Abstract
Background:
Recent studies suggest that lipid-lowering agents (LLA) may reduce chronic periodontitis, but it is unknown whether this benefit extends to people with type 2 diabetes (T2D).
Objective:
We assessed the association between LLA use and periodontitis in Hispanic adults with T2D.
Design:
This was a cross-sectional observational study.
Methods:
We assessed the association of LLA use and periodontal parameters in 253 Puerto Ricans 40–65 years with T2D who participated in the Lipid-Lowering agents use in Periodontitis and Diabetes Study study. Participants were classified as (a) none- or <1 year, (b) 1–4 years, or (c) >4 years. The primary outcome consists of a tertile percent of sites with probing pocket depth (PPD) ⩾ 4 mm and the secondary outcome includes tertiles of percent sites with clinical attachment loss (CAL) ⩾ 4 mm. Multinomial logistic regression models adjusted for age, gender, smoking status, education, waist circumference, glycosylated hemoglobin A1C (HbA1c), bleeding on probing, examiner, and anti-inflammatory agents were used to estimate the association.
Results:
LLA (92.5%, statins) was used by 52% of participants. LLA use 1–4 years was associated with lower odds of PPD ⩾ 4 mm (OR: 0.22, p = 0.005; high versus low tertile) or lower odds of CAL ⩾ 4 mm (OR: 0.33, p = 0.02, middle versus low tertile), compared to those with LLA minimal or no use. This association was lost for participants who used LLA for >4 years. LLA users for >4 years with periodontal disease had elevated HbA1c (OR: 1.36, p = 0.05).
Conclusion:
The use of LLA for 1–4 years was associated with lower values of periodontal parameters versus minimal LLA use. This association was not present among people using LLA > 4 years users, but these participants had poorer glycemic control compared to other participants. In this cross-sectional study, the finding that LLA use 1- 4 years is associated with lower values of periodontal parameters of severity in T2D individuals may help clarify some of the controversies regarding the benefit of these medications in this population.
Plain language summary
• Lipid-lowering agents (LLA), such as statins, are reported to reduce chronic periodontitis, making individuals with diabetes ideal candidates for this treatment due to their high prevalence of oral disease. • The association between LLA use and periodontitis was assessed among Hispanic adults with type 2 diabetes (T2D). • Short-term LLA use (but not long-term use) was associated with lower values of periodontal parameters in individuals with T2D. • For long-term LLA use, high glycemic level was associated with high values of periodontitis. • Thus, under the study design limitations, the finding may provide clinical implications and may help clarify some of the controversies regarding the benefits of LLA use in humans. • Furthermore, the higher glycemic levels for periodontal patients with long-term LLA use points to the need to monitor these patients carefully.
Keywords
Introduction
Periodontal disease (PD) is due to bacteria-induced inflammation that can lead to long-lasting gingival inflammation that affects the soft connective tissue and supporting bone. PD ranges from simple reversible gingivitis to a more serious chronic periodontitis formerly known as adult periodontitis. Although bacterial pathogens are the major cause of PD development, their presence alone is not sufficient for the disease to occur. The mechanism of action is dependent on a host-mediated tissue injury caused by the immune response that plays an important role in chronic periodontal tissue destruction.1,2
PD is a major cause of tooth loss, which can compromise mastication, esthetics, self-confidence, and quality of life. 3 The prevalence of PD is approximately 42% in dentate Americans aged 30–79 years 4 and is even greater among the Hispanic population. According to the available data, nearly 45% of older Puerto Ricans aged 70–97 years have moderate/severe PD, while the corresponding estimate for all Americans aged 75 years or older is 21%. 5 Yet, PD is preventable, especially at earlier age. Periodontal disease is associated with systemic conditions linked to increased chronic inflammation such as cardiovascular disease (CVD), diabetes, or rheumatoid arthritis. 6
An association between diabetes mellitus and PD has been recognized for over 70 years. 7 Animal experiments and human studies have demonstrated that diabetes is a risk factor for PD.8–11 Mechanistically, diabetes enhances periodontal inflammation, and animal studies have shown that it alters the oral microbiome to enhance its pathogenicity.12,13 Recent studies suggest the association between these diseases is bi-directional as PD can worsen glycemic control. 10
According to recent meta-analysis findings, individuals with type 2 diabetes (T2D) have a 34% increase in the risk of developing PD (incidence) compared to individuals without T2D. 10 In specific populations, such as the Pima Indians, who may be related to the Caribbean Taino Indians of Puerto Ricans 14 , the evidence of the association is even stronger: The odds of having PD is three-to-four fold higher in individuals with T2D compared to normoglycemic individuals. 15 Early and prolonged progression of T2D increases the extent and severity of PD. 16 The impact of diabetes on PD severity and progression is inversely proportional to the degree of glycemic control. 7 The prevalence of T2D in 2019 was 16.7% in Puerto Rico as compared to 10.7% in the US mainland. 17 Given the increasing prevalence of T2D, it is likely that PD will also continue to increase,8,18 highlighting the critical need to develop strategies to treat or prevent PD in individuals with T2D to improve their oral health and quality of life.
Lipid-lowering agents (LLA) such as statins, fibrates, and omega-3 fatty acids have beneficial effects on chronic inflammatory diseases, including periodontitis.19–22 Of these, statins are likely to have the greatest potential for treatment of PD as they have pleiotropic anti-inflammatory and immunomodulatory, antibacterial, as well as positive effects on bone metabolism, epithelization, and wound healing.23–26 In vitro and some in vivo experimental or observational studies have reported beneficial effects of statins in the treatment of periodontitis or an association between statin use and improved periodontal outcomes.24–27 Clinical trials involving statins and PD have largely focused on local delivery and the results have been controversial. 24 The potential association between LLA and periodontitis in individuals with T2D have been less explored, even though diabetic populations have a higher prevalence of PD and tooth loss and are more likely to be prescribed LLA. Therefore, our main objective was to assess whether LLA usage was associated with better periodontal status in individuals with T2D.
