Abstract
Non-alcoholic fatty liver disease (NAFLD) and non-alcoholic steatohepatitis (NASH) have been reported as a novel worldwide epidemic, very often associated with obesity, metabolic syndrome, and type 2 diabetes. Both conditions have also been shown to be associated with a number of endocrine pathologies. Despite the epidemic, the complex pathophysiology and major complications, ranging from metabolic disturbances (diabetes and more) to cardiovascular disease, people with NASH are left with very few management options. The best and most approved therapeutic option is lifestyle intervention.
Although pharmacotherapies based on pathophysiological background are in development, response rates appear modest, mainly for fibrosis treatment, which is the reason for lack of approved drug therapy. Previous drugs analyzed, such as pioglitazone and vitamin E, show weak efficacy. From different phase II trials, antidiabetic (injectable) drugs seem to be promising, both in mono- or bitherapy. Also, derivatives of peroxisome proliferator-activated receptors may have an interesting future, as well. For that reason, more focus should be given on prevention of this novel disease entity.
In view of this booming epidemic, with a background of obesity and type 2 diabetes, and the important medical consequences, early recognition, prevention and intervention of NAFLD/NASH seems appropriate. In this review, we will focus on the different current and future therapeutic intervention options, taking into consideration the complex pathophysiology of this disease.
Keywords
Introduction
Alongside the worldwide epidemic of obesity, metabolic syndrome and its cardiovascular complications,1,2 non-alcoholic fatty liver disease (NAFLD) is also rising globally, and is the primary hepatic complication of metabolic syndrome.3–5 NAFLD is defined as the presence of hepatic fat content, mostly triglycerides, greater than 5% of hepatic weight, in the absence of other causes of liver steatosis or chronic liver disease, e.g. viral and/or autoimmune hepatitis, hemochromatosis, alpha-1 antitrypsin deficiency, Wilson’s disease, steatogenic drug use or significant alcohol consumption.6,7 NAFLD is an umbrella pathology encompassing a histological spectrum ranging from simple or isolated steatosis to non-alcoholic steatohepatitis (NASH), with substantial liver-cell damage (evidenced on the liver biopsy as ballooning of hepatocytes) and inflammatory changes as key histological characteristics. NASH can be accompanied by progressive fibrosis and may ultimately lead to cirrhosis in up to 20% of cases. Cirrhosis, which is irreversible in the majority of cases, can be complicated by decompensation or by the development of a hepatocellular carcinoma, but the latter has also been described in earlier disease stages.2,7,8 Global prevalence of NAFLD is estimated ranging between 10% and 30%, with its highest prevalence in the Middle East and South America and lowest in Africa. 9 In the United States, NASH has been recognized as one of the leading causes of cirrhosis, and NASH-related cirrhosis is currently the second most common indication for liver transplantation. 10 Assuming the current trends in the prevalence of diabetes and obesity, it has been estimated that the overall liver-graft donation in 2030 will fall from 78% to 44%. 11 As the prevalence of fatty liver disease is increasing, it is expected that a higher proportion of potential donors will have steatosis or steatohepatitis and might be declined for liver-transplant purpose.
NAFLD is associated with several metabolic comorbidities including obesity in approximately 50% of NAFLD patients, type 2 diabetes (T2D) in more than 20%, and dyslipidemia in almost 70%. In patients with more advanced stages of liver disease such as NASH, prevalence estimates for T2D reach up to 43%, obesity prevalence to over 80%, and hyperlipidemia in over 70% of subjects. 10
Conversely, prevalence of NAFLD is high in patients with components of the metabolic syndrome and is reported in over 75% of patients with T2D and in over 90% of severely obese patients undergoing bariatric surgery. 9 Diabetes in individuals with NAFLD is a risk factor for progression toward NASH, cirrhosis, and for increased mortality, and poor glycemic control increases the risk of fibrosis in NASH. 12 Heart disease is one of the leading causes of death for NAFLD patients. 10 NAFLD and its severity has been suggested a major determinant of cardiovascular disease (CVD) outcome. However, when corrected for conventional risk factors, this link seems to disappear, 13 but subjects with both NAFLD and T2D carry a higher overall risk. 14
NAFLD patients have an almost twice-higher risk of incidental T2D compared with non-NAFLD patients and this risk increases with expanding waist circumference. 15 T2D and NAFLD are mutually, closely, and bi-directionally associated. 16
From a pathophysiological point of view, the liver is particularly prone to the accumulation of ectopic lipids when subcutaneous adipose-tissue-expanding capacity is reached. 17 Increased dietary fat intake, adipose-tissue-derived free fatty acids (FFAs) and de novo lipogenesis in the liver can all contribute to excess liver fat storage.18,19 Weight gain and fat excess in the liver contribute to the induction of hepatic insulin resistance. The liver continuously produces glucose (gluconeogenesis), regulated by insulin. In an insulin-resistant state, inhibition of hepatic glucose production is tempered, leading to slight hyperglycemia. These higher insulin levels enhance carbohydrate-induced liver lipogenesis, leading to more fat accumulation and inducing a vicious cycle. 20 Dietary carbohydrates, especially derived from fructose, further stimulate de novo lipogenesis, additionally contributing to liver steatosis and its metabolic consequences. 21
Although the association between overweight, alterations in fat distribution with visceral and ectopic fat accumulation, and metabolic complications are obvious in epidemiological studies, the exact pathophysiological pathways underlying these consequences are not fully elucidated. Several major abnormalities are currently being suggested as possible pathophysiological mechanisms.17–20,22 First, the lipid overflow theory proposes that the incapacity of subcutaneous adipose tissue to store fat leads to accumulation of fat in the visceral fat depots and of triglycerides within cells of non-adipose tissue [e.g. liver, muscle, heart, pancreatic beta (β) cell], defined as ectopic fat. Second, these ectopic fat deposits are associated with the formation of toxic metabolites that increase insulin resistance, and are potentially cytotoxic to the fat-loaded non-adipose tissues (lipotoxicity). 23 Third, obesity, metabolic syndrome and T2D are also characterized by a state of chronic inflammation, caused by infiltration of macrophages in the adipose tissue. This leads to the production of inflammatory molecules that have been associated with insulin resistance, pancreatic β-cell dysfunction, liver inflammation and CVD. Finally, fat cells secrete a variety of adipokines acting as an endocrine organ and exerting autocrine, paracrine, and endocrine actions. Large adipocytes, as found in visceral fat, secrete less anti-inflammatory and protective but more harmful adipocytokines, further disturbing overall metabolism. 17
In view of this booming epidemic, with a background of obesity and T2D, and the important medical consequences, early recognition and intervention of NAFLD/NASH seems appropriate. In this review, we will focus on the different actual and future therapeutic intervention options, taking into consideration the complex pathophysiology of this disease.
