Abstract
Background:
Type 2 diabetes mellitus (T2DM) is a significant public health concern in India, driven by rapid urbanization, increased consumption of ultra-processed foods, and changing dietary habits. These factors necessitate updated, real-world data to guide dietary interventions and public health strategies.
Aims:
This study aims to evaluate macronutrient intake as a percentage of total energy consumption in adults with and without T2DM. Secondary objectives include assessing dietary patterns, meal frequency, water intake, and associations with biochemical markers such as glycosylated hemoglobin (HbA1c), fasting blood glucose (FBG), postprandial blood glucose (PPBG), and lipid profiles.
Methods:
The I-STARCH study (Indian Study to Assess Real World CarboHydrate Consumption) employs a multicentric, cross-sectional observational design across 33 healthcare centers in India, targeting 1,000 participants. Data collection includes a structured 3-day dietary recall, demographic and clinical data from authenticated medical records, and standardized case report forms (CRFs). Statistical analysis will examine dietary patterns, their relationship with metabolic outcomes, and subgroup differences (e.g., urban vs. semi-urban).
Expected Outcomes:
The study will provide critical insights into contemporary dietary behaviors, facilitating personalized dietary recommendations and contributing to strategies for effective diabetes prevention and management.
Summary:
The I-STARCH study explores how diet affects blood glucose and lipid levels in Indian adults with and without Type 2 diabetes, aiming to guide personalized nutrition strategies.
Keywords
Abbreviations
BMI: Body mass index
CRF: Case report form
FBG: Fasting blood glucose
HbA1c: Glycated hemoglobin
HDL-C: High-density lipoprotein cholesterol
LDL-C: Low-density lipoprotein cholesterol
PPBG: Postprandial blood glucose
SD: Standard deviation
T2DM: Type 2 diabetes mellitus
TC: Total cholesterol
TG: Triglycerides
Introduction
Type 2 Diabetes Mellitus (T2DM) is a growing public health concern globally, with 537 million adults living with diabetes as of 2021, a number projected to rise significantly by 2045 (International Diabetes Federation, 2021). 1 In India, the burden is particularly alarming, with 101 million individuals diagnosed with diabetes and an additional 136 million with prediabetes. 2 India accounts for one in seven adults with diabetes globally, underscoring the urgent need for effective interventions to manage this epidemic. 1
Dietary patterns play a pivotal role in the prevention and management of T2DM.3–6 Balanced diets rich in whole grains, fruits, vegetables, legumes, and lean proteins have been shown to improve glycemic control, while diets high in refined carbohydrates and unhealthy fats exacerbate diabetes risk.7–10 However, traditional Indian diets, dominated by cereal-based carbohydrates like white rice and wheat, contribute to high glycemic loads and remain a significant challenge.11,12 Socio-economic shifts, the proliferation of ultra-processed foods, and food delivery platforms have further disrupted dietary habits, particularly over the past decade. 13 The COVID-19 pandemic has accelerated these trends, fostering sedentary lifestyles and greater reliance on calorie-dense convenience foods.
Existing studies, such as the STARCH study, highlighted high carbohydrate consumption and insufficient protein intake among individuals with T2DM, with only 14.8% of participants meeting their protein requirements. 14 However, these findings may no longer reflect current dietary realities, given the rapid changes in dietary behaviors and food environments in India. Updated, granular data on macronutrient intake and dietary patterns are crucial to addressing these gaps.
Due to high patient volumes, limited consultation times, and inadequate access to dietitians, clinicians encounter significant challenges when addressing dietary issues among individuals with T2DM. As a result, pharmacological management often takes precedence over dietary counseling, and even when it is provided, it may lack specificity or depth due to time constraints.
This study aims to address these gaps by capturing up-to-date data on dietary intake in individuals with and without T2DM. By analyzing macronutrient intake, meal frequency, and associated biochemical markers, this study seeks to provide insights for tailored dietary recommendations and public health policies that reflect contemporary Indian lifestyles.
Study Objectives
The primary objective of this study is to assess the macronutrient composition of dietary intake—including carbohydrates, proteins, fats, and dietary fiber—as a percentage of total energy consumption among adults with and without T2DM.
Secondary objectives include evaluating broader dietary patterns, such as the relative proportions of simple and complex carbohydrates, meal frequency, and daily water intake. Additionally, biochemical markers such as glycated hemoglobin (HbA1c) and fasting blood glucose (FBG), postprandial blood glucose (PPBG), and lipid profiles (total cholesterol, LDL, HDL, and triglycerides) will be analyzed to explore potential associations between dietary behaviors and metabolic health.
