Abstract
Metabolic dysfunction–associated steatotic liver disease (MASLD) has attained epidemic proportions in recent years, affecting nearly one-third of the general population and two-thirds of patients with type 2 diabetes (T2DM). It encompasses a wide spectrum of pathophysiological entities, ranging from simple steatosis to steatohepatitis to fibrosis and cirrhosis, with both hepatic and extrahepatic implications, including cardiovascular disease. The possibility of MASLD should be considered in high-risk groups such as T2DM, obesity, metabolic syndrome, polycystic ovarian syndrome (PCOS), etc., with/ without evidence of hepatic steatosis on imaging or persistently raised liver enzymes. Initial assessment should include a comprehensive evaluation of comorbidities, exclusion of secondary causes of steatosis, and fibrosis risk stratification. The presence of NASH and fibrosis is the most important predictor of adverse clinical outcomes, and non-invasive risk assessment has been an area of intense research in recent decades. FIB-4 can be used as a primary tool to effectively rule out significant fibrosis, followed by vibration-controlled transient elastography (VCTE) or enhanced liver fibrosis (ELF) tests as secondary tools if the FIB-4 score is indeterminate or high. The management of MASLD patients has shifted from a hepatocentric to a multicentric approach, with a focus on lifestyle interventions, disease-modifying pharmacotherapy in selected patients, and optimized management of comorbidities and complications.
Introduction
Metabolic dysfunction–associated steatotic liver disease (MASLD), earlier known as non-alcoholic fatty liver disease (NAFLD), is a chronic condition that has attained epidemic proportions in the current health landscape. It is characterized by the presence of macrovesicular steatosis in ≥5% of hepatocytes and encompasses a broad range of pathophysiological entities, ranging from steatosis to steatohepatitis to fibrosis and cirrhosis. 1 Non-alcoholic steatohepatitis (NASH), accounting for about 20% of NAFLD, represents a more aggressive form of NAFLD characterized by hepatic inflammation and fibrosis, with a higher propensity to progress to cirrhosis and hepatocellular carcinoma (HCC).
MASLD is the second most common cause of liver transplantation after hepatitis C and is one of the most common causes of hepatocellular carcinoma (HCC).2-4 It has far-reaching implications beyond the liver, including an elevated risk of cardiovascular disease (CVD), obstructive sleep apnea, osteoporosis, chronic kidney disease, and extrahepatic malignancies such as carcinomas of the colon, stomach, pancreas, uterus, and breast. 5 CVD is the principal cause of death, affecting approximately 40% of patients, while malignant and nonmalignant liver diseases account for deaths in less than 10% of patients. 6
MASLD affects nearly 30% of the general population, with significant regional variations. 7 Shalimar et al. conducted a meta-analysis to report a higher disease burden in India, with a pooled prevalence of 38.6% in Indian adults. 8 On the other hand, NASH affects up to 12–14% of the general population. 9 There has also been a secular trend towards an increasing prevalence of NAFLD over the decades, with an estimated yearly increase of 0.7%.10,11 Patients with diabetes comprise a particularly high-risk group, with reported prevalence of NAFLD and NASH being as high as 55–70% and 30–40%, respectively. On the other hand, nearly half of NAFLD patients are diabetic, highlighting the bidirectional relationship between the two conditions. 10