Diagnostic procedures to diagnose NAFLD
Liver biopsy still remains the gold standard for the diagnosis of NAFLD, since histology allows exact measurements of hepatocyte fat content, inflammation, fibrosis rate, and hepatocyte damage. 6 However, due to its invasive nature with a small, but significant risk of complications and high cost, it is not suitable for daily clinical practice and routine follow up during treatment. Therefore, reliable, accurate, and non- or minimally invasive biomarkers and non-invasive procedures are being explored. 24 Liver enzymes are unreliable or inaccurate predictors, since up to 80% of NAFLD patients have normal serum liver tests. 25
In a general population, indices combined with biological parameters, can offer an adequate diagnostic efficacy [area under the receiver-operator curve (AUROC): 0.7, 95% confidence interval (CI) 0.53–0.87] and relate positively to hepatocellular liver content, insulin sensitivity and β-cell function. 26 However, although these indices can accurately diagnose NAFLD, none of them can be used to quantitate liver fat and evaluate treatment effect. 26
Ultrasound-based techniques such as transient elastography (TE) and controlled attenuation parameter (CAP) are techniques enabling the simultaneous measurement of steatosis (by CAP) and liver stiffness (by TE), the latter reflecting fibrosis. The AUROC for CAP to diagnose mild (S1), moderate (S2), and severe steatosis (S3) is 0.96 [standard error (SE) 0.01], 0.82 (SE 0.03), and 0.70 (SE 0.02) respectively. 27
With advanced magnetic resonance imaging (MRI) of proton-density fat fraction (PDFF), hepatic fat content across the entire liver can be measured in an accurate and reproducible manner. MRI-PDFF has been validated against liver histology, and shows more sensitivity (optimal cut-off point 29.8%) in detecting changes in hepatic fat content and treatment response. 28 However, although non-invasive imaging techniques such as MRI-PDFF are evolving at increasing pace and provide early diagnosis and prognostic information on NAFLD, MRI is not largely available in common clinical practice. Furthermore, MRI-PDFF does not provide reliable evaluation of the presence of NASH, nor fibrosis.
Methodology
The authors executed a literature search using the PubMed, Embase and Web of Science databases. The authors used the following MeSH terms: ‘NonAlcoholic Steatohepatitis’[MeSH] OR ‘Non-alcoholic Fatty Liver Disease’[MeSH]) AND ‘Therapy’[Subheading]; ‘NonAlcoholic Steatohepatitis’[MeSH] OR ‘Non-alcoholic Fatty Liver Disease’[MeSH] AND ‘Pathophysiology’[Subheading] AND ‘Drug’; ‘Pharmacological therapy’[MeSH] AND ‘Non-alcoholic Fatty Liver Disease’[MeSH]. Articles were ranked, and abstracts were screened for relevance. Language was restricted to English. Authors provided additional articles based on relevance for this paper from a personal archive or search strategy. We searched for articles published between April 2019 and April 2021. References were screened for additional papers. Abstract books from relevant conferences were screened for unpublished trials. Clinical trials were further identified by consulting ClinicalTrials.gov.
Therapeutic approaches
Lifestyle measures by diet adaptation
There is a general consensus that 5–10% weight loss will reduce the majority of risk factors leading to glucose disturbance, hepatic fat accumulation, and CVD.28–31 It is also long known that a calorie-restricted diet rapidly decreases liver fat and can completely normalize hepatic insulin sensitivity in obese individuals with or without T2D.32,33 Macronutrient composition of the diet, high versus low carbohydrate, high versus low fat and high versus low protein, seems to be of less importance than total caloric deficit in determining fat loss. Among different macronutrient diets, no differences were noted in body composition, abdominal or hepatic fat loss. 34 A more recent trial found that both a low-carbohydrate, high-fat diet and an intermittent calorie-restrictive diet were more effective compared with general lifestyle advice from a hepatologist. 33
The current American Association for the Study of Liver Diseases (AASLD) and European Association for the Study of the Liver guidelines recommend weight loss achieved by hypocaloric diets in conjunction with increased physical activity for treatment of NAFLD/NASH.6,7 While lifestyle modification consisting of diet, exercise, and weight loss is advocated to treat patients with NAFLD/NASH, such modifications are difficult for subjects to sustain in long term. 35 Moreover, weight loss alone may not always suffice for the majority of patients, particularly those with advanced hepatic fibrosis, and as it is known from obesity management, challenges exist with implementing lifestyle recommendations.
Many studies exist that analyzed the effects of lifestyle and weight loss on NAFLD and the risk of subsequent NASH, including MRI spectroscopy studies. 36 Results in overweight patients with biopsy-proven NAFLD could confirm initial findings, also offering the opportunity to assess important changes in inflammation, lobular changes, and fibrosis. 37 Improvements in lifestyle can have immediate and sometimes significant effects on the liver in NAFLD patients, and may contribute to the high rate of ‘placebo response’ observed in NASH trials. 38 Lifestyle modification remains key to managing NASH even after medical interventions become available.