Methods
Study Design and Rationale
This multicentric, cross-sectional observational study aims to evaluate dietary patterns and their association with metabolic health outcomes among adults with and without T2DM. A cross-sectional design was chosen for its efficiency in capturing diverse dietary habits and clinical characteristics within a short timeframe while minimizing bias by assessing habitual practices without influencing participant behavior. The study workflow is summarized in Figure 1.
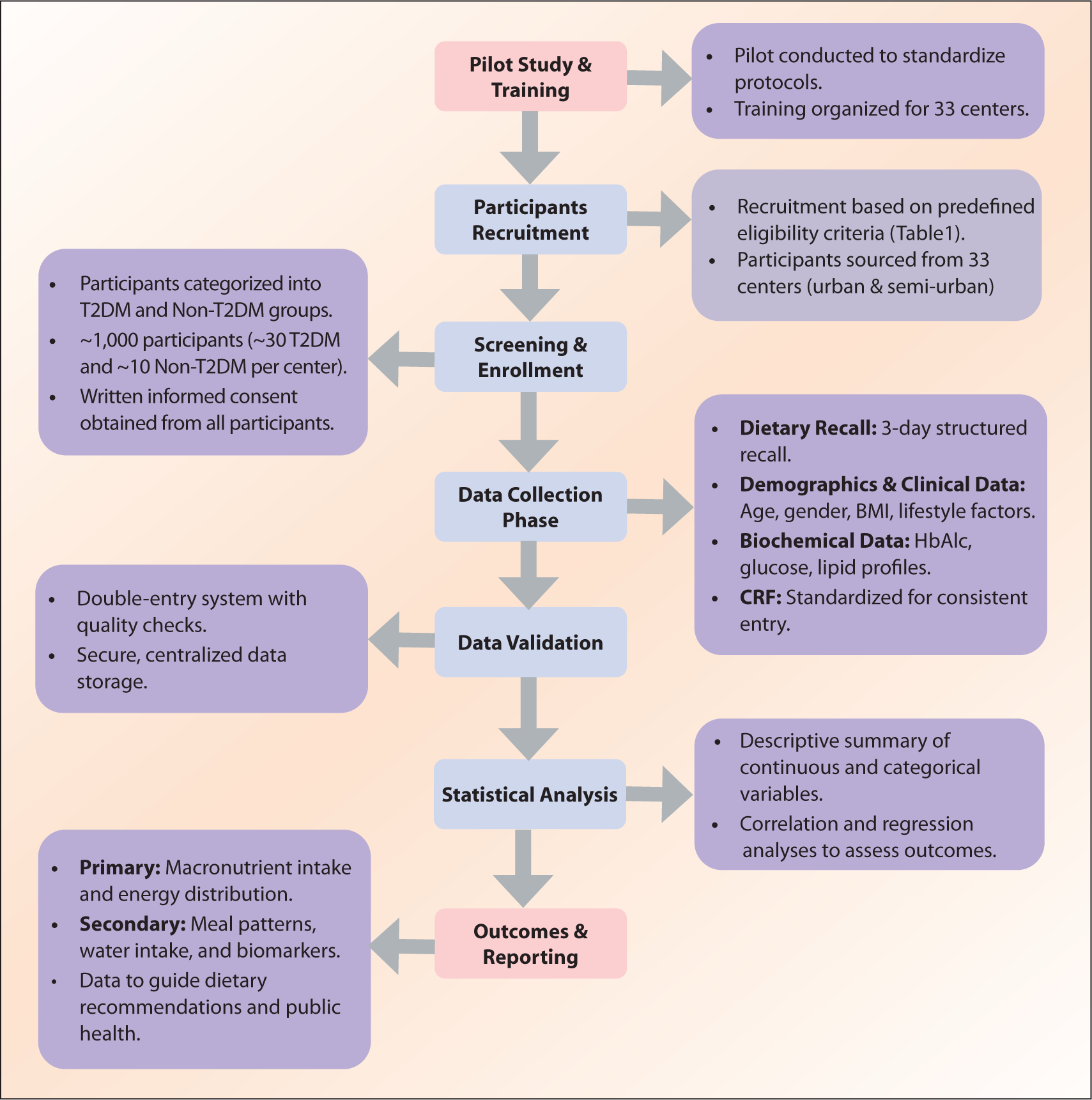
Study Timeline
The I-STARCH study (Indian Study to Assess Real World Carbohydrate Consumption) spans over a year and is divided into distinct phases with specific timelines for each key activity. The phases include a pilot phase for feasibility testing and method refinement, a training phase for procedural standardization across investigators and centers, and the ongoing main enrollment and data collection phase. This structured approach ensures methodological rigor and reliable data collection.
