Abstract
Background
Stroke remains a concern after transcatheter aortic valve replacement (TAVR). Several studies attempted to identify correlates of stroke but there remains a need for better predictors.
Objective
This study aims to characterize the aortic arch to identify potential correlates of stroke after TAVR using a patient cohort that includes cases with and without stroke.
Methods
A retrospective analysis was performed on 52 patients who underwent TAVR at Mayo Clinic and Maria Cecilia Hospital. Of these patients, 26 had a stroke after TAVR and 26 did not (control). Anatomical characterization was performed on each patient. Patient-specific 3D digital models of the ascending aorta, aortic arch, and calcium were created for each patient. Geometrical measurements, including cross-sectional areas, volumes, tortuosity, curvature, and length of the model were recorded.
Results
There was no significant difference in the various area measurements between the two groups, but the aortic arch cross-sectional area ratio (cerebral ostia cross-sectional areas/arch cross-sectional area) was significantly higher in the stroke group. Additionally, there was no significant difference in tortuosity, curvature, aortic volume, and length of the aorta. Aortic valve calcium score, aorta calcium, and aortic arch type (I, II, or III) were not significantly different between the groups. The prevalence of a bovine arch (BA) configuration was significantly higher in the stroke group (46% vs 8% control, p < 0.05).
Conclusion
Aortic arch cross-sectional area ratio and BA variant were the most significant correlates of stroke after TAVR.
Keywords
Key Points
Stroke is a major concern after transcatheter aortic valve replacement (TAVR). Several studies have attempted to identify stroke predictors after TAVR with some success. However, better predictors are still needed and will likely require a combination of geometric and patient-specific anatomical features. A bovine arch variant and the ratio of the branching aortic arch arteries’ cross-sectional areas to the descending aorta's cross-sectional area were strong predictors of stroke. Other geometric and anatomical predictors, although not significant in this cohort, may still be of importance and warrant further investigation in a larger study.
Introduction
Transcatheter aortic valve replacement (TAVR) is a minimally invasive procedure that aims to replace the diseased native aortic valve with a transcatheter bioprosthetic valve for patients with aortic stenosis.1,2 Stroke is a major concern for TAVR patients; results of the PARTNER 3 and PARTNER 2A trials showed 30-day stroke rates of 5.5% in intermediate-risk 3 and 0.6% in low-risk 4 patients. Retrospective studies have shown an increase in 30-day mortality for stroke positive patients.5,6
Embolization of calcific plaque can occur during a TAVR procedure, resulting in periprocedural strokes. Due to calcification of the aortic valve, the use of rigid, large delivery catheters, and the interaction between the TAV stent and the native valve during deployment, embolism may occur.7–9 The mechanism of embolic events may differ between valve types, where emboli are created during positioning (ballon-expandable) or deployment (self-expanding).10,11 The flow dynamics downstream of different valves are also different,12–14 leading to various transport mechanisms that can correlate with stroke. Although stroke rates after TAVR are decreasing, studies have shown that post-TAVR silent ischemic embolic lesions are almost always present.15,16 In addition to embolic debris originating from the aortic valve, aortic wall calcium can also embolize when the delivery catheter system interacts with it. Patients with extensive calcium in the aorta may require alternative access sites for TAVR, though some of these sites have been associated with increased stroke risk. 17
Studies have shown that significant high-intensity transient signals (HITS) occur during valve crossing in TAVR procedures, potentially indicating a high risk of stroke. Kahlert et al 18 reported that during TAVI, intracoronary HITS were observed in all cases and during every defined procedural step. The highest numbers of HITS were observed during the initial crossing of the native valve and during the positioning of the bioprosthesis. 18 This study suggested that the valve crossing stage of TAVR procedures may indeed represent a period of heightened stroke risk due to the high number of HITS observed. However, it's important to note that the study found no significant correlations between the amount of HITS and post-procedural increases in troponin I, indicating that the relationship between HITS and actual myocardial or cerebral injury is complex and requires further investigation.
To reduce the risk of periprocedural stroke, embolic protection devices have been developed to shield at least two of the major cerebral vessels branching from the aortic arch, effectively capturing calcific debris generated during the TAVR procedure. Several of these devices are commercially available, including the Boston Scientific SENTINEL, Keystone Heart Triguard, and Edwards Lifesciences Embrella. The results of the DEFLECT I study showed the TriGuard device did not have a significant effect on the occurrence of strokes, and the PROTECTED TAVR clinical trial had a similar finding for the SENTINEL device.19,20 This highlights the importance of identifying predictors of stroke.