The majority of high-risk patients present to general practitioners, physicians, and endocrinologists, which necessitates adequate knowledge of the entity to enable early identification and initiate appropriate management and timely referrals. The landscape of MASLD has been in a constant flux, with the emergence of new research on simplified non-invasive identification of at-risk groups, research on newer disease-modifying drugs, and a shift from a hepato-centric approach to holistic multidisciplinary care. Our article aims to provide a brief overview of the recent developments, diagnosis, and management of MASLD.
What’s in a Name?
The term “non-alcoholic steatohepatitis” (NASH) was first used by Jurgen Ludwig in 1980 to describe hepatic steatosis and injury mimicking alcoholic hepatitis in overweight and obese individuals. 12 This subsequently became a part of the umbrella term “non-alcoholic fatty liver disease” (NAFLD), and this nomenclature has since served as the framework for use in clinical practice and research alike.
However, the nomenclature has been revisited in recent years in the search of a terminology that accurately describes the pathophysiology, acknowledges the overlapping disease mechanisms, and is inclusive and non-stigmatizing. The “non-alcoholic” part of the nomenclature does not acknowledge the etiopathogenesis of the condition and the potential overlap with alcohol-related liver disease. Additionally, the use of the word “fatty” can be stigmatizing and deter people from seeking medical attention. Hence, the American Association for the Study of Liver Disease (AASLD) and the European Association for the Study of the Liver (EASL), along with relevant stakeholders, published a consensus for revised nomenclature, the salient features of which have been summarized in Table 1. 13
Revised Nomenclature of MASLD and Related Entities. 13
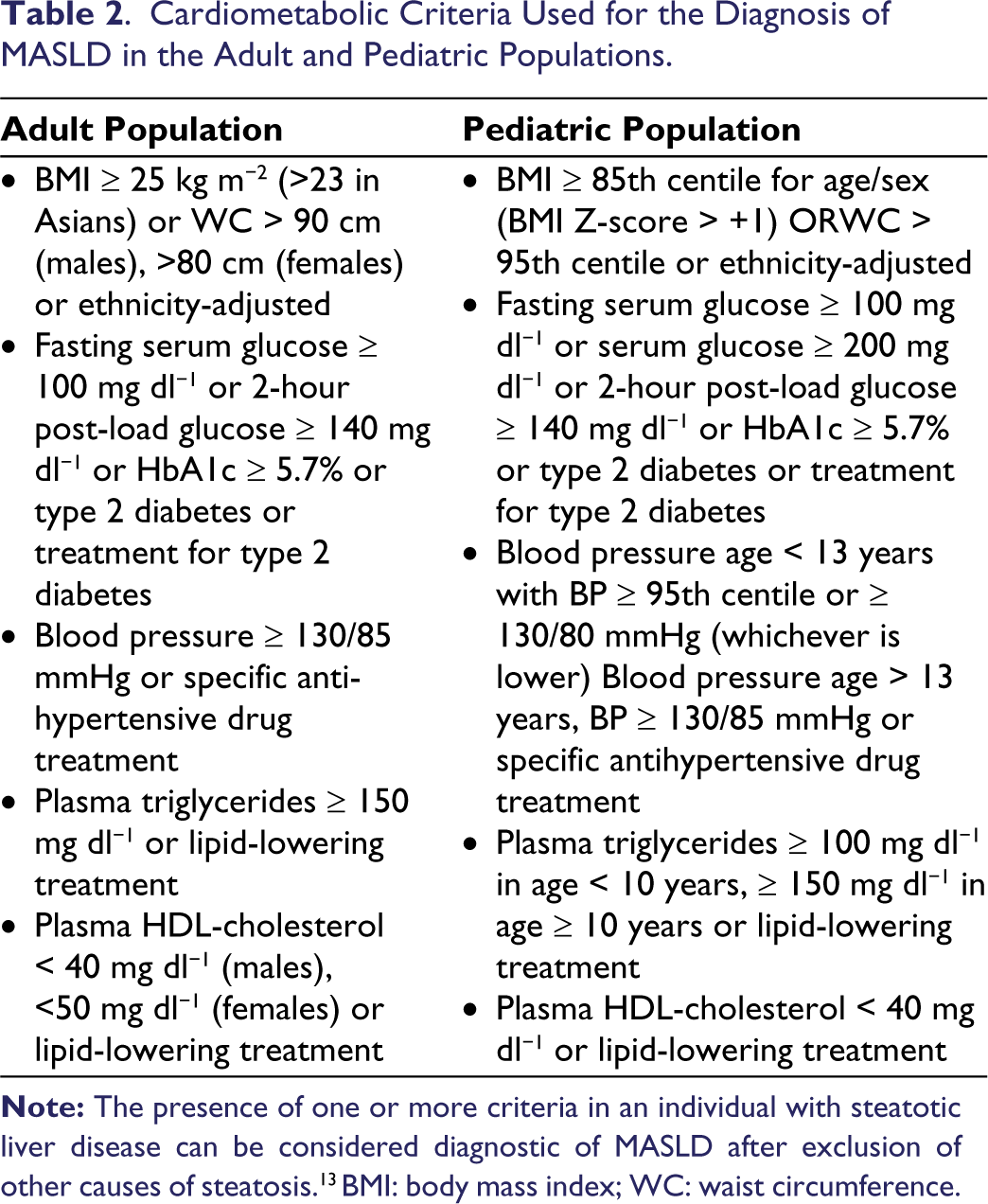
The cardiometabolic criteria used for the diagnosis of MASLD in adults and pediatric age groups are represented in Table 2.
Cardiometabolic Criteria Used for the Diagnosis of MASLD in the Adult and Pediatric Populations.
We will be using the term MASLD interchangeably with NAFLD in the article, as the vast majority of scientific literature has been described with the older terminologies.
Diagnosis of MASLD
The diagnosis of MASLD is based on the presence of hepatic steatosis in addition to a lack of significant alcohol intake and the exclusion of other liver diseases, as outlined above. Abdominal ultrasound is not required to diagnose steatosis in patients at a high risk of MASLD, and clinicians can directly proceed to the initial evaluation and risk stratification for fibrosis after excluding secondary causes of steatosis, irrespective of transaminase levels.
14
Targeted screening for MASLD and advanced fibrosis is recommended in the following high-risk groups:14,15
Obesity and/or features of metabolic syndrome Type 2 DM (T2DM), prediabetes PCOD NAFLD with moderate alcohol use First-degree relative of a patient with cirrhosis due to NAFLD/NASH Hepatic steatosis in any imaging study Persistently elevated transaminases over six months
The most common clinical scenario remains that of a patient presenting with incidentally detected steatosis on abdominal imaging or raised transminases. It should be noted that alanine transferase (ALT) levels should be interpreted as per the normal range of 29–33 and 19–25 U L−1 in males and females, respectively, which is lower than the usual ranges reported by laboratories. 16 General population-based screening for NAFLD is not recommended.
The approach to a patient with suspected/confirmed NAFLD should be two-pronged:
Comprehensive evaluation of associated comorbidities and complications Assessment of the risk of fibrosis
Initial Approach in MASLD Patients
The initial workup in patients with suspected/confirmed MASLD should include a comprehensive evaluation for attendant comorbidities and complications, as outlined in Table 3.
Initial Evaluation in Patients with Suspected/Confirmed MASLD.

OGTT: oral glucose tolerance test; OSA: obstructive sleep apnea.
