycaemia and weight for empagliflozin.
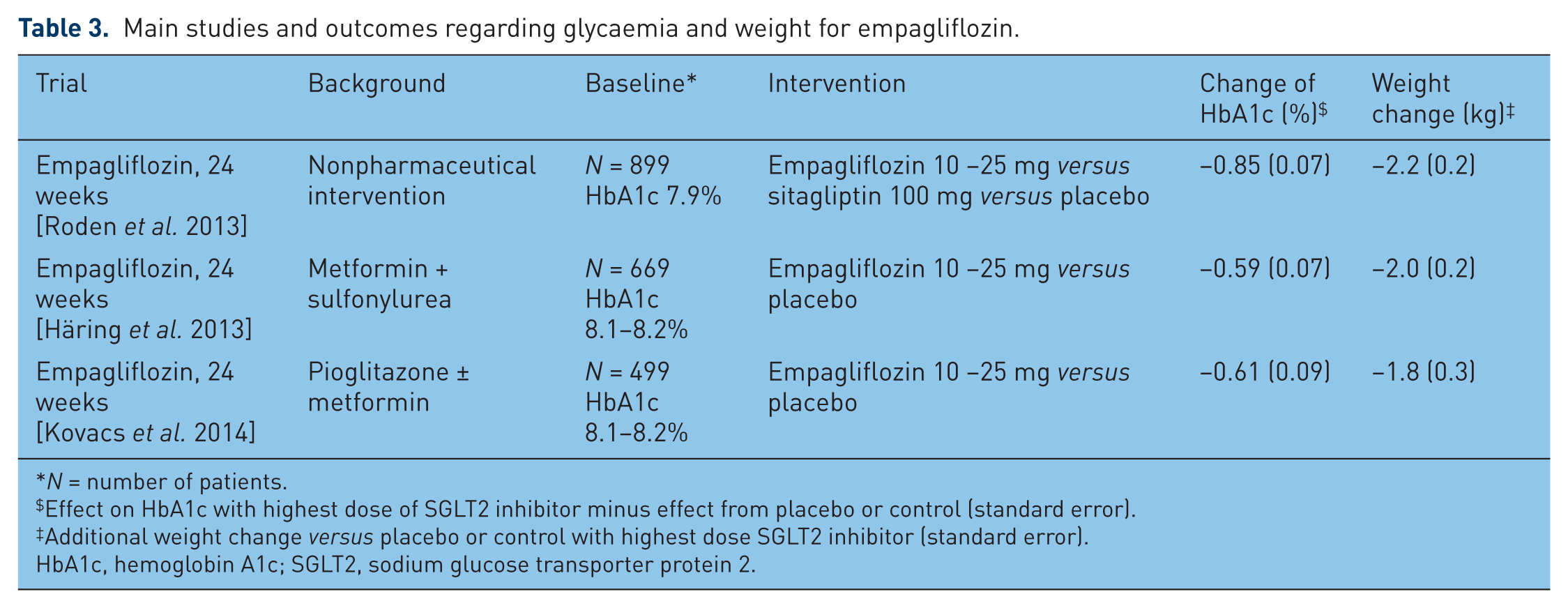
N = number of patients.
Effect on HbA1c with highest dose of SGLT2 inhibitor minus effect from placebo or control (standard error).
Additional weight change versus placebo or control with highest dose SGLT2 inhibitor (standard error).
HbA1c, hemoglobin A1c; SGLT2, sodium glucose transporter protein 2.