In a sub-analysis of the LOOK-AHEAD study after 12 months, participants allocated to an intensive lifestyle intervention lost significantly more weight than participants receiving therapeutic support and education only and showed a greater decline in hepatic steatosis. 36 In addition, 26% of those on conventional therapy without NAFLD at baseline, developed steatosis. 36 In the COUNTERPOINT (COUNTERacting Pancreatic inhibitiOn of INsulin secretion by Triglyceride) study, designed to study insulin secretion after short-term weight loss with a 600-kcal-restricted diet, and after withdrawal of metformin, patients with T2D achieved a weight loss of 15.3 kg over an 8-week period. Within 7 days, liver fat levels and hepatic insulin sensitivity returned to levels seen in weight-matched non-diabetic controls. After 8 weeks, pancreatic fat content decreased as well, and the first-phase insulin response normalized. 39
Paired liver biopsy studies revealed that 5% loss of body weight is associated with significant reductions in hepatic steatosis, over 7% weight loss is associated with reduction in hepatic inflammation, and over 10% weight loss, with reduction in fibrosis. 40
The Diabetes Remission Clinical Trial(DiRECT) included T2D patients with a body mass index (BMI) of 27–45 kg/m² and a disease duration for up to 6 years. The mean weight loss in the intervention group at 12 months was 10 kg. Diabetes remission, defined as glycated hemoglobin (HbA1c) < 6.5% without the use of antidiabetic drugs, was achieved in 46% of patients after 1 year, and in 36% of patients after 2 years.41,42 In a post hoc analysis of the whole-study population, of those participants who maintained at least 10 kg weight loss (24% of the patients in the intervention group), 64% achieved diabetes remission. Liver fat content fell to 3.3% after rapid weight loss and remained at 3.0% after 12 months of follow up, even though mean BMI remained at 31.5 kg/m² after weight loss. 42 Very-low-density lipoprotein 1 (VLDL1), triglyceride production, and intrapancreatic fat also decreased after weight loss. In those who relapsed after initial remission, a greater VLDL1–triglyceride content, re-accumulation of intrahepatic fat, and loss of the first-phase insulin response was observed. 43 The finding that liver fat accumulation remains low, despite the high residual BMI, confirms the concept that ectopic fat accumulation only occurs when an individual threshold of body fat burden is exceeded. Moreover, the timeframe of reversal and regain of intrahepatic fat and lowering and increasing of VLDL–triglyceride content, with pancreatic fat content and effects on first-phase insulin secretion, confirms that weight-related disturbed fat metabolism drives the development and reversal of T2D. 44
In a recent systematic review, including six studies comprising 317 patients, reduction in hepatic steatosis was statistically significant, concomitantly with weight loss, mainly in Mediterranean diets but also with one low-carbohydrate-, one intermittent-fasting- and one low-fat-based intervention. 45 Despite this promising outcome, it remains unclear what will be the optimal nutritional approach in this population. Indeed, little is known of popular weight-loss diets such as low-carbohydrate diets and the recent ketogenic diets. Other review articles have previously shown similar findings.46,47 Further randomized trials of longer duration, exclusively focusing on NAFLD individuals and on sustainability, are needed to evaluate the most appropriate nutritional plan.
Systematic reviews have also shown that exercise can reduce liver steatosis in patients with NAFLD, independent of weight loss and dietary intake.48,49 The main results suggest that, regardless of type, exercise can reduce the amount of intrahepatic fat. In a separate exercise study of short duration, Houghton et al. 50 reported the reduction of hepatic triglyceride content with limited effects on markers of inflammation and fibrosis. There is no consensus as to whether physical exercise may have antifibrotic effects, but Linden et al. 51 reported that liver fibrosis in a fatty rat model may benefit from exercise by altered stellate-cell activation.
Lifestyle intervention with mainly aerobic exercise was more recently studied with spectroscopy techniques to analyze the effects of exercise on hepatic fat content. A randomized controlled trial with over 100 participants with prediabetes from China studied whether exercise alone or in combination with a fiber-enriched diet could have an effect on fat content. The authors found that aerobic exercise was able to reduce the liver fat content by 24.4%, increasing to almost 48% when a fiber-enriched diet was combined, confirming the previous data that combination therapy may augment the outcome effects. 52
It is generally accepted that exercise at moderate level (three to four times a week, at 20–40 min per session, achieving 70% maximal oxygen uptake volume) is ideal for mobilizing fat from the liver. Although the effects of exercise are generally known to act protectively on overall metabolism, relatively little is known about the underlying molecular and metabolic mechanisms; reduction of inflammation through adipokine modulation could be one of the explanations. 48 An important limitation is that the majority of studies on exercise have solely analyzed intrahepatic fat, without assessing factors of inflammation and ballooning as part of NASH.
Since a link between gut-microbiome-derived metabolites and steatosis has been suggested, a proof-of-concept study indicated that probiotics could improve liver fat in NASH patients.12,53,54 More recent research is ongoing, studying probiotics as potential new strategies in the treatment of NAFLD. 55
Bariatric surgery
Bariatric surgery, as an alternative option for patients with severe obesity and T2D, is known to be an effective intervention for weight reduction and diabetes control in many individuals. 56 To date, it is the only procedure able to restore glucose tolerance. The effects of these procedures reach far beyond isolated reduction of body fat, explaining why it is referred to as metabolic surgery. 57
Long-term diabetes incidence reduction is reported, with slight differences between surgical procedures. An early reported meta-analysis by Buchwald et al. 58 already indicated diabetes improvement and resolution by 86% and 77%, respectively.
Metabolic surgery, by reducing subcutaneous and visceral adipose-tissue volumes, can reduce HbA1c numerically by over 3%, as was reported in several outcome studies among obese individuals with T2D.59–62 Remission, defined as return to normal glucose tolerance without medication, was found to be dependent on the duration of the disease in a subanalysis form the Swedish SOS study. 63 In addition, surgical procedures also improve both micro- and macrovascular complications, and the reduction of microvascular complications was found to occur when early intervention (<4 years) was achieved. 63 When compared with intensive medical therapy, the 5-year outcome in the STAMPEDE trial clearly showed superiority in diabetes control and a reduction in the need of glucose-lowering medication, even in the lower BMI subgroups. 61
Metabolic surgery also reduces ectopic fat, hepatic fat in particular, contributing to a marked improvement of NAFLD/NASH manifestations.42,64,65 The development of non-invasive imaging techniques (myocardial, hepatic and pancreatic triglyceride content) has made it possible to accurately quantify ectopic fat accumulation. In an MRI-based study, 89% of individuals with the diagnosis of NAFLD at baseline, were cleared from NAFLD after 6 months. 66 This effect is likely due to the reduction of FFAs circulating to the liver. 67 In several studies, after only a few weeks, early changes in liver fat fractions are described. 68 In another study, the reduction rates of intrahepatocellular lipid levels in obese women were up to 80% after 12 months following surgery. With biopsy-proven NASH, metabolic intervention led to the resolution of NASH in nearly 85% of patients.