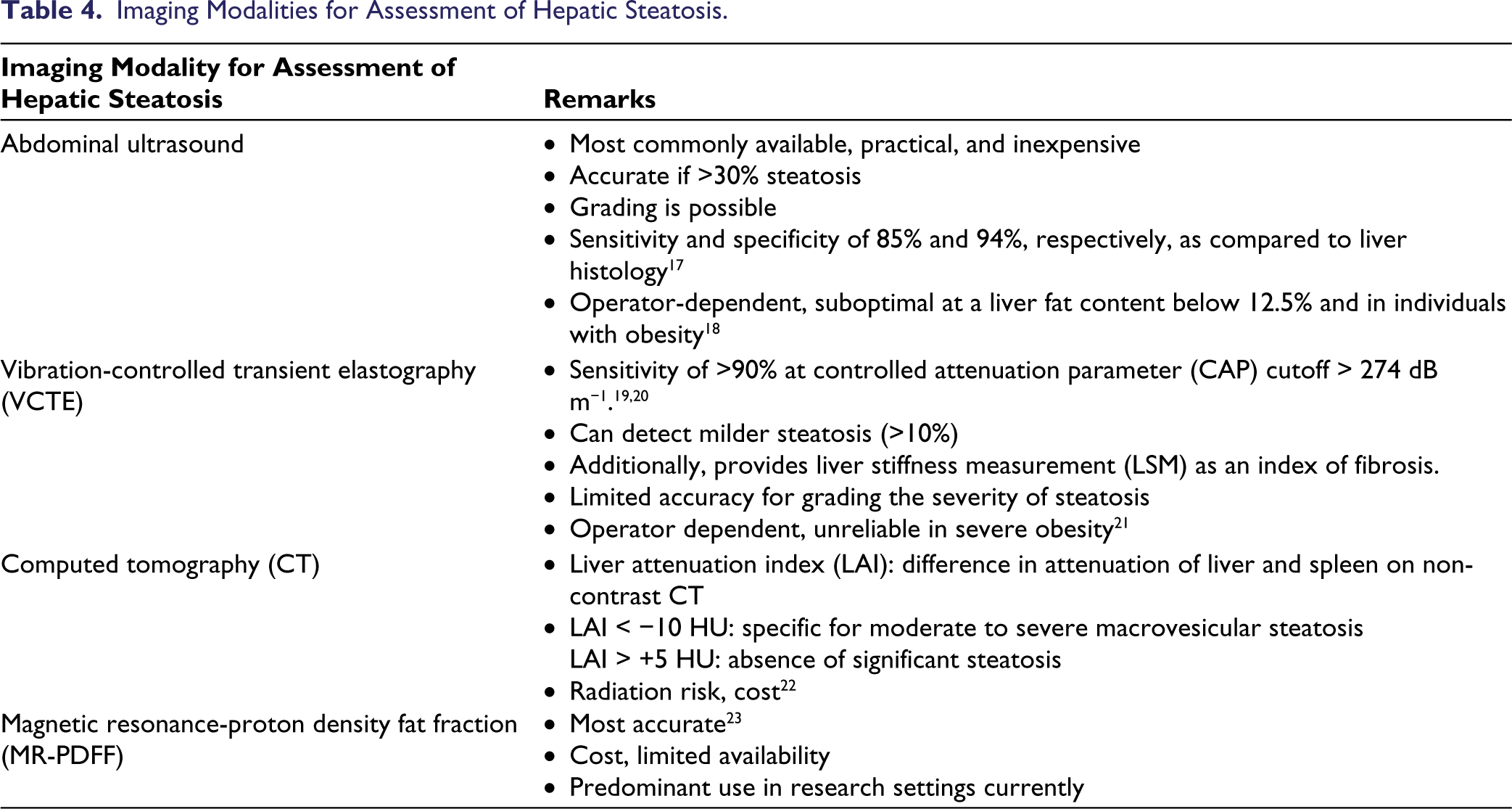
Imaging Modalities for Assessment of Hepatic Steatosis
The most common presentation is an incidental finding of hepatic steatosis on an abdominal ultrasound. Table 4 summarizes the imaging modalities that can be used to detect hepatic steatosis.
Imaging Modalities for Assessment of Hepatic Steatosis.
Non-invasive Assessment of Hepatic Inflammation and Fibrosis
Serum transaminases, often used in clinical practice for assessment of hepatic inflammation, have been found to have a poor correlation with hepatic inflammation in patients with MASLD. 24 Other non-invasive biomarkers for hepatic inflammation include cytokeratin-18 (CK18), integrated scores like Fibroscan-AST (FAST score), and MEFIB score (combination of MR elastography and fibrosis-4 score).25-27 As steatohepatitis is a major driver of fibrosis progression, clinical algorithms have instead incorporated non-invasive biomarkers to accurately detect clinically significant fibrosis (≥F2) in lieu of those that detect active inflammation.