Study Population
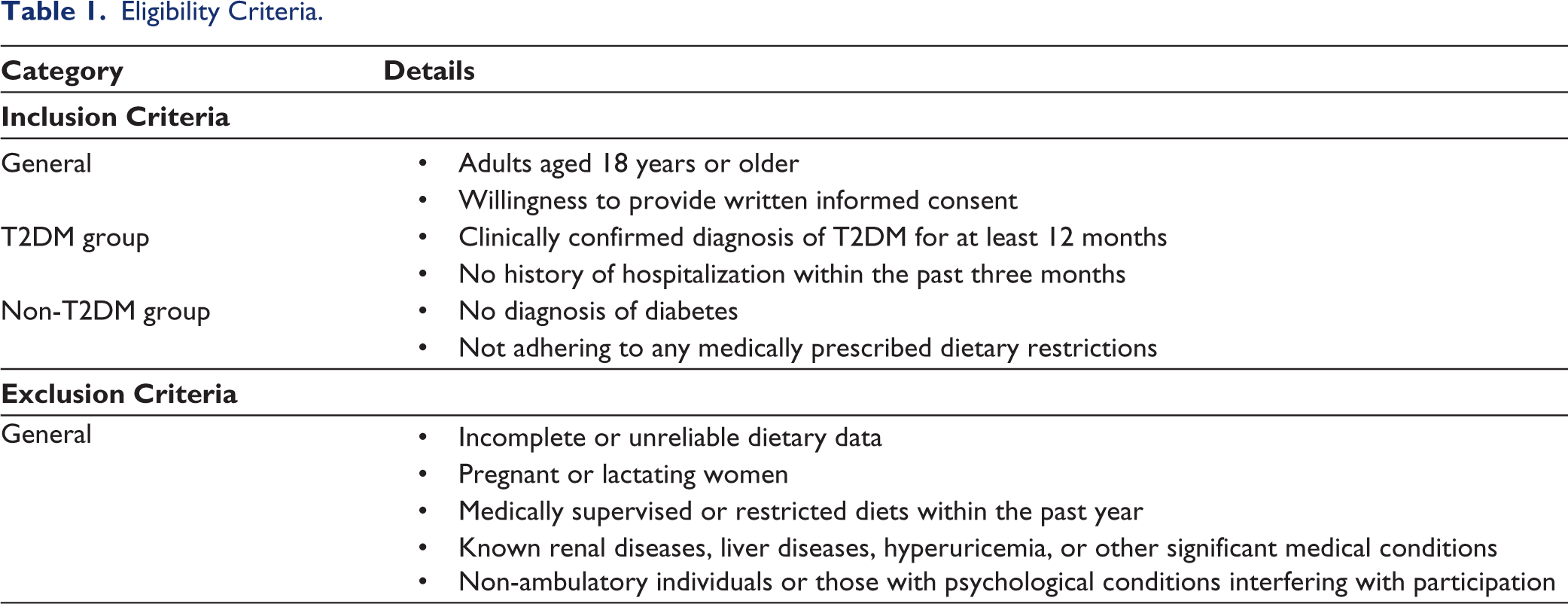
The study includes adults recruited from healthcare centers and community outreach programs to ensure a representative sample of individuals with and without T2DM. Participants will be drawn from diverse socio-economic and cultural contexts, with efforts to include urban and semi-urban populations. Additionally, healthcare professionals and corporate employees will be recruited to capture dietary patterns specific to professional and non-clinical settings, enhancing the findings’ relevance across varied groups. The detailed inclusion and exclusion criteria for participant eligibility are presented in Table 1.
Eligibility Criteria.
Recruitment Strategy
Participants will be recruited from 33 healthcare centers and community outreach programs across India, supplemented by enrollment from non-clinical settings, such as conferences and corporate offices. The study aims to recruit 1,000 participants, with at least 30 individuals with T2DM and 10 without T2DM from each center. Recruitment will employ convenience sampling, enrolling individuals who voluntarily agree to participate and provide informed consent.