Materials and methods
Study population
The present study is a cross-sectional observational study. The study population consisted of 253 non-institutionalized, residents of Puerto Rico who were primarily of Hispanic origin and were recruited for the ‘Lipid-lowering agents use in Periodontitis and Diabetes Study’ (LLIPDS). Participants were drawn from the Puerto Rico Center for Diabetes (PRCD) (50%); from the general population (45%) among which, 7% of them came from the San Juan Overweight Adults Longitudinal study (SOALS); and from COSSMA (5%), a private decentralized healthcare organization located in Puerto Rico.
Eligibility criteria
To allow for outcome comparisons with our original study, 28 most of the following criteria derived from the previous SOALS’ study: inclusion criteria: (a) age 40–65 years, (b) at least four teeth for a valid periodontal measurement, and (c) T2D based on physician diagnosis, medication(s), or fasting blood glucose and glycosylated hemoglobin A1C (HbA1c) levels as described below; and exclusion criteria: (a) orthodontic appliances or gross oral pathology that might impair periodontal measurements; (b) regular use of steroids, anti-inflammatory drugs (except aspirin at doses of ⩽150 mg/day), immunosuppressants, thiazolidinediones, or glitazones; (c) systemic complications, such as hemophilia or other bleeding disorders, chronic inflammatory diseases, or infectious disease within 6 months; (d) use of antibiotic therapy for any medical or dental condition prior to the clinical examination; or (e) use of any medication that affects periodontal status for 2 weeks or more within 1 month prior to the clinical examination; (f) diagnosis of congenital or chronic heart diseases, endocarditis, or rheumatic fever; (g) undergoing active dialysis treatment; (h) receiving anticoagulant therapy; (i) had undergone procedures related to CVD, including pacemaker or defibrillator implantation, or surgery on the heart or vessels involving the use of prosthetic material (e.g., stent); (j) had hip or other joint replacement surgery; or (k) were diagnosed with cancer with active radio/immunotherapy.
Ascertainment of diabetic status
Participants who met the main eligibility criteria that were assessed during the pre-clinical phone call screening and verbally consented to participate in the study were invited and scheduled for a clinical examination. Participants were asked to bring the written documentation from physicians, laboratory results, or use of diabetes medications labeled with their names at the clinical examination visit to confirm the presence of T2D. Participants from SOALS with likely T2D diagnosis had HbA1c ⩾ 6.5% or fasting blood glucose ⩾ 126 mg/dl). 29
All eligible participants from the pre-clinical phone call screening were scheduled to attend the clinical examination. Prior to any study procedures, eligibility criteria were further confirmed, participants were explained again about purposes of the study, the study procedures, voluntarily participation in the study, participant’s right to withdrawal at any time during the study procedures, and participant’s consent for the use of the data for research purposes. Written and signed informed consent was obtained from all participants prior to performance of the study procedures. The study was approved by the Institutional Review Board of the University of Puerto Rico (IRB # B0930116). The study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2013.
Enrollment in the study
The LLIPDS study was conducted between April, 2017 and March, 2020. A total of 595 participants were screened and of these 267 were eligible and enrolled in the study (Figure 1). A total of 14 participants initially enrolled in the study were later excluded for protocol deviations leaving 253 participants for analyses. A sensitivity analysis determined that exclusion of 17 participants who had taken anti-inflammatory drugs did not significantly impact the outcome measurements.

Selection of the 253 study participants, LLIPDS (26 April 26 2017 to 9 March 9 2020).
Periodontal status and tooth loss outcomes measurement
Periodontal status was assessed at six sites per tooth for all teeth except the third molars 30 using a periodontal probe with 2 mm markings (Hu Friedy PCP2). Probing pocket depth (PPD) was rounded down if the reading fell between two probe markings. 30 Examinations were performed by one of two dental examiners before participants’ interview to avoid bias. Using the same periodontal probing protocol procedures, both dental examiners 1 and 2 were trained and calibrated for measurements of periodontal pocket depth by two different gold standards provided at two different consecutive time periods (BD, NHANES; HH, Forsyth Institute). They achieved a 96% and 97% agreement within 1 mm of PPD measurement, respectively. Prior to the switch in the tasks in the project, examiner 2 additionally received a training and calibration with examiner 1 to achieve at least 96% within 1 mm of PPD measurement on inter-examiner agreement to ensure the data quality. The PPD measure performed by the dental examiner 1 was compared with the PPD performed by the dental examiner 2. There was no statistical difference in the percentage of sites with PPD ⩾ 4 mm between the two dental examiners (p = 0.342). The primary outcome was percent of sites with PPD ⩾ 4 mm, 31 which was categorized in low (reference), medium, or high tertiles based on its distribution. Likewise, the secondary outcome included tertile of percent of sites with clinical attachment loss (CAL) ⩾ 4 mm categorized in low (reference), medium, and high tertiles. Alternative periodontal outcomes included tertiles of number of teeth lost or having at least one site (or tooth) with PPD ⩾ 5 mm, with the latter indicating the severity of disease. Silness and Loe Plaque Index, 32 bleeding on probing (BOP) on any two (one from buccal and one from lingual surfaces) of the six sites examined per tooth was recorded. 33
LLA and other data collection