Several studies have identified potential predictors for early stroke based on patient characteristics and previous medical history.21–26 Although this can provide insight into what can potentially increase the risk of stroke, some of these predictors have been inconclusive or contradictory between different studies. For example, several studies identified female sex and bilateral carotid stenosis as predictors of stroke,21,27,28 and others showed that these factors are insignificant.25,29 Additionally, some studies reported that early stroke risk was higher after transapical versus transfemoral access TAVR (SAPIEN devices with either 23Fr or 24Fr sheaths).25,30 However, other studies reported similar stroke rates between the two access routes (SAPIEN or CoreValve with no mention of delivery system size).24,31 As a result of the inconsistencies in proposed stroke correlates, there is still a need in the field to identify more correlates to assist cardiologists with post-procedural patient management in the absence of fixed guidelines.
The aim of this study is to identify geometric and anatomical correlates of stroke after TAVR using a patient cohort that includes cases with and without stroke.
Methods
Patient Data
This multicenter retrospective study used a database of 52 patients who underwent TAVR at Mayo Clinic (Rochester, MN, USA) and Maria Cecilia Hospital (Cotignola, Italy). Of these patients, 26 had a stroke after TAVR and 26 did not (control). The dataset included computed tomography (CT) images which were used to create patient-specific models of the ascending aorta and aortic arch. Also included were patient characteristics, anatomical features, valvular calcium score, and procedure methods and outcomes. This study was performed under an approved Institutional Review Board (IRB) protocol and data usage agreement (DUA) between all institutions.
Digital 3D Model Construction
The provided CT images were imported in Mimics (v23.0; Materialise, Belgium) for the development of patient-specific 3D digital models and post processing was performed using 3-Matic (v17.0; Materialise, Belgium) similar to previous work.32,33 The workflow starts by applying a mask to the CT images of each patient using a Hounsfield unit (HU) threshold that isolates blood and calcium deposits from the surrounding soft tissue. Specifically, calcium was isolated by adjusting the threshold to only include higher HUs outside the range of blood. The mask is then segmented to extract the digital aorta model. Each model included the ascending aorta and aortic arch, brachiocephalic artery ostium, left common carotid artery ostium, and left subclavian artery ostium. In addition, the models were trimmed at sinotubular junction (STJ), brachiocephalic artery ostium, left common carotid artery ostium, left subclavian artery ostium, and the aortic arch just after the left subclavian artery (Figure 1) to ensure no bias was caused by the amount of the aorta and branching vessels visible in each CT image stack. A centerline was calculated for each model (Figure 1) using Mimics, which was used to measure the length of each model, maximum and mean curvature of the aorta, and tortuosity from the STJ to the ostium of the brachiocephalic and left subclavian arteries. Measurements were recorded for aorta volume, aorta calcium volume, STJ cross-sectional area, aortic arch cross-sectional area, ascending aorta cross-sectional area, and the ostium areas of the three branching arteries. Each cross-sectional area was taken normal to the centerline and their locations are shown in Figure 1. Area ratios were calculated by taking the sum of branching artery's ostium areas and dividing by the cross-sectional area of the STJ (Equation 1), ascending aorta (Equation 2), and aortic arch (Equation 3).