Dapagliflozin
Trials show that dapagliflozin monotherapy is effective in reducing glycaemia in naïve patients with T2DM [hemoglobin A1c (HbA1c) reductions versus placebo of a minimum of 0.66% and a maximum of 0.84%] [Bailey et al. 2012; Ferrannini et al. 2010; Ji et al. 2014] (Table 1). Combination therapy with metformin and dapagliflozin 10 mg was more effective than monotherapy with either metformin or dapagliflozin [Henry et al. 2012]. Dapagliflozin is also effective when added to other oral antidiabetic drugs such as metformin, glimepiride, pioglitazone and sitagliptin, with HbA1c reductions compared with placebo varying from 0.23% at 24 weeks to 0.8% at 102 weeks in the group of dapagliflozin 10 mg combined with metformin [Bailey et al. 2013; Nauck et al. 2011]. Dapagliflozin was also proved to be noninferior to glipizide in addition to metformin [Nauck et al. 2011]. A meta-analysis concluded that dapagliflozin was as effective in reducing HbA1c when added to monotherapy with metformin as are Dipeptidyl Peptidase (DPP4) inhibitors, thiazolidinediones or sulfonylurea [Goring et al. 2013]. The addition of dapagliflozin to insulin also produced a significant reduction in HbA1c, which is an interesting insulin-sparing effect [Wilding et al. 2012, 2014]. These beneficial effects were also maintained in the long term, as shown in studies extended up to 2 years [Bailey et al. 2013; Bolinder et al. 2013; Wilding et al. 2014].
Canagliflozin
Canagliflozin was effective in reducing glycaemia when started as monotherapy and in combination with diverse antidiabetic agents. Canagliflozin 300 mg has been shown to reduce HbA1c by 1.16% when compared with placebo in treatment-naïve patients [Stenlöf et al. 2013] (Table 2). Significant HbA1c reductions are obtained when canagliflozin is added to monotherapy with metformin (HbA1c reduction of 0.77% at 26 weeks with canagliflozin 300 mg) or in combination with other antidiabetic agents [Lavalle-González et al. 2013]. Canagliflozin was also proven to be noninferior to DPP4 inhibitors and sulfonylurea. Canagliflozin 300 mg induced an additional HbA1c reduction of 0.12% compared with a mean dose of 6 mg glimepiride and an additional HbA1c reduction of 0.15% compared with sitagliptin 100 mg, both at week 52 added to metformin [Cefalu et al. 2013; Lavalle-González et al. 2013]. These results were statistically significant, although in routine practice this might not be clinically relevant. Clinical superiority was however obtained in one study in which an additional HbA1c reduction of 0.37% was observed for canagliflozin 300 mg versus sitagliptin 100 mg at week 52 on a background of metformin + sulfonylurea [Schernthaner et al. 2013]. Currently there are no long-term studies with a duration longer than 52 weeks.
Empagliflozin
Empagliflozin is the newest of the three reviewed molecules (Table 3). Therefore fewer trials have been published and the trial durations are also shorter. Empagliflozin was proven to be effective at reducing glycaemia in monotherapy at a dose of 25 mg with an additional HbA1c reduction of 0.85% for empagliflozin 25mg versus placebo at week 24 [Roden et al. 2013]. On a background of metformin + sulfonylurea or metformin + pioglitazone, empagliflozin 25 mg produced additional HbA1c reductions of 0.59% and 0.61% respectively compared with placebo at week 24 [Häring et al. 2013; Kovacs et al. 2014]. A 78-week extension study of empagliflozin 25 mg as monotherapy and combined with metformin showed HbA1c reductions versus baseline of 0.47% and 0.63% respectively [Ferrannini et al. 2013].
In conclusion, SGLT2 inhibitors are effective at reducing glycaemia in monotherapy as well as in combination with other antidiabetic agents. The effect is comparable to other oral hypoglycemic drugs with an expected HbA1c reduction of about 0.5%. This is confirmed in a recent meta-analysis which studied SGLT2 inhibitors as a class and withheld a weighted mean difference versus placebo for HbA1c of −0.79% when used as monotherapy and −0.61% when used as add-on therapy. Compared with other antidiabetic agents, the differences are minimal (weighted mean difference for HbA1c of 0.05% and 0.16% for monotherapy and add-on treatment respectively) [Vasilakou et al. 2013]. Hypoglycemia is a common side effect of many antidiabetic drugs, such as insulin, with a negative effect on quality of life [Davis et al. 2005; Lundkvist et al. 2005]. A recent meta-analysis showed that the odds ratio for hypoglycemia with SGLT2 inhibitors was only 1.28 compared with placebo, confirming that the risk of hypoglycemia was only slightly higher than with placebo [Vasilakou et al. 2013]. Compared with other antidiabetic agents, the risk of hypoglycemia with SGLT2 inhibitors is similar, with an odds ratio of 1.01 in the same meta-analysis. Nonetheless when compared with drugs which have a high intrinsic hypoglycemia risk, such as sulfonylurea, this risk is substantially lower [Cefalu et al. 2013; Nauck et al. 2011]. The risk for hypoglycemia is also dependent on the background therapy, with an expected higher risk when SGLT2 inhibitors are added to insulin or sulfonylurea [Schernthaner et al. 2013; Wilding et al. 2014].