Participants were asked to bring their current medications and/or a list of all their current medications at the visit and completed a thorough interview-based questionnaire that collected information on LLA and other medications, dosage, and duration of use. In addition, to prove medication compliance, up to three pharmacy addresses where participants usually bought their medications were recorded, and the study personnel verified the pharmacy information within 2–3 weeks after the visit from approximately the first 30 eligible participants at the beginning of the study. Types of LLA included statins, fibrates, niacin, bile acid binding resins, cholesterol absorption inhibitor, omega-3 fatty acids, combinations, and injectable medications. The intake of food items, such as milk enriched with omega-3 milk fatty acids can provide benefits similar to LLA to lower cholesterol, but this information was not available. Data were directly recorded in RedCap. 34 Participants were classified as LLA users if they were currently taking LLA and LLA non-users if they reported no current or previous use of LLA. Duration of LLA use was categorized as follows: (a) none or minimal <1 year, (b) 1–4 years, and (c) >4 years based on previous reports.35,36 Statin-potency was categorized as low, moderate, or high based on its dosage and low-density lipoprotein-cholesterol reduction goals. 37 The questionnaire also gathered demographic data, and diabetes duration (years). Lifestyle health habits included smoking status defined as never smoked (i.e. report to never smoked or has smoked <100 cigarettes in lifetime), former smoker (had smoked at least 100 cigarettes but had stopped smoking prior to the study visit), and current smoker (smoked at least 100 cigarettes and continue smoking). 38 Alcohol consumption was defined as ‘current drinker’ (had at least 12 drinks of any type of alcoholic beverage in lifetime and had at least one drink in past 12 months) or ‘non-current drinker’ (i.e. lifetime abstainer: had fewer than 12 drinks in lifetime; former drinker: had at least 12 drinks in lifetime but no drinks in the past 12 months). 39 The questionnaire also collected exercise information (yes/no, ‘Have you exercised in the past 12 hrs.?’). The diabetes duration of the SOALS participants was based on the date they were provisionally diagnosed with diabetes up to the date when their glucose levels were remeasured in the current study. Waist circumference and height were measured two to three times to the nearest 0.1 cm, and averaged. Weight was measured two to three times to the nearest 0.5 kg using a standard calibrated stadiometer TANITA, Made in Japan, Tanita Corporation of America Inc. (Analyzer-TBF-310A), and averaged as well. Body mass index (BMI) was computed from the weight and the height measures (kg/m2). Blood pressure was taken three times after 1–2 min intervals, 40 and the average was computed. Participants were classified as hypertensive if they reported a physician-diagnosed hypertension, currently took antihypertensive medications, or had an average SBP ⩾ 130 mm Hg or average DBP ⩾ 80 mm Hg at the clinical examination. 41 HbA1c was measured using a Siemens Kit, Made in UK, Siemens Healthcare Diagnostics Inc. for DCA 2000 and DCA Vantage Analyzer. Other biochemical measures, such as fasting serum glucose, insulin, and a lipid panel, were performed at Clendo Reference Laboratories in Puerto Rico. 42 Homeostatic model assessment (HOMA) of insulin resistance (IR) was computed as [fasting insulin (mU/L) × fasting glucose (mmol/L)/22.5]. 43
Statistical analyses
A brief summary of the general characteristics of the study population was provided. Characteristics of the study population by periodontal status were described using mean (± standard deviation), median (25th, 75th percentiles) or frequency (percent) as appropriate. LLA and other variables were assessed by tertile of percent of sites with PPD ⩾ 4 mm. We assessed potential correlations between the covariates and avoided including two highly correlated covariates in the same model. Multinomial or binary logistic regression models were used to assess the associations between LLA duration of use and periodontal or tooth loss parameters. We controlled for major risk factors for periodontitis including age, gender, years of education, BMI or waist circumference, smoking status, alcohol consumption, and exercise. We also adjusted for plaque index or BOP. Other potential confounders, including levels of lipid panels, fasting glucose or HbA1c, duration of diabetes, self-reported use of anti-inflammatory agents, or dental examiners were considered for inclusion in the model using change in estimate procedures.
Results
The general characteristics, oral health status, and the distribution of periodontal outcomes and alternative periodontal parameters of the study population are described in Table 1. The average age of the study population was approximately 55 (±5.93) years. A total of 44% of the participants were male gender, 36% former or current smokers, and 43% classified to have current alcohol intake. The duration of their diabetic status ranged from 1 to 37 years with the mean duration of 10 years. Most of the participants were obese (mean BMI 34.92 ± 10.67), and nearly 66% of them were hypertensive.
Distribution of the general and oral health characteristics and alternative periodontal parameters of the study population (N = 253 participants, N = 34,274 sites).

BMI, body mass index; BOP, bleeding on probing; CAL, clinical attachment loss; CDC/AAP, Centers for Disease Control and Prevention/American Academy of Periodontology; HbA1c, glycosylated hemoglobin A1C; HDL-C, high-density lipoprotein cholesterol; HOMA-IR, homeostatic model assessment of insulin resistance; LDL-C, low-density lipoprotein cholesterol; LLA, lipid-lowering agents; PPD, probing pocket depth; yr, year; yrs, years.
All the LLA used were oral medications. Approximately 52% of the participants were current LLA users. Among LLA users, the median duration of use was 4.21 (1.17, 6.5) years. Statins were the most used LLA (92.5%), followed by fibrates (5.3%), and the remaining 2.2% (others) were omega-3, cholestyramine, or ezetimibe (data not shown). Among statin users, approximately 28% (33/118) took high-potency statins (7 participants in LLA use <1 year; 9 in LLA use 1–4 years; and 17 in LLA > 4 years).
The highest tertile of percent of sites with PPD ⩾ 4 mm ranged from nearly 4% to 86.5% and for percent of sites with CAL ⩾ 4 mm from 7.6 to 88%; 52.6% of the participants had tooth loss ranging from 2 to 14 (medium/high tertiles), and 29.6% of the participants had at least one tooth with PPD ⩾ 5 mm. Based on the Centers for Disease Control and Prevention (CDC)/American Academy of Periodontology (AAP) periodontitis case definition, 44 approximately 62% of the participants had moderate or severe forms of this disease.