Concomitant with fat in the liver, pancreatic fat reduction could be shown in a study with an MRI evaluation, as well.66,69 This reduction was associated with improved β-cell function and glucose control. 70 Improvement of ectopic fat in the heart, e.g. epicardial or intracardial steatosis, has been documented with several reports.65,71,72 Differences have indeed been demonstrated concerning epicardial fat reduction with less effect on myocardial fat reduction. Such anatomic effects were associated with significant changes in left ventricle mass and cardiac output. 65
A recent report revealed a rapid decrease of cardiac dietary fatty acid partitioning (cardiac fatty acid uptake and systemic spillover) after sleeve gastrectomy and biliopancreatic diversion. 73
Insights into mechanisms of ectopic fat changes added the potential enhancing effect on adenosine monophosphate (AMP)-activated protein kinase (AMPK) phosphorylation and chaperone-mediated autophagy translating into removal of lipid droplets, leading to an increase in lipolysis and amelioration of hepatic insulin resistance. 64
A few systematic reviews concluded that bariatric surgery is associated with significant reduction of a number of histological features of NAFLD, mainly after Roux-en-Y gastric bypass graft (RYBG) surgery, with the most pronounced effect on steatosis, ballooning, and inflammation, and the lowest beneficial effect on fibrosis. 74 A recent meta-analysis of 20 studies compared RYBG and sleeve gastrectomy based on four indices: alanine transaminase, aspartate transaminase, NAFLD activity score, and NAFLD fibrosis score. All criteria improved after either surgery, but superiority of any procedure could not be demonstrated. 75 However, a novel study, not included in the aforementioned analysis, compared 16 patients who underwent RYBG with eight patients with sleeve gastrectomy. This study found that RYBG provided better results according to NAFLD activity score compared with sleeve. 76 Studies on the effect of bariatric surgery on NAFLD stratified per procedure are summarized in Tables 1–3.
Studies on the effect of RYBG on NAFLD and its aspects.

↓, ameliorated; 0, no effect; NA, not applicable; NAFLD, non-alcoholic fatty liver disease; NASH, non-alcoholic steatohepatitis; RYBG, Roux-en-Y gastric bypass graft.
Studies on the effect of gastric banding on NAFLD and its aspects.

Unknown how many exactly had a follow-up biopsy, > 80% according to the authors.
↑, deteriorated; ↓, ameliorated; 0, no effect; NA, not applicable; NAFLD, non-alcoholic fatty liver disease; NASH, non-alcoholic steatohepatitis.
Studies on the effect of sleeve gastrectomy on NAFLD and its aspects.

↑, deteriorated; ↓, ameliorated; 0, no effect; NA, not applicable; NAFLD, non-alcoholic fatty liver disease; NASH, non-alcoholic steatohepatitis.
The safety of bariatric surgery for patients with NASH, and particularly NASH cirrhosis, is not well established. 105 Some reports mentioned an increased mortality in obese individuals with NASH undergoing bariatric surgery, suggesting a reduced survival time. 106 On the contrary, in a large propensity-matched analysis, bariatric surgery was associated with reduction of hepatocellular carcinoma. 107 Despite the improvements in NASH histology after bariatric surgery, the 2018 AASLD guidelines state that it is premature to consider foregut bariatric surgery as an established option to treat NASH. 7 In our opinion, this statement is certainly valid for patients with NASH cirrhosis. Larger-scale outcome studies will be necessary, also indicating optimal indication and timing of intervention, to identify the full long-term outcome.
Pharmacological intervention
Currently available pharmacological treatment options
Several drugs, not specifically licensed for the treatment of NASH but with a potential benefit based on their mode of action, have been tested in several trials. Actual and future pharmacological interventions may tackle different pathophysiological background mechanisms, such as insulin sensitivity, inflammation, weight lowering, antifibrotic and lipid-lowering mechanisms. To date, no drugs are officially licensed for the treatment of NAFLD. Multiple drugs, initially introduced in other fields of medicine, mainly T2D, are extensively being explored for their potential direct or indirect, through weight-loss effects on NAFLD. 108 From the hepatologist’s viewpoint, ursodeoxycholic acid (UDCA), a hepatoprotective bile acid used in cholestatic disease, did not show histological benefit in two long-term trials and is therefore not recommended. 108
Since the strong entanglement between NAFLD, insulin resistance and visceral adipose tissue (VAT), a great proportion of drugs originating from the metabolic arsenal are investigated for NAFLD due to their insulin-sensitizing activity and impact on glycemic control.
Metformin was the first molecule to be tested for NASH, since its beneficial effect on insulin resistance, but no histological effects were witnessed, and as a consequence, metformin is not indicated to treat NAFLD. 109 New recent data however in animal studies revealed that metformin could attenuate the onset of NAFLD in association with changes of intestinal microbiota. 110
Another antidiabetic class of drugs that has been thoroughly tested for NASH are the thiazolidinediones (TZDs) or glitazones. These drugs are agonists of peroxisome-proliferator-activated receptor (PPAR), gamma (γ) isotype, a nuclear receptor that exerts a pivotal role in glucose and lipid homeostasis, but also influences inflammation and fibrogenesis.111,112 TZDs exert their hepatoprotective effect not only by increasing insulin sensitivity and subcutaneous fat accumulation, but also by enhancing adiponectin production and decreasing pro-inflammatory cytokine production in adipose tissue and macrophages. Glitazones have been approved for the treatment of diabetes, but were also shown to be effective in improving histological lesions of NASH in several trials.16,113–115 The improvement in liver histology is probably driven by both direct intra- and extrahepatic effects that subsequently benefit the liver, most importantly, an improvement in adipose-tissue dysfunction. 116 Safety concerns have been raised for some molecules. 116
Pioglitazone has been shown to reduce hepatic steatosis, inflammation, and ballooning, although effects on fibrosis were less consistent. 114 There is some weight gain with PPAR-γ, but mostly due to a translocation from fat from visceral to subcutaneous adipose tissue, which is less harmful. 116 Pioglitazone clearly improved cardiovascular outcomes in diabetic patients and has a more favorable safety profile, but is nevertheless not frequently used. 117 There are some concerns regarding the possibility of eliciting heart failure in predisposed individuals, although, as mentioned, an overall significant cardiovascular benefit has been recently demonstrated.
