Abstract
This is a visual representation of the abstract.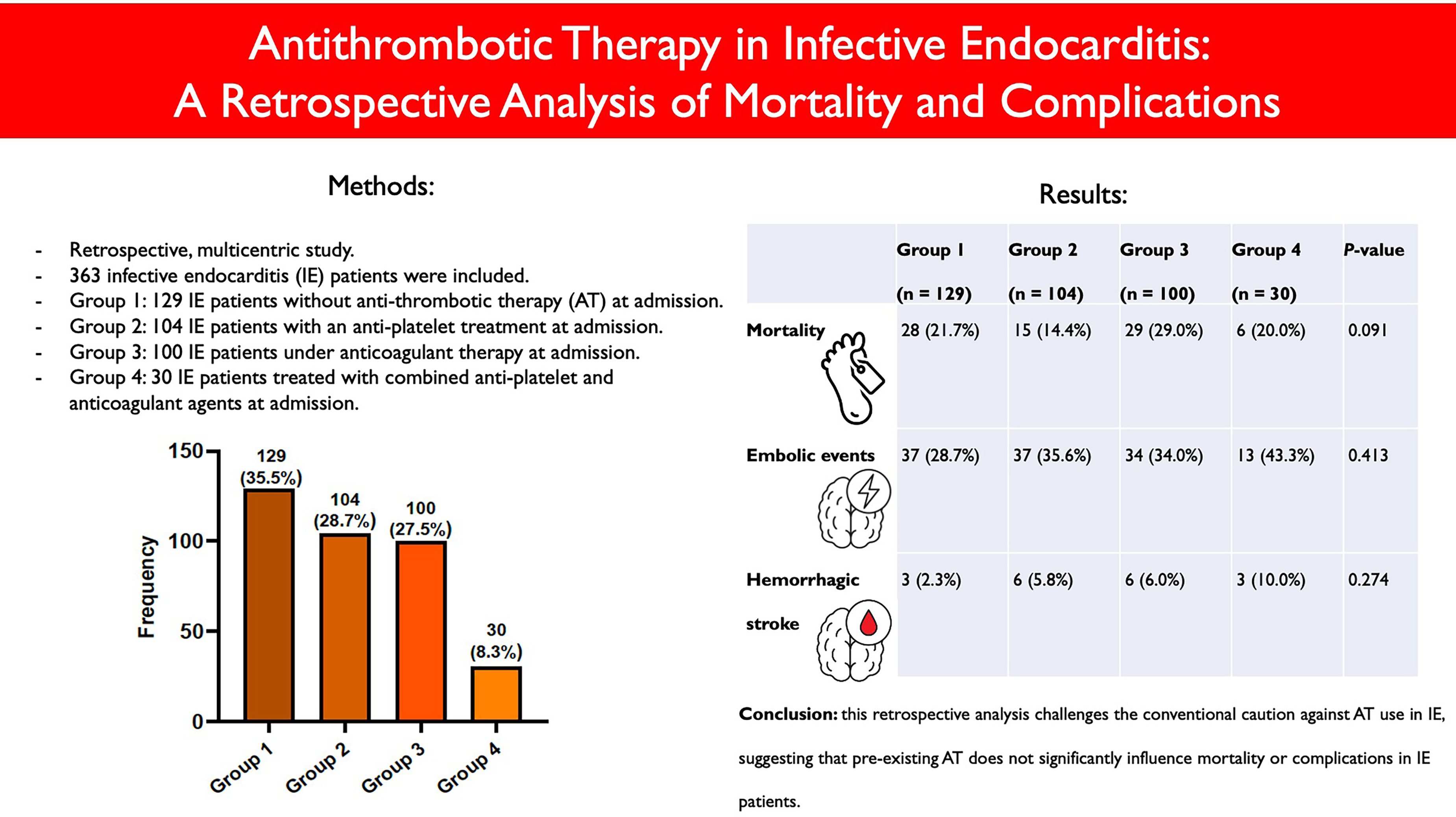
Key Points
This analysis found no significant differences in in-hospital mortality among patients with infective endocarditis (IE) with or without pre-existing antithrombotic therapy (AT).
There were no significant differences in thromboembolism or hemorrhagic stroke between patients with IE with or without pre-existing AT.
This study challenges current guidelines advising against AT use in IE management because of bleeding risks.
Introduction
Infective endocarditis (IE) and its complications remain a diagnostic and therapeutic challenge. Thromboembolism (TE) arises in 20% to 50% of patients with IE, significantly contributing to morbidity and mortality rates.1,2 Antimicrobial therapy is the only medical treatment known to reduce the risk of TE linked with IE.1,3 While the pathogenesis of IE includes the formation of vegetations with platelet-fibrin clots, antithrombotic therapy (AT), that is, antiplatelet and anticoagulant treatment, has not consistently shown to prevent TE.3–6 Moreover, because of the increased risk of cerebral bleeding, current European and American IE guidelines advise against using AT in IE management.1,7 However, many patients with IE already receive AT at admission due to pre-existing cardiovascular conditions (eg, atrial fibrillation, prosthetic heart valves, peripheral arterial disease, or after percutaneous coronary intervention) and the numbers of such individuals are rising. 7 In such cases, current guidelines generally recommend to carefully continue AT in the absence of major bleedings, weighing the potential risks and benefits.1,7 However, the level of evidence underlying these recommendations is low and data about complications and mortality due to AT in IE is limited. Moreover, there are currently no studies that directly compare characteristics of patients with IE under antiplatelet, anticoagulant, and combination therapy at admission. Therefore, this retrospective, observational, multicentric study aims to provide contemporary insights into the in-hospital mortality and complications of patients with IE under various regimens of pre-existing AT.