The general and oral health characteristics of the study population by tertiles of percent of sites with PPD ⩾ 4 mm are shown in Table 2. T2D participants with the highest tertile percent of sites with PPD ⩾ 4 mm were more likely to be males (p < 0.01), former or current smokers (overall, p = 0.06), and were less likely to be educated (p < 0.001) than their counterparts with low or medium tertile of percent of sites with PPD ⩾ 4 mm. T2D participants had higher levels of HbA1c (p = 0.03), higher LDL-cholesterol (p = 0.01), higher fasting glucose (p = 0.03), insulin (p = 0.01), or HOMA-IR (p < 0.001) across the tertiles of percent of sites with PPD ⩾ 4 mm. There was no statistically significant difference in HDL-cholesterol across the tertile of percent of sites with PPD ⩾ 4 mm. The LLA other than statins taken ⩾ 1 year were removed from the data yielding a total of N = 238 (i.e. instead of 253), and the reference group was the same as the initial group of ‘None or LLA < 1 year.’ There was no statistical difference in the distribution of the statin potency across the three tertile groups either. Overall, there was a difference in the plaque index (p < 0.01), number of sites with BOP (p < 0.001) and number of missing teeth (p = 0.03) across the three tertile groups (Table 2). Likewise, there was an overall difference in periodontitis CDC/AAP case definition (p < 0.001), number of teeth with PPD ⩾ 4 mm and BOP (p < 0.001), or number of teeth with CAL ⩾ 3 mm & PPD ⩾ 4 mm at the same site (p < 0.001), across the three tertiles of percent of sites with PPD ⩾ 4 mm.
General and oral health characteristics by tertiles of percent of sites with PPD ⩾ 4 mm (N = 253 participants, N = 34,274 sites).

BMI, body mass index; BOP, bleeding on probing; CAL, clinical attachment loss; CDC/AAP, Centers for Disease Control and Prevention/American Academy of Periodontology; HbA1c, glycosylated hemoglobin A1C; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; HOMA-IR, homeostatic model assessment of insulin resistance; LLA, lipid-lowering agents; PPD, probing pocket depth; yr, year; yrs, years.
Using analysis of variance (ANOVA), Chi-square, Kruskal–Wallis H, or K-sample equality-of-median test.
P-value in bold indicates the level of significance of p < 0.05.
The crude and adjusted odds ratios (adjusted for age, gender, education, smoking status, alcohol consumption, waist circumference, HbA1c, BOP, examiner, and anti-inflammatory agents) of the associations between the LLA users and medium or high tertile percent of sites with PPD ⩾ 4 mm showed no significant reduction compared to the first tertile (Table 3). Participants taking LLA between 1 and 4 years had 78% lower odds of having high tertile percent of sites with PPD ⩾ 4 mm than those with none or minimal LLA use (adj. OR: 0.22, 95% CI: 0.08–0.64). There was no association between LLA use between 1 and 4 years and lower or medium tertile percent sites with PPD ⩾ 4 mm as compared to those with minimal LLA use. Moreover, there was no association between LLA use >4 years and tertiles of percent of PPD ⩾ 4 mm (Table 3). The sensitivity analysis excluding enrolled participants reporting use of anti-inflammatory agents (N = 17) showed similar results (i.e. reference: LLA non-users or users <1 year: LLA 1–4 years and high tertile of PPD ⩾ 4 mm: adj. OR: 0.23, 95% CI: 0.08–0.70; no association between LLA use 1–4 years and medium percent of sites with PPD ⩾ 4 mm; and no association between LLA use >4 years and the percent of sites with PPD ⩾ 4 mm). Likewise, additional adjustment for covariates with missing values, such as ‘diabetes duration’ (10 missing) or ‘exercise’ (2 missing) provided similar patterns (Supplemental Table S1) and replacing the covariate ‘BOP’ in the models with that of ‘plaque index’, which had seven missing values provided similar estimates (Supplemental Table S2). Due to the low number of high-potency statin use in each LLA duration of use category and several switches in dosage primarily due to drug intolerance, we could not compare the statin-potency between the LLA categories.
Association between LLA use and tertiles of percent of sites with PPD ⩾ 4 mm (N = 253 participants, N = 34,274 sites).

Adjusted for age, gender, education (⩽12 yrs, 12 yrs), smoking status (never or smoker, current), alcohol consumption (non-current, current), waist circumference, HbA1c, BOP, examiner, and anti-inflammatory agents.
Anti-inflammatory agents (n = 17) excluded from the data.
BOP, bleeding on probing; CAL, clinical attachment loss; 95% CI, 95% confidence interval; HbA1c, glycosylated hemoglobin A1C; LLA, lipid-lowering agents; OR, odds ratio; yr, year; yrs, years.
P-value in bold indicates the level of significance of p < 0.05.
Participants taking LLA for 1–4 years had an adjusted OR = 0.33 (95% CI: 0.13, 0.86) for medium (not the highest) percent of sites with CAL ⩾ 4 mm compared to participants with minimal LLA usage (Table 4). Similarly, there was no association between LLA use >4 years and the tertiles percent of sites with CAL ⩾ 4 mm. The sensitivity analysis by excluding from the data participants reporting taking anti-inflammatory agents (N = 17) after being enrolled showed consistent similar results. Tertiles of number of teeth lost (Supplemental Table S3) did not show any association with LLA use. The association between LLA >4 years and high tertile of teeth lost was borderline (adj. OR: 11.90, 95% CI: 0.91–3.97). The outcome of at least one tooth with PPD ⩾ 5 mm for a disease severity (Table 5) showed similar patterns as the primary or secondary outcomes (LLA use for 1–4 years: adj OR: 30, 95% CI: 0.11–0.78); no association with LLA use >4 years).
Association between LLA use and tertiles of percent of sites with CAL ⩾ 4 mm (N = 253 participants, N = 34,274 sites).