Efforts will focus on capturing a diverse and representative sample, including participants from urban and semi-urban populations and various socio-economic backgrounds, ensuring the findings reflect the heterogeneity of dietary behaviors and metabolic health profiles across India. Recruitment will be facilitated by trained study investigators and staff, who will ensure clear communication of the study’s purpose, procedures, and the voluntary nature of participation.
Data Collection Methods
Data will be collected using a structured 3-day dietary recall, medical records, and standardized case report forms (CRFs).
3-day dietary recall: Trained staff will conduct in-person interviews to document participants’ detailed dietary intake, including food items, portion sizes, meal timings, and beverages. This method, chosen for its balance between practicality and accuracy, provides a comprehensive snapshot of habitual dietary patterns. Standardized tools such as measuring cups and spoons will be used to ensure consistency.
Medical records: Biochemical data, including HbA1c, FBG, PPBG, and lipid profiles, will be extracted from authenticated medical records dated within the past three months.
CRFs: A standardized CRF will capture participant demographics (height, weight, body mass index, waist circumference), dietary intake, lifestyle factors, and biochemical markers. Insights from the pilot study refined the CRF to ensure it effectively captures relevant data for the study objectives. The survey forms were reviewed by five subject matter experts (SMEs) for content validity.
Training and Refinement
Comprehensive training sessions, standardized data collection procedures will be carried out across all 33 centers. Investigators and dietitians will be trained on dietary recall methods, CRF completion, and clinical data extraction. Real-time demonstrations and post-training support materials ensure familiarity with study protocols. Pilot study insights, such as the need for precise portion measurement and clarity in CRF fields, guided these refinements.
Data Validation and Quality Assurance
To maintain data integrity, all CRFs will undergo double data entry into a secure electronic system. Embedded algorithms will flag discrepancies, which will be reviewed and resolved by the study team. Regular site monitoring visits will ensure adherence to protocols, and inconsistencies will be corrected using a documented process with proper verification. Data will be stored on password-protected servers with restricted access to safeguard confidentiality. These measures, informed by pilot study findings, aim to uphold the validity and reliability of study outcomes.
Endpoints
The study aims to evaluate dietary patterns and metabolic health through clearly defined primary and secondary endpoints. These endpoints are closely aligned with the study objectives and will be measured using validated methods.
Primary endpoints
Percentage composition of total energy intake from macronutrients (dietary carbohydrates, proteins, fats, and dietary fiber) among participants with T2DM and non-T2DM.
Secondary endpoints
Proportion of total energy intake derived from simple and complex carbohydrates. Frequency and distribution of meals across the day. Daily water intake. Biochemical markers, including:
HbA1c. FBG. PPBG. Lipid profile components: Total cholesterol, LDL, HDL, and triglycerides. Distribution of calories and macronutrients (carbohydrates, proteins, fats, dietary fiber) across different meals.
Statistical Analysis Plan
This study will recruit 1,000 participants from 33 healthcare centers to ensure sufficient statistical power to detect differences in dietary patterns and energy intake between T2DM and non-T2DM groups, accounting for demographic and dietary diversity.
Continuous variables, such as macronutrient intake and biochemical markers (e.g., HbA1c, fasting glucose), will be summarized as mean ± standard deviation (SD) or median with interquartile range (IQR), depending on data distribution. Categorical variables, such as meal frequency and dietary preferences, will be presented as percentages. Group comparisons will utilize t-tests or Mann-Whitney U tests for continuous data and Chi-square tests for categorical data. Subgroup analyses (e.g., urban vs. semi-urban) will employ ANOVA or Kruskal-Wallis tests, as appropriate.
Associations between dietary patterns and clinical outcomes will be evaluated using Pearson or Spearman correlation coefficients, with multivariable regression models adjusting for confounders like age, gender, and BMI. Missing data will be addressed through imputation for minor gaps, while participants with significant missing data will be excluded. Sensitivity analyses will test the robustness of findings. Statistical analyses will be conducted using R or SPSS software, with a significance threshold of P < .05.
Strengths and Limitations
This study’s multicentric design, spanning 33 healthcare centers, is a major strength, ensuring a diverse and representative sample that reflects varied socio-economic and geographic contexts. The use of structured tools like the 3-day dietary recall and standardized CRFs enhances the reliability and consistency of data collection. The large sample size will allow for robust subgroup analyses and meaningful insights into dietary patterns and metabolic health.