Weight loss and body composition
Another important advantage of SGLT2 inhibitors in patients with diabetes is the associated weight reduction. Most clinical trials with SGLT2 inhibitors have demonstrated significant weight loss for patients treated with SGLT2 inhibitors compared with treatment with control or placebo (Tables 1 and 3). A recent meta-analysis, which includes most of the available SGLT2 studies, reports an additional weight loss of 1.74 kg compared with placebo and 1.11 kg compared with treatment with an active control. Studies with canagliflozin, which reported a percentage, showed a weight reduction of 2.37% compared with placebo and 2.14% compared with an active control [Vasilakou et al. 2013]. This weight reduction is maintained in the long term, lasting 102 weeks with dapagliflozin on a background with metformin and lasting 104 weeks with dapagliflozin on a background with insulin. The weight reduction reached a plateau of −3.1 kg and −3.3 kg respectively [Bailey et al. 2013; Wilding et al. 2014]. A fast decline in body weight is seen during the first weeks, which evolves to a more gradual decline which then often reaches a plateau phase. In contrast, in one study with dapagliflozin combined with metformin, weight loss was still ongoing and had not yet reached a plateau phase at week 102 [Bolinder et al. 2013].
As SGLT2 inhibition induces glycosuria, this weight loss could be due to osmotic diuresis which entails or is due to caloric loss, a logic consequence of glycosuria, and amounts to about 200−300 kcal per day. It is generally believed that osmotic diuresis is responsible for the faster decline in weight initially, though caloric loss and subsequent loss of fat mass are responsible for the long-term gradual decrease in body weight. Measurement of fat mass (FM) using dual energy X-ray absorptiometry in patients treated with dapagliflozin or placebo on a background of metformin showed a consisted decrease of FM of 75% compared with only 50% in the placebo group. Both weight reduction and FM reduction are associated with increased glycosuria, proving the assumption that sustained weight loss is due to urinary loss of calories. Moreover magnetic resonance imaging demonstrated that visceral adipose tissue decreased more than subcutaneous adipose tissue at week 24 [Bolinder et al. 2012]. Given the known driving force of visceral adipositas in T2DM, this is a very interesting finding [Kahn et al. 2014]. Furthermore it was shown that dapagliflozin when combined with other antidiabetic agents, such as sulfonylurea, thiazolidinediones or insulin, can mitigate their known side effect of weight gain [Rostenstock et al. 2011; Strojek et al. 2011; Wilding et al. 2014]. Weight loss reduction by SGLT2 inhibitors also seems to have a positive influence on health-related quality of life [Grandy et al 2014].
Blood pressure effect
T2DM is frequently associated with arterial hypertension, which also contributes to the micro- and macrovascular complications of diabetes [Ferrannini and Cushman, 2012]. Trials with SGLT2 inhibitors have shown a reduction of blood pressure. A recent meta-analysis of these trials has shown a statistically significant decrease of 4 mmHg in systolic blood pressure for SGLT2 inhibitors compared with the control groups. Reduction in diastolic blood pressure was also significant with a weighted mean difference of −1.6 mmHg compared with controls [Baker et al. 2014]. These results are also in line with findings of an older meta-analysis [Vasilakou et al. 2013]. The most recent meta-analysis showed no significant increase in orthostatic hypotension, while the older study showed a small increase in hypotension for SGLT2 inhibitors compared with other antidiabetic agents (odds ratio of 2.68), but not compared with placebo [Baker et al. 2014; Vasilakou et al. 2013]. The exact mechanisms of blood pressure reduction remain to be elucidated, but the osmotic diuretic effect and the weight loss are probably contributing factors.