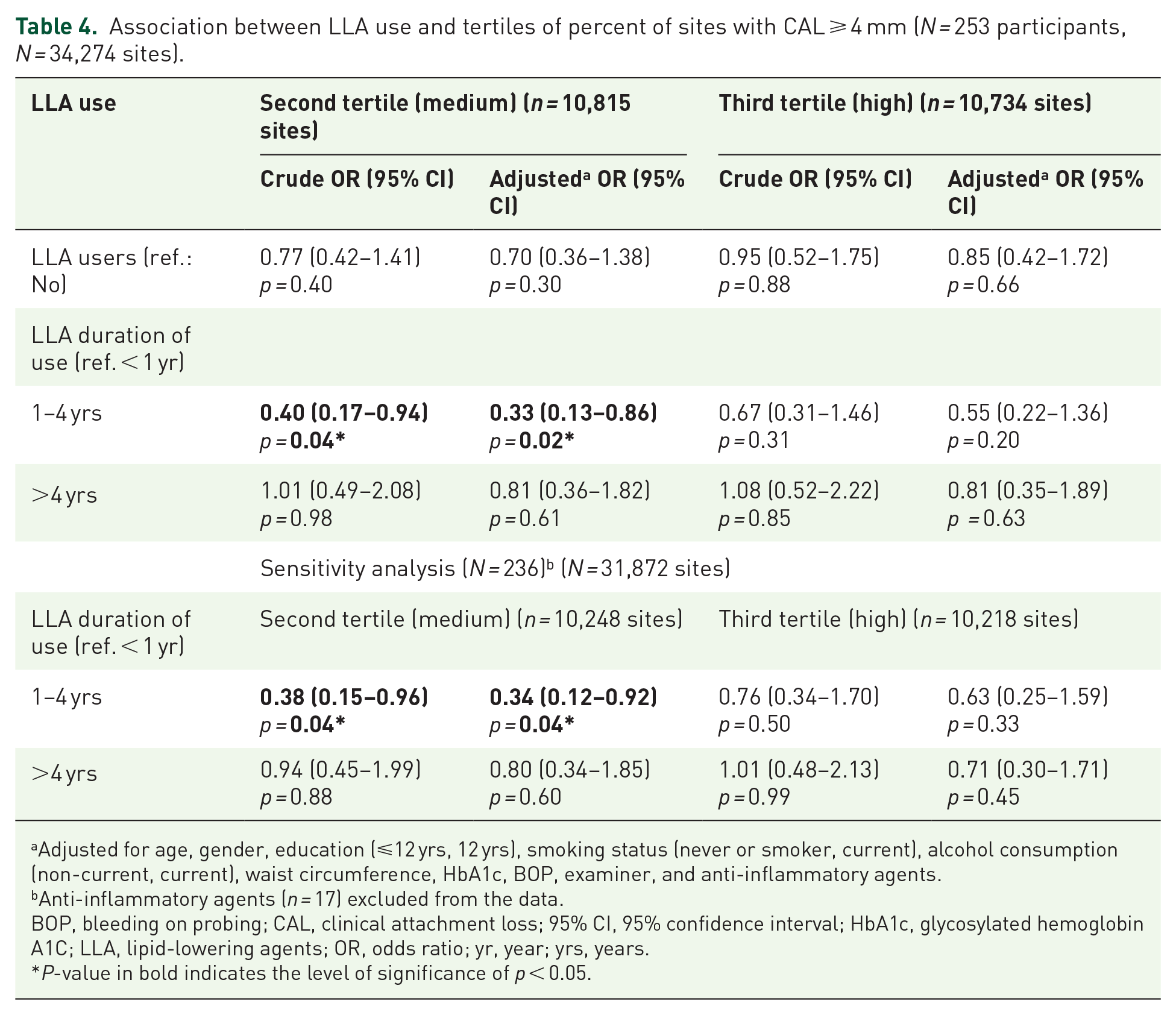
Adjusted for age, gender, education (⩽12 yrs, 12 yrs), smoking status (never or smoker, current), alcohol consumption (non-current, current), waist circumference, HbA1c, BOP, examiner, and anti-inflammatory agents.
Anti-inflammatory agents (n = 17) excluded from the data.
BOP, bleeding on probing; CAL, clinical attachment loss; 95% CI, 95% confidence interval; HbA1c, glycosylated hemoglobin A1C; LLA, lipid-lowering agents; OR, odds ratio; yr, year; yrs, years.
P-value in bold indicates the level of significance of p < 0.05.
Association between LLA use and having at least one tooth with PPD ⩾ 5 mm (N = 253 participants, N = 6483 teeth).