Novel PPAR agonists, acting on one or more of the PPAR isoforms, including pan-PPAR agonists, are being studied for their potential use in the treatment of NAFLD.118,119
Fibroblast growth factor-21 (FGF21) has insulin-mimetic properties that beneficially affects carbohydrate and lipid metabolism. Although mainly secreted by the liver, FGF21 is also secreted by adipocytes and acts in an autocrine fashion in adipocytes. FGF21 is required to mediate the effects of the PPAR-γ agonists. Besides FGF21, adiponectin also seems to play a role in the mechanistic benefit of pioglitazone in NASH. 120
More recent molecules with effects on glycemic control and weight loss also show the potential to improve NASH.
Dipeptidyl peptidase-4 (DPP4) inhibitors are molecules that inhibit the rapid breakdown of incretins like glucagon-like peptide-1 (GLP-1). They are used for T2D and are tested for NASH as well, but to date, no large phase IIb trials with histological endpoints have been reported. 121 DPP4 inhibitors only have short-acting effects, acting indirectly on GLP-1. Therefore, direct GLP-1 agonists, or incretin mimetics, are introduced due to their longer-lasting effects. They have been approved for the treatment of T2D. Exenatide, an exendin derived GLP-1 receptor agonist was initially studied in NAFLD patients. 122 Liraglutide in a dose up to 3 mg per day is also approved for the treatment of obesity in the absence of diabetes, since they reduce weight by more than 5% when applied on top of dietary measures and can normalize glucose intolerance.123,124 Despite significant weight losses with GLP-1-receptor-agonist therapy, the effects of incretin mimetics on ectopic fat are conflicting. Some studies show dose-dependent decreases in visceral and ectopic adiposity and liver fat reduction.124,125 In a south Asian diabetic population, 26-week treatment with liraglutide decreased VAT volume, and the reduction in VAT was associated with an improved HbA1c. 124 On the other hand, in other studies, treatment with liraglutide reduced subcutaneous adipose-tissue volume without effects on VAT volume or did not reduce visceral, epicardial, paracardial or pericardial fat mass, and had no effect on hepatic or myocardial steatosis compared with placebo. 125
Liraglutide improves liver enzymes and has been reported in several trials to beneficially affect liver histology in subjects with NAFLD. The effect may be partially indirect due to the accompanied weight loss, although direct hormonal effects on the liver might coexist. Multiple phase II and phase III studies with GLP-1 agonists are currently ongoing, but to date, they are not (yet) approved to treat NASH, except in individuals with coexisting T2D and/or obesity. Liraglutide (at a dose of 1.8 mg four times daily) has been reported to beneficially affect liver histology in a small randomized controlled trial (RCT) with 23 patients in each arm. 126 It induced NASH resolution without worsening of fibrosis significantly more than placebo, and there was a trend for less fibrosis progression. It is likely the induced weight loss is one of the main drivers of the histological benefit, although other hormonal effects might also contribute to the overall effect on liver histology. Until further data become available (several other GLP-1 analogs are currently being studied, including semaglutide), its use should be restricted to the approved indications. However, the addition of NASH to the list of comorbidities of obesity that justify pharmacological treatment of obesity is increasingly proposed. 116 Most studies that explored the effects of GLP-1 receptor agonists on liver histology in patients with NAFLD/NASH were performed in patients with T2D. In this population, histological evaluation of the treatment with the GLP-1-receptor agonist showed that hepatic steatosis, lobular inflammation, hepatocyte ballooning, and fibrosis were reduced.127,128
With the newer molecule semaglutide, promising effects have been reported on liver enzymes and inflammation parameters. 129 Recently, in a large phase IIb study, semaglutide at a daily dose of 0.4 mg significantly induced resolution of NASH without worsening of fibrosis after 72 weeks of treatment, but it did not induce an improvement of fibrosis stage > 1. 130 Studies with the once-weekly approach have shown promising effects on body weight, and an outcome trial is ongoing.
Sodium–glucose cotransporter 2 (SGLT-2) inhibitors or gliflozins are another new class of antidiabetic drugs that inhibit glucose reabsorption from the urinary ultrafiltrate leading to increased glucosuria and subsequent metabolic control. 131 They are also associated with weight loss and cardiorenal protection. 132 Outside diabetes, SGLT-2 inhibitors are increasingly investigated for their beneficial effects in patients with heart failure, even without diabetes, as shown in the EMPEROR-Reduced and DAPA-HF trials.133,134 Multiple phase II and phase III studies are currently ongoing in NAFLD patients since early preclinical data suggested that SGLT-2 inhibitors reduce liver fat content in NAFLD patients with T2D. 135 A trial of 50 patients with T2D randomized to empagliflozin versus placebo on top of their treatment showed significant reduction in liver fat content and improvement of liver enzymes. 131 Using MRI analyses in subjects with T2D, there was a significant > 30% reduction from baseline in liver fat (p = 0.007) and > 10% reduction in adipose-tissue volumes (p < 0.01) with dapagliflozin plus saxagliptin/metformin at week 52 versus glimepiride plus metformin. 136 A Swedish study with 84 participants with T2D showed a significant reduction in hepatic fat content in those taking a combination of dapagliflozin and omega-3 carboxylic acids compared with placebo, but not in those taking dapagliflozin in monotherapy. 137 A recent meta-analysis of RCTs analyzing liver parameters and steatosis confirmed these preliminary findings in patients with T2D. 138 So far, there are no published RCTs available evaluating histological endpoints, so SGLT-2s are not yet established in NASH therapy. 135 One prospective trial in five NAFLD patients without controls using paired biopsies showed histopathological improvement. 139
Combinations of SGLT-2 inhibitors with GLP-1 receptor agonists (exenatide and dapagliflozin) have been shown to improve markers of steatosis and fibrosis in T2D patients, but effects on histological endpoints are still unknown.140,141
Vitamin E improved liver histology in non-diabetic NASH patients without cirrhosis, so its use can be recommended in this specific patient category, dosed at 800 IU/day according to the PIVENS trial data.116,142 Indeed, vitamin E was ineffective in treating NASH in patients with T2D, while the combination of vitamin E and pioglitazone was significantly better than placebo. Although a recent large meta-analysis did not confirm earlier safety issues, the risk of prostate cancer in males is an issue of concern. 143
Drugs in development
There is currently no pharmacological treatment that has NASH in its label. There is, however, a large pipeline of drugs being tested, some of them already in phase III.