Methods
Study Design and Data Collection
Patient data from 2 tertiary care hospitals in Belgium (Algemeen Ziekenhuis Maria Middelares Gent and Universitair Ziekenhuis Brussel), admitted for IE between 2009 and 2022, were retrospectively analyzed. Inclusion criteria were adult patients of more than 18 years of age treated for definite or possible IE, based on the European Society of Cardiology (ESC) 2015 IE guidelines, as detailed in Supplemental Table 1. 8 Exclusion criteria were patients with IE of less than 18 years of age, and with nonbacterial, blood-culture negative thrombotic endocarditis, for example, due to antiphospholipid syndrome, auto-immune disease (like systemic lupus erythematosus), or terminal cancer. Native valve endocarditis (NVE) was defined as an infection of the heart's natural (nonprosthetic) valves. Prosthetic or repaired valve infective endocarditis (PRVE) was defined as IE involving prosthetic heart valves or previously repaired native valves. Intracardiac device-related infective endocarditis (CDRIE) was defined as IE of implanted cardiac devices (pacemakers or defibrillators), often involving the leads. Pre-existing AT at the time of admission was recorded for all patients. AT was defined as treatment with anticoagulants (vitamin K antagonist (VKA), intravenous unfractionated heparin (UFH), subcutaneous low-molecular-weight heparin (LMWH) or direct oral anticoagulants (DOAC)), or with antiplatelet agents (acetylsalicylic acid (ASA), thienopyridines (eg, clopidogrel) or triazolopyrimidine (ticagrelor)). Other collected patient data comprised: demographics (age and sex) and medical history (cardiovascular and comorbidities), clinical characteristics at admission (signs and symptoms), microbiology (blood cultures and serology), imaging (echocardiography, computed tomography (CT), 18F-FDG positron emission tomography/computed tomography (18F-FDG PET/CT), and magnetic resonance imaging (MRI)), complications during hospitalization (TE, hemorrhagic events, and shock), cardiac surgery, and in-hospital mortality. Cerebral, splenic, pulmonary, peripheral, renal, coronary, and hepatic TE was considered. These events included both symptomatic and asymptomatic cases identified through imaging. The study was approved by the local Ethical Committee of both hospitals and was carried out in accordance with the ethical principles for medical research involving human subjects established by Helsinki's Declaration, protecting the privacy of all participants, as well as the confidentiality of their personal information.
Data Management and Statistical Analysis
Data were retrospectively collected at the participating sites and entered in an online electronic case report form (CRF). Data quality was monitored by the principal investigator who had full access to all study data and takes full responsibility for its integrity and the data analysis. Missing data were not replaced. Continuous variables are expressed as mean ± standard deviation. Comparisons among groups were performed using analysis of variance (ANOVA) for parametric data and Kruskal-Wallis H test for nonparametric data. Categorical variables are expressed as frequency and percentages. Among-group 2 × 2 comparisons were made using Pearson's Chi-squared χ2 test or Fisher's exact test if any expected cell count was <5. Univariable logistic regression and survival analyses were conducted for both continuous and categorical variables. Variables with a P-value < .100 in the univariable analysis were subsequently included in the multivariable logistic regression and survival analyses. A forward stepwise Cox proportional hazards regression was performed to assess variables associated with mortality. Statistical significance was considered for a P-value < .05. When required, the Bonferroni correction was applied to control for the increased risk of type I errors due to multiple comparisons. Specifically, the significance level was adjusted by dividing the original α (0.05) by the number of comparisons conducted (n) and a corrected P-value threshold of α/n was used to determine statistical significance. Analyses were conducted using IBM SPSS Statistics software version 29.0.1.0 (171) (Chicago, IL, USA). For reporting, the STROBE cross-sectional reporting guidelines were applied. 9
Results
Overall, 363 patients with IE were retrospectively included. 129 control patients with IE and without AT (group 1) were compared to 104 patients with IE with an antiplatelet treatment (group 2), 100 patients with IE under anticoagulant therapy (group 3), and 30 patients with IE treated with combined antiplatelet and anticoagulant agents at admission (group 4). Subclasses of AT per group are shown in Supplemental Figure 1. In group 2, most patients with IE were treated with ASA (n = 95; 91.3%), while in group 3, VKA were most often used (n = 48; 48.0%). Finally in group 4, ASA with VKA was the most frequent combination (n = 15; 50.0%). The 4 groups and the main findings were evenly distributed between the 2 participating centers.
IE Characteristics, Patient Demographics, and Comorbidities
IE characteristics are shown in Table 1, while a condensed version of patient demographics and comorbidities is shown in Table 2. A full version of Table 2 can be found in Supplemental Table 2. The control IE group 1 was significantly younger than the AT IE groups 2 to 4 (P < .001).
Infective Endocarditis Characteristics per Group.
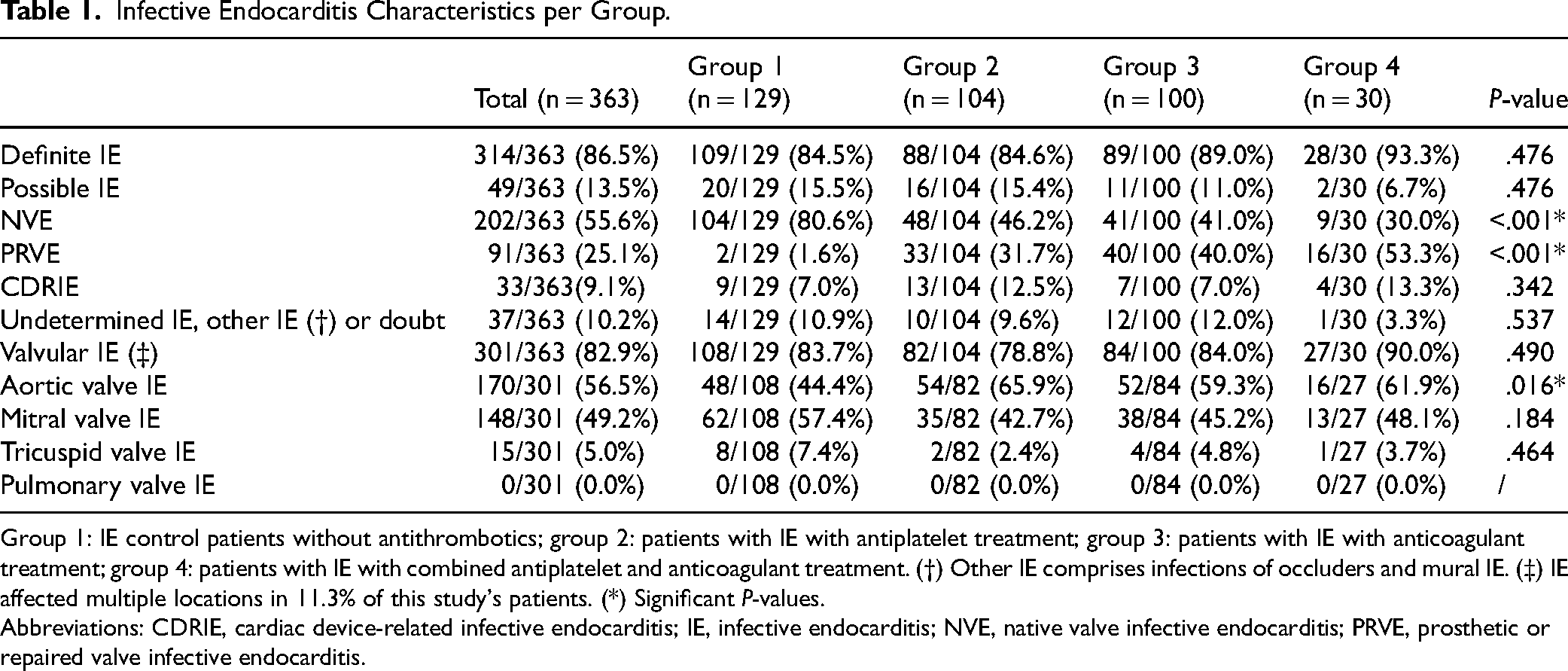
Group 1: IE control patients without antithrombotics; group 2: patients with IE with antiplatelet treatment; group 3: patients with IE with anticoagulant treatment; group 4: patients with IE with combined antiplatelet and anticoagulant treatment. (†) Other IE comprises infections of occluders and mural IE. (‡) IE affected multiple locations in 11.3% of this study's patients. (*) Significant P-values.