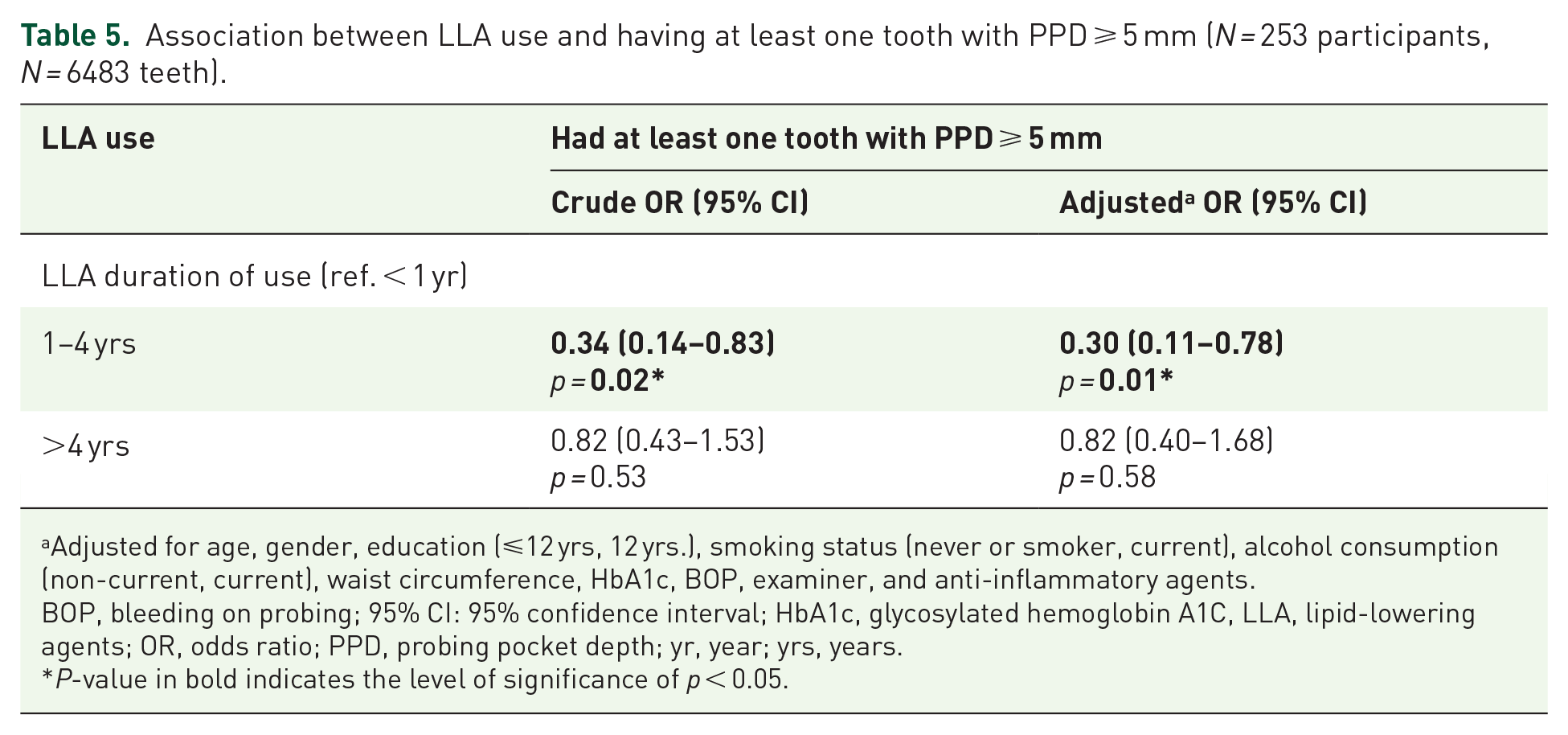
Adjusted for age, gender, education (⩽12 yrs, 12 yrs.), smoking status (never or smoker, current), alcohol consumption (non-current, current), waist circumference, HbA1c, BOP, examiner, and anti-inflammatory agents.
BOP, bleeding on probing; 95% CI: 95% confidence interval; HbA1c, glycosylated hemoglobin A1C, LLA, lipid-lowering agents; OR, odds ratio; PPD, probing pocket depth; yr, year; yrs, years.
P-value in bold indicates the level of significance of p < 0.05.
To further validate the results a number of alternative periodontal parameters were measured. They included periodontitis CDC/AAP case definition, a continuous form of the primary outcome percent of sites with PPD ⩾ 4 mm (log-transformed), number of teeth with PPD ⩾ 4 mm and BOP on the same tooth (log-transformed), or number of teeth with CAL ⩾ 3 mm and PPD ⩾ 4 mm at the same site (log-transformed), were done (Table 6). The trends remained consistent even though the statistical significance in some of the subgroups of the periodontal parameters was lost due probably to the lower proportions of the participants within them.
Association between LLA use and periodontitis CDC/AAP definition or other periodontal parameters (N = 253 participants, N = 34,274 sites, N = 6483 teeth).

Adjusted for age, gender, education (⩽12 yrs, 12 yrs), smoking status (never or smoker, current), alcohol consumption (non-current, current), waist circumference, HbA1c, BOP (or plaque index for teeth with PPD ⩾4 mm and BOP), examiner, and anti-inflammatory agents.
Log-transformed of the periodontal variables and use of multiple linear regression models with robust standard errors.
β, beta coefficient; BOP, bleeding on probing; CAL, clinical attachment loss; CDC/AAP, Centers for Disease Control and Prevention/American Academy of Periodontology; 95% CI, 95% confidence interval; HbA1c, glycosylated hemoglobin A1C; LLA, lipid-lowering agents; OR, odds ratio; PD, periodontal disease; PPD: probing pocket depth; SE: standard error; yrs, years.
P-value in bold indicates the level of significance of p < 0.05.
It is striking that among LLA users >4 years, the mean HbA1c was significantly greater than for participants who used LLA for less than 4 years (Table 7). Participants with an increase in HbA1c level had 36% higher odds of having medium/high percent of sites with PPD ⩾ 4 mm (adj. OR: 1.36, 95% CI: 1.00–1.85) than with a lower mean of HbA1c level.
Association between glycemic or insulin measures and medium/high tertiles of percent of PPD ⩾ 4 mm (reference: lowest tertile) by duration of LLA use (N = 253 participants, N = 34,274 sites).

Each glycemic measure evaluated in a separate model.
Adjusted for age, gender, education (⩽12 years, 12 years), smoking status (never or smoker, current), alcohol consumption (non-current, current), waist circumference, BOP, and anti-inflammatory agents.
BOP, bleeding on probing; 95% CI, 95% confidence interval; HbA1c, glycosylated hemoglobin A1C; HOMA-IR, homeostatic model assessment of insulin resistance; LLA, lipid-lowering agents; OR, odds ratio; PPD, probing pocket depth.
P-value in bold indicates the level of significance of p < 0.05. The value number was rounded-up to 0.05.