Indeed, numerous drugs are currently being tested for the treatment of NASH. The development is complex, as different endpoints can be defined and a variety of targets proposed. Efficacy must be present on the following clinical endpoints: development of cirrhosis, cirrhosis-related complications and all-cause mortality.116,144 Due to the long duration of these phase III–IV trials, drugs can be applied for conditional approval based on histological benefit, which is considered a reasonable surrogate for later clinical outcomes. Histological benefit is defined as resolution of NASH without worsening of fibrosis, or improvement in at least one stage of fibrosis without worsening of NASH. 144
An important consequence of these considerations is the necessity of histological proof of efficacy and serial biopsies. In the following paragraphs we will briefly discuss the most important drug classes currently under investigation. An extensive review of all potential targets is beyond the scope of this review, so we will focus on the major classes. The most important phase II and III trials are summarized in Table 4.
New therapeutic drugs targeting NASH in phase II and III trials.
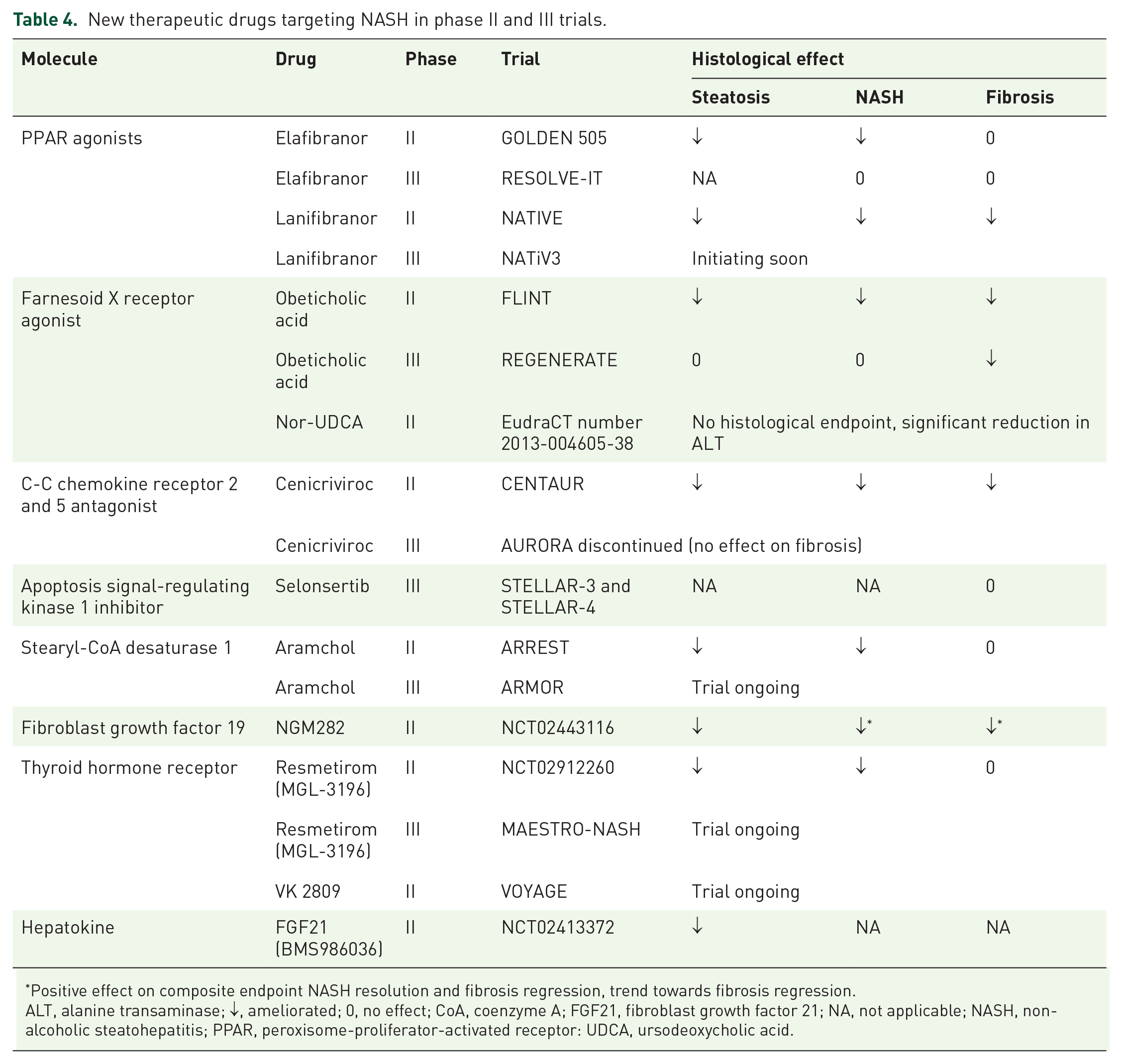
Positive effect on composite endpoint NASH resolution and fibrosis regression, trend towards fibrosis regression.
ALT, alanine transaminase; ↓, ameliorated; 0, no effect; CoA, coenzyme A; FGF21, fibroblast growth factor 21; NA, not applicable; NASH, non-alcoholic steatohepatitis; PPAR, peroxisome-proliferator-activated receptor: UDCA, ursodeoxycholic acid.