Abbreviations: CDRIE, cardiac device-related infective endocarditis; IE, infective endocarditis; NVE, native valve infective endocarditis; PRVE, prosthetic or repaired valve infective endocarditis.
Demographics and Comorbidities of Infective Endocarditis Patients per Group (Condensed Version).
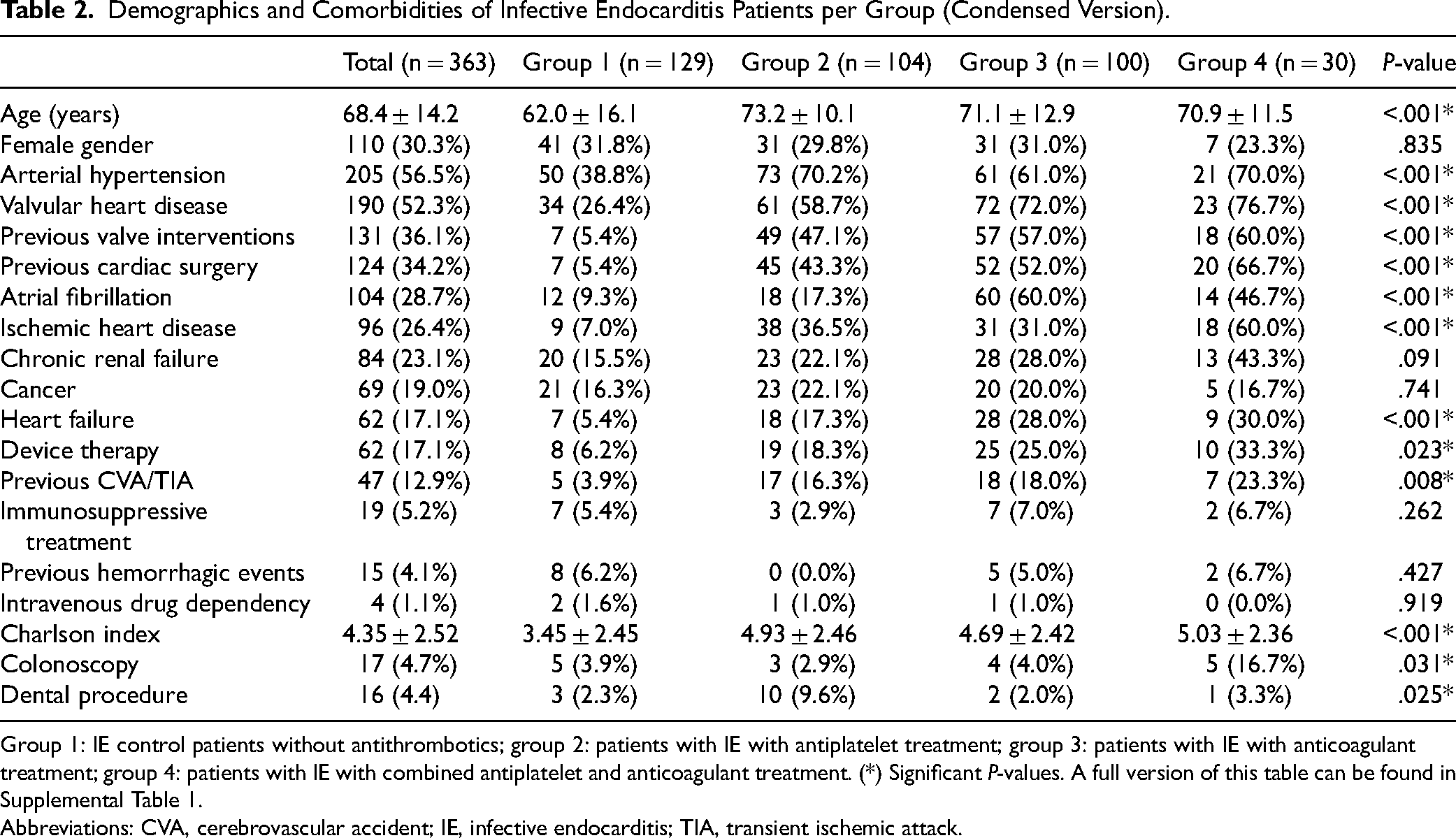
Group 1: IE control patients without antithrombotics; group 2: patients with IE with antiplatelet treatment; group 3: patients with IE with anticoagulant treatment; group 4: patients with IE with combined antiplatelet and anticoagulant treatment. (*) Significant P-values. A full version of this table can be found in Supplemental Table 1.
Abbreviations: CVA, cerebrovascular accident; IE, infective endocarditis; TIA, transient ischemic attack.
Clinical Features at Admission and Microbiology
Clinical features at admission are detailed in Supplemental Table 3. Fever (54.5%), a cardiac murmur (38.8%), and congestive heart failure (CHF) (14.0%) were common symptoms at admission. Notably, CHF at admission was significantly more prevalent in the older patients with IE treated with AT (P = .032).
Microbiological results can be found in Table 3. Blood cultures were positive in 89.3% of patients with IE. The most frequently identified microorganisms were Staphylococcus aureus (22.9%), viridans streptococci (19.8%), coagulase negative staphylococci (CoNS) (17.4%), and enterococci (10.7%). Gram-negative HACEK infections (2.2%) and Coxiella burnetii infections (0.3%) were rare. There were significantly more viridans streptococci infections in control group 1 (27.1%, P = .012). Variables significantly associated with all microorganisms by logistic multiple regression analysis, adjusted for cancer, were age (odds ratio (OR) = 0.97; 95% confidence interval (CI) [0.95-0.99]; P = .002) and acute kidney injury (AKI) (OR = 2.02; 95% CI [1.15-3.54]; P = .014).