Discussion
We found that administration of LLA such as statins for 1–4 years is associated with lower values of periodontal measures in individuals with T2D, but the benefit is lost for LLA use beyond 4 years. Moreover, the loss of long-term benefits from statins may be influenced by poorer glycemic control that enhances risk factors for periodontitis among T2D-participants. The results of this study may have important clinical ramifications for individuals with T2D and PD who are treated with LLA.
HMG-CoA reductase inhibitors, referred to as statins, are the most prescribed lipid-lowering drugs used to prevent/treat CVD.45,46 Previous studies suggest that statins might improve periodontal parameters based on their anti-inflammatory and antibacterial properties.24,25 Few clinical studies have reported the impact of orally administered statins on PD or tooth loss.24,26,36,47–50 Our study showed that individuals with T2D who reported taking LLA between 1 and 4 years have lower values of periodontal parameters compared to T2D participants with minimal LLA use. However, this benefit was lost for participants taking LLA for more than 4 years.
Similar studies in individuals with no T2D have been conflicting. Some studies reported associations between statin use and lesser periodontitis severity26,27,48,50; two studies showed little relationship between statin use and tooth loss.49,51 A retrospective study by Cunha-Cruz et al. from 1021 individuals reported that any statin use during the first 3 years after the initial periodontal exam was associated with a 48% reduction in tooth loss in subsequent years. Meisel et al 47 showed statin use over 5 years was associated with nearly 28% reduction in the risk of tooth loss in a population-based longitudinal study in Pomerania but did not show a reduction in PD progression probably due to the limited periodontal analysis. A recent prospective study based of a large population-based electronic database from the Korean National Health Insurance Service-Health Screening (KNHIS-HS) cohort reported an increase in the risk of developing chronic periodontitis with long-term use of statins (>1 year) compared to short-term users (⩽1 year). 52 However, the interpretation of the results is tempered by determination of periodontitis using an insurance code rather than clinical measurements.
Our previous findings from the baseline data of the SOALS, which included overweight or obese individuals showed LLA to be associated with both lower systemic and gingival/periodontal inflammation. 53 In the present study, among individuals with T2D in whom systemic inflammation is common, oral intake of LLA appeared to be associated with a clinically healthier periodontium, but only for 1–4 years duration. The reasons for the limited benefits may be due to the small sample size power of the analysis or the impact of statins on T2D onset or severity or an unanticipated consequence of LLA treatment. We found that LLA > 4 years was linked to higher HbA1c levels in participants with periodontitis. This is consistent with reports that statins are associated with an increased risk of new-onset of T2D 54 and/or worsened glucose control. 55
In the present study, LLA use of 1–4 years was associated with lower odds of having high tertile percent of sites with PPD ⩾ 4 mm, and lower odds of having medium percent of sites with CAL ⩾ 4 mm as compared to low tertile of respective outcomes. Oral clinical parameters, PI and BOP were not treated as outcomes, and were used to adjust the statistical models to assess the magnitude of the estimates of the association between LLA use and PDs or tooth loss. We defined BOP based on the presence of bleeding at the buccal and lingual surface of each tooth when any site on those surfaces was bleeding after probing. 56 Ideally, we could have used any of the six tooth sites with BOP (interproximal distal at buccal side, mid-buccal, and interproximal mesial at buccal side, interproximal mesial at lingual side, mid-lingual, and interproximal distal at lingual side). However, when there was high level of bleeding while probing, the flow of the blood from one site contaminated the other sites in the pocket, and the exact source of the blood flow was difficult to determine. Thus, we only recorded bleeding from any random two of the six sites at the lingual and buccal surfaces of each tooth to increase measurement accuracy.
We understand that controlling for too many covariates in addition to data stratification by outcome levels may cause overfit of the multivariable model when the study aims to assess the association between an exposure and clinical outcome.57,58 However, the crude estimates of the associations by the outcome category were similar to the adjusted estimates. Moreover, the estimates obtained from reduced models by dropping other covariates, such as ‘examiner ID, and anti-inflammatory agents’ were similar to full models carried out in this work. It should be noted that some investigators prefer maximizing the covariate analysis, 59 especially in retrospective studies, to make the independent association more reliable.
The present study used periodontal parameters derived from the original proposal, which was submitted and accepted back in 2016, and the data were collected based on the original protocol. Thus, the latest classification of periodontitis from 2018 was not used. 60 Note that each periodontal parameter or case definition of PD has its own advantages or drawbacks, and so far, no perfect and/or satisfying PD case definition has been established. Recent findings, such as those from the study by Nascimento et al. reported the new classification system not to adequately capture the periodontal response to therapy in their study population. 61 Nonetheless, we have attempted to use the periodontitis CDC/AAP definition, which is based on both PPD and CAL parameters collected at the four interproximal sites of each tooth, which is used for population surveillance or prevalence studies. 44 We measured PPD and CAL at six sites per tooth. Although CAL on the direct buccal and lingual may be due to gingival recession, we included these measurements in the data analysis since periodontal inflammation, a focus of our study, is one of the primary factors contributing to gingival recession.62–66 Moreover, two other approaches were utilized,67–70 which included the increase in number of teeth with PPD ⩾ 4 mm and BOP on the same tooth, 67 or increase in number of teeth with CAL ⩾ 3 mm and PPD ⩾ 4 mm at the same site 68 to assess the associations. The periodontal parameter of ‘having a site with CAL ⩾ 3 mm and PPD ⩾ 4 mm at the same site’ would help to reduce, at least partially, the potential confounding of gingival recession at the buccal and lingual sites, although that would create a new bias with missing of sites with CAL but no pocket depth at the interproximal areas. Likewise, the parameter ‘having a tooth or site with PPD ⩾ 4 mm and BOP’ has quite the same drawbacks as the primary outcome ‘site with PPD ⩾ 4 mm’, which reflects the presence of periodontal inflammatory status without considering the CAL.