However, potential limitations include reliance on self-reported dietary data, which may lead to recall bias, particularly among participants with T2DM. The observational nature of the study limits the ability to establish causal relationships between dietary patterns and metabolic outcomes. Additionally, geographic and cultural heterogeneity in the participant pool may introduce variability, complicating direct comparisons. Efforts to address these challenges include rigorous standardization of data collection processes, stratified analyses to account for variability, and robust validation protocols for non-clinical data sources.
Discussion
This protocol outlines a multicentric, cross-sectional observational study aiming to address significant knowledge gaps in dietary patterns and metabolic health outcomes in Indian adults with and without T2DM. By capturing real-world data on macronutrient intake, meal frequency, water consumption, and biochemical markers, this study provides a unique opportunity to analyze the interplay between diet and metabolic health in diverse populations.
The multicentric approach is a key strength, enabling the study to reflect India’s diverse socio-economic and geographic contexts. The inclusion of healthcare professionals and corporate employees broadens the scope, making the findings relevant to varied occupational and non-clinical settings. Furthermore, the use of structured tools like the 3-day dietary recall and standardized CRFs ensures data reliability and comparability across centers.
However, the reliance on self-reported dietary data introduces the potential for recall bias. Efforts to mitigate this include detailed investigator training and the use of validated dietary recall methods. The cross-sectional design limits causal inferences but is well-suited for exploring associations and generating hypotheses for future longitudinal studies.
The expected outcomes include critical insights into dietary behaviors and their implications for diabetes prevention and management. The findings are anticipated to guide personalized dietary recommendations and public health strategies, contributing to scalable interventions in resource-constrained settings.
Study Status
The study will be open for recruitment from January 30th, 2025. We expect to complete the recruitment of all the participants by May 30th, 2025. The protocol version was approved on January 17th, 2025.
Publication Plan
The study findings will be disseminated through a core manuscript detailing macronutrient intake patterns and their association with metabolic markers, targeting a high-impact peer-reviewed journal. Additional focused articles will address specific observations, such as meal frequency, water intake, and regional dietary variations, to guide public health strategies and clinical practices. Results will also be presented at academic conferences and public health forums to engage stakeholders and promote actionable insights.
Footnotes
Acknowledgements
The authors acknowledge the contributions of the team at USV Private Limited, including Dr. Abhijit Pedenekar, Dr. Amit Gupta, Ms. Shashikala Borhade, Mr. Vinod Darade, Mr. Keshav Sawant, and Mr. Raj Patwa, for their roles in protocol development, operational planning, and data management setup.
The authors would also like to express their gratitude to the steering committee members for their guidance in conceptualizing this protocol. The operational support provided by Ms. Natasha Vora and Ms. Shefa Syed from Nurture Health Solutions, along with the Mediclin Clinical Research Organization (CRO) team, is deeply appreciated.
Author Contributions and Consent for Publication
SS and TA drafted the initial manuscript with inputs from SK, NK, ND, PS and SG. SS and TA were involved in the study design discussions and finalization, as well as the write-up of the article, along with NK and ND. All authors have read and approved the final version of the manuscript and consent to its submission for publication.
Data Availability
The full study protocol, case report form, and pilot study details are available from the primary author upon reasonable request. Requests can be made in writing to Dr. Sanjay Kalra.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approvals and Participant Rights
The study protocol (Protocol No. MCR/CT/0424/04) was reviewed and approved by the Shah Lifeline Hospital and Heart Institute Ethics Committee. It complies with the Indian Council of Medical Research (ICMR) guidelines, the Declaration of Helsinki, and International Conference on Harmonization—Good Clinical Practice (ICH-GCP) standards.
Participants will be fully informed about the purpose, procedures, and potential implications of the study. Written informed consent will be obtained from all participants before enrollment. For individuals unable to read or write, an impartial witness will be present during the consent process to ensure transparency and understanding. Participation in the study is entirely voluntary and participants will have the right to withdraw at any point without providing a reason and without any consequences.
Data confidentiality and privacy will be rigorously maintained through the de-identification of participant information, secure electronic storage on password-protected servers, and restricted access to authorized personnel. Regular audits will be conducted to ensure compliance with ethical standards and data security protocols. The ethics committee will be updated on study progress, any protocol amendments, and the occurrence of serious adverse events (SAEs). A final report will be submitted upon study completion, summarizing key findings and adherence to ethical guidelines.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The development and implementation of this study protocol are facilitated by USV Private Limited, Mumbai, India.