Hepatic fibrosis remains the most important factor for prognosis, treatment, and outcomes in NAFLD. 6 While liver biopsy remains the gold standard for diagnosis of fibrosis and cirrhosis, several non-invasive serum-based markers (indirect and direct) as well as imaging-based biomarkers have been demonstrated to reliably predict clinically significant (≥F2), advanced (F3–F4) fibrosis, and cirrhosis (F4 fibrosis), and a few of them have been integrated into clinical practice algorithms. The available non-invasive biomarkers have been summarized in Table 5.5,14,15,19
Non-invasive Serum and Imaging Biomarkers for Fibrosis Risk Stratification.

Role of Liver Biopsy in MASLD
The gold standard for the diagnosis of steatosis, steatohepatitis, fibrosis, and cirrhosis is the histopathology of liver biopsy. However, liver biopsy remains an invasive procedure, with attendant risks like post-procedural pain in up to 50% of cases and potentially life-threatening complications such as hemorrhage in 0.6–1% of cases. 30 Additionally, it requires specialized equipment, trained staff, cost, and other logistical challenges. Hence, its utility is currently limited to patients with competing etiologies for hepatic steatosis, discordant results on non-invasive tests, and as an endpoint in clinical trials. 5
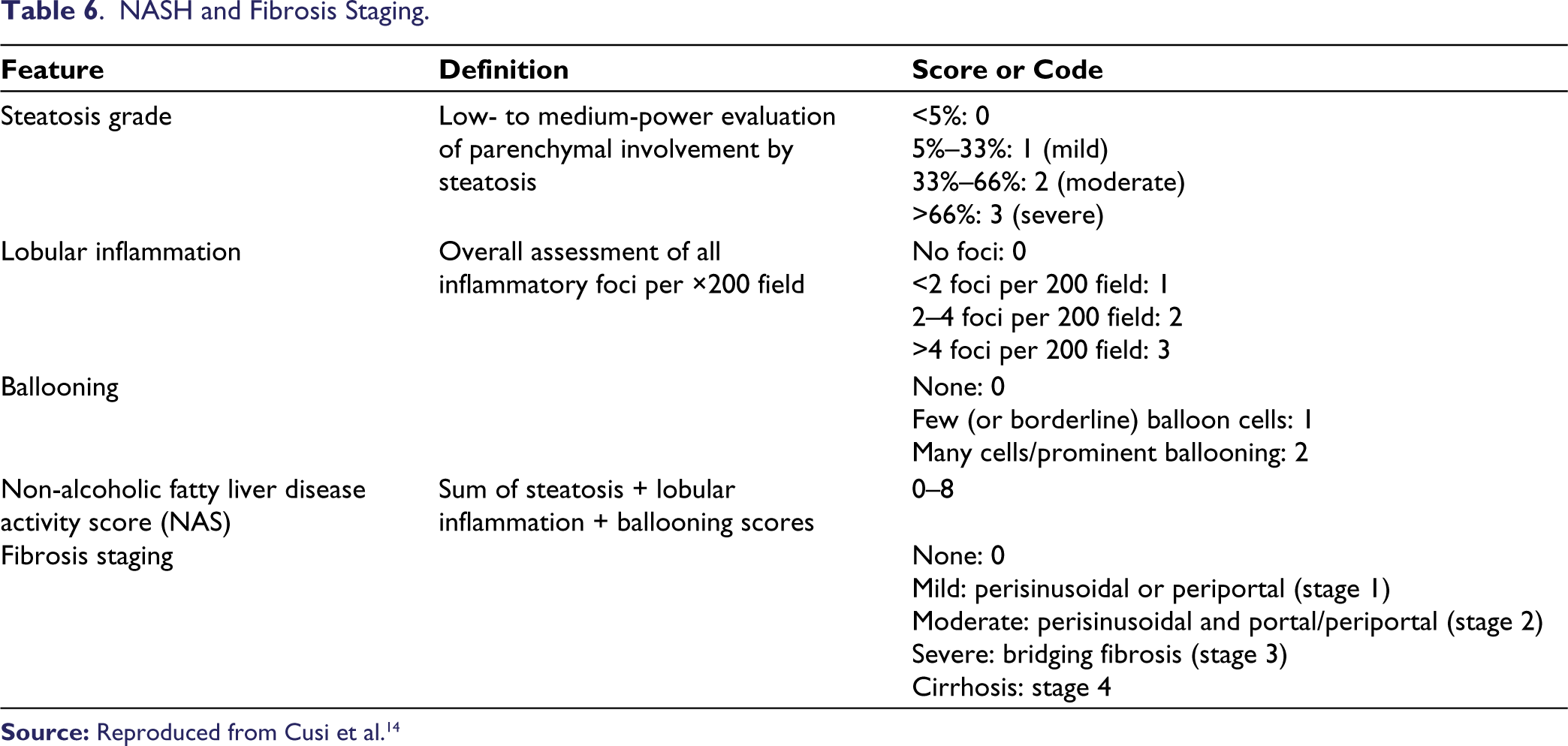
Several staging systems have been proposed, of which the non-alcoholic fatty liver disease activity score (NAS) is the most commonly used and validated score. Salient features have been described in Table 6.
NASH and Fibrosis Staging.
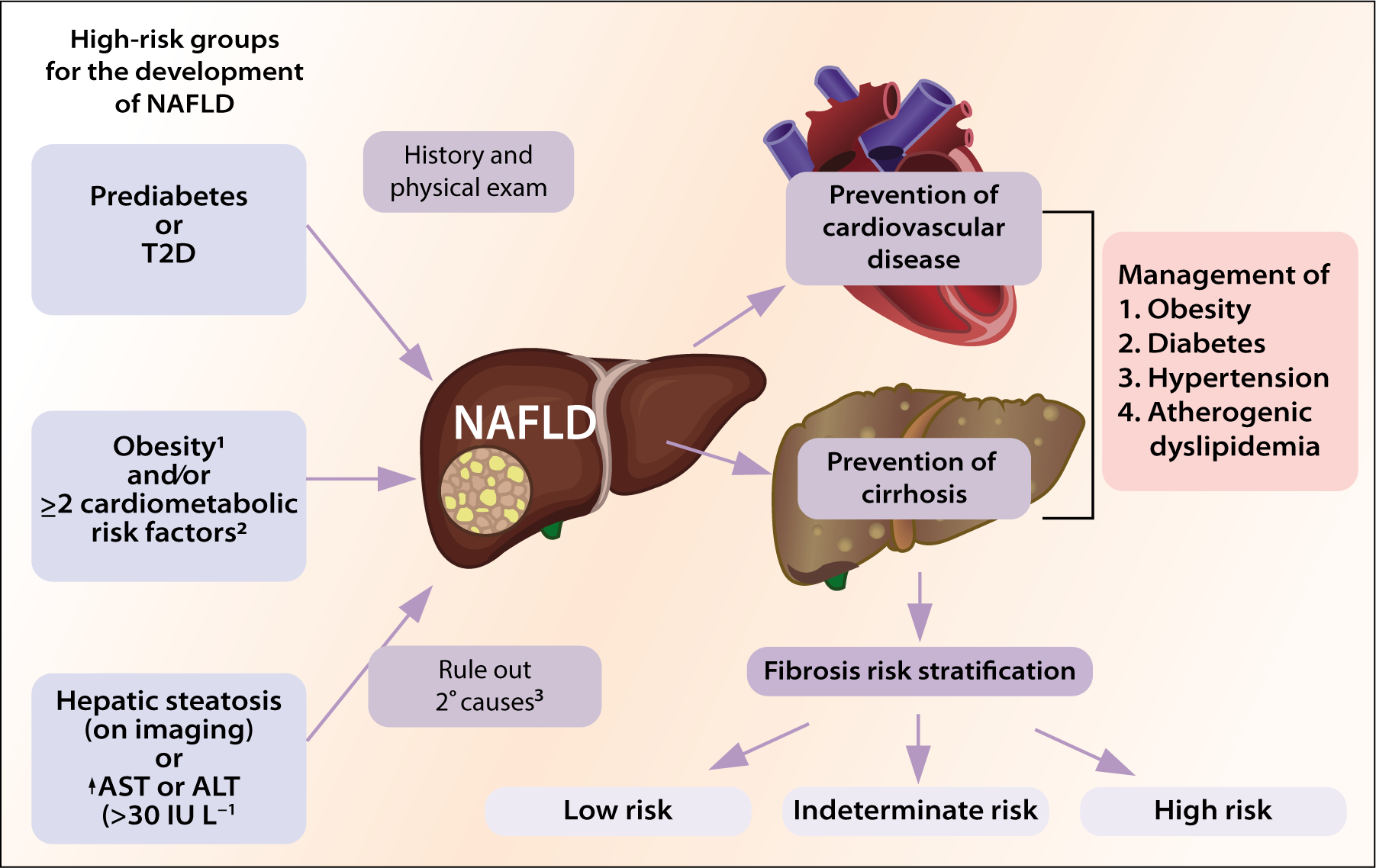
Diagnostic Approach in MASLD
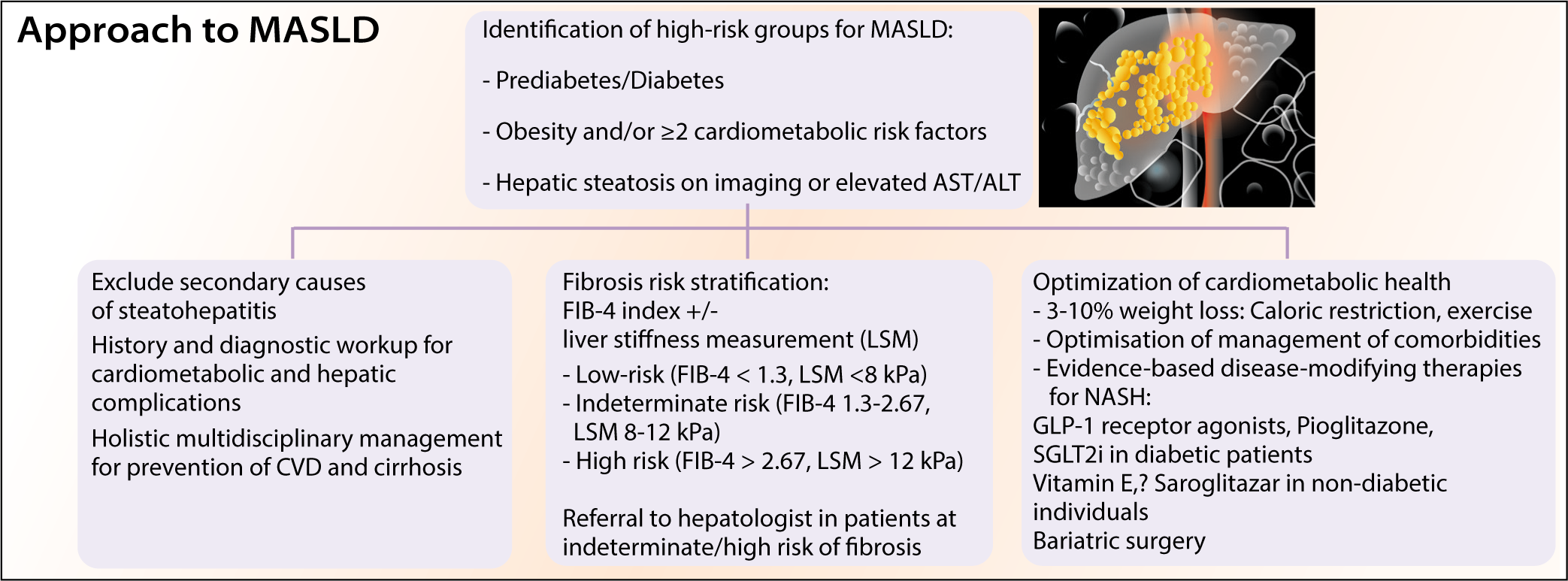
All patients with hepatic steatosis or clinical suspicion of MASLD based on the presence of metabolic risk factors should have a detailed initial assessment as detailed in Table 3 and fibrosis risk stratification as outlined in Figure 1.
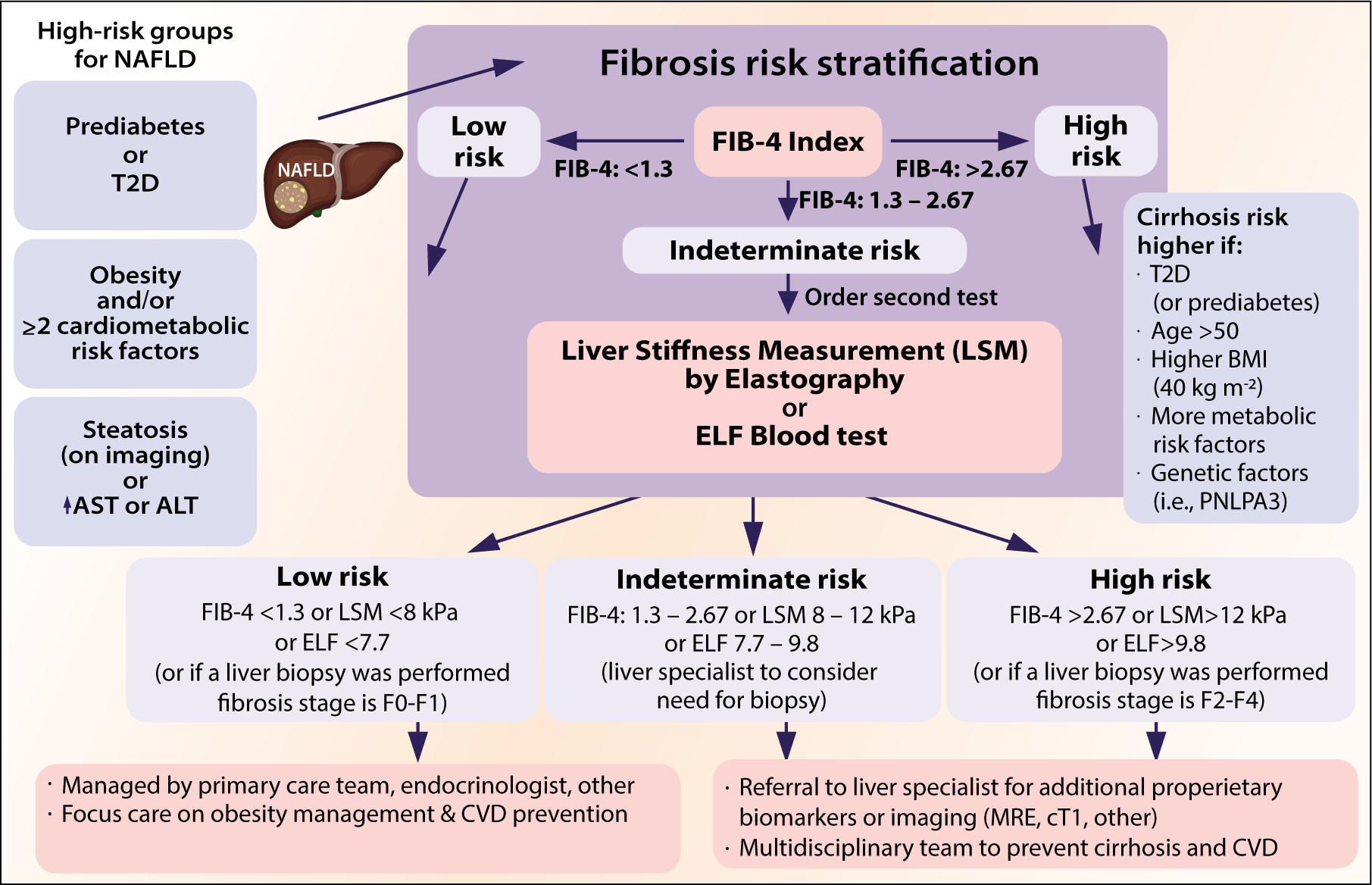
Primary risk assessment for fibrosis can be easily done in primary care settings with FIB-4. FIB-4 < 1.3 effectively excludes significant fibrosis, and patients can be followed up periodically in the primary care setting. Patients with an indeterminate (1.3–2.67) or increased (>2.67) FIB-4 index can be followed up with a secondary risk assessment with vibration-controlled transient elastography (VCTE) or enhanced liver fibrosis (ELF) panel. The diagnostic algorithm and recommendations have been outlined in Figure 2.