Microbiology of Infective Endocarditis Patients per Group.
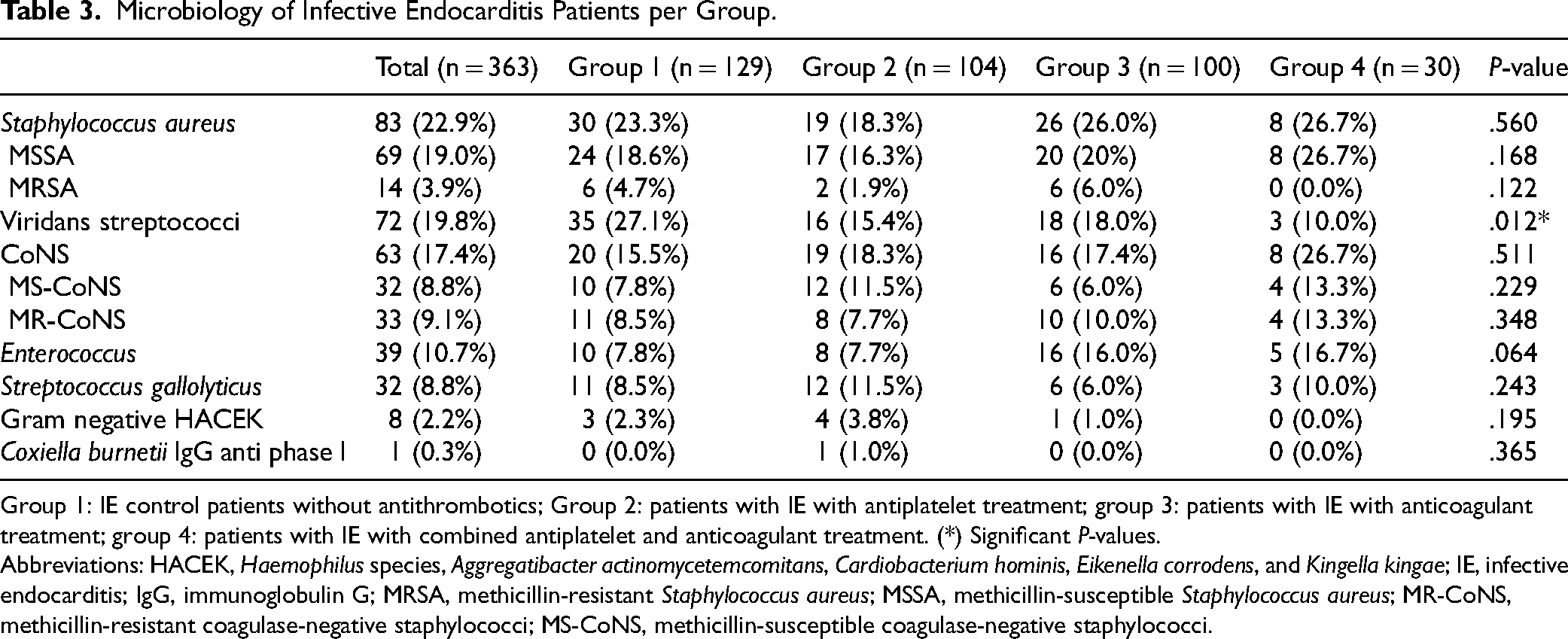
Group 1: IE control patients without antithrombotics; Group 2: patients with IE with antiplatelet treatment; group 3: patients with IE with anticoagulant treatment; group 4: patients with IE with combined antiplatelet and anticoagulant treatment. (*) Significant P-values.
Abbreviations: HACEK, Haemophilus species, Aggregatibacter actinomycetemcomitans, Cardiobacterium hominis, Eikenella corrodens, and Kingella kingae; IE, infective endocarditis; IgG, immunoglobulin G; MRSA, methicillin-resistant Staphylococcus aureus; MSSA, methicillin-susceptible Staphylococcus aureus; MR-CoNS, methicillin-resistant coagulase-negative staphylococci; MS-CoNS, methicillin-susceptible coagulase-negative staphylococci.
Imaging
Echocardiography was performed at least once in 99.5% of patients with IE, transthoracic echocardiography (TTE) in 83.0% and transoesophageal echocardiography (TOE) in 86.0% of cases. There were no significant differences in usage between groups, neither significant differences in the echocardiographic assessment of vegetation presence (group 1: 76.0%; group 2: 72.1%; group 3: 69.0%; group 4: 70.0%; P = 0.491; combined groups 2 to 4 with AT: 70.5%; vs group 1: P = .079) and size (group 1: 14.4 ± 6.9 mm; group 2: 15.2 ± 6.4 mm; group 3: 12.0 ± 5.1 mm; group 4: 13.1 ± 6.0 mm; P = 0.112; combined groups 2 to 4 with AT: 13.7 ± 6.0 mm; vs group 1: P = .538) or other local IE complications. Variables significantly associated with a vegetation length of more than 10 mm by multivariable logistic regression analysis were mitral valve IE (OR = 3.56; 95% CI [2.03–5.51]; P = <.001) and indication for surgery (OR = 2.10; 95% CI [1.12–3.95]; P = .021). 18F-FDG positron emission tomography/computed tomography (18F-FDG PET/CT) was performed at least once in 131 of 363 (36.1%), multislice CT in 287 of 363 (79.0%) (of which 136 of 363 (37.5%) brain CT) and MRI in 78 of 363 (21.5%) (of which 69 of 363 (19.0%) brain MRI), without statistically significant differences between groups.
In-Hospital Complications Under Antibiotic Treatment
All patients received intravenous antibiotic treatment. The main in-hospital complications are shown in Table 4. AKI (42.7%) was the most frequent in-hospital complication, possibly due to the older patients with IE with regularly underlying chronic renal failure (23.1%). No significant differences in complications were observed between IE groups in this study. Particularly, this study found no significant differences in TE rates between the AT-treated IE groups 2, 3 and 4 (84 of 234, 35.9%) and the younger control group 1 without AT (37 of 129, 28.7%; P = .163). Similarly, no significant differences were found in the incidence of hemorrhagic stroke (groups 2 to 4: 15 of 234, 6.4%; group 1: 3 of 129, 2.3%; P = .086). Multivariable logistic regression analysis identified mitral valve IE to be significantly associated with TE (OR = 2.54; 95% CI [1.39–4.64]; P = .002). Cerebral hemorrhagic events were significantly associated with cerebrovascular embolism by logistic multiple regression analysis (OR = 2.55; 95% CI [1.00–6.49]; P = .049).