Effect on serum lipids and overall cardiovascular effects
In different studies small, though significant changes in serum lipid concentrations were shown with the use of SGLT2 inhibitors. In an analysis of multiple phase II and III trials with dapagliflozin 10 mg, a mean placebo-subtracted increase of low-density lipoprotein (LDL) and high-density lipoprotein (HDL) cholesterol was seen of approximately 4% [FDA, 2014]. For canagliflozin 300 mg results were similar with a placebo-subtracted increase of LDL of 4.6–12.2% in phase III trials and a placebo-subtracted increase in HDL of 0.8–8.5% [FDA, 2013]. There were no important differences in LDL/HDL ratio for both dapagliflozin and canagliflozin. Data from individual studies for empagliflozin showed increases of LDL and HDL cholesterol comparable to dapagliflozin and canagliflozin [Häring et al. 2013; Kovacs et al. 2014; Roden et al. 2013]. An etiology for these increases in serum lipids has not been identified, though hemoconcentration is a possible explanation. Nonetheless this increase might have a negative effect on cardiovascular outcome [Go et al. 2013]. However, SGLT2 inhibitors have a favorable effect on glycaemia, bodyweight and blood pressure which are all established risk factors for cardiovascular disease. It is reassuring to see that multiple meta-analyses show no increase in cardiovascular events [FDA, 2013, 2014; EMA, 2014]. Nonetheless further study is warranted, and multiple cardiovascular outcome trials with the different SGLT2 inhibitors are currently ongoing.
Urinary tract and genital infections
T2DM is associated with an increased risk for urinary tract infections (UTIs) and nonsexually transmitted genital infections such as vaginitis and balanitis [Benfield et al. 2007; de Leon et al. 2002]. This is partially caused by glycosuria, but immune dysfunction and altered glycosylation of uroepithelium cells in diabetes also appear to play a role [Geerlings et al. 2014]. As SGLT2 inhibitors induce glycosuria, an increase in these infections can be expected. This was indeed confirmed in trials with an odds ratio of 1.34 for UTI and an odds ratio of 3.50 for genital infections in the dapagliflozin group versus the placebo group [Vasilakou et al. 2013]. Most data are available for dapagliflozin, with data from 12 randomized, placebo-controlled trials being pooled [Johnsson et al. 2013a,b]. UTI rates were indeed slightly higher in the dapagliflozin group (5.7% and 4.3% in the 5 mg and 10 mg dapagliflozin group respectively versus 3.7% in the placebo group). These results were only significant for dapagliflozin 5 mg. It was noted that the first event of UTI usually occurred relatively early in the trial and that UTI was also more frequent in women. Most UTIs were classified as mild to moderate, responded well to standard treatment and generally did not lead to discontinuation of dapagliflozin. Pyelonephritis was very rare and its incidence was similar in the placebo and intervention groups (0.1%) [Johnsson et al. 2013a]. Genital infection rates in the same population were 5.7 and 4.8% for dapagliflozin 5 mg and 10 mg respectively versus 0.9% for placebo. These infections were again more frequent in women, generally occurred early in the trial, responded well to standard treatment and did not lead to discontinuation of treatment [Johnsson et al. 2013b].
Results for canagliflozin are very similar in a grouped dataset from several placebo-controlled studies with UTI incidences of 4.0 and 5.1% for placebo and canagliflozin (100 mg and 300 mg combined). In the same dataset, the incidence of genital infections in women was 13.9% for canagliflozin versus 3.2% for placebo, and in men 3.9% versus 0.6% for placebo [FDA, 2013]. The EMA reports similar results in their grouped analysis of empagliflozin trials [EMA, 2014].
Cancer risk
Malignancy rates during trials with SGLT2 inhibitors were low, with generally no significant differences in rates between intervention and placebo or comparator groups in the separate trials. Nonetheless, in an analysis of several phase II and III trials for dapagliflozin, an imbalance was found in bladder and breast malignancies, leading to an initial refusal by the FDA.