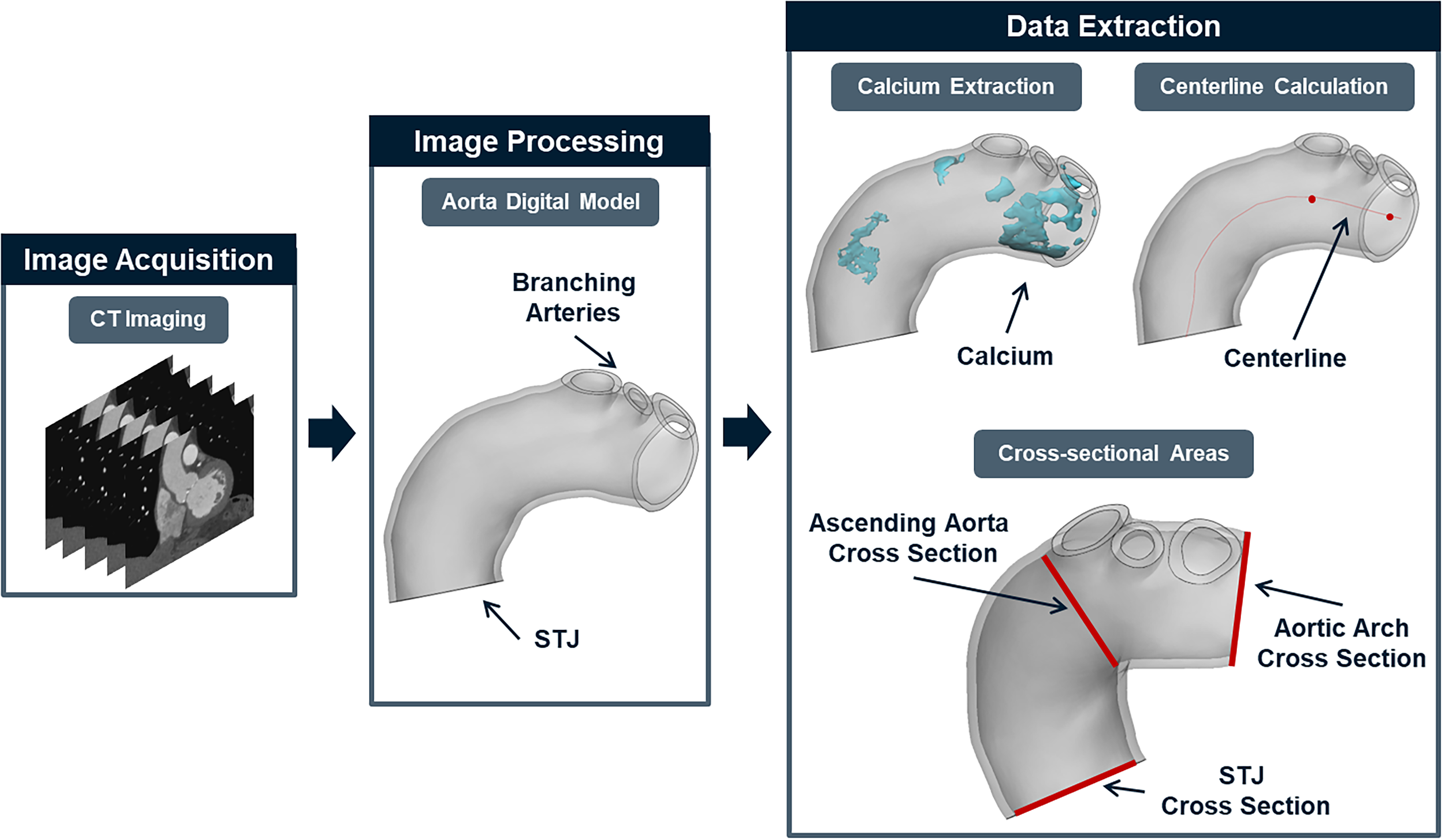
Aortic modeling and geometry measurements.
This method uses circumcircles to approximate curvature along the centerline, where pi, pi−1, and pi+1 represent consecutive points on the centerline. Tortuosity was calculated with Mimics based on Equation 5.
Statistics
Statistical analysis was performed using JMP Pro (v16.0.0; SAS Institute Inc, Cary, NC). Student's t test was used to compare the means (if the distribution was normal) and the Wilcoxon test was used for non-normal data distributions. p < 0.05 was considered statistically significant.
Results
Patient characteristics, anatomical features, and valvular calcium score are listed in Table 1. The presence of a bicuspid aortic valve (BAV) was similar between both groups. There were significantly more patients with a bovine arch (BA) in the stroke group (46% vs 8% control, p < 0.05). The distribution of aortic arch type I and II was the most different between the groups but was not significant. Aortic valve calcium score and aortic calcium volume (within the bounds of the model detailed in the methods section) were not significantly different between both groups.
Differences in Anatomy and Calcium Metrics for the Stroke and Control Groups.

Geometric measurements are listed in Table 2. Both groups had similar aortic volumes (within the bounds of the model detailed in the methods section). Tortuosity from the STJ to the left subclavian artery ostium was similar between both groups, but from the STJ to the brachiocephalic artery ostium, tortuosity was higher in the stroke group (p = 0.0591). The mean aortic curvature was similar between both groups, but the maximum curvature was higher in the control group, though the difference was not significant. All area measurements were similar between the two groups, except for the brachiocephalic ostium area, which was larger in the stroke group, though not significantly. The proposed area ratios were all higher in the stroke group, with the aortic arch ratio being significant (p < 0.05), while the cross-sectional ratio measurement was nearly significant (p = 0.0614).