In-Hospital Complications of Infective Endocarditis Patients per Group.
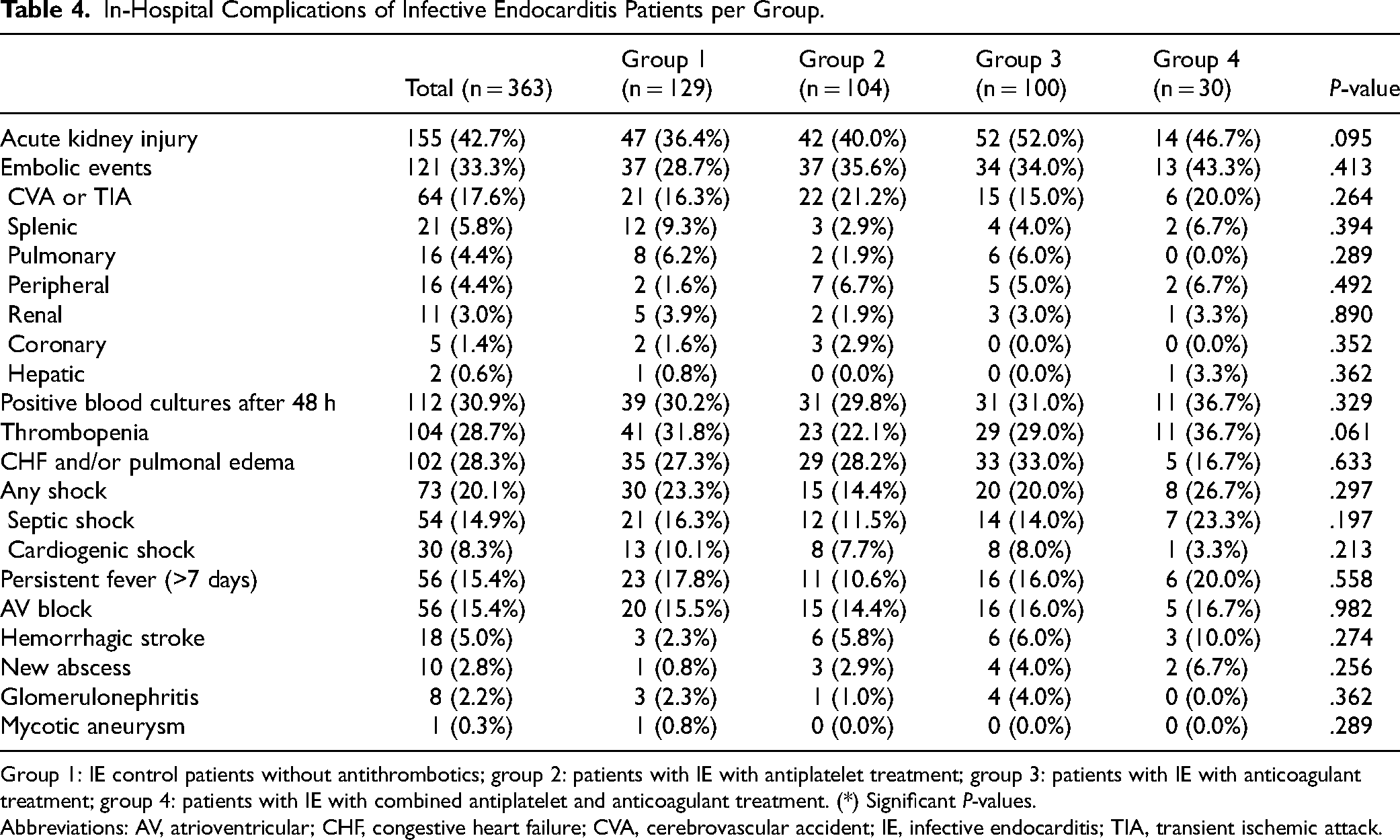
Group 1: IE control patients without antithrombotics; group 2: patients with IE with antiplatelet treatment; group 3: patients with IE with anticoagulant treatment; group 4: patients with IE with combined antiplatelet and anticoagulant treatment. (*) Significant P-values.
Abbreviations: AV, atrioventricular; CHF, congestive heart failure; CVA, cerebrovascular accident; IE, infective endocarditis; TIA, transient ischemic attack.
Cardiac Surgery and Mortality
ESC guidelines-derived theoretical indications for cardiac surgery (213 of 363, 58.7%) were not significantly different across the IE groups (P = .432).1,8 The most frequent indication in all groups was an uncontrolled infection (43.3%). Surgery was performed when theoretically indicated in 179 of 213 (84.0%) of patients with IE, without significant differences between IE groups with and without AT (P = .133). The most common reason for not performing surgery was a high surgical risk (15.4%, P = .078). Bioprostheses were most often implanted (aortic valve position: 79.4% versus 14.7% mechanical; mitral valve position: 42.3% versus 24.3% mechanical). Mitral valve repair was performed in 30.8% of mitral valve IE.
In-hospital death occurred in 78 of 363 (21.5%) patients with IE, without significant differences between groups (group 1: 28 of 129 (21.7%); group 2: 15 of 104 (14.4%); group 3: 29 of 100 (29.0%); group 4: 6 of 30 (20.0%); P = .091). This study found no significant difference in all-cause mortality between the older, AT-treated IE groups 2, 3 and 4 (50 of 234, 21.4%) and the younger control group 1 without AT (28 of 129, 21.7%; P = 1.000). In contrast, there was a significant difference in cardiovascular death between the 4 groups (P = .029), mainly driven by heart failure. Causes of mortality are shown in Supplemental Table 4. The predominant cause of mortality was noncardiovascular (11.6%; P = .607), primarily from uncontrolled sepsis (10.2%). Independent variables significantly associated with mortality in patients with IE, identified through multivariable Cox regression analysis adjusted for age, are presented in Table 5. The results of the univariable analysis are shown in Supplemental Table 5.
Variables Significantly Associated with Mortality by Multivariable Cox Regression Analysis.