Management of MASLD
The role of the clinician caring for patients with NAFLD is to reduce the morbidity and mortality associated with the condition, so management has to move beyond a “hepato-centric” approach to a multidisciplinary holistic approach that includes the following:
Lifestyle education and intervention Liver-directed therapies for MASLD Assessment and optimization of T2DM and other cardio-metabolic risk factors
Lifestyle Interventions for MASLD
One of the first-line recommendations for NAFLD management is caloric restriction and exercise to induce a net weight loss. The histological improvement in MASLD has a dose–response relationship with the degree of weight reduction. Overweight or obese patients with MASLD should be encouraged to lose at least 7–10% of their body weight; even modest weight loss of 3–5% results in improvement in steatosis, but greater weight loss (>10%) is required to improve NASH and fibrosis.40,41
Sustained weight loss improves insulin sensitivity and adipose tissue stress. Only about one-third of overweight or obese adults with NAFLD receiving usual care achieve >5% weight reduction over 2 years of follow-up, and weight regain is common. 42
Dietary Interventions
Weight loss can be achieved by a calorie deficit of 500–1,000 kcal day−1 while ensuring adequate protein intake (1.5 g kg−1 body weight). The benefits are sustained irrespective of whether the calorie deficit is achieved by acute, very low-calorie diets (VLCD) or more modest reductions in calorie intake over an extended period.5,15,43 Caloric restriction is of prime importance versus the type of diet per se. Dietary composition should be modified to minimize the intake of saturated fats, refined carbohydrates, fructose, and sugar-sweetened beverages.
Exercise
Studies have shown that aerobic exercise—30 to 60 minutes of moderate- to high-intensity exercise performed 2–5 days per week—results in a significant reduction in liver fat content despite minimal or no weight loss, which is particularly relevant in patients with lean NAFLD. 44 Overall, there is clear evidence demonstrating that exercise, be it aerobic, resistance, or modified high-intensity interval training (HIIT) training, has the ability to lower liver fat content to a similar degree.14,45
Alcohol Use
Alcohol use can act synergistically with insulin resistance to accelerate fibrosis progression, especially with moderate to heavy alcohol use (>20 and >30 g day−1 in women and men, respectively). Hence, alcohol intake should be assessed at regular intervals in all MASLD patients, and alcohol abstinence should be recommended in these patients, especially with clinically significant fibrosis (≥F2). 15
Pharmacotherapy in MASLD
The therapeutic landscape in NAFLD is rapidly evolving and can be conceptualized into two broad mechanisms of action:
Restoration of normal adipose tissue biology, with fat redistribution from hepatic and visceral fat to subcutaneous fat (e.g., PPAR agonists) or Weight loss agents, where reduction of excess adipose tissue also restores adipose tissue biology/function and responsiveness to insulin (e.g., glucagon-like peptide-1 receptor agonists [GLP-1RAs] and new dual agonists)
Although several agents have undergone clinical trials, there is currently no FDA-approved medication for management of NAFLD.
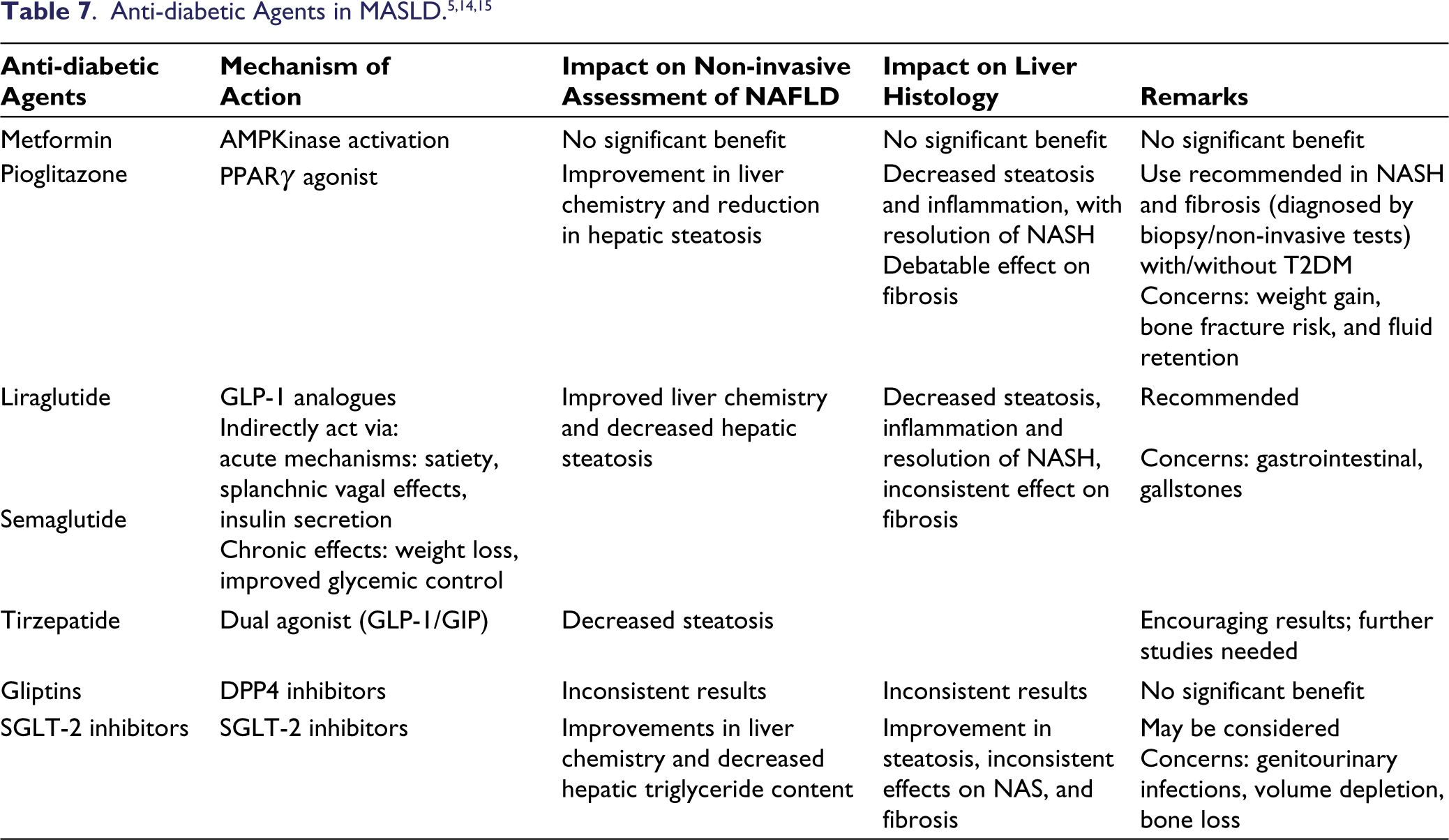
Anti-diabetic Agents in MASLD
T2DM occurs in nearly half of patients with MASLD. On the other hand, clinically significant liver fibrosis may be present in up to one-fourth of T2DM patients.46,47 Due to the close bidirectional relationship, it is important to understand the interactions of various antidiabetic agents on the natural course of MASLD and choose appropriate agents that can serve the dual purpose of addressing hyperglycemia as well as MASLD.