The initial pool of patients submitted to the FDA comprised 5501 patients treated with dapagliflozin and 3184 who received placebo. Nine patients in the dapagliflozin group (0.16%, all male) were diagnosed with bladder cancer versus one patient in the placebo group (0.03%). Incidence risk ratio (IRR) for men only was 5.38 (95% confidence interval 0.84–122), not statistically significant. However, since the IRR was over 5, clearly higher than the expected rate of two cases of bladder cancer in an age-matched male reference population, a negative advice was given by the FDA pending further investigation. At the time of the safety update 30 months after initial submission, there was only one additional case of bladder cancer in the dapagliflozin group, now comprising 6045 patients (incidence 0.17%) versus no new patients in the placebo group of 3512 patients (incidence 0.03%). It was noted that of the 10 subjects in the dapagliflozin group who had developed bladder cancer during treatment, 6 subjects already had hematuria at baseline, with another 3 developing hematuria within the first six months of treatment. Furthermore it was noted that all cases were reported in the first 2 years of exposure, with no additional cases between 2 and 4 years of exposure. This seems to suggest that dapagliflozin was not the cause of these tumors and that the reported cases were patients who already had cancer at the moment of inclusion in the trials. Based on these data, dapagliflozin received approval by the FDA, after it had already been approved by EMA [FDA, 2014; EMA, 2012].
Regarding breast cancer, in the final FDA report there were 12 cases in 2693 female patients of the dapagliflozin group (incidence 0.45%) versus 3 cases in 1439 women in the placebo group (incidence 0.21%). It has to be noted that in a comparable age group the incidence of breast cancer is expected to be 0.25%, and that patients with diabetes are expected to have an even higher incidence. This combined with the fact that 10 out of 12 breast cancer cases in the dapagliflozin group were diagnosed within 1 year of randomization led to the conclusion that there is probably no causative role for dapagliflozin in the development of breast cancer [FDA, 2014; EMA, 2012]. Further postmarketing surveillance for cancer risk is ongoing.
In the authorization process for canagliflozin, neither the EMA nor the FDA could identify a significant difference in malignancies [FDA, 2013; EMA, 2013]. In the grouped analysis of the studies undertaken for authorization of empagliflozin, EMA found an imbalance for bladder cancer (two cases with empagliflozin versus no cases with placebo), though given the very small numbers this was not deemed to be of enough significance to stop authorization. Furthermore a small imbalance was seen for melanoma, though given the very small numbers and the absence of a mechanistic rationale this was not considered relevant. Again, further postmarketing surveillance is ongoing [EMA, 2014].
Renal impairment, renal effects
Chronic renal disease is a common comorbidity in diabetes, with a prevalence of about 40% in patients with T2DM in the USA [Molitch et al. 2003]. In patients with acute renal injury or chronic kidney disease, in whom the glomerular filtration rate is decreased, the effect of SGLT2 inhibitors is also expected to be decreased. In a study with patients with an estimated glomerular filtration rate (eGFR) greater than 30 and up to 60 ml/min, there was no significant difference in HbA1c reduction compared with placebo for dapagliflozin 10 mg at 24 or 104 weeks. Subgroup analyses showed that there was a significant effect compared with placebo in the group with an eGFR of at least 45 and less than 60 ml/min with a reduction in HbA1c of 0.33% for dapagliflozin 10 mg [Kohan et al. 2013]. For canagliflozin 300 mg there was a significant HbA1c reduction of 0.40% compared with placebo in subjects with an eGFR of at least 30 and less than 50 ml/min [Yale et al. 2013]. Finally for empagliflozin 25 mg a significant HbA1c reduction of 0.42% compared with placebo was found in patients with an eGFR of at least 30 and less than 60 ml/min [Barnett et al. 2014]. Though there is a significant effect in some cases, this is clearly less than the effect observed in a population without renal impairment.