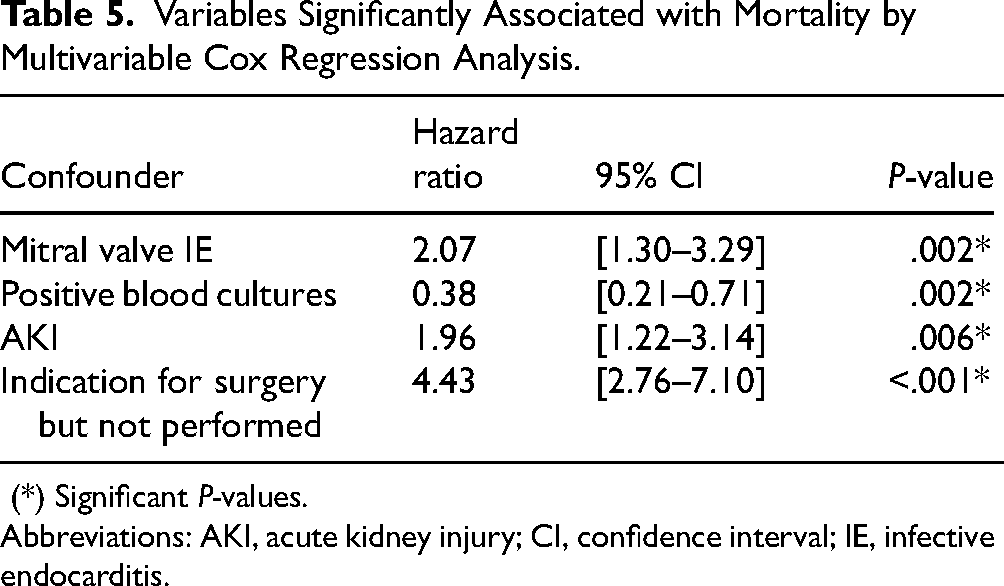
(*) Significant P-values.
Abbreviations: AKI, acute kidney injury; CI, confidence interval; IE, infective endocarditis.
Additionally, 173 of 363 (47.7%) patients with IE met a composite outcome of TE, cerebral bleeding, and all-cause in-hospital mortality, without significant differences between groups (group 1: 57 of 129 (44.2%); group 2: 49 of 104 (47.1%); group 3: 50 of 100 (50.0%); group 4: 17 of 30 (56.7%); P = .608).
Discussion
The management of AT (antiplatelet and anticoagulant treatments) in IE remains debated. 10 This is the first study that provides a direct comparison between patients with IE treated with various regimens of pre-existing AT at admission. The following key findings arise from this retrospective, multicentric analysis: first, no significant difference in in-hospital mortality between patients with IE treated with and without pre-existing AT was found. Second, this study did not find pre-existing AT in patients with IE to be associated with a significant difference in TE or cerebral hemorrhage. Third, there is a nonsignificant trend toward a reduced vegetation size in the anticoagulant-treated IE groups.
In-Hospital Mortality
In-hospital all-cause mortality was slightly higher in this study (78 of 363, 21.5%) compared to the European Endocarditis registry (EURO-ENDO) (17.1%), likely due to the older age of our population (68.4 ± 14.2 years vs 59.3 ± 18.0 years old in EURO-ENDO). 2 Conversely, the current analysis found no significant difference in all-cause mortality between the older, AT-treated IE groups 2, 3, and 4 (50 of 234, 21.4%) and the younger control group 1 without AT at admission (28 of 129, 21.7%; P = 1.000), which may challenge the traditional caution against AT use in IE. This finding aligns with other retrospective and prospective studies on the use of ASA and VKA in patients with IE,5,6,11–14 but contrast with an older study linking anticoagulant therapy to death in S. aureus PVE. 15 Additionally, no significant differences were observed between IE groups when analyzing a combined outcome of TE, cerebral bleeding, and all-cause mortality (P = .608). Interestingly, this analysis showed a nonsignificant trend toward lower all-cause mortality in the antiplatelet treated group 2 (15 of 104, 14.4%) compared to control group 1 (28 of 129, 21.7%; Bonferroni-corrected P = .154), despite group 2 being older (73.2 ± 10.1 years vs group 1: 62.0 ± 16.1 years). Univariable Cox regression analysis confirms pre-existing antiplatelet therapy to be a significant independent, protective predictor of all-cause mortality (hazard ratio (HR) = 0.56; 95% CI [0.32–0.98]; P = .041). After adjustment for age, AT at admission is not predictive of all-cause mortality using multivariable Cox regression analysis, suggesting that pre-existing AT does not significantly influence mortality in patients with IE. Meanwhile, AKI, mitral valve IE, positive blood cultures and especially unperformed cardiac surgery when indicated (HR = 4.43; 95% CI [2.76–7.10], P < .001) remained independent variables significantly associated with mortality by multivariable analysis. Positive blood cultures upon IE diagnosis are protective by enabling targeted treatment based on the antibiogram. 16 In contrast, higher mortality rates have likewise been observed previously in patients with a surgical indication not undergoing surgery.2,17,18 In this analysis, the primary reason for denying surgery was a high surgical risk, associated with advanced age, frailty, and comorbidities.
In-Hospital Thrombo-Embolic Events and Haemorrhagic Stroke
At admission, there were no significant differences in the incidence of TE (12.1%, P = .053) and hemorrhagic stroke (1.4%, P = .326) between IE groups with or without pre-existing AT. Despite the initiation of antibiotic therapy, additional TE during hospitalization was detected in 33.3% of IE cases, consisting mainly of cerebrovascular accident (CVA) and transient ischemic attack (TIA) (in 17.6%). These TE rates are higher than those reported in EURO-ENDO (20.6%), possibly due to better detection because of the more frequent use of imaging in our study (CT: 78.8% vs 53.1% in EURO-ENDO). 2 While no significant differences in TE rates during hospitalization were observed between the older, AT-treated IE groups 2, 3, and 4 (84 of 234, 35.9%) and the younger control group 1 without AT at admission (37 of 129, 28.7%; P = .163), there was a trend toward a lower frequency of CVA and TIA in the older, anticoagulant treated group 3 at admission (5.0%, Bonferroni-corrected P = .157), and during in-hospital follow up (15.0%, Bonferroni-corrected P = .792) compared to the younger control group 1 (10.1% and 16.3%, respectively). However, no causal benefit of AT in the prevention of TE can be inferred from these data. Additionally, although not statistically significant, the total TE rates were higher in the combined AT groups 2 to 4 compared to the non-AT group 1. This could potentially be explained by the older age of these patients or by vegetations becoming more friable under AT therapy, as has been previously suggested.4,19 Previous clinical research, mostly retrospective, has yielded discrepant results, with some studies showing a reduced risk of TE in patients with IE under anticoagulation as well as under antiplatelet therapy.3,5,12,14 A meta-analysis indicated that the risk of TE was significantly higher in patients with IE who discontinued anticoagulant therapy. 20 In contrast, other studies have reported an increased incidence of CVA with VKA treatment, and no reduction in TE among patients with IE on ASA (administered after IE diagnosis).13,21 A prospective, randomized, placebo-controlled trial found no difference in TE between ASA- and placebo-treated IE groups. 4
During in-hospital follow up, there were no significant differences in the incidence of hemorrhagic stroke between the older, AT-treated IE groups 2, 3, and 4 (15 of 234, 6.4%) and the younger control group 1 without AT at admission (3 of 129, 2.3%; P = .086). These results align with previous studies of AT in IE.6,11,12,14,22 Historically, safety concerns about the potential risk of hemorrhagic conversion of ischemic cerebral lesions or rupture following septic arteritis, have discouraged the concurrent use of AT during IE treatment.1,21 However, the current data do not demonstrate a significant influence of pre-existing AT on the incidence of cerebral hemorrhage in patients with IE.