PPAR agonists
Three isoforms of PPARS exist.145,146 PPAR alpha (α) is mainly expressed in hepatocytes but also in many other cell types, including muscle cells. PPARα agonists like fibrates have not been extensively studied, but smaller studies did not show a histological benefit. 108 We demonstrated previously that PPARα expression is inversely correlated to the severity of NASH and that NASH improvement is associated with increased PPARα expression, giving rationale to a PPARα-targeted treatment despite the negative data with fibrates. 145 Elafibranor is a hepatotropic dual PPARα-δ agonist, hence targeting not only PPARα but also PPARδ that is expressed in stellate cells and several other cell types. In a large phase IIb trial including 276 patients, elafibranor was able to induce resolution of NASH without worsening of fibrosis in significantly more patients compared with placebo if baseline NASH was sufficiently severe. 118 The drug had a very good safety profile and also improved serum lipids and HbA1c, reducing the calculated overall risk of CVD. Interim analysis of the elafibranor phase III trial failed to show a benefit of the drug on liver histology and further development was halted. Several other PPAR drugs are in development, including lanifibranor (a pan-PPAR agonist potentially combining positive effects of the glitazones with PPARα-δ agonism,73,93,94 saroglitazar (a PPARα-γ dual agonist) and seladelpar (a PPARδ agonist).119,147,148
Lanifibranor, as studied in the phase II NATIVE trial [ClinicalTrials.gov identifier: NCT03008070], showed significant results on both NASH resolution without worsening of fibrosis, and fibrosis improvement without worsening of NASH, and is thus the first compound to report efficacy in both these histological endpoints. The drug showed significant results on the composite endpoint of NASH resolution and fibrosis improvement, improved lipid profile and HbA1c. Based on these efficacy data and accompanying safety data, the drug received the breakthrough therapy designation by the US Food and Drug Administration and is currently moving into phase III (NATiV3). Saroglitazar is also further studied as individual compound in the EVIDENCES IV trial [ClinicalTrials.gov identifier: NCT03061721].
Farnesoid X receptor (FXR) agonist
FXR is a nuclear receptor in enterocytes and hepatocytes that plays in important role in bile-acid metabolism but also impacts several metabolic, inflammatory, and fibrogenic pathways. FXR is present in the liver and the intestine, with some differences in effect, depending on the site. 149
Bile acids are the natural ligands of FXR. UDCA has no FXR agonistic effect, but the bile acid obeticholic acid (OCA) is a potent FXR agonist and resulted in a significant response compared with placebo, as defined by at least a 2-points reduction in NAFLD activity score (NAS) in the FXR Ligant obetIcholic acid in NASH Treatment (FLINT) trial in 110 treated versus 109 placebo patients with paired biopsies. 150 There was also a beneficial effect on fibrosis and a trend for resolution of NASH, both secondary endpoints. Because of the significant benefit in terms of the primary endpoint, the study was stopped prematurely and the drug went on to phase III. Pruritus is a known side effect of OCA, which is currently already licensed for the treatment of primary biliary cholangitis. Furthermore, OCA decreased high-density-lipoprotein cholesterol levels and did not improve glycemic control. OCA is currently tested in a large phase III trial (REGENERATE). The interim analysis showed a significant effect of OCA on at least one-stage improvement of liver fibrosis without worsening of NASH after 18 months of treatment. The NASH resolution endpoint was not met. Based on these interim results, the drug was filed for conditional approval but has not yet been granted by the regulatory authorities. 151
Several other bile-acid FXR agonists are currently investigated, with nor-UDCA (phase II) being the most advanced. Furthermore, several non-bile-acid FXR agonists are being developed. Since they have different effects on intestinal or hepatic FXR and exhibit variable pharmacokinetic and pharmacodynamic properties, their net effects on the liver and metabolism as well as their safety profile are curiously awaited.
Cenicriviroc
Cenicriviroc is a C-C chemokine receptor (CCR)2 and -5 dual antagonist. CCR2 and CCR5 play an important role in macrophage recruitment and differentiation and have been implicated in NASH pathogenesis. A large 2-year phase II trial studying the efficacy and safety of cenicriviroc for the treatment of NASH in adult subjects with liver fibrosis (CENTAUR) including 289 patients reported after 1 year a significant decrease in systemic inflammation, but this did not clearly ameliorate NASH. 152 By contrast, there was a significant benefit of cenicriviroc over placebo with regards regression of fibrosis. The data after 24 months corroborates antifibrotic findings after 1 year of treatment in the group that started on placebo, but switched to active drug after 1 year. The majority of participants who achieved fibrosis response after 1 year could maintain it, with greater efficacy in patients with advanced fibrosis at baseline. 153 Based on these data, the drug entered phase III with reduction in fibrosis as the primary endpoint. Interim analysis showed that cenicriviroc failed to achieve this primary endpoint, which resulted in terminating the study.
Apoptosis signal-regulating kinase 1 inhibitor (selonsertib)
Selonsertib is an apoptosis signal-regulating kinase 1 (ASK1) inhibitor involved in response to various stresses that was tested in a 6-month trial in combination with or without simtuzumab in an antifibrotic strategy. When the other simtuzumab trials produced negative results, simtuzumab was considered the placebo. In this new setting, selonsertib was superior to placebo in terms of fibrosis regression, without an effect on steatohepatitis or on the metabolic features. It was subsequently tested in two phase III trials, one for cirrhotic patients and one in advanced fibrosis patients (STELLAR trials). Harrison et al. 154 recently reported the results of these two STELLAR studies, indicating that selonsertib monotherapy had no antifibrotic effect in patients with bridging fibrosis or compensated cirrhosis due to NASH.
Stearyl-CoA desaturase 1 (aramchol)
Besides the molecules currently in phase III, several other molecules are under investigation. Aramchol is a compound of which the phase II ARREST study data in 247 patients showed higher rates of NASH resolution without worsening of fibrosis, and fibrosis amelioration without worsening of NASH, leading to the initiation of the phase III ARMOR trial [ClinicalTrials.gov identifier: NCT04104321]. Aramchol is a bile-acid–fatty-acid conjugate acting as a stearyl-coenzyme A desaturase 1 (the rate-limiting enzyme in the synthesis of unconjugated fatty acids) inhibitor. Other attempts, like ezetimibe, have failed in that regard. Different approaches, like fatty-acid synthase inhibition and other targets of cholesterol and triglyceride metabolism are tested in earlier phases of clinical development.