Differences in Geometry and Centerline Metrics for the Stroke and Control Groups.

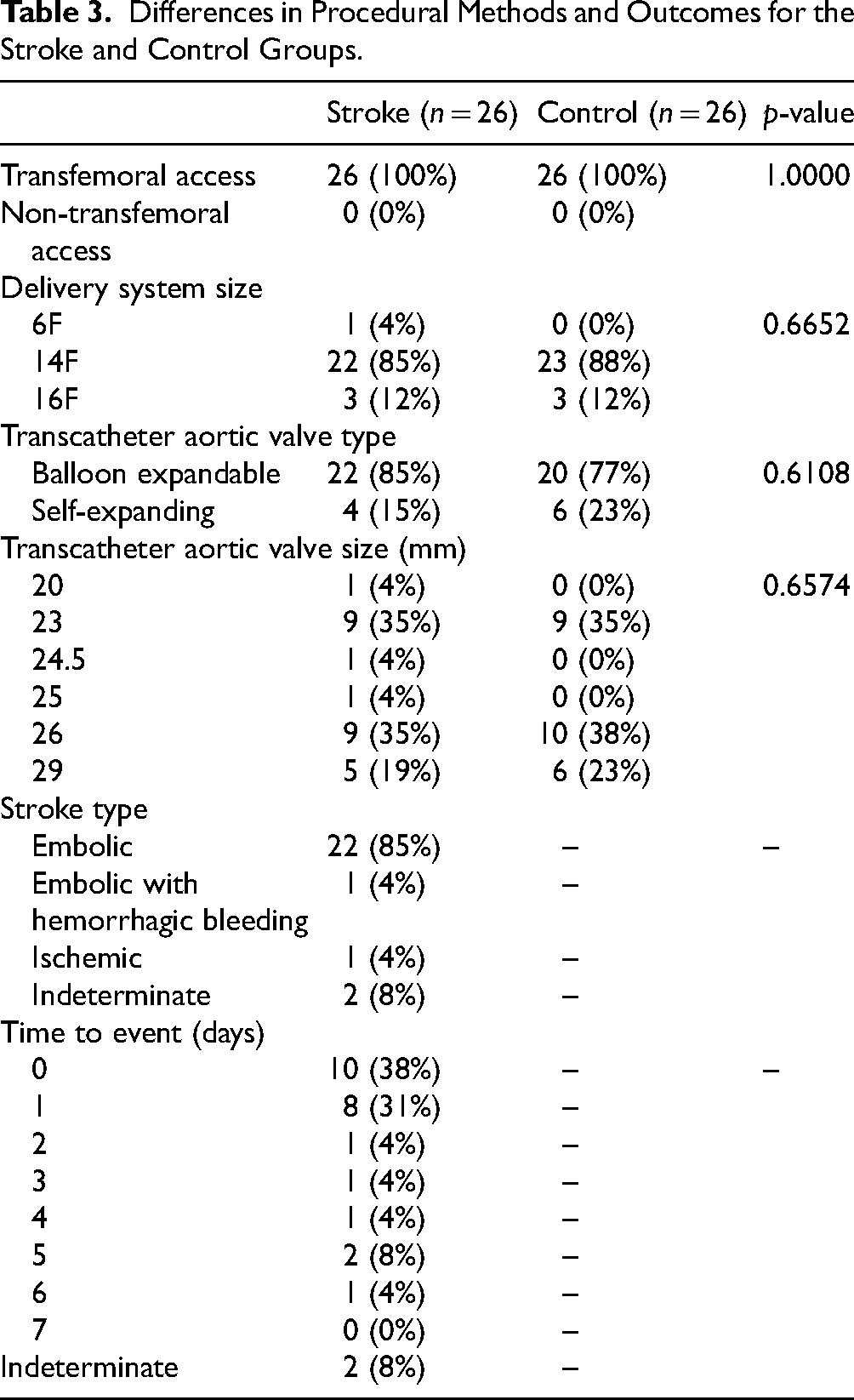
The procedural methods and outcomes are listed in Table 3. Transfemoral access was used in every case. The most common delivery system size for both groups was 14F, and the most frequently used valve type was balloon expandable. Embolic stroke occurred in 85% (n = 22) of the stroke group, with only one patient having embolic stroke and hemorrhagic bleeding. The time to a stroke event most often occurred within one day (n = 10).