Pioglitazone, a PPARγ agonist, has demonstrated beneficial effects on liver transaminases, hepatic steatosis, and inflammation in several RCTs, with improvement in fibrosis noted in fewer studies and a recent meta-analysis.48-50 GLP-1 receptor agonists have changed the landscape of diabetes and obesity, and there is a growing interest in their role in the management of NASH. A recent meta-analysis of Liraglutide at doses used in diabetic patients reported efficacy in lowering plasma aminotransferases and liver fat content and improving liver histology in patients with MASLD.51,52 Semaglutide, at daily doses of 0.1, 0.2, or 0.4 mg, was reported to improve NASH resolution, without worsening of fibrosis, to a similar degree in people with or without type 2 diabetes. 53 Tirzepatide, a novel dual agonist at GLP-1 and GIP receptors, has demonstrated impressive results in the SURPASS series of trials with respect to weight loss and control of diabetes. In a substudy of SURPASS-3 in which tirzepatide was administered subcutaneously once weekly at doses of 5, 10, and 15 mg, it demonstrated a significant reduction in liver fat content versus insulin degludec in T2DM patients as evaluated by MRI-PDFF, with a treatment difference of −4.71% (95% CI −6.72 to −2.7, p < .0001). 54
Sodium glucose cotransporter-2 inhibitors (SGLT2i) comprise another class of agents that offer significant cardio-renal benefits in addition to the management of hyperglycemia. They decrease the severity of hepatic steatosis in individuals with T2DM and NAFLD by approximately 10–39%. It was also noted that the reduction in hepatic steatosis was more than expected with the modest weight loss seen with SGLT2i, suggestive of mechanisms beyond weight loss for their hepatic effects.55-58 A meta-analysis by Sinha et al. confirmed a reduction in liver enzymes and liver fat content with SGLT2i, suggestive of a salutary effect of SGLT-2i on NAFLD in T2DM, probably driven by an improvement of glycemia and body weight, which in turn attenuates hepatic inflammation and hepatic fat accumulation. 59
The impact of various anti-diabetic agents on MASLD has been summarized in Table 7.
Non-diabetic Medications in MASLD
Vitamin E is a potent antioxidant that has shown a reduction in transaminases, steatosis, inflammation, and cellular ballooning on biopsy in non-diabetic patients with NASH in doses of 800 IU day−1, with no impact on fibrosis. 50 There is some evidence to suggest an increased risk of hemorrhagic stroke and all-cause mortality, although the data on the relationship to prostate cancer risk is less clear. 60
Saroglitazar is a dual potent PPAR-α/γ agonist. The synergistic effect of improved lipid oxidation and improved insulin resistance by PPAR-α and PPAR-γ, respectively, makes its use in NASH/NAFLD rational. Real-world data from India showed Saroglitazar 4 mg day−1 is effective and safe for improving biochemical parameters, LSM, and CAP values, irrespective of weight reduction, with a debatable effect on fibrosis. It may be used in compensated cirrhotics with close monitoring for side effects. 61 The Drug Controller General of India (DCGI) has approved its use in NASH with F1–F3 fibrosis and in NAFLD with comorbidities (obesity, T2DM, metabolic syndrome, dyslipidemia).