Fibroblast growth factor 19 (FGF19)
FGF19 is released by the intestinal cells upon FXR stimulation and will, after reaching the liver via the portal vein, exerts its actions on bile-acid metabolism via the FGF-receptor 4/β-klotho complex and also impacts lipid and glucose metabolism. Via the interleukin (IL)-6/STAT3 pathway, however, it also drives tumorigenesis. NGM282, a recently engineered FGF19 analog that lacks effect on the STAT3 pathway and hence most likely lacks tumorigenic effect of FGF19, demonstrated a significant reduction in liver fat content in a phase II study including 82 NASH patients. Data of single-arm studies have also been released recently. This injectable drug, named Aldafermin, appeared to have an acceptable safety profile and was further investigated in a phase II study. This study reported a significant reduction in absolute liver fat content compared with placebo after 24 >weeks, alongside a trend toward fibrosis improvement 155
Thyroid hormone receptors
Thyroid hormones increase energy expenditure and have catabolic properties, acting via the thyroid hormone receptor (THR), a nuclear receptor with different isoforms. An intrahepatic hypothyroidism has been shown to be present in NASH and potentially contributes to its pathophysiology. 156 This intrahepatic hypothyroidism is potentially attributable to alterations in hepatic deiodinase expression because of repair-related Hedgehog activation. 156 The THR agonist resmetirom (MGL-3196) has a selectivity for the THR-β1 receptor mainly expressed in the liver and the kidney and therefore most likely lacks some potentially important side effects; among others, on bone metabolism. A study of 348 patients found that resmetirom treatment resulted in a significant reduction of liver fat content after 12 weeks and 36 weeks of treatment compared with placebo. 157 In addition, liver enzymes were reduced, and biomarkers (adiponectin, pro-C3 and cytokeratin-18) associated with inflammation and fibrosis were statistically significantly changed by resmetirom. 158 In secondary analyses, liver biopsy data after 36 weeks showed reduction in NAS and NASH resolution, together with significant improvement of a number of atherogenic lipids and lipoproteins. Based on the provided data, this compound also entered a phase III clinical trial (MAESTRO-NASH). VK2809, a compound with similar effects, is also currently in phase II to evaluate its effects on NASH [ClinicalTrials.gov identifier: NCT04173065].
Hepatokines
FGF21 is a so-called hepatokine, a peptide hormone predominantly produced by the liver (but also by multiple other organs, but circulating levels are mainly determined by hepatic production), regulating sugar intake, glucose homeostasis, and energy expenditure. Interestingly, in view of the PPAR drugs in the pipeline, its expression in the liver is regulated by PPARα. Animal data suggest enhanced NASH and associated metabolic derangements upon FGF21 deficiency and improvement upon administration. 114 Human data are conflicting, with increased FGF21 levels in NASH patients suggesting FGF21 resistance. 69 Recent data demonstrated a beneficial effect on liver fat content of BMS986036, an injectable pegylated analog of human FGF21, along with a reduction in biomarkers of liver injury and fibrosis in a placebo-controlled phase IIa trial. 159
As with cenicriviroc, some drugs that mainly target inflammatory mechanisms are even so tested. BI 1467335, an oral small-molecule inhibitor of amine-oxidase copper-containing 3 (AOC3), also called vascular adhesion protein 1 (VAP-1), has been tested in phase II; results are pending. AOC3 plays an important role in the recruitment of various inflammatory cell types to the site of inflammation and was shown to play a role in NASH pathogenesis in preclinical models. Its soluble variant showed a correlation with NAFLD severity. Despite the study meeting the trial targets, including clinically relevant changes in NASH biomarkers, Boehringer Ingelheim decided not to further develop the drug. Interestingly, VAP-1 has been implicated in atherosclerosis and cardiovascular prognosis, reinforcing the link between NAFLD and CVD and the rationale for AOC3 antagonists in NASH. 160
Inhibition of caspases to interfere with inflammatory and apoptotic processes is another of the many pathways that can be targeted in the attempt to stop/regress the NASH–fibrosis process.
While we are still waiting for more phase III proof of efficacy of single drugs, several combinations of drugs are already being tested. As outlined before, disease pathophysiology is heterogeneous and complex, offering a rationale for combining drugs with different modes of action that can have additive or even synergistic effects. Beside testing combinations of individual drugs (e.g. tropifexor and cenicriviroc), several molecules that combine different structures are engineered and tested: dual peptide molecules that combine a GLP-1 receptor agonist with a glucagon receptor agonist or GIP (tirzepatide) are actually in clinical development. Tirzepatide has shown very promising strong metabolic effects (both glucose and weight lowering) in individuals with T2D and is now studied in phase IIb and III (SURPASS CVOT) and in NASH individuals (SYNERGY-NASH). 161 The triple GLP-1/GIP/glucagon receptor agonist HM15211 is currently under development, but the first trials in non-diabetic obese subjects with NAFLD showed significantly decreases in liver fat content and body weight. 162 Another potential future bimolecule approach is the semaglutide/cilofexor (FXR agonist) combination.
Conclusion
NAFLD is a worldwide epidemic associated with metabolic and endocrine pathologies. The most evidenced approach is lifestyle intervention, which lacks long-term efficacy. Therefore, prevention merits more attention, since the pharmaceutical options currently available are limited. Although multiple pharmacotherapies are in development, response rates appear modest, mainly for fibrosis treatment. From different phase II trials, antidiabetic drugs seem promising, both in mono- or bitherapy. Derivatives of PPAR molecules may have an interesting future, as well. As a general recommendation, we propose early screening and recognition of the problem, to deliver optimal treatment once an approved pharmacological armamentarium becomes available.
Footnotes
Author contribution(s)
Conflict of interest statement
Luc Van Gaal declares to be member of the Advisory Board and/or Speakers Bureau of AstraZeneca, Boehringer Ingelheim, Eli Lilly, MSD, and Novo Nordisk.
Jonathan Mertens reports no conflicts of interest.
Sven Francque has a senior clinical research mandate from the Fund for Scientific Research (FWO) Flanders (1802154N) and has acted as adviser and/or lecturer for Roche, Gilead, Abbvie, Bayer, BMS, MSD, Janssen, Actelion, Astellas, Genfit, Inventiva, Intercept, Genentech, Galmed, Promethera, Coherus and NGM Bio.
Christophe De Block reports consulting fees and honoraria for speaking for Abbott, AstraZeneca, Boehringer Ingelheim, Menarini Diagnostics, Eli Lilly, Medtronic, Novo Nordisk and Roche.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