Differences in Procedural Methods and Outcomes for the Stroke and Control Groups.
Discussion
In this study, we investigated different geometric and anatomical parameters of the aorta for correlates of stroke after TAVR. The most significant geometric difference was the aortic arch cross-sectional area ratio (the summation of the branching arteries ostia areas divided by the cross-sectional area of the aortic arch at a point just after the left subclavian ostium, see Fig. 1), while the ascending aorta cross-sectional area and the tortuosity from the STJ to the brachiocephalic and left subclavian artery ostia were close to achieving statistical significance. Another major finding of this study was the significant amount of a BAs in the stroke group (46% vs 8% control).
The risk of TAVR-related periprocedural stroke,34,35 and the consequent increase in morbidity and mortality,5,36,37 is elevated by embolic debris originating from the aortic valve. Elevated calcium scores of the aortic valve have been associated with an increased risk of periprocedural stroke. 38 In addition, increased calcific burden of the aorta and larger calcium thickness is also associated with increased stroke risk in TAVR patients.39,40 Bjursten et al 41 conducted a study to assess calcification patterns of the aortic valve to identify patients at a higher risk of periprocedural stroke during TAVR but did not find any predisposing patterns. The present study has shown a similar aortic valve calcium score and aorta calcium volume between both groups. Despite this, the underlying mechanism for debris transport and overall anatomy of the aorta in some patients may contribute to stroke risk but a larger patient cohort is necessary to investigate this further. Given that blood flow is a vehicle for debris transportation, it is important to consider its role in stroke development. Specific fluid mechanics studies on this topic are lacking, but fundamental fluid mechanic concepts may provide some insight. Centrifugal forces act on the blood in the aortic arch, creating Dean vortices and shifting the velocity profile away from the central axis.42–44 This results in higher blood velocity near the outer wall of the aorta, where the brachiocephalic, left common carotid, and left subclavian arteries are located. Consequently, the transport of debris from the aortic valve and ascending aorta may be redirected toward this wall, increasing the likelihood of it entering the branching arteries and causing embolization. A computational study from Carr et al 45 showed that medium sized particles, about 1 mm in diameter, were more likely to enter the branching arteries due to their inertia carrying them to the outer wall of the aortic arch. Debris that do not enter these arteries, often greater than 1 mm in diameter, can be transported to the whole body. 45 Peripheral emboli are often asymptomatic and undetected, but they can still pose a potential risk. A comprehensive fluid dynamics analysis of post-TAVR flow changes and particle tracking is necessary to better understand the transport of calcific debris, not only to the branching arteries but throughout the whole body.
Tortuosity has been a useful measurement to determine the difficulty of catheter guidance for TAVR. 46 Although there was no significant difference in tortuosity of the aorta between the groups in the present study, it is possible that patients with higher tortuosity may be at an increased risk of stroke due to the interaction of the catheter system with calcium in the aortic arch and ascending aorta. This risk is potentially intensified when considering the amount of calcium that may be present in these regions. Although no difference in aortic calcium volume was observed in this study, some patients may have higher calcium levels than others, potentially increasing their risk in cases that are tortuous. Given the constraints of our model to maintain consistency, tortuosity from a distal point of the descending aorta or branching arteries relative to the aortic valve was not measured. However, clinical investigations have identified an association between increased iliofemoral tortuosity scores and bleeding complications, 47 though it remains unclear whether this also increases the risk of peripheral emboli. In addition, the interaction of the catheter system with aortic wall thrombosis may increase the risk of thromboembolic events after TAVR. 48 Non-transfemoral access sites, such as transcarotid and transsubclavian, have been associated with an increased risk of stroke. 17 The use of these alternative access sites in patients with highly tortuous vessels may increase the interaction between the delivery system and calcium deposits in these regions, potentially elevating embolic risk but further studies are needed on this subject.