This study identified mitral valve IE to be significantly associated with TE by logistic regression analysis. Prior research has shown that mitral valve vegetations are associated with a higher incidence of TE, likely due to the frequent destabilization of attached vegetations by a more rapid and forceful movement during mitral valve closure. 23 Additionally, mitral valve IE has been linked to larger vegetations, which further increases the risk of TE. 24 CVA and TIA are significantly associated with hemorrhagic events by logistic regression analysis, likely due to the afore mentioned hemorrhagic transformation of pre-existing ischemic lesions. 25
IE Characteristics, Patient Demographics, and Comorbidities
In this study, the prevalence rates of PRVE (25.1%) and CDRIE (9.1%), as well as the distribution of valvular IE locations, closely match those documented in the global IE population.2,17 NVE was significantly more common in the control IE group 1, while PRVE was significantly more prevalent in AT-treated IE groups 2, 3, and 4. Patients who undergo cardiac valve interventions face an elevated risk of prothrombotic complications, hence current guidelines generally advocate for AT usage in this population. 26 AT-treated IE groups 2, 3, and 4 also showed a significantly higher incidence of aortic valve IE (P = .016), due to the more frequent occurrence of aortic valve PRVE (61, 16.8%) compared to mitral valve PRVE (17, 4.7%) and tricuspid valve PRVE (0, 0.0%) in this analysis.
The control IE group was significantly younger than the AT IE groups (P < .001), likely because AT are predominantly prescribed to elderly individuals with more frequently underlying cardiovascular comorbidities. In this study, patients with IE commonly retained a history of arterial hypertension (56.5%), valvular heart disease (52.3%), atrial fibrillation (28.7%), and ischemic heart disease (26.4%). Significant differences in comorbidities between groups generally align with recognized medical indications for long-term AT.
Microbiology
In this study, only 10.7% of IE cases were blood culture negative, in contrast to around 20% in the global EURO-ENDO population. 2 This discrepancy may possibly be attributed to the more rapid and liberal hemoculture collection prior to antibiotic administration at our facilities. The most frequently identified microorganism was S. aureus (22.9%), followed by viridans streptococci (19.8%), CoNS (17.4%), and Enterococcus (10.7%). This contrasts with the global EURO-ENDO data, where Enterococcus surpassed viridans streptococci IE (15.8% vs 12.4%, respectively). 2
Previous studies have shown that S. aureus and certain CoNS can adhere to the heart valves by binding to von Willebrand factor (vWF).27,28 AT might potentially influence the vWF binding capacity of these microorganisms, by diminishing thrombin and/or platelet activation and indirectly reducing platelet-vWF interactions.29,30 An experimental study has indeed shown that combined ASA and ticlopidine treatment provides protection against S. aureus in a rat IE model. 31 Additionally, in vitro models have shown that ticagrelor exhibits bactericidal activity against S. aureus, and that dabigatran interferes with S. aureus coagulase activity, providing protection against IE as well.32,33 Despite these previous findings, our study showed no significant difference in the incidence of S. aureus (P = .560) or CoNS infections (P = .511) between the control IE group 1 and the AT-treated IE groups 2, 3, and 4. In line with previous studies, there was a significantly higher prevalence of viridans streptococci IE in the younger control group 1 (27.1%, P = .012), while the older AT IE groups were more commonly affected by Enterococcus (P = .064). 34 As such, logistic multiple regression analysis identified age, but also AKI to be significantly associated with the infecting microorganisms. An association between S. aureus bacteremia and AKI has previously been reported, potentially due to the nephrotoxicity of certain antibiotics. 35
Imaging
The wider use of imaging techniques that followed the publication of the 2015 ESC guidelines was clear in this study, as TEE was performed in 86.0% (vs 58.1% in the EURO-ENDO global population) and 18F-FDG PET/CT in 36.0% (vs 16.6%) of cases.2,8 The presence and size of vegetations on echocardiography have previously been identified as predictors of increased TE risk.1,36 In this study, a nonsignificant trend toward fewer and smaller vegetations was observed in the anticoagulant-treated IE groups 3 and 4 (Bonferroni-corrected P = .299). AT could potentially reduce the formation of platelet-fibrin thrombus and inhibit vegetation growth.5,11 In vitro and animal studies examining the effects of ASA, ticlopidine and dabigatran on vegetation development have previously shown promising results.37–40 Conversely, other studies have failed to observe a reduction in vegetations when preventively using ASA in rodents.31,39,41 Importantly, a prospective, randomized controlled trial failed to demonstrate any significant effect of ASA on the number and size of vegetations in patients with IE. 4 In the current study, multivariable analysis identified mitral valve IE and a surgical indication to be significantly associated with a vegetation size of more than 10 mm. Indeed, current guidelines recommend surgery for patients with IE with large vegetations due to their association with an elevated risk of TE and mortality.1,7 Furthermore, previous research has indicated that vegetations might be more frequently detected in patients with mitral valve IE, and these vegetations tend to be significantly larger. 42
Limitations
Measured as well as unmeasured confounders may bias the results of this retrospective study. Notably, there is a considerable heterogeneity in patients’ age and a disproportionate prevalence of some comorbidities and microorganisms (eg, viridans streptococci) among the 4 groups in this study, which are independently associated with complications and mortality. The disparity in the incidence of NVE (not on anticoagulants) and PRVE (on anticoagulants) between the control patients with IE (group 1) and AT-treated patients with IE (groups 2–4) may also affect complications and mortality rates.5,13 Moreover, this study includes patients with CDRIE (33 of 361, 9.1%) and isolated tricuspid valve IE (7 of 361, 1.9%), which are associated with a lower risk of developing TE. 43 However, a limited subanalysis restricted to patients with aortic valve IE and mitral valve IE (n = 274) demonstrated no significant differences in the main outcome results, as detailed in Supplemental Table 6.