We used PPD as the primary outcome. Since PPD measures may include pseudo-pockets, we also examined CAL and tooth loss. These alternative outcome measurements also have disadvantages as sites with CAL may reflect gingival recession without formation of periodontal pockets and there are multiple reasons for tooth loss. 71
Due to the very small number of participants taking fibrates (5%) or other medications, such as omega-3, cholestyramine, or ezetimibe (2%), we could not present the results by these types of LLA use. However, these medications have the same indication of use as the statins to treat dyslipidemia even though the mechanisms of actions and the properties may differ, 72 and we combine them with the statins to be under the group of LLA as defined in the original proposal of the present study. We ensured valid and reliable data with rigorous LLA assessment and periodontal measures conducted using the NHANES protocol.30,73 We assessed and adjusted for important potential confounders, including sociodemographic data, lifestyle habits, oral hygiene measures (BOP or plaque index), and diabetes medications (metformin or insulin) and/or duration. Further adjustment of the models for factors related to other medications, such as blood pressure or neurological medications; serum lipid (i.e. HDL-C, LDL-C, total cholesterol, and triglycerides), glucose, or insulin levels; brushing or flossing teeth; dental exams or treatment, such as participants’ reports on the reason(s) to visit dental office within the last 12 months, which included routine dental examination (96/253), dental cleaning (165/253), diagnosis of PD (6/253); or history of previous periodontal treatment did not significantly change the estimates of the associations. The extensive exclusion criteria were challenging for recruitment, but necessary to ensure the homogeneity of the sample population, reduce confounding factors, assure ethical principles, and improve the study validity.
Certain limitations need to be considered. Since this is a convenience sample, it is possible that the findings will not be generalizable. However, there is little reason to expect that the relationship between LLA and periodontitis/tooth loss would be different for a random population. Recall bias is inherent to a cross-sectional design. However, meticulous questions, including past and present detailed history of medication use (e.g., switch to other medications, the reason(s) to stop taking the medications, etc.), were administered in each subsequent year, which might have helped to boost participants’ memory. Healthy user bias might have occurred as well rendering to the beneficial effect of LLA use, that is, those who reported taking LLA might have likely been more aware of their health and engaged in a healthier lifestyle. However, further adjustment for lifestyle factors (e.g., smoking or alcohol drinking status, oral hygiene or doing exercise, etc.) did not alter the estimates of the associations. Another limitation included the definition of ‘former smoker’, which was defined as having smoked at least 100 cigarettes, but had stopped smoking prior to the study visit regardless of the duration of non-smoking given the impact of smoking cessation on periodontitis. 74 However, due to the low quality and complexity of the information on participant’s report on several times of attempts to stop smoking, we regrouped and used the smoking status in category of ‘current’ versus non-current smoking throughout the data analysis. Finally, the cross-sectional nature of the study of the association does not enable a causal interpretation. Both LLA and periodontal status were measured at one time point, and we cannot disentangle the temporal sequence. The study of the independent association between LLA use and the values of periodontal parameters in individuals with T2D is challenging due to the interaction and/or correlation between the various conditions, such as obesity, lipid measures, glycemic control, and periodontitis. Moreover, obesity, T2D, and PD may share underlying common biological mechanisms, as these chronic health conditions often occur in the same individuals revealing a comorbid condition,75,76 making it difficult to disentangle the independent effect of one on others.
Conclusion
This cross-sectional study suggests LLA use for 1–4 years to be associated with lower values of periodontal parameter outcomes in patients with T2D. Large, prospective clinical studies or trials are needed to explore this further. Interestingly, our study also supports a relationship between poorer glycemic control and PD as HbA1c levels were significantly higher in individuals with PPD ⩾ 4 mm among LLA users >4 years. Under the study design limitations, the finding that short-term LLA use is associated with lower values of periodontal parameters in a diabetic population, but not long-term use has clinical ramifications and may help clarify some of the controversies regarding the benefit in humans. Furthermore, the poorer glycemic control for periodontal patients with long-term LLA use points to the need to monitor these patients carefully.
Supplemental Material
sj-docx-1-taj-10.1177_20406223231213252 – Supplemental material for Hispanic adults with type 2 diabetes mellitus using lipid-lowering agents have better periodontal health than non-users
Supplemental material, sj-docx-1-taj-10.1177_20406223231213252 for Hispanic adults with type 2 diabetes mellitus using lipid-lowering agents have better periodontal health than non-users by Oelisoa M. Andriankaja, Kaumudi J. Joshipura, Michael A. Levine, Margarita Ramirez-Vick, Julio A. Rivas-Agosto, Jorge S. Duconge and Dana T. Graves in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
The authors also acknowledge the LLIPDS team (Dr. Francisco Jiménez, Mr. Francisco Muñoz-Torres, Mr. Abdiel Castillo, Ms. Claudia Díaz, Mr. Alexis Acevedo, Ms. Patricia Serrano, Mr. Alejandro Llera, and all who participated in the study) the Alliance (formerly PRCTRC) personnel, who contributed to the conduct/oversight/planning of data collection of the study (administrative and regulatory affairs: Ms. Antonia Ortiz, Ms. Ivette Molina, and Ms. Adelma Rivera; nurses: Ms. Bárbara Guzmán, Ms. Sheyla Garced, Ms. Ladimila De Lima, and Mr. Robert Pinder; laboratory work: Mrs. Nilda González and Ms. Carola López-Cepero), and Dr. Hatice Hasturk, Director of the Center for Clinical and Translational Research at the Forsyth Institute, who provided the training and calibration of the dental examiner. The authors also acknowledge the Puerto Rico Diabetes Center, COSSMA, and all participants, who contributed to and participated in the study, and Dr. Yan Huang at the Medical University of South Carolina, College of Medicine for his additional help in the data interpretation.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