A bovine arch (BA) is an anatomical variant that deviates from the conventional arch anatomy of three main branches, instead there are only two branches due to a common origin between the brachiocephalic and left common carotid arteries. In the general population, this variant has been shown to have an increase in acute stroke risk of 22–25%.49,50 Lo Russo et al 26 found a 2-fold increase in TAVR periprocedural stroke and higher incidence of stroke at later follow-ups for patients with a BA, which accounted for 18% of the studied population. The present study has shown a BA prevalence of 46 and 8% in the stroke and control groups, respectively. Sun et al 51 reported that BAV patients are more likely to have a BA. However, in our study, only one patient had both a BAV and BA; larger studies are needed to further investigate this relationship. Stroke outcomes at 30 days after TAVR for low-risk BAV are mixed, some studies found a significantly higher rate of stroke52,53 whereas others did not.54,55 Increased stroke risk in BAV patients may result from increased calcification of the leaflets, the complex calcium patterns which can contribute to embolic debris during TAVR,56–58 and the even more complicated flow dynamics. 59 In addition, excessive calcium volume and calcification of the raphe can also complicate TAVR procedures, potentially increasing stroke risk.58,60 The results of this study show differences in the aortic arch cross-sectional area ratio between the stroke and control groups, with a significantly higher ratio in the stroke group (0.89 ± 0.37 vs 0.68 ± 0.14 control, p = 0.0243). This suggests that relative sizes of the aortic branching arteries ostia to the aortic arch area may be associated with stroke risk. When the ratio value approaches or exceeds 1, we believe that pressure gradients will be induced leading to promoting the movement of embolic debris into cerebral arteries with lower resistance. In a previous study, we demonstrated how the aortic arch affects pressure distribution downstream of an aortic valve, however, the study did not account for the branching arteries. 44 This needs to be assessed thoroughly using flow dynamics simulations. The ascending aorta cross-sectional area ratio showed a near-significant difference between the stroke and control groups (0.57 ± 0.22 vs 0.47 ± 0.11, p = 0.0614) too. Although not statistically significant, this may suggest that the relative size of the branching arteries ostia compared to the ascending aorta area may play a role in stroke risk, but larger studies are needed to fully realize this. The conclusion here, keeping in mind the low number of total patients (n = 52), is that a BA appears to be a potential correlate of stroke after TAVR. Again, the work of Lo Russo et al, which included a total of 2775 patients, also found indications of a BA being a potential correlate of stroke. Aortic arch type (I, II, or III) was not found to be significantly associated with stroke, consistent with another study that included 4590 patients. 61
Clinical Implications
Results from cerebral embolic protection devices have shown no difference in their reduction of stroke.62,63 At the time of writing, the BHF PROTECT-TAVI 64 study has been stopped prematurely, which now leaves previous studies19,20 as the current findings for protection devices. Based on the findings of the present study and those of Foley et al, 38 patients with a high amount of calcium may benefit from the use of a protection device. As shown in this study, BA patients may benefit from embolic protection devices given the elevated prevalence of BA variant in the stroke cohort, the findings of Lo Russo et al 26 further reinforce these findings. Moreover, embolic protection devices, such as the Sentinel (Boston Scientific, MA, USA), are particularly suited for BA variants. 65 This study has identified area ratios as a quick and simple method to stratify patients at a higher stoke risk than those that aren’t. A larger aortic arch cross-sectional area ratio that approaches or exceeds 1 correlates to an increased stroke risk.
Limitations
This study has several limitations, the first being the small sample size. Given that the significant findings of this study can be performed in clinic, a much larger study could be adopted to further assess these findings. The second limitation is the exclusion of the branching arteries of the aortic arch and the descending aorta. Despite studies showing stroke risk from parameters measured in the descending aorta,39,40,48 in this study, the consistency of the model boundaries (STJ to just after the left subclavian ostium) helps to reduce any differences in patients that may have been observed due to how much of the anatomy is present in each CT image stack. The last limitation is that not all patients were matched by age and sex due to the quality of some CT images and visibility of the aortic arch in CT for modeling purposes. Therefore, we have opted to show a global comparison between the 2 groups as opposed to a direct comparison of each patient.
Conclusion
Our hypothesis is that there are geometric features that correlate with stroke risk in patients undergoing TAVR. We explored that in patient specific models in 26 pairs of stroke and matched controls. We found that the most significant feature was the aortic arch cross-sectional area ratio, and that the bovine arch was the anatomical feature that correlates with stroke. There were also several other geometric features (brachiocephalic tortuosity and ascending aorta cross-sectional area ratio) that were close to significance and may still be clinically relevant. Further studies are needed to assess the flow dynamics in these patients, especially the BA patients, to identify potential flow indices that correlate with stroke after TAVR.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially funded by the National Science Foundation (NSF) award # 2301649.
Division of Chemical, Bioengineering, Environmental, and Transport Systems, (grant number 2301649).