Additionally, the study covers a period of 15 years, during which changes in AT regimens have occurred, including the introduction of DOACs. Consequently, patients with IE in 2009 may have received different treatment regimens compared to those in 2022 despite similar clinical conditions. Due to the small patient populations, patients with concomitant use of VKA and DOAC-treated IE were combined into group 3, potentially blunting differences in outcomes. However, a limited subanalysis comparing VKA-treated patients with IE (n = 60) and DOAC-treated patients with IE (n = 40) showed no significant differences in all-cause mortality (P = .096), TE (P = .276), or hemorrhagic stroke (P = .327). Furthermore, the small sample size of particularly group 4 makes statistical comparisons less reliable. Moreover, it could be debated whether patients on short-term UFH or LMWH at the time of admission should be included in groups 3 and 4, as the exact duration and underlying reason for this anticoagulation is often unclear and may be associated with an initial clinical manifestation of IE (ie, TE). However, only 14 patients received LMWH in this study, and none were ultimately treated with UFH, as shown in Supplemental Figure 1. A limited subanalysis excluding the 14 patients treated with LMWH demonstrated that the main outcome results remained consistent with the overall findings, as presented in Supplemental Table 7.
Most importantly, precise information on the duration and dose of AT prior to IE development, as well as INR values and the (dis)continuation of AT treatment during hospitalization (possibly influenced by findings on imaging), are lacking in this retrospective study. As a result, patients could only be classified based on their pre-existing AT. However, it can be inferred that AT management would be revised in the presence of in-hospital complications due to IE, as recommended by the current guidelines. 1 Therefore, a future prospective, observational IE registry should consider documenting the evolution of AT-usage from baseline through hospitalization, at discharge, and during follow up.
Furthermore, only a small number of cerebral hemorrhagic events were detected in this study. Selection bias is possible in this retrospective study, as patients at higher pre-existing bleeding risk may have been less likely to receive AT. Second, reliable INR values were unavailable, and lower INR levels may have contributed to the low incidence of hemorrhagic complications among anticoagulated patients. Third, physicians may have discontinued AT during hospitalization to mitigate bleeding risk. Finally, in line with other IE studies, TE and hemorrhagic stroke may have been underdiagnosed due to the lack of routine (cerebral) imaging. Although sepsis was the dominant cause of all-cause mortality, overall patient survival could have been influenced by subtle (and possibly missed) bleeding or TE. Future, prospective studies should consider routine screening for silent complications, and it would be of interest to distinguish and systematically register rates of symptomatic and asymptomatic events to provide more accurate and clinically meaningful insights.
While acknowledging these limitations, our findings may nonetheless contribute to the evolving landscape of innovative IE management. The results specifically advocate for a tailored approach to AT therapy in the context of IE, considering individual patient risk factors and clinical presentation, in close consultation with the Endocarditis Team as recommended by the current guidelines. 1 Given the challenge of increasing rates of infections by antibiotic-resistant bacterial strains in IE, the use of AT as an adjunct therapy to prevent vegetation growth warrants further investigation. 39 Specifically, large, prospective registries are needed to investigate the impact of newer AT, such as DOACs and ticagrelor, on clinical outcomes and to determine the influence of AT on surgical timing in IE. However, recruitment will remain challenging due to the low incidence of IE.
Conclusion
This is the first study to directly compare mortality and complications of patients with IE receiving pre-existing antiplatelet therapy, anticoagulation therapy, or a combination of both, against patients with IE not receiving any AT at admission. Our findings indicate no significant difference in in-hospital mortality between patients with IE treated with pre-existing AT and those without it. Additionally, AT at admission in patients with IE does not seem to lead to a significant difference in TE or cerebral hemorrhage. Although limited by its retrospective nature, this analysis challenges the traditional caution against AT use in IE.
Supplemental Material
sj-docx-1-hvs-10.1177_30494826251336319 - Supplemental material for Antithrombotic Therapy in Infective Endocarditis: A Retrospective Analysis of Mortality and Complications
Supplemental material, sj-docx-1-hvs-10.1177_30494826251336319 for Antithrombotic Therapy in Infective Endocarditis: A Retrospective Analysis of Mortality and Complications by Bram Roosens, Sarah Brewée, Johan De Sutter, Andreea Motoc, Anne-Marie Willems, Delphine Vervloet, Xavier Galloo, Philippe Unger, Edgard Prihadi, Steven Droogmans, Gilbert Habib and Bernard Cosyns in Journal of the Heart Valve Society
Supplemental Material
sj-docx-2-hvs-10.1177_30494826251336319 - Supplemental material for Antithrombotic Therapy in Infective Endocarditis: A Retrospective Analysis of Mortality and Complications
Supplemental material, sj-docx-2-hvs-10.1177_30494826251336319 for Antithrombotic Therapy in Infective Endocarditis: A Retrospective Analysis of Mortality and Complications by Bram Roosens, Sarah Brewée, Johan De Sutter, Andreea Motoc, Anne-Marie Willems, Delphine Vervloet, Xavier Galloo, Philippe Unger, Edgard Prihadi, Steven Droogmans, Gilbert Habib and Bernard Cosyns in Journal of the Heart Valve Society
Footnotes
Abbreviations
Acknowledgments
The authors would like to thank Ingrid Lemoine and Kurt Barbé for their support.
ORCID iDs
Ethical considerations
The study was approved by the local Ethical Committee of both hospitals and was carried out in accordance with the ethical principles for medical research involving human subjects established by Helsinki's Declaration, protecting the privacy of all participants, as well as the confidentiality of their personal information.
Consent to participate
Not applicable. This is a retrospective, observational study. The Ethical Committee has confirmed that no informed consent was required.
Author contributions/CRediT
Bram Roosens, Johan De Sutter, Philippe Unger, Steven Droogmans, Gilbert Habib, and Bernard Cosyns contributed to the study conception and design. The methodology was conceptualized by Bram Roosens, Johan De Sutter, Andreea Motoc
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data presented in this study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