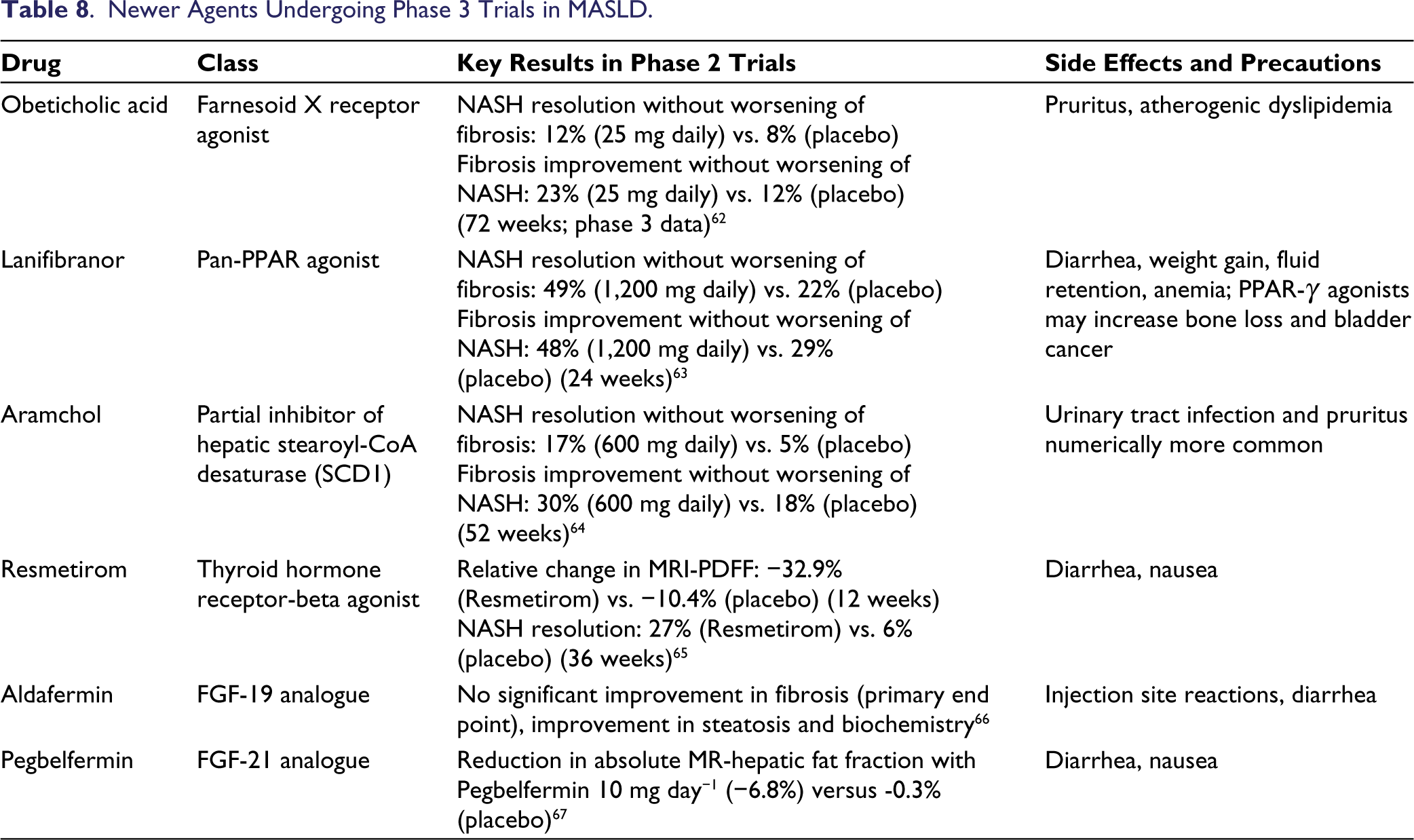
Newer drugs that modify the disease pathogenesis are in the pipeline for phase 3 trials in patients with NASH. Salient features have been summarized in Table 8.
Newer Agents Undergoing Phase 3 Trials in MASLD.
Bariatric Surgery
Metabolic surgery, including bariatric surgery, is an effective therapy for obesity and T2DM. A recent systematic review and meta-analysis showed that, in addition to weight loss, bariatric surgery significantly reduces the risk of progression to cirrhosis in obese patients as compared to matched controls. 68 Bariatric surgery more reliably and predictably achieves a >10% loss in total body weight, which is associated with NASH resolution and fibrosis improvement, than lifestyle and pharmacological treatments of obesity. 69 A meta-analysis by Lee et al. confirmed a significantly improved liver histology, including steatosis, ballooning degeneration, inflammation, and fibrosis, after bariatric surgery. However, bariatric surgery is associated with new or worsening features of NAFLD, like fibrosis, in 12% of the patients. 70 Caution has to be exercised in patients with advanced fibrosis and cirrhosis, as these patients are at a higher risk of hepatic decompensation and mortality. 14
Hence, bariatric surgery should be considered as an option to treat MASLD and improve cardiometabolic health in MASLD patients with a BMI ≥ 35 kg m−2 (≥32.5 kg m−2 in the Asian population), especially with coexisting T2DM. Additionally, it can also be considered in individuals with a BMI of ≥30–34.9 kg m−2 (≥27.5–32.4 kg m−2 in Asian population). 14
Optimization of Management of Comorbidities and Complications
The management of dyslipidemia should be optimized for cardiovascular risk reduction. Statins are safe for CVD risk reduction across the spectrum of NAFLD, except in decompensated cirrhosis. Hypertriglyceridemia can be managed by lifestyle changes, omega-3 fatty acids, icosapent ethyl, or fibrates.
Hypertension can be managed as per prevailing guidelines, with ACE inhibitors/ARBs as first-line agents; their use has to be avoided in decompensated cirrhosis. Calcium channel blockers, beta blockers, diuretics, and other classes of antihypertensive agents can be added as per clinical requirements.
Last but not least, liver cirrhosis can be associated with complications such as portal hypertension, gastroesophageal varices, hepatocellular carcinoma, and osteoporosis, all of which would need specific management under the care of a gastroenterology team. Hence, the management of MASLD requires a collaborative approach from primary as well as specialized care for holistic management of this truly multisystem disease.
Conclusion
The awareness of MASLD remains low among patients and clinicians, and there is an increasing need to develop simplified clinical algorithms for early diagnosis of these individuals and appropriate management of these patients. MASLD should be considered in high-risk groups such as T2DM/ prediabetes, obesity, metabolic syndrome, etc., followed by appropriate workup. Recommendations from major societies have also highlighted the need for a shift in the management strategy to holistic personalized care, involving primary care physicians, endocrinologists, nutritionists, and gastroenterologists.
Weight loss and exercise are central to any successful cardio-metabolic management strategy. The risk of hepatic and extra-hepatic complications associated with NASH should prompt considering the use of agents with therapeutic benefits backed by evidence. The presence of MASLD in diabetic patients should prompt a tailored management strategy, much akin to the establishment of treatment pathways in diabetic patients with cardiovascular disease, heart failure, and renal disease. Diabetic patients with NASH can benefit from the use of GLP-1 receptor agonists and pioglitazone, while vitamin E can be used in non-diabetic patients with NASH. We anticipate that novel pharmacological agents will become available soon and transform the current NASH management landscape, in which a combination of current “dual diabetes-NASH pharmacological agents” such as pioglitazone, GLP-1RAs, and SGLT2 inhibitors plus novel NASH pharmacological agents will create the framework for a more comprehensive treatment strategy.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.