During these trials, a small decrease in eGFR was observed in the first weeks of treatment, which gradually returned to baseline. This is probably an early repercussion of the diuretic effect of SGLT2 inhibitors. However, in the longest follow-up trial, after the initial decline, a long-term stabilization of renal function was observed. Meanwhile the eGFR in the placebo group continued to decline slowly. Furthermore, there was a trend towards a reduction in albumin–creatinine ratio in the dapagliflozin-treated patients [Kohan et al. 2013]. These remarkable findings seem to support recent claims of a possible nephron-protective effect of SGLT2 inhibitors, which would be a very exciting new way of preventing diabetic nephropathy. This may be done by modulating the tubuloglomerular feedback. Tubuloglomerular feedback is a mechanism where glomerular filtration rate and electrolyte reabsorption are finely coordinated with an important role for the macula densa which acts as a sensor. In situations where there is an increased NaCl delivery to the macula densa, there is an associated decrease in single nephron glomerular filtration (SNGFR) through vasoconstriction in the afferent arteriole and vasodilatation in the efferent arteriole. In patients with diabetes the opposite happens, with a decreased NaCl delivery to the macula densa and a compensatory increase in SNGFR. This decrease in NaCl delivery is due to upregulated SGLT2 transporters and increased glucose reabsorption, with the coupled Na reabsorption in the proximal tubule. The increase in SNGFR is caused by vasodilatation in the afferent arteriole and vasoconstriction in the efferent arteriole, leading to intraglomerular hypertension which drives the increase in SNGFR. This is one of the driving forces in progressive diabetic nephropathy, usually countered by the use of angiotensin-converting enzyme inhibitors or angiotensin II receptor blockers, which leads to reduced vasoconstriction in the efferent arteriole. Blocking SGLT2, and therefore normalizing NaCl delivery to the macula densa has the potential of blocking this maladaptive mechanism at its roots, as has been proven in mouse models. This effect is independent of the blood glucose lowering effect of SGLT2 inhibition [De Nicola et al. 2014; Gilbert, 2013]. Furthermore, the improvement of hyperglycemia by itself could contribute to the nephron-protective effect. For instance, studies have demonstrated that treatment with an SGLT2 inhibitor reduces glomerular hypertrophy and markers of renal inflammation. It is thought that these effects are mainly secondary to the reduction of blood glucose, and not an intrinsic effect of SGLT2 inhibition [Gembardt et al. 2014; Terami et al. 2014]. Lastly, the blood pressure lowering effect can also have a protective effect for the kidney, as the detrimental effect of hypertension on renal function has been well established. Further studies on this exciting new preventive option to prevent progression to, or from, diabetic nephropathy are ongoing [De Nicola et al. 2014; Gilbert, 2013].
For the moment, however, the FDA limits the use of dapagliflozin to patients with an eGFR greater than 60 ml/min. For canagliflozin the FDA advises a reduced dose of 100 mg in patients with an eGFR of at least 45 and less than 60 ml/min, and the drug is contraindicated if eGFR is less than 45 ml/min. The EMA advises against initiation of canagliflozin in patients with an eGFR less than 60 ml/min, yet allows patients whose eGFR declines after initiation to at least 45 and less than 60 ml/min to continue the drug on a reduced dose of 100 mg. Similarly, the EMA limits initiation of empagliflozin to patients with an eGFR greater than 60 ml/min. If the eGFR declines after initiation of therapy, a reduced dose of 10 mg is allowed with an eGFR greater than 45 and less than 60 ml/min, and treatment should be discontinued if eGFR drops to less than 45 ml/min.
Conclusion and future perspectives
SGLT2 inhibitors are an exciting novel class of antidiabetic agents which are effective at reducing glycaemia, induce weight loss and reduce blood pressure with no severe adverse effects. The most frequent adverse events are an increase in urinary and genital infections, though this rarely leads to treatment discontinuation. SGLT2 inhibitors also have a very low intrinsic capacity to cause hypoglycemia. Moreover, the mechanism of action of SGLT2 inhibitors is independent of insulin secretion or action. The favorable metabolic effects of the SGLT2 inhibitors makes this class of oral antidiabetic agents an attractive new tool in the battle against T2DM. Nevertheless there are some issues such as cardiovascular safety and cancer risk which require close monitoring post marketing. Finally, the effect of SGLT2 inhibition in patients with impaired kidney function needs more research. There might be a protective effect on kidney function which would be an exciting new way to prevent diabetic nephropathy. However, the efficacy of the drug seems to decline as kidney function becomes more impaired. Further studies are also ongoing investigating the effect of SGLT2 inhibition in patients with type 1 diabetes, when this might also be protective for the kidney [Cherney et al. 2014].
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